ASCO and EHA 2019 Carfilzomib and AMG 420

ASCO and EHA 2019 Carfilzomib and AMG 420 Abstracts Selected Carfilzomib Data from Amgen-sponsored, Other Company-sponsored or Investigator-sponsored Studies and Other Abstracts of Interest SC-CH-CARFILZOMI-00165 Amgen (Europe) Gmb. H Suurstoffi 22 CH-6343 Rotkreuz Switzerland www. amgen. com Do not copy or distribute. © 2019 Amgen. All rights reserved
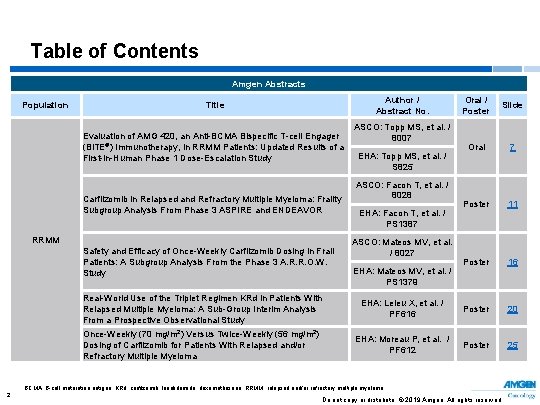
Table of Contents Amgen Abstracts Population Author / Abstract No. Title Evaluation of AMG 420, an Anti-BCMA Bispecific T-cell Engager (Bi. TE®) Immunotherapy, in RRMM Patients: Updated Results of a First-in-Human Phase 1 Dose-Escalation Study Carfilzomib in Relapsed and Refractory Multiple Myeloma: Frailty Subgroup Analysis From Phase 3 ASPIRE and ENDEAVOR RRMM Safety and Efficacy of Once-Weekly Carfilzomib Dosing in Frail Patients: A Subgroup Analysis From the Phase 3 A. R. R. O. W. Study ASCO: Topp MS, et al. / 8007 EHA: Topp MS, et al. / S 825 ASCO: Facon T, et al. / 8028 EHA: Facon T, et al. / PS 1387 ASCO: Mateos MV, et al. / 8027 EHA: Mateos MV, et al. / PS 1379 Oral / Poster Slide Oral 7 Poster 11 Poster 16 Real-World Use of the Triplet Regimen KRd in Patients With Relapsed Multiple Myeloma: A Sub-Group Interim Analysis From a Prospective Observational Study EHA: Leleu X, et al. / PF 616 Poster 20 Once-Weekly (70 mg/m 2) Versus Twice-Weekly (56 mg/m 2) Dosing of Carfilzomib for Patients With Relapsed and/or Refractory Multiple Myeloma EHA: Moreau P, et al. / PF 612 Poster 25 BCMA, B-cell maturation antigen; KRd, carfilzomib, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma. 2 Do not copy or distribute. © 2019 Amgen. All rights reserved
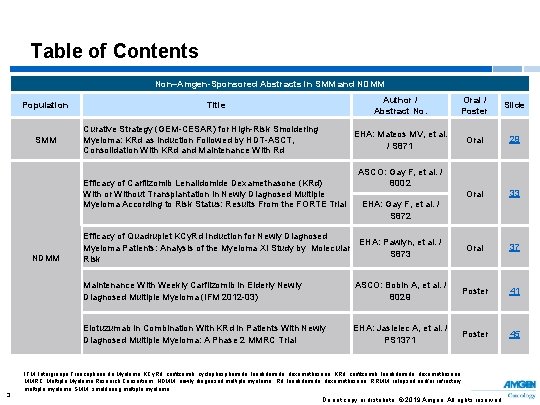
Table of Contents Non–Amgen-Sponsored Abstracts in SMM and NDMM Population SMM Title Curative Strategy (GEM-CESAR) for High-Risk Smoldering Myeloma: KRd as Induction Followed by HDT-ASCT, Consolidation With KRd and Maintenance With Rd Efficacy of Carfilzomib Lenalidomide Dexamethasone (KRd) With or Without Transplantation in Newly Diagnosed Multiple Myeloma According to Risk Status: Results From the FORTE Trial NDMM 3 Author / Abstract No. Oral / Poster Slide EHA: Mateos MV, et al. / S 871 Oral 29 Oral 33 Oral 37 ASCO: Gay F, et al. / 8002 EHA: Gay F, et al. / S 872 Efficacy of Quadruplet KCy. Rd Induction for Newly Diagnosed EHA: Pawlyn, et al. / Myeloma Patients: