Asisted Reproductive TechniquesEmbryology Embryology Laboratory Practices Oocyte Pick

Asisted Reproductive Techniques-Embryology

Embryology Laboratory Practices • Oocyte Pick Up (OPU) • Microinjection-Insemination • Embryo Culture • Embryo Transfer • Cryopreservation • PGD • Assisted Hatching

n n n OPU (Oocyte Pick Up) This process is carried out by vaginal route through a needle placed in an ultrasound probe. Follicular fluid which is surrounded by cumulus cells, examined under a stereo microscope and then the oocytes collected. Cumulus-oocyte complex maturation is evaluated before it is removed to incubator.

Microinjection n n After a period of incubation at least 2 -3 hours, the cumulus cells are removed by enzymatic and mechanical procedure. Selected by the microinjection process, a single sperm cell is injected into the oocytes. Microinjection procedure is performed with the assistance of micropipettes attached to an inverted microscope. Oocytes are controlled for fertilization after 14 -16 hours. Fertilization rates increased from 80% to 90 with the microinjection.
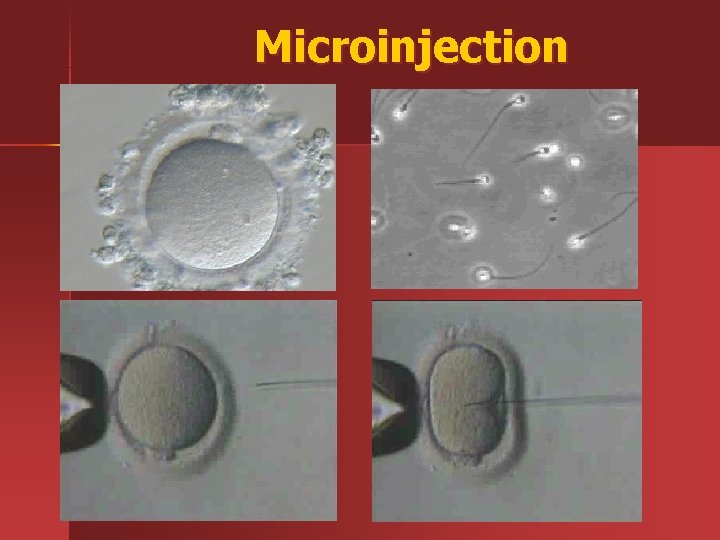
Microinjection
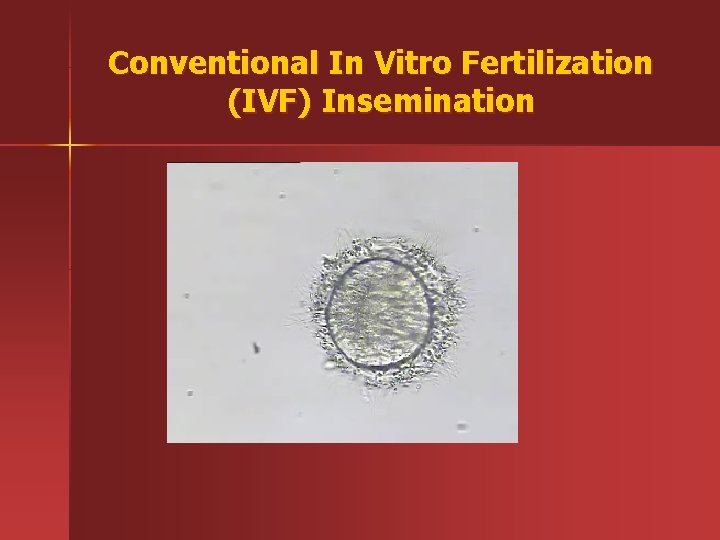
Conventional In Vitro Fertilization (IVF) Insemination
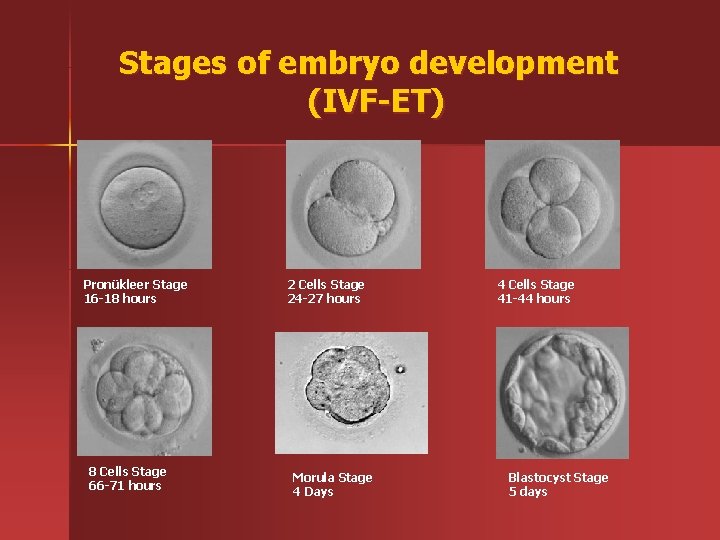
Stages of embryo development (IVF-ET) Pronükleer Stage 16 -18 hours 8 Cells Stage 66 -71 hours 2 Cells Stage 24 -27 hours Morula Stage 4 Days 4 Cells Stage 41 -44 hours Blastocyst Stage 5 days
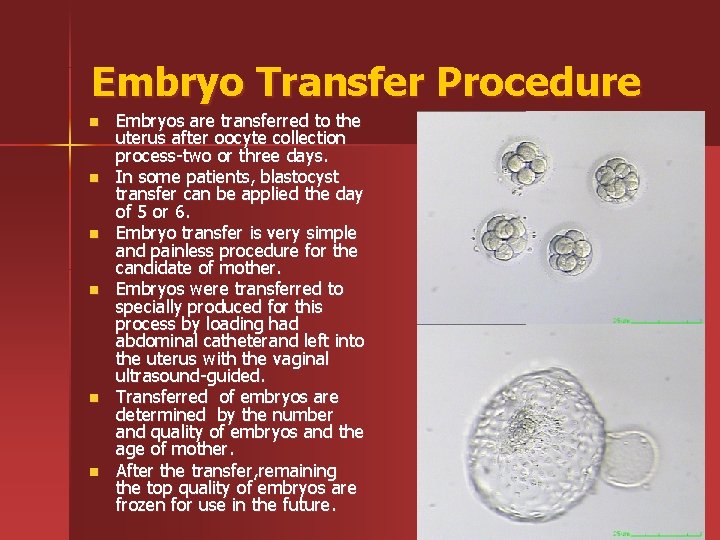
Embryo Transfer Procedure n n n Embryos are transferred to the uterus after oocyte collection process-two or three days. In some patients, blastocyst transfer can be applied the day of 5 or 6. Embryo transfer is very simple and painless procedure for the candidate of mother. Embryos were transferred to specially produced for this process by loading had abdominal catheterand left into the uterus with the vaginal ultrasound-guided. Transferred of embryos are determined by the number and quality of embryos and the age of mother. After the transfer, remaining the top quality of embryos are frozen for use in the future.

