Assemblage Diagrams and the Effects of Volatiles Dr

Assemblage Diagrams and the Effects of Volatiles Dr. Stephen Crabtree Sept. 24, 2018
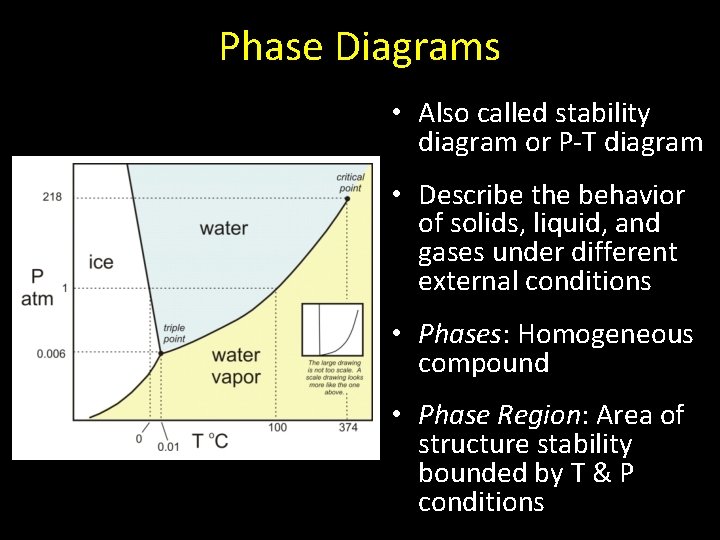
Phase Diagrams • Also called stability diagram or P-T diagram • Describe the behavior of solids, liquid, and gases under different external conditions • Phases: Homogeneous compound • Phase Region: Area of structure stability bounded by T & P conditions

Stability and Equilibrium • Coexisting minerals (phases) can sometimes be used to define the pressure and temperature of formation – Equilibrium formation if there are no reaction rims – Better yet if the grains are touching each other (but not always necessary) – Typically need additional evidence to prove equilibrium
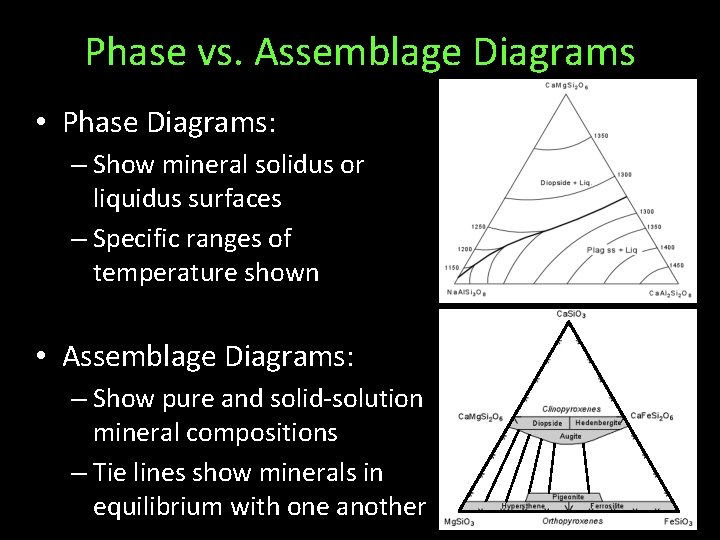
Phase vs. Assemblage Diagrams • Phase Diagrams: – Show mineral solidus or liquidus surfaces – Specific ranges of temperature shown • Assemblage Diagrams: – Show pure and solid-solution mineral compositions – Tie lines show minerals in equilibrium with one another mol% ternary assemblage diagram
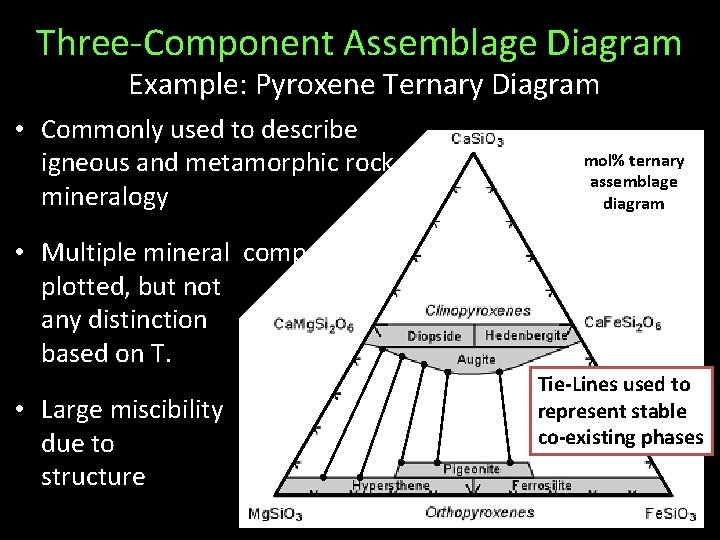
Three-Component Assemblage Diagram Example: Pyroxene Ternary Diagram • Commonly used to describe igneous and metamorphic rock mineralogy mol% ternary assemblage diagram • Multiple mineral compositions plotted, but not any distinction based on T. • Large miscibility due to structure crystal gaps Tie-Lines used to represent stable co-existing phases