Analysis of the Myeloma XI Study by Molecular S 873 Risk Maintenance With Weekly Carfilzomib in Elderly Newly Diagnosed Multiple Myeloma (IFM 2012 -03) ASCO: Bobin A, et al. / 8029 Poster 41 Elotuzumab in Combination With KRd in Patients With Newly Diagnosed Multiple Myeloma: A Phase 2 MMRC Trial EHA: Jasielec A, et al. / PS 1371 Poster 45 IFM, Intergroupe Francophone du Myélome; KCy. Rd, carfilzomib, cyclophosphamide, lenalidomide, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; MMRC, Multiple Myeloma Research Consortium; NDMM, newly diagnosed multiple myeloma; Rd, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma; SMM, smoldering multiple myeloma. Do not copy or distribute. © 2019 Amgen. All rights reserved
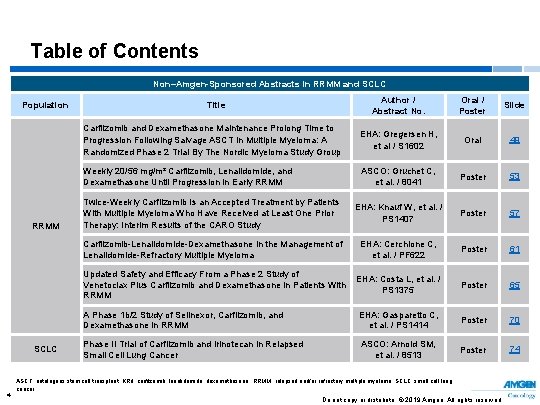
Table of Contents Non–Amgen-Sponsored Abstracts in RRMM and SCLC Population RRMM SCLC 4 Author / Abstract No. Oral / Poster Slide Carfilzomib and Dexamethasone Maintenance Prolong Time to Progression Following Salvage ASCT In Multiple Myeloma: A Randomized Phase 2 Trial By The Nordic Myeloma Study Group EHA: Gregersen H, et al / S 1602 Oral 49 Weekly 20/56 mg/m² Carfilzomib, Lenalidomide, and Dexamethasone Until Progression in Early RRMM ASCO: Gruchet C, et al. / 8041 Poster 53 Twice-Weekly Carfilzomib Is an Accepted Treatment by Patients With Multiple Myeloma Who Have Received at Least One Prior Therapy: Interim Results of the CARO Study EHA: Knauf W, et al. / PS 1407 Poster 57 Carfilzomib-Lenalidomide-Dexamethasone in the Management of Lenalidomide-Refractory Multiple Myeloma EHA: Cerchione C, et al. / PF 622 Poster 61 Updated Safety and Efficacy From a Phase 2 Study of EHA: Costa L, et al. / Venetoclax Plus Carfilzomib and Dexamethasone in Patients With PS 1375 RRMM Poster 65 A Phase 1 b/2 Study of Selinexor, Carfilzomib, and Dexamethasone in RRMM EHA: Gasparetto C, et al. / PS 1414 Poster 70 Phase II Trial of Carfilzomib and Irinotecan in Relapsed Small Cell Lung Cancer ASCO: Arnold SM, et al. / 8513 Poster 74 Title ASCT, autologous stem cell transplant; KRd, carfilzomib, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma; SCLC, small cell lung cancer; Do not copy or distribute. © 2019 Amgen. All rights reserved

Amgen Abstracts 5 Do not copy or distribute. © 2019 Amgen. All rights reserved