Top Quality Embryo Selection Criteria n n The day of 2 (after 41 -44 hours) have at least 4 or 5 blastomeres The day of 3 (after 66 -71 hours) have at least 7 blastomeres Cytoplasmic fragmentation<%20 Blastomere lack of multinucleation (Van Royen et. al. 1999, 2001, Gerris et. al. 1999)
Assisted Hatching n n The embryo continues to grow and divide into the uterus. After a period of time to rip around layer of the zona pellucida and implant into the uterus. n Before being transferred the embryo, the thinning zona pellucida, makes it easier to hold the uterus. n Specially; -Women over the age of 35 -In case of a thick zona pellucida -Recurrent implantation failure -In frozen embryo transfer. In our institution, the application time is too short, reliable, and reproducible technique using a laser high pregnancy and rates can be achieved.

What is a co-culture? n n In some patients, the treatment of the previous IVF attempts, embryo development was slow and poor quality can not be observed and achieved repeated attempts to pregnancy. In this case of patients for the development of better embryo, endometrial cells instead of synthetic feed solution was prepared by supporting a culture medium (co-culture) is used. Previous trials of patients with this method is applied its own center than enables the development of good-quality embryos and pregnancy rates increase. However, to apply this method is not used in several centers in the laboratory for requires additional labor and technical support.