Three-Component Assemblage Diagram Example: Silica-Nepheline-Kalsilite Ternary Diagram • Horizontal lines on this diagram show solid solutions • Tie-lines show phases coexisting with one another • Note: Si. O 2 cannot coexist with the feldspathoids nepheline or leucite, but feldspars can Si. O 2 Tridymite mol% ternary assemblage diagram KAl. Si 3 O 8 Orthoclase Na. Al. Si 3 O 8 Albite 1000 °C KAl. Si 2 O 6 Leucite *Na. Al. Si 2 O 6 Jadeite Na. Al. Si. O 4 Nepheline Fig 11. 12 from Klein, 23 nd Ed, p. 260 KAl. Si. O 4 Kalsilite

Three-Component Diagram Example: Calcite-Siderite-Magnesite Ternary Diagram Ternary diagram of metamorphic carbonate Siderite Only one region of triple coexistence among phases ~400 °C Ankerite Ca. Fe(CO 3)2 • Complete solid-solution between magnesite and siderite • Incomplete solid solution by calcite or dolomite Calcite Dolomite Magnesite
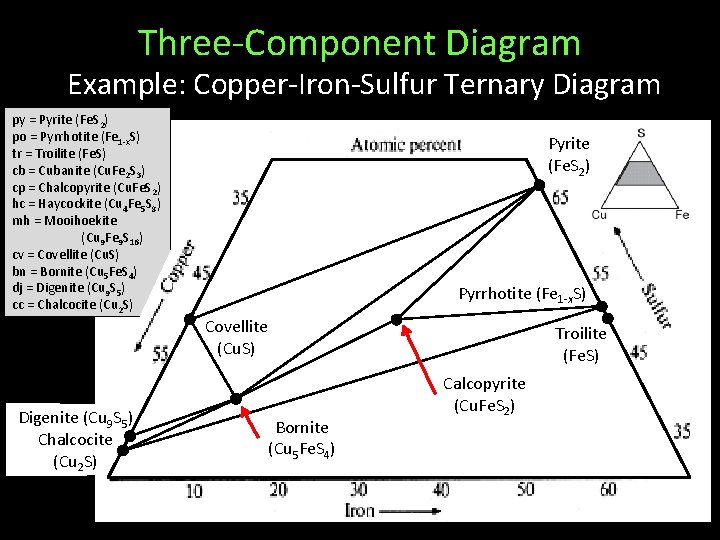
Three-Component Diagram Example: Copper-Iron-Sulfur Ternary Diagram py = Pyrite (Fe. S 2) po = Pyrrhotite (Fe 1 -x. S) tr = Troilite (Fe. S) cb = Cubanite (Cu. Fe 2 S 3) cp = Chalcopyrite (Cu. Fe. S 2) hc = Haycockite (Cu 4 Fe 5 S 8) mh = Mooihoekite (Cu 9 Fe 9 S 16) cv = Covellite (Cu. S) bn = Bornite (Cu 5 Fe. S 4) dj = Digenite (Cu 9 S 5) cc = Chalcocite (Cu 2 S) Digenite (Cu 9 S 5) Chalcocite (Cu 2 S) Pyrite (Fe. S 2) Pyrrhotite (Fe 1 -x. S) Covellite (Cu. S) Troilite (Fe. S) Bornite (Cu 5 Fe. S 4) Calcopyrite Solid-Solutions (Cu. Fe. S 2) Not Shown Many, many coexistence tie-lines