Carfilzomib in Relapsed and Refractory Multiple Myeloma: Frailty Subgroup Analysis From Phase 3 ASPIRE and ENDEAVOR Facon T, et al. Poster Presentation, ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8028. Facon T, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1387. 6 Do not copy or distribute. © 2019 Amgen. All rights reserved
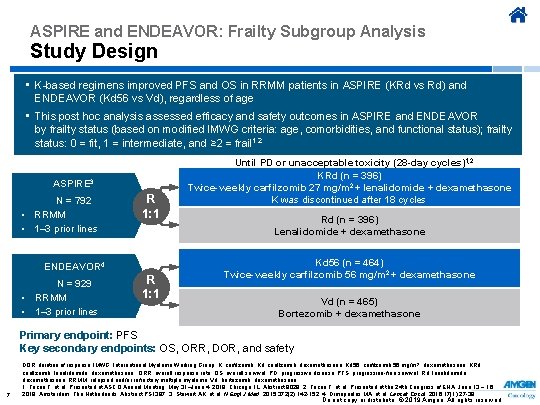
ASPIRE and ENDEAVOR: Frailty Subgroup Analysis Study Design • K-based regimens improved PFS and OS in RRMM patients in ASPIRE (KRd vs Rd) and ENDEAVOR (Kd 56 vs Vd), regardless of age • This post hoc analysis assessed efficacy and safety outcomes in ASPIRE and ENDEAVOR by frailty status (based on modified IMWG criteria: age, comorbidities, and functional status); frailty status: 0 = fit, 1 = intermediate, and ≥ 2 = frail 1, 2 ASPIRE 3 N = 792 • RRMM • 1– 3 prior lines R 1: 1 ENDEAVOR 4 N = 929 • RRMM • 1– 3 prior lines R 1: 1 Until PD or unacceptable toxicity (28 -day cycles)1, 2 KRd (n = 396) Twice-weekly carfilzomib 27 mg/m 2 + lenalidomide + dexamethasone K was discontinued after 18 cycles Rd (n = 396) Lenalidomide + dexamethasone Kd 56 (n = 464) Twice-weekly carfilzomib 56 mg/m 2 + dexamethasone Vd (n = 465) Bortezomib + dexamethasone Primary endpoint: PFS Key secondary endpoints: OS, ORR, DOR, and safety 7 DOR, duration of response; IMWG, International Myeloma Working Group; K, carfilzomib; Kd, carfilzomib, dexamethasone; Kd 56, carfilzomib 56 mg/m 2, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; ORR, overall response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; Rd, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma; Vd, bortezomib, dexamethasone. 1. Facon T, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8028. 2. Facon T, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract PS 1387. 3. Stewart AK, et al. N Engl J Med. 2015; 372(2): 142 -152. 4. Dimopoulos MA, et al. Lancet Oncol. 2016; 17(1): 27 -38. Do not copy or distribute. © 2019 Amgen. All rights reserved
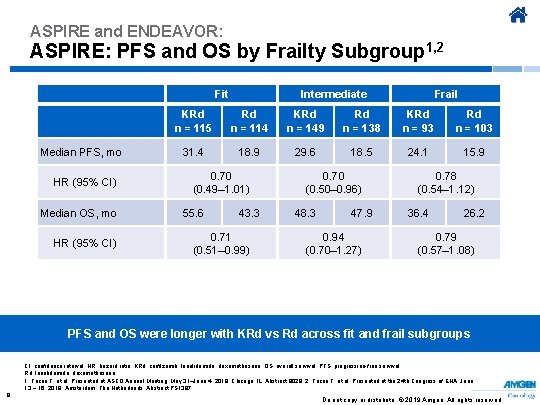
ASPIRE and ENDEAVOR: ASPIRE: PFS and OS by Frailty Subgroup 1, 2 Fit Median PFS, mo HR (95% CI) Median OS, mo HR (95% CI) Intermediate Frail KRd n = 115 Rd n = 114 KRd n = 149 Rd n = 138 KRd n = 93 Rd n = 103 31. 4 18. 9 29. 6 18. 5 24. 1 15. 9 0. 70 (0. 49– 1. 01) 55. 6 43. 3 0. 71 (0. 51– 0. 99) 0. 70 (0. 50– 0. 96) 48. 3 47. 9 0. 94 (0. 70– 1. 27) 0. 78 (0. 54– 1. 12) 36. 4 26. 2 0. 79 (0. 57– 1. 08) PFS and OS were longer with KRd vs Rd across fit and frail subgroups CI, confidence interval; HR, hazard ratio; KRd, carfilzomib, lenalidomide, dexamethasone; OS, overall survival; PFS, progression-free survival; Rd, lenalidomide, dexamethasone. 1. Facon T, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8028. 2. Facon T, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract PS 1387. 8 Do not copy or distribute. © 2019 Amgen. All rights reserved