How to make co-culture? n n n The luteal phase of the menstrual cycle of 20 -23 days in the last period of endometrial samples are taken with the assistance of Pipelle. After this mechanical and enzymatic digestion, stromal and glandular cells are separated in flasks and incubated. 3 days before OPU, cells surrounds the entire surface of flasks (confluent) fourwell (300, 000 viable cells / well) are planted. After the formation of PN, embryos are taken on these cells and incubated there until the blastocyst stage. In this method, embryos are being extended on the endometrium like to the body's and growth factors in the environment supports the development of the embryo. In addition, toxins that can damage the embryo, removed from the environment by the antioxidants.

Pre-implantation Genetic Diagnosis(PGD) Point; n Prevent transmission of chromosomal abnormalities n Reduce the risk of abort n To select genetically healthy embryos

Who Applies to PGD? Quantitative chromosomal abnormalities Anöploidi (Trisomies, monozamiler, nullizomiler), Triploidi, haploidi n Structural chromosomal abnormalities Deletions, microdeletions (Di. George, Kallman etc. ) Translocations (Reciprocal and Robertsonian) Inversion and insertion Sex-linked recessive diseases Duchenne muscular dystrophy, fragile X syndrome, hemophilia, Hunter syndrome, Wiskott-Aldrich syndrome, Lesch-Nyan syndrome, etc. n Single gene defects Autosomal recessive (cystic fibrosis, beta-Thalasemia, Spinal muscular atrophy (type I), etc. Autosomal dominant (Myotonic dystrophy, Marfan's syndrome, etc. ) n

Who Applies to PGD? IVF n Recurrent implantation failure n Advanced female age n Recurrent miscarriages n Patients with previous pregnancies, detected aneuploidy
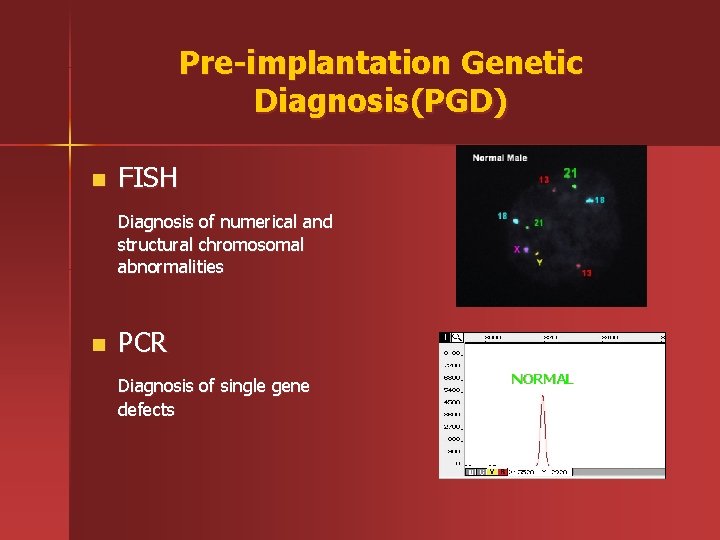
Pre-implantation Genetic Diagnosis(PGD) n FISH Diagnosis of numerical and structural chromosomal abnormalities n PCR Diagnosis of single gene defects NORMAL
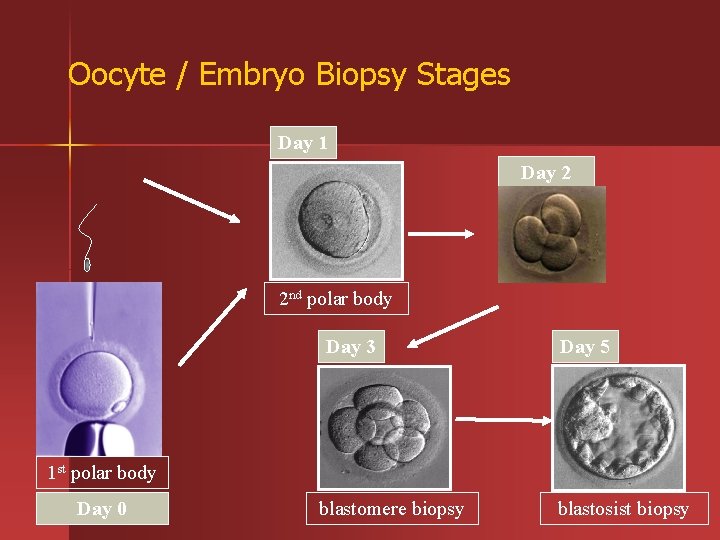
Oocyte / Embryo Biopsy Stages Day 1 Day 2 2 nd polar body Day 3 Day 5 1 st polar body Day 0 blastomere biopsy blastosist biopsy
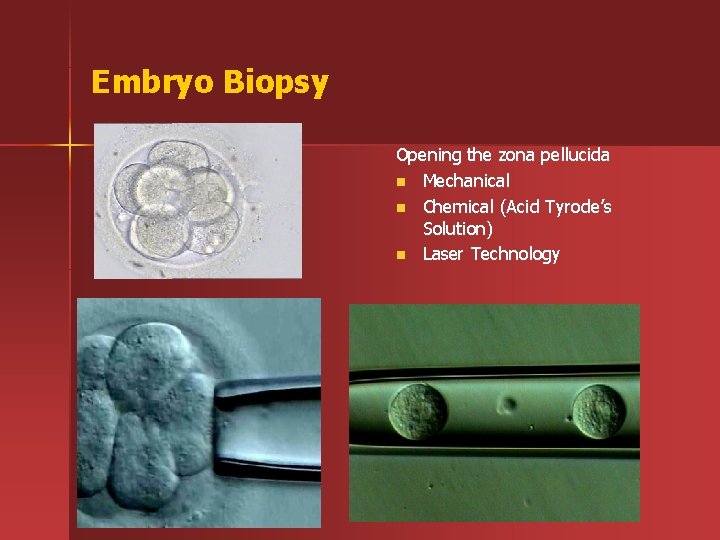
Embryo Biopsy Opening the zona pellucida n Mechanical n Chemical (Acid Tyrode’s Solution) n Laser Technology

