Assign 13 1 Dynamic Equilibrium and Equilibrium Constants

Assign. #13. 1 Dynamic Equilibrium and Equilibrium Constants

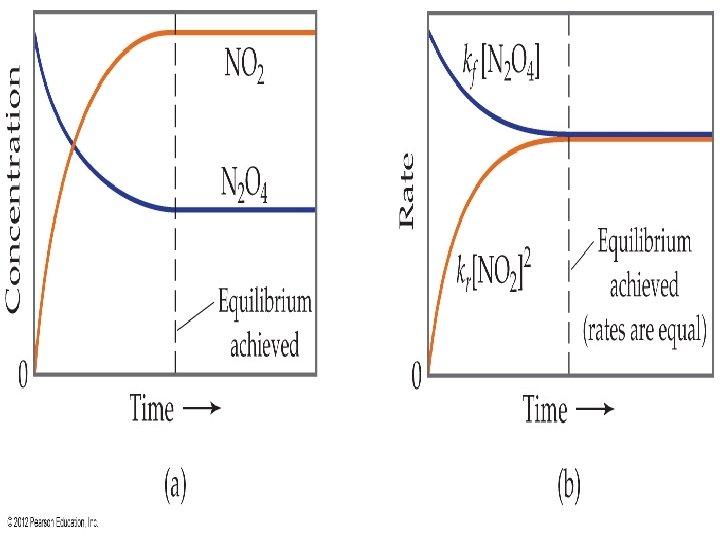
Equilibrium and Rates • The equilibrium below is two reactions: N 2 O ⇌ 2 NO 2 • There is the forward reaction: • The reverse reaction is: • At equilibrium the rate of the forward reaction is equal to the reverse reaction:
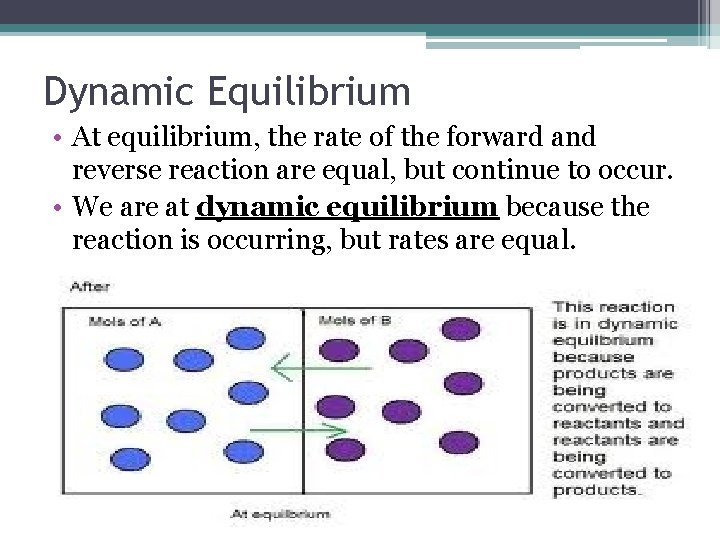
Dynamic Equilibrium • At equilibrium, the rate of the forward and reverse reaction are equal, but continue to occur. • We are at dynamic equilibrium because the reaction is occurring, but rates are equal.

Equilibrium Constants (K) • The ratio of products to reactants in equilibrium is known as the equilibrium constant (K) • Only written for aqueous and gaseous substances. Liquids and gases do not participate in equilibrium. • For the general reaction: a. A (aq) + b. B (aq)⇌ c. C (aq) + d. D (aq) • The equilibrium constant (Kc) for all aqueous substances is:

Gaseous Equilibrium Constants (Kp) • When the substances in an equilibrium, we can calculate the equilibrium constant using partial pressures. • This is represented as Kp • Kp≠Kc • For the equilibrium: a. A (g) + b. B (g) ⇌ c. C (g) + d. D (g)

Class Example • Write the equilibrium constant for the following equilibrium: 2 NO (g) + Cl 2 (g) ⇌ 2 NOCl (g)

Table Talk • Write the equilibrium constant for the following equilibrium: Ag+ (aq) + 2 NH 3 (aq) ⇌ Ag(NH 3)2+ (aq)

Quick Write • Which side of the equilibrium do you think is favored in the reaction below based on the value of Kp. Justify your response. CO (g) + Cl 2 (g) ⇌ COCl 2 (g) Kp = 4. 56 x 109

What K Tells Us • A large K indicates that there are more products than reactants at equilibrium • A small K indicates that there are more reactants than products at equilibrium • If K >> 1, then it is products favored • If K << 1, then it is reactants favored

Class Example • After mixture of hydrogen and nitrogen gases in a reaction vessel are allowed to attain equilibrium at 472 o. C, it is found to contain 7. 38 atm H 2, 2. 46 atm N 2, and 0. 166 atm NH 3. From these data calculate the equilibrium constant, Kp, and state whether it is product or reactants favored: N 2 (g) + 3 H 2 (g) ⇌ 2 NH 3 (g)
- Slides: 11