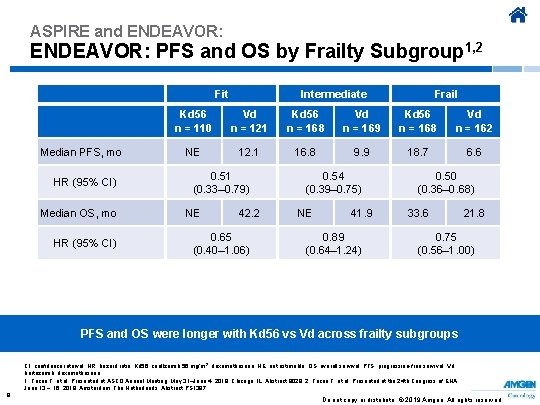
ASPIRE and ENDEAVOR: PFS and OS by Frailty Subgroup 1, 2 Fit Median PFS, mo HR (95% CI) Median OS, mo HR (95% CI) Intermediate Frail Kd 56 n = 110 Vd n = 121 Kd 56 n = 168 Vd n = 169 Kd 56 n = 168 Vd n = 162 NE 12. 1 16. 8 9. 9 18. 7 6. 6 0. 51 (0. 33– 0. 79) NE 42. 2 0. 65 (0. 40– 1. 06) 0. 54 (0. 39– 0. 75) NE 41. 9 0. 89 (0. 64– 1. 24) 0. 50 (0. 36– 0. 68) 33. 6 21. 8 0. 75 (0. 56– 1. 00) PFS and OS were longer with Kd 56 vs Vd across frailty subgroups CI, confidence interval; HR, hazard ratio; Kd 56, carfilzomib 56 mg/m 2, dexamethasone; NE, not estimable; OS, overall survival; PFS, progression-free survival; Vd, bortezomib, dexamethasone. 1. Facon T, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8028. 2. Facon T, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract PS 1387. 9 Do not copy or distribute. © 2019 Amgen. All rights reserved
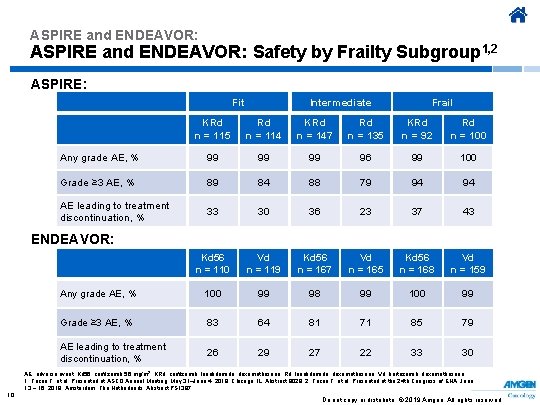
ASPIRE and ENDEAVOR: Safety by Frailty Subgroup 1, 2 ASPIRE: Fit Intermediate Frail KRd n = 115 Rd n = 114 KRd n = 147 Rd n = 135 KRd n = 92 Rd n = 100 Any grade AE, % 99 99 99 96 99 100 Grade ≥ 3 AE, % 89 84 88 79 94 94 AE leading to treatment discontinuation, % 33 30 36 23 37 43 Kd 56 n = 110 Vd n = 119 Kd 56 n = 167 Vd n = 165 Kd 56 n = 168 Vd n = 159 Any grade AE, % 100 99 98 99 100 99 Grade ≥ 3 AE, % 83 64 81 71 85 79 AE leading to treatment discontinuation, % 26 29 27 22 33 30 ENDEAVOR: AE, adverse event; Kd 56, carfilzomib 56 mg/m 2; KRd, carfilzomib, lenalidomide, dexamethasone; Rd, lenalidomide, dexamethasone; Vd, bortezomib, dexamethasone. 1. Facon T, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8028. 2. Facon T, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract PS 1387. 10 Do not copy or distribute. © 2019 Amgen. All rights reserved

Safety and Efficacy of Once-Weekly Carfilzomib Dosing in Frail Patients: A Subgroup Analysis From the Phase 3 A. R. R. O. W. Study Mateos MV, et al. Poster Presentation, ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8027. Mateos MV, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1379. 11 Do not copy or distribute. © 2019 Amgen. All rights reserved