Paternal Lymphocyte Vaccine POİNT n With Lymphocyte Vaccine Treatment Method, 'block antibody, called antibodies that damage the developing baby in the uterus, suppression of the targeted cells.

Paternal Lymphocyte Vaccine Who can Apply? n Unexplained Infertility n Recurrent İmplantation Failure n Recurrent Abortions

Paternal Lymphocyte Vaccine Necessary before the; n n n Candidates for ‘Dad’ analyzed of strong Hepatitis and HIV in terms. People whose tests (+) blood from, are not used to prepare the vaccine. In cases where the mother is Rh-negative, father Rh+positive, to prevent problems in the future due to blood incompatibility with the given Rhogam. Lymphocyte vaccine is not any harm in pregnant woman and the developing baby. Mothers whose treated with lymphocyte vaccine, there is not increase in congenital anomalies in babies or thrive.
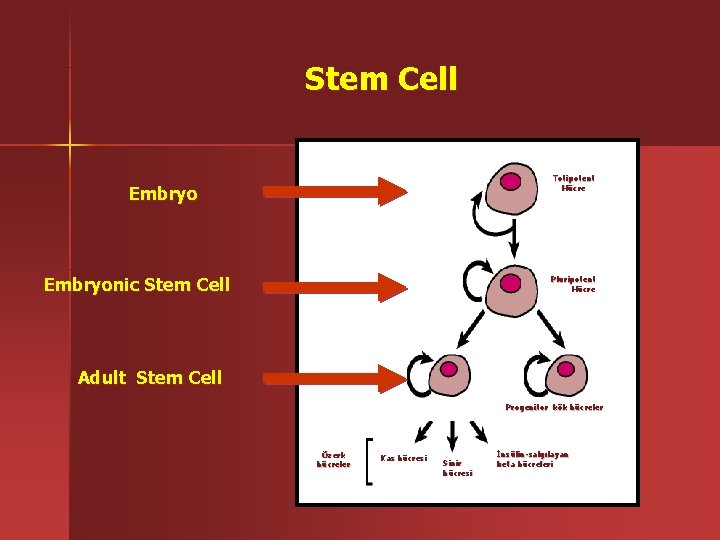
Stem Cell Totipotent Hücre Embryonic Stem Cell Pluripotent Hücre Adult Stem Cell Kan Hücreleri Özerk hücreler Kas hücresi Sinir hücresi Progenitor kök hücreler İnsülin-salgılayan beta hücreleri
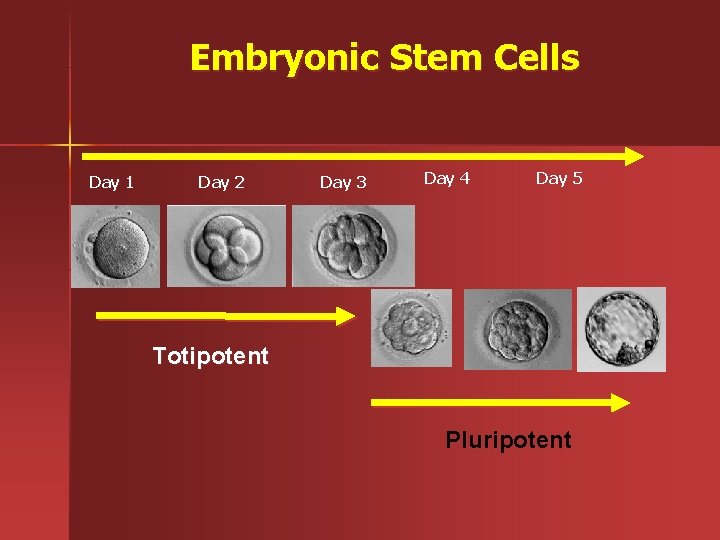
Embryonic Stem Cells Day 1 Day 2 Day 3 Day 4 Day 5 Totipotent Pluripotent
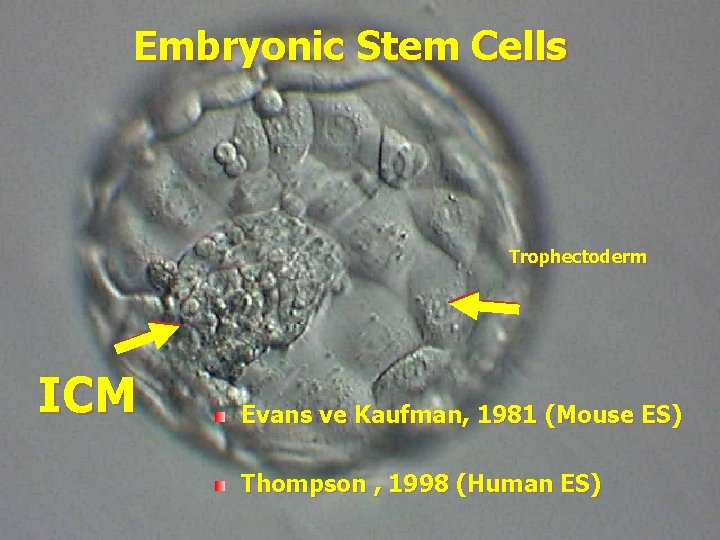
Embryonic Stem Cells Trophectoderm ICM Evans ve Kaufman, 1981 (Mouse ES) Thompson , 1998 (Human ES)

Embryonic Stem Cells In laboratory conditions can be improved continuously, without any structural change. Can be create any type of adult cell in body. Like bone, blood, muscle and liver tissues, such as creating an important source in the near future will be used for treatment purposes.