Three-Component Diagram Example: Copper-Iron-Sulfur Ternary Diagram What phases can coexist with Chalcopyrite? Pyrite (Fe. S 2) What phases cannot coexist with Bornite? Pyrrhotite (Fe 1 -x. S) Covellite (Cu. S) Digenite (Cu 9 S 5) Chalcocite (Cu 2 S) Troilite (Fe. S) Bornite (Cu 5 Fe. S 4) Calcopyrite Solid-Solutions (Cu. Fe. S 2) Not Shown Many, many coexistence tie-lines
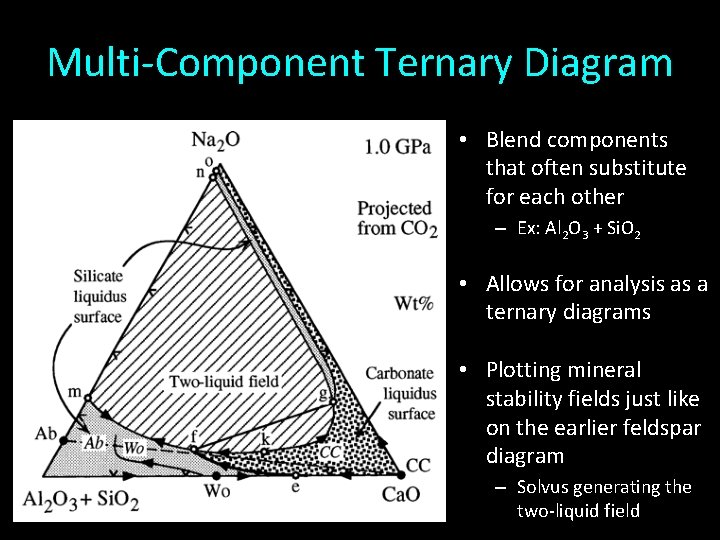
Multi-Component Ternary Diagram • Blend components that often substitute for each other – Ex: Al 2 O 3 + Si. O 2 • Allows for analysis as a ternary diagrams • Plotting mineral stability fields just like on the earlier feldspar diagram – Solvus generating the two-liquid field
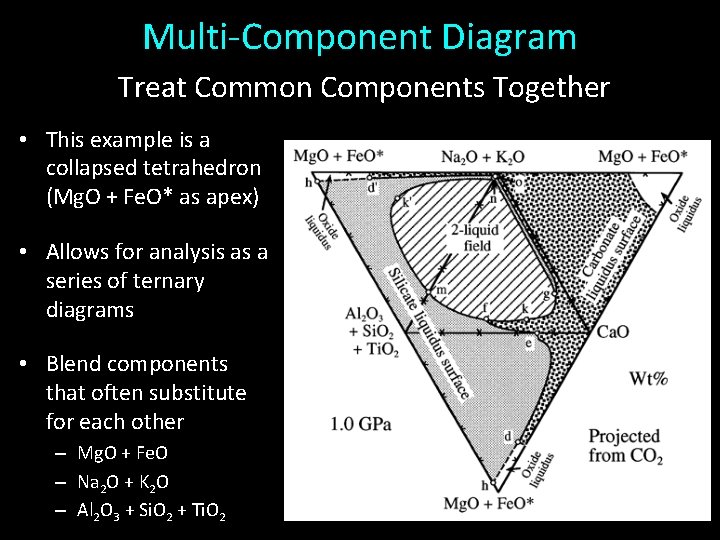
Multi-Component Diagram Treat Common Components Together • This example is a collapsed tetrahedron (Mg. O + Fe. O* as apex) • Allows for analysis as a series of ternary diagrams • Blend components that often substitute for each other – Mg. O + Fe. O – Na 2 O + K 2 O – Al 2 O 3 + Si. O 2 + Ti. O 2