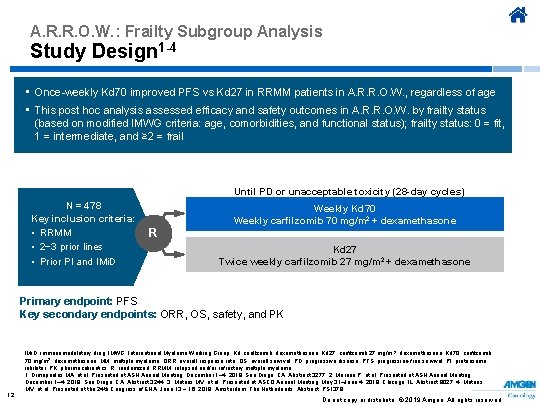
A. R. R. O. W. : Frailty Subgroup Analysis Study Design 1 -4 • Once-weekly Kd 70 improved PFS vs Kd 27 in RRMM patients in A. R. R. O. W. , regardless of age • This post hoc analysis assessed efficacy and safety outcomes in A. R. R. O. W. by frailty status (based on modified IMWG criteria: age, comorbidities, and functional status); frailty status: 0 = fit, 1 = intermediate, and ≥ 2 = frail Until PD or unacceptable toxicity (28 -day cycles) N = 478 Key inclusion criteria: • RRMM • 2− 3 prior lines • Prior PI and IMi. D R Weekly Kd 70 Weekly carfilzomib 70 mg/m 2 + dexamethasone Kd 27 Twice weekly carfilzomib 27 mg/m 2 + dexamethasone Primary endpoint: PFS Key secondary endpoints: ORR, OS, safety, and PK 12 IMi. D, immunomodulatory drug; IMWG, International Myeloma Working Group; Kd, carfilzomib, dexamethasone; Kd 27, carfilzomib 27 mg/m 2, dexamethasone; Kd 70, carfilzomib 70 mg/m 2, dexamethasone; MM, multiple myeloma; ORR, overall response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PI, proteasome inhibitor; PK, pharmacokinetics; R, randomized; RRMM, relapsed and/or refractory multiple myeloma. 1. Dimopoulos MA, et al. Presented at ASH Annual Meeting; December 1– 4, 2018; San Diego, CA. Abstract 3277. 2. Moreau P, et al. Presented at ASH Annual Meeting; December 1– 4, 2018; San Diego, CA. Abstract 3244. 3. Mateos MV, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8027. 4. Mateos MV, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract PS 1379. Do not copy or distribute. © 2019 Amgen. All rights reserved
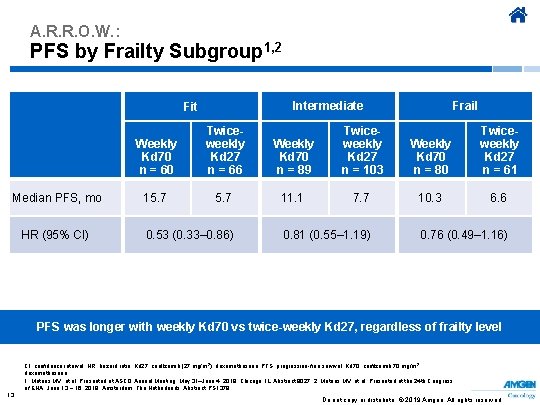
A. R. R. O. W. : PFS by Frailty Subgroup 1, 2 Intermediate Fit Median PFS, mo HR (95% CI) Frail Weekly Kd 70 n = 60 Twiceweekly Kd 27 n = 66 Weekly Kd 70 n = 89 Twiceweekly Kd 27 n = 103 Weekly Kd 70 n = 80 Twiceweekly Kd 27 n = 61 15. 7 11. 1 7. 7 10. 3 6. 6 0. 53 (0. 33– 0. 86) 0. 81 (0. 55– 1. 19) 0. 76 (0. 49– 1. 16) PFS was longer with weekly Kd 70 vs twice-weekly Kd 27, regardless of frailty level CI, confidence interval; HR, hazard ratio; Kd 27, carfilzomib (27 mg/m 2), dexamethasone; PFS, progression-free survival; Kd 70, carfilzomib 70 mg/m 2, dexamethasone. 1. Mateos MV, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8027. 2. Mateos MV, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract PS 1379. 13 Do not copy or distribute. © 2019 Amgen. All rights reserved