Human Embryonic Stem Cell Culture Cell Sources After the procedure, the unwanted frozen embryos donated for research purposes. •
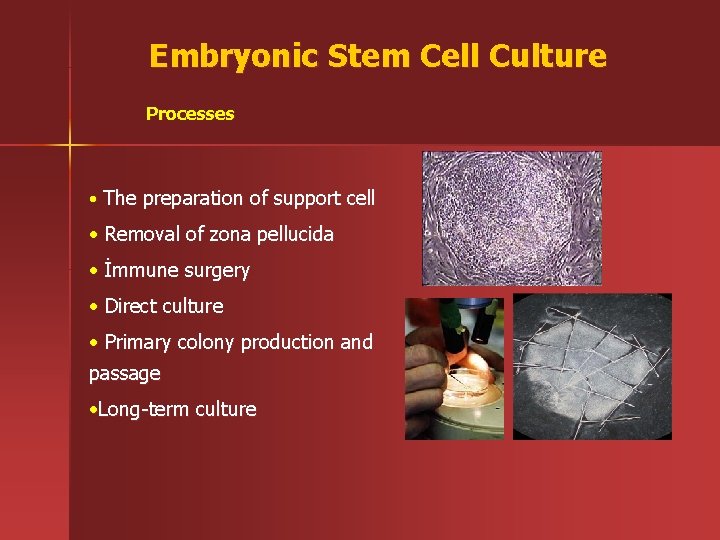
Embryonic Stem Cell Culture Processes • The preparation of support cell • Removal of zona pellucida • İmmune surgery • Direct culture • Primary colony production and passage • Long-term culture

Embryonic Stem Cell Culture NIH embryonic stem cell identification criteria • Normal karyotype • Immunocytochemical Analysis • SSEA-3 • SSEA-4 • TRA-1 -60 • TRA-1 -81 • Alkaline phosphatase expression • Telomerase expression • Creating a SCID feature of mice teratoma
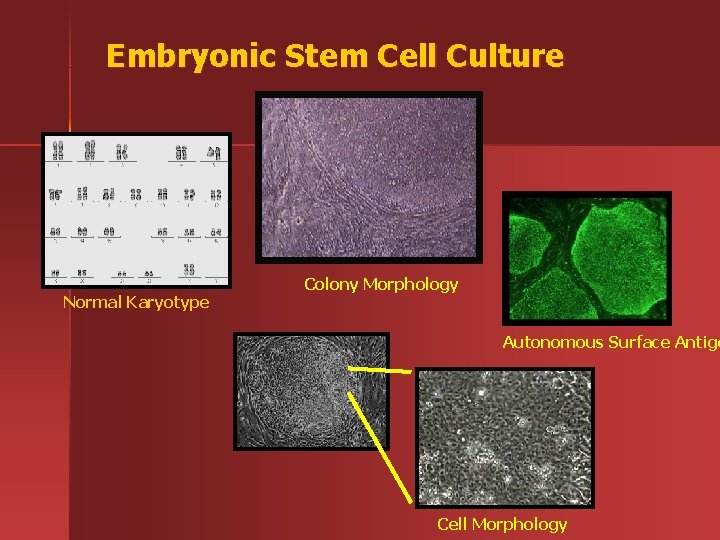
Embryonic Stem Cell Culture Normal Karyotype Colony Morphology Autonomous Surface Antige Cell Morphology