Four-Component Tetrahedral Diagram • Mapping mineral compositions and tie -lines in 3 D space • Not applying for any crystallization paths towards eutectic or peritectic points – Simply used as an assemblage diagram • Able to plot many minerals on diagram
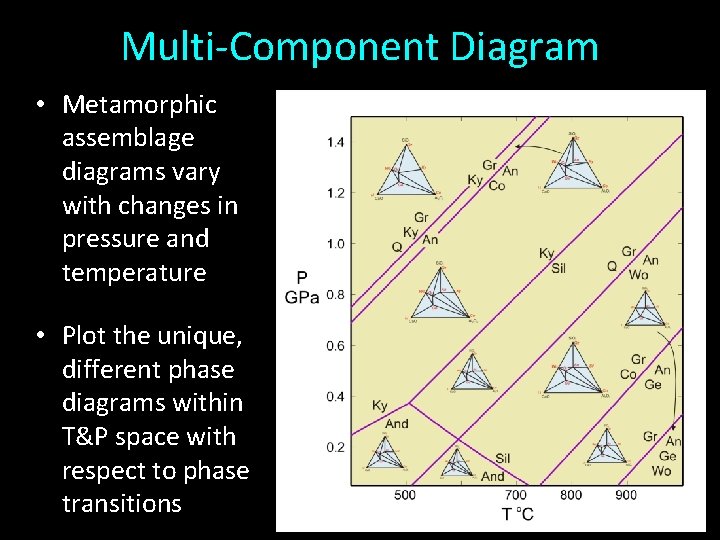
Multi-Component Diagram • Metamorphic assemblage diagrams vary with changes in pressure and temperature • Plot the unique, different phase diagrams within T&P space with respect to phase transitions
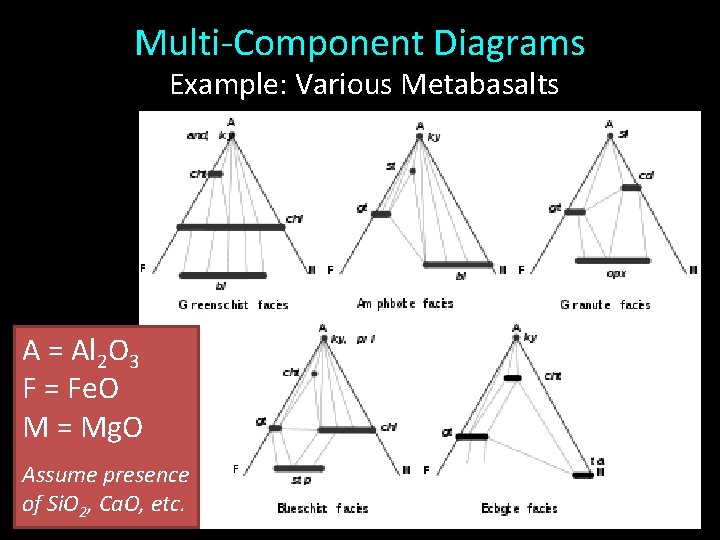
Multi-Component Diagrams Example: Various Metabasalts A = Al 2 O 3 F = Fe. O M = Mg. O Assume presence of Si. O 2, Ca. O, etc.
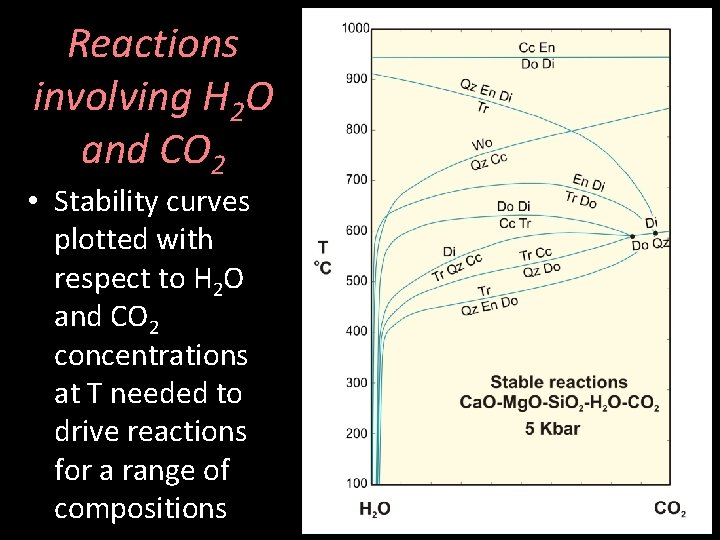
Reactions involving H 2 O and CO 2 • Stability curves plotted with respect to H 2 O and CO 2 concentrations at T needed to drive reactions for a range of compositions