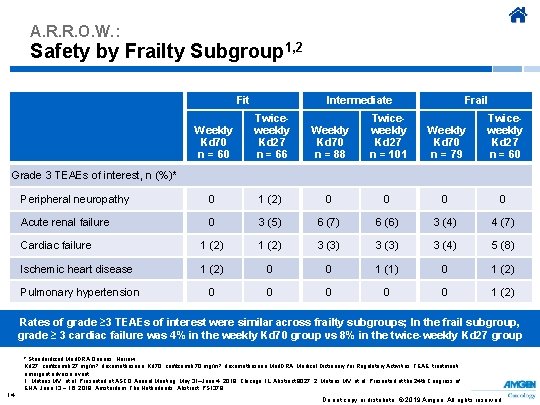
A. R. R. O. W. : Safety by Frailty Subgroup 1, 2 Intermediate Fit Weekly Kd 70 n = 60 Twiceweekly Kd 27 n = 66 Weekly Kd 70 n = 88 Twiceweekly Kd 27 n = 101 Frail Weekly Kd 70 n = 79 Twiceweekly Kd 27 n = 60 Grade 3 TEAEs of interest, n (%)* Peripheral neuropathy 0 1 (2) 0 0 Acute renal failure 0 3 (5) 6 (7) 6 (6) 3 (4) 4 (7) Cardiac failure 1 (2) 3 (3) 3 (4) 5 (8) Ischemic heart disease 1 (2) 0 0 1 (1) 0 1 (2) Pulmonary hypertension 0 0 0 1 (2) Rates of grade ≥ 3 TEAEs of interest were similar across frailty subgroups; In the frail subgroup, grade ≥ 3 cardiac failure was 4% in the weekly Kd 70 group vs 8% in the twice-weekly Kd 27 group * Standardized Med. DRA Queries: Narrow. Kd 27, carfilzomib 27 mg/m 2, dexamethasone; Kd 70, carfilzomib 70 mg/m 2, dexamethasone Med. DRA, Medical Dictionary for Regulatory Activities; TEAE, treatmentemergent adverse event. 1. Mateos MV, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8027. 2. Mateos MV, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract PS 1379. 14 Do not copy or distribute. © 2019 Amgen. All rights reserved

Real-World Use of the Triplet Regimen KRd in Patients With Relapsed Multiple Myeloma: A Sub-Group Interim Analysis From a Prospective Observational Study Leleu X, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PF 616. KRd, carfilzomib, lenalidomide, dexamethasone. 15 Do not copy or distribute. © 2019 Amgen. All rights reserved

Real-World Use of KRd in RRMM Patients Study Design 1, 2 • This observational study aimed to characterize the KRd patient population and describe K use and benefit-risk profile in a real-world setting in 10 countries in the EU and Israel Key inclusion criteria: • Relapsed MM • ≥ 1 prior line of therapy • ≥ 1 carfilzomib dose in routine care KRd (N = 178) Dosing and administration schedule used in routine clinical practice Primary objective: Describe carfilzomib utilization in routine clinical practice, including dosage, administration schedule, regimen, duration of treatment, and reason for discontinuation K, carfilzomib; Kd, carfilzomib, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; MM, multiple myeloma; RRMM, relapsed and/or refractory multiple myeloma. 1. Leleu X, et al. Presented at: European Hematology Association Annual Meeting, June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PF 616. 2. Clinical. Trials. gov. NCT 03091127. Accessed May 19, 2019. 16 Do not copy or distribute. © 2019 Amgen. All rights reserved