Embryonic Stem Cell Culture In Vitro Differentiation n n n Three embryonic germ layers is developing to differentiation of cell types spontaneously or adding growth factors ( Reubinoff et al. Nature Biotechnology 2000, Schuldiner et al. PNAS 2000) Neuron ( Reubinoff et al Nature 2001, Schuldiner et al. Brain res. 2001) Cardiomyocyte( Kehat et al. J clin İnvest 2001, Xu et al. Circ. Res. 2002, Mummery et al. Circ. 2003) The insulin-producing cells( Assady et al. Diabetes 2001, Odorico, Kaufmann and Thomson, Stem Cells 2001) Blood Cells( Kaufmann et al PNAS 2001) Endothelial Cells( Levenberg et al. PNAS 2002)
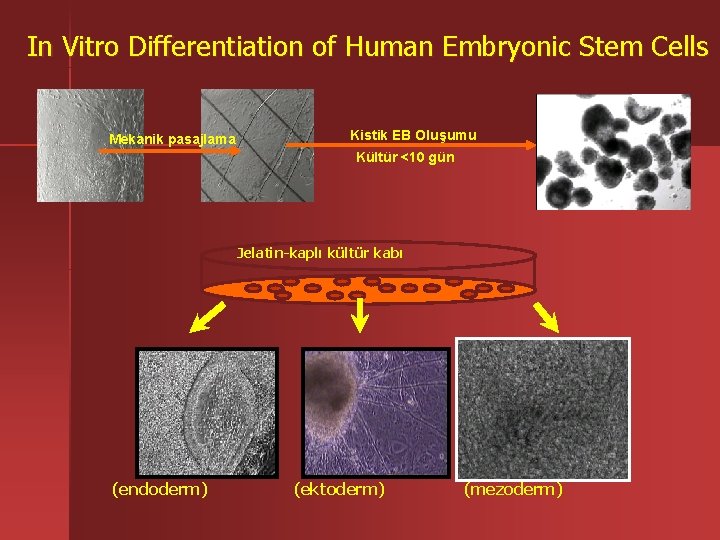
In Vitro Differentiation of Human Embryonic Stem Cells Mekanik pasajlama Kistik EB Oluşumu Kültür <10 gün Jelatin-kaplı kültür kabı (endoderm) (ektoderm) (mezoderm)
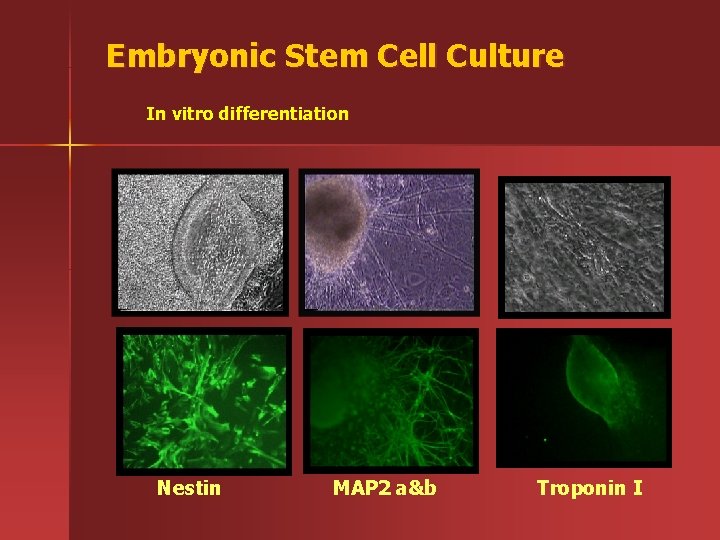
Embryonic Stem Cell Culture In vitro differentiation Nestin MAP 2 a&b Troponin I

Cardiomyocyte differentiation-TEM Study Erken Faz Sarkomer Z Bandı Mitokondri Miyofibriller

Embryonic Stem Cell Culture Areas of Use Examination of embryonic development İn Farmakology'de tissue-specific drug testing Toxicology in the identification of new agents For Therapeutic Use

Embryonic Stem Cells Areas of Use n Parkinson's and Alzheimer's disease n MS n Paralysis, Spinal Cord Injuries Diabet n Joint and Bone damage n Liver damage n Muscular dystrophy n Cardiac failure, Myocardial infarction n Canser n Hematologic diseases

Embryonic Stem Cells Clinical Therapeutic Use Preliminary studies : n After transplantation, undifferentiated human embryonic stem cells to motor neuropathy paralyzed rats, recovery was observed in. Kerr DA, Llado J 2003 n After injection of ESCs to experimental myocardial infarction in rats was determined improvement in contractile function and reduction of infarct size. Denice M. Hodgson, 2004 n When were given Insulin-producing cells from embryonic stem cells in diabetic mice returned to normal levels of glycemia. Soria B, 2000 n Parkinson modeli oluşturulmuş farelerde embriyonik kök hücre kaynaklı dopaminerjik nöronların fonksiyon gösterdiği bulundu. Parkinson's model was created in mice and was found thats functional of dopaminergic neurons from embryonic stem cells. Kim JH, Auerbach JM, 2002

Problems that need to be resolved before the clinical trial: The use of materials from animal sources Support systems, of human origin(HFF) The immune system is the problem of compliance Cloning for therapeutic purposes Genetic manipulation Stem cell bank Differentiation of protocols incompatibility The protection of life after transplantation The risk of tumor Ethical problems

Result and Discussion Embryonic Stem Cells can be unlimited culture under laboratory conditions, this properties an important source for cell therapy. These cells in the near future, especially by solving the existing ethical and technical problems, heart disease, endocrine diseases, including neurodegenerative and may be the source for the treatment of many other diseases.
- Slides: 38