Reactions involving H 2 O and CO 2 • Increased solubility at higher temps. and higher pressures

Reactions involving H 2 O and CO 2

Reactions involving H 2 O and CO 2 • Many common minerals have structurally-bound water – Amphiboles, Micas, Clays – Gypsum, Limonite, Azurite, Carnotite, Borates, etc… • Hydrous conditions can change the reactions and the products from those reactions
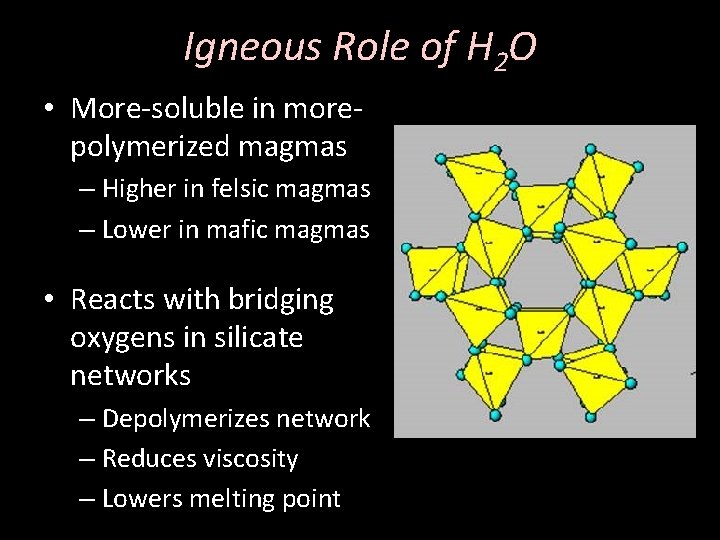
Igneous Role of H 2 O • More-soluble in morepolymerized magmas – Higher in felsic magmas – Lower in mafic magmas • Reacts with bridging oxygens in silicate networks – Depolymerizes network – Reduces viscosity – Lowers melting point
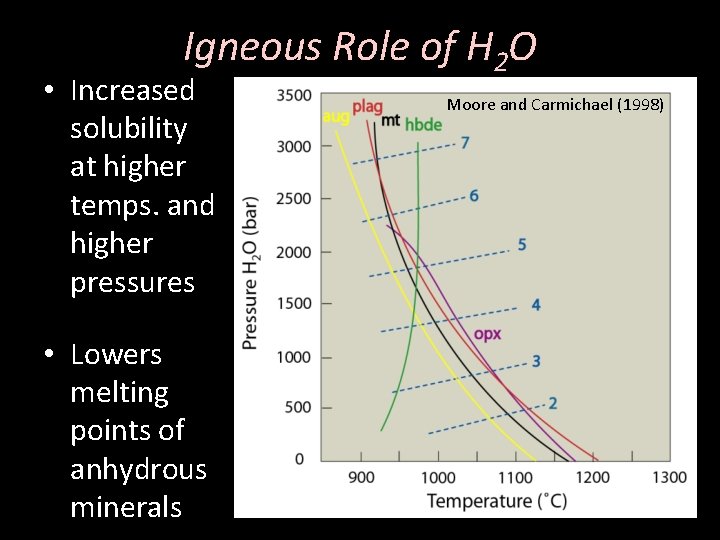
Igneous Role of H 2 O • Increased solubility at higher temps. and higher pressures • Lowers melting points of anhydrous minerals Moore and Carmichael (1998)

Summary Reactions Involving H 2 O • Hydrous minerals are more stable at lower temperatures than anhydrous minerals – Increasing partial-pressure of H 2 O raises the stability limit of these minerals – Decreasing partial-pressure of H 2 O prevents the formation of hydrous minerals