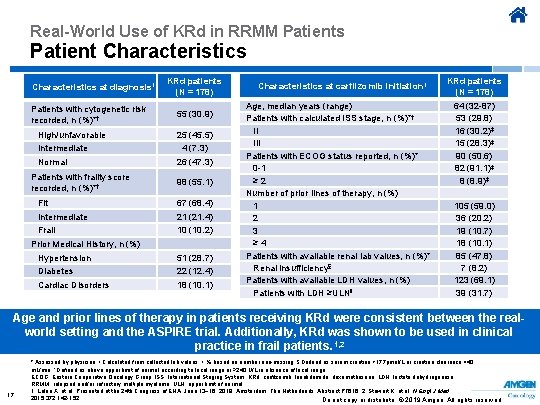
Real-World Use of KRd in RRMM Patients Patient Characteristics at diagnosis 1 Patients with cytogenetic risk recorded, n (%)*† High/unfavorable Intermediate Normal Patients with frailty score recorded, n (%)*† KRd patients (N = 178) 55 (30. 9) 25 (45. 5) 4 (7. 3) 26 (47. 3) 98 (55. 1) Fit 67 (68. 4) Intermediate 21 (21. 4) Frail 10 (10. 2) Prior Medical History, n (%) Hypertension 51 (28. 7) Diabetes 22 (12. 4) Cardiac Disorders 18 (10. 1) Characteristics at carfilzomib initiation 1 Age, median years (range) Patients with calculated ISS stage, n (%)*† II III Patients with ECOG status reported, n (%)* 0 -1 ≥ 2 Number of prior lines of therapy, n (%) 1 2 3 ≥ 4 Patients with available renal lab values, n (%)* Renal insufficiency§ Patients with available LDH values, n (%) Patients with LDH ≥ULN‖ KRd patients (N = 178) 64 (32 -87) 53 (29. 8) 16 (30. 2)‡ 15 (28. 3)‡ 90 (50. 6) 82 (91. 1)‡ 8 (8. 9)‡ 105 (59. 0) 36 (20. 2) 19 (10. 7) 18 (10. 1) 85 (47. 8) 7 (8. 2) 123 (69. 1) 39 (31. 7) Age and prior lines of therapy in patients receiving KRd were consistent between the realworld setting and the ASPIRE trial. Additionally, KRd was shown to be used in clinical practice in frail patients. 1, 2 17 * Assessed by physician. † Calculated from collected lab values. ‡ % based on number non-missing. § Defined as serum creatine >177µmol/L or creatine clearance <40 m. L/min. ‖ Defined as above upper limit of normal according to local range or ≥ 240 U/L in absence of local range. ECOG, Eastern Cooperative Oncology Group; ISS, International Staging System; KRd, carfilzomib, lenalidomide, dexamethasone; LDH, lactate dehydrogenase; RRMM, relapsed and/or refractory multiple myeloma; ULN, upper limit of normal. 1. Leleu X, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 616. 2. Stewart K, et al. N Engl J Med. 2015; 372; 142 -152. Do not copy or distribute. © 2019 Amgen. All rights reserved
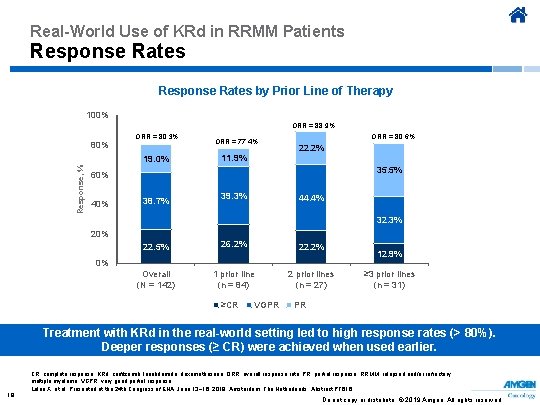