Metamorphosis from Clays at High Pressure to produce H 2 O Example: Al 2 Si 2 O 5(OH)4 Al 2 Si. O 5 + Si. O 2 + 2 H 2 O kaolinite andalusite + quartz + H 2 O (clay)
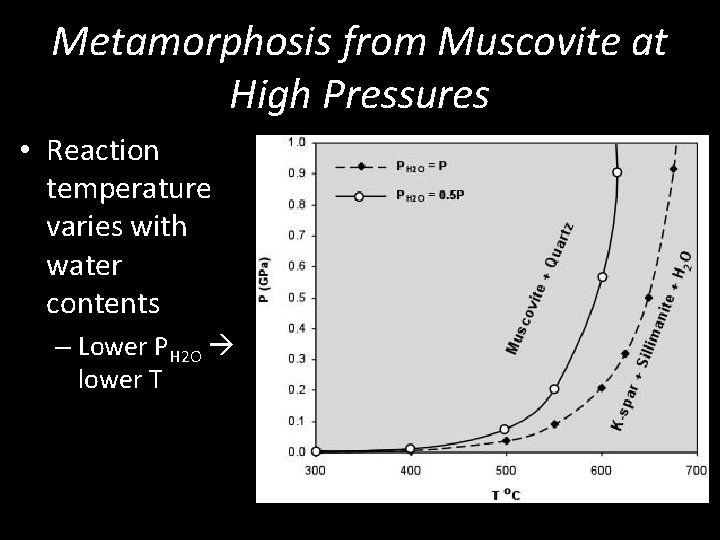
Metamorphosis from Muscovite at High Pressures • Reaction temperature varies with water contents – Lower PH 2 O lower T
Igneous Role of CO 2 • More soluble in less • Dissolution of CO 2 in polymerized melts silicate melts increases polymerization – More non-bridging oxygens in mafic melts
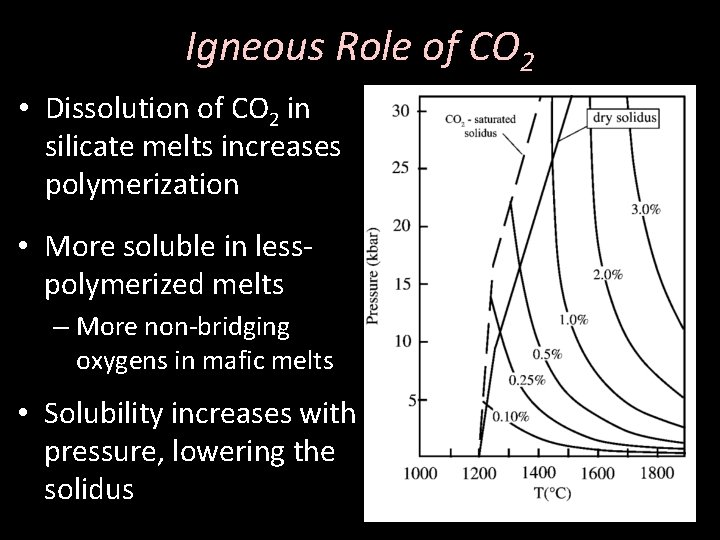
Igneous Role of CO 2 • Dissolution of CO 2 in silicate melts increases polymerization • More soluble in lesspolymerized melts – More non-bridging oxygens in mafic melts • Solubility increases with pressure, lowering the solidus

Metamorphosis of Carbonates may produce both CO 2 and H 2 O Example: 2 Talc + 3 Calcite Dolomite + Tremolite + CO 2 + H 2 O Tremolite in Marble, Franklin, NJ
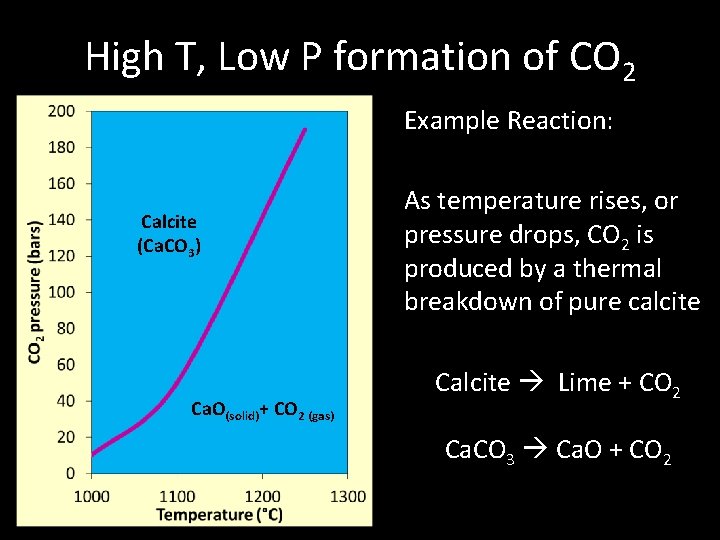
High T, Low P formation of CO 2 Example Reaction: Calcite (Ca. CO 3) Ca. O(solid)+ CO 2 (gas) As temperature rises, or pressure drops, CO 2 is produced by a thermal breakdown of pure calcite Calcite Lime + CO 2 Ca. CO 3 Ca. O + CO 2
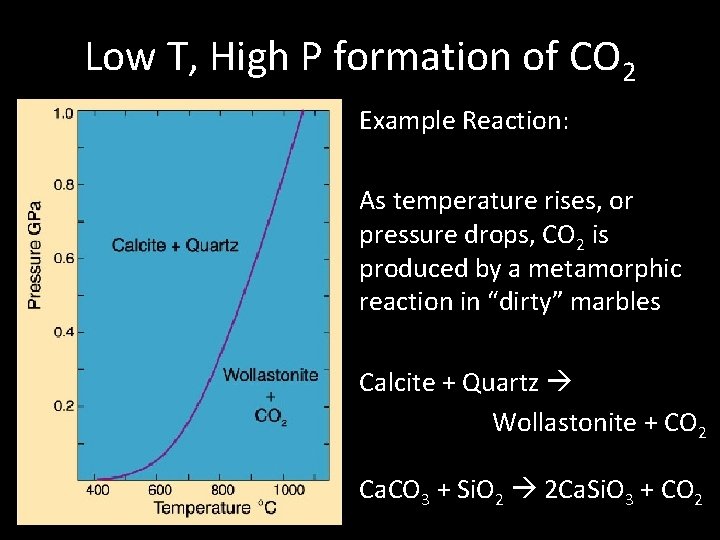
Low T, High P formation of CO 2 Example Reaction: As temperature rises, or pressure drops, CO 2 is produced by a metamorphic reaction in “dirty” marbles Calcite + Quartz Wollastonite + CO 2 Ca. CO 3 + Si. O 2 2 Ca. Si. O 3 + CO 2
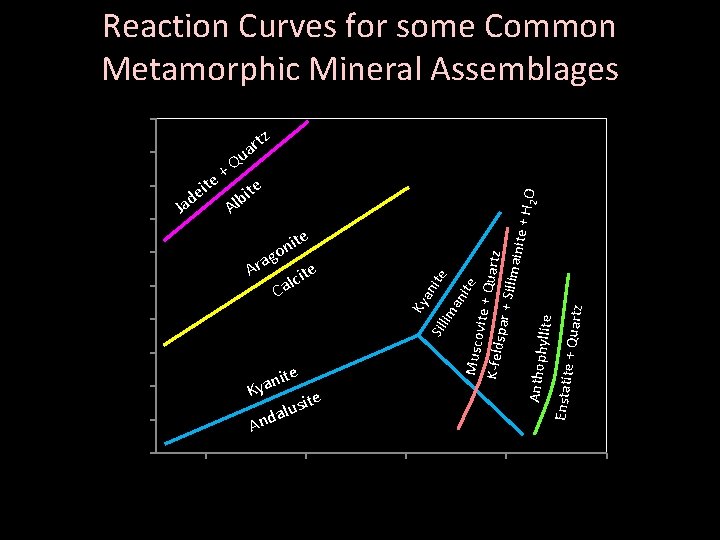
Reaction Curves for some Common Metamorphic Mineral Assemblages 10 tz 9 Q + ite e ite d b l Ja A 7 e Sil 4 3 ite n a Ky site u l a And 2 1 0 an 5 lim g a r A ite c l Ca an 6 ite it on Ky Pressure (kilobars) 8 0 200 ite Musco vite + Q uartz K-felds par + S illimain ite + H 2 O Anthoph yllite Enstatite + Quartz r ua 400 600 Temperature (°C) 800

Effect of p. H and Eh • p. H is a measure of solution acidity. • Technically, the negative log of H+ ion concentrations • p. H = 0 -7 : Acidic • p. H = 7 : Neutral • p. H = 7 -14: Basic

Effect of p. H and Eh • Eh is a measure of oxidation potential • Higher Eh values indicate oxidizing conditions (rusting/ tarnishing) – loss of electrons • Lower Eh numbers indicate reducing conditions – gain of electrons
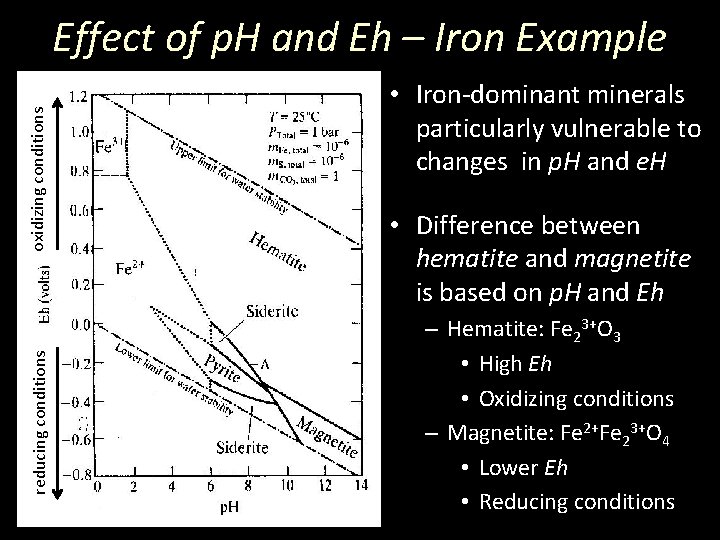
reducing conditions oxidizing conditions Effect of p. H and Eh – Iron Example • Iron-dominant minerals particularly vulnerable to changes in p. H and e. H • Difference between hematite and magnetite is based on p. H and Eh – Hematite: Fe 23+O 3 • High Eh • Oxidizing conditions – Magnetite: Fe 2+Fe 23+O 4 • Lower Eh • Reducing conditions

reducing conditions oxidizing conditions Effect of p. H and Eh – Iron Example • Iron-dominant minerals particularly vulnerable to changes in p. H and e. H • Difference between pyrite and pyrrhotite is based on p. H and Eh – Pyrite: Fe. S 2 • High e. H, Low p. H • Oxidizing conditions – Pyrrhotite: Fe 1 -x. S • Lower e. H, High p. H • Reducing conditions

Effect of p. H and Eh – Iron Example • Equilibrium lines show if such minerals can coexist • Knowledge of the oxidation conditions during crystallization can tell something about the setting or environment

Effect of p. H and Eh – Mn Example • Most-oxidized has the highest Eh value • Pyrolusite is the most oxidized natural oxide mineral (Mn 4+O 2) • Bixbyite (Mn 3+2 O 3) • Manganosite (Mn 2+O) and Pyrochroite (Mn 2+(OH)2) formed in reducing conditions

Effect of p. H and Eh – Iron Example • Modern manganese nodules are found on the ocean floor • Knowledge of the conditions under which they grow may aid in defining growth rates and viability as a future economic resource
Summary of Key Diagram Types • Phase Diagrams: – Show mineral solidi surfaces – Specific ranges of temperature shown • Assemblage Diagrams: – Show pure and solid-solution mineral compositions – Tie lines show minerals in equilibrium with one another mol% ternary assemblage diagram

Summary of H 2 O and CO 2 Reactions • Solubility of H 2 O and CO 2 are variable with T and P • Enrichment in volatiles can change the reactions and the products from those reactions – Changes in melting points – Changes in polymerization – Changes in viscosity

Summary of p. H and Eh • p. H is a measure of solution acidity. – Solubility of certain metallic ions may vary based on p. H • Eh is a measure of oxidation potential – Most-oxidized has the highest Eh value

Next Time… • Polymorphism and Polytypism • Exsolution Processes • Metamicts and Mineraloids **p. 282 -283 • Pseudomorphism **p. 284 -285
- Slides: 40