Real-World Use of KRd in RRMM Patients Response Rates by Prior Line of Therapy 100% ORR = 88. 9% 80% ORR = 80. 3% Response, % 19. 0% ORR = 77. 4% 11. 9% ORR = 80. 6% 22. 2% 35. 5% 60% 40% 38. 7% 39. 3% 44. 4% 32. 3% 20% 22. 5% 26. 2% 22. 2% Overall (N = 142) 1 prior line (n = 84) 2 prior lines (n = 27) 0% ≥CR VGPR 12. 9% ≥ 3 prior lines (n = 31) PR Treatment with KRd in the real-world setting led to high response rates (> 80%). Deeper responses (≥ CR) were achieved when used earlier. 18 CR, complete response; KRd, carfilzomib, lenalidomide, dexamethasone; ORR, overall response rate; PR, partial response; RRMM, relapsed and/or refractory multiple myeloma; VGPR, very good partial response. Leleu X, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 616. Do not copy or distribute. © 2019 Amgen. All rights reserved
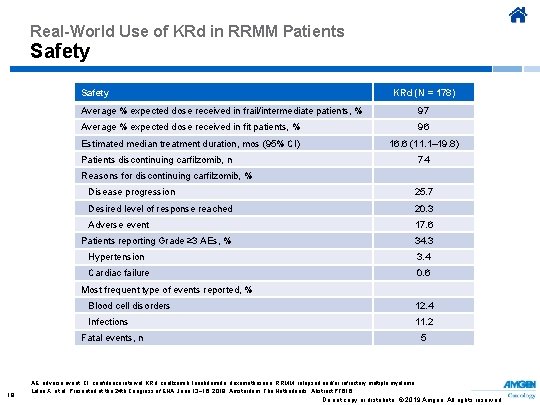
Real-World Use of KRd in RRMM Patients Safety KRd (N = 178) Average % expected dose received in frail/intermediate patients, % 97 Average % expected dose received in fit patients, % 96 Estimated median treatment duration, mos (95% CI) 16. 6 (11. 1– 19. 8) Patients discontinuing carfilzomib, n 74 Reasons for discontinuing carfilzomib, % Disease progression 25. 7 Desired level of response reached 20. 3 Adverse event 17. 6 Patients reporting Grade ≥ 3 AEs, % 34. 3 Hypertension 3. 4 Cardiac failure 0. 6 Most frequent type of events reported, % Blood cell disorders 12. 4 Infections 11. 2 Fatal events, n 19 5 AE, adverse event; CI, confidence interval; KRd, carfilzomib, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma. Leleu X, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 616. Do not copy or distribute. © 2019 Amgen. All rights reserved

Once-Weekly (70 mg/m 2) Versus Twice-Weekly (56 mg/m 2) Dosing of Carfilzomib for Patients With Relapsed and/or Refractory Multiple Myeloma Moreau P, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PF 612. 20 Do not copy or distribute. © 2019 Amgen. All rights reserved
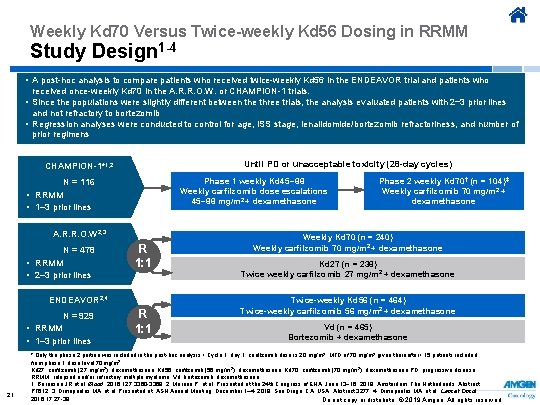
Weekly Kd 70 Versus Twice-weekly Kd 56 Dosing in RRMM Study Design 1 -4 • A post-hoc analysis to compare patients who received twice-weekly Kd 56 in the ENDEAVOR trial and patients who received once-weekly Kd 70 in the A. R. R. O. W. or CHAMPION-1 trials. • Since the populations were slightly different between the three trials, the analysis evaluated patients with 2− 3 prior lines and not refractory to bortezomib • Regression analyses were conducted to control for age, ISS stage, lenalidomide/bortezomib refractoriness, and number of prior regimens Until PD or unacceptable toxicity (28 -day cycles) CHAMPION-1*1, 2 Phase 1 weekly Kd 45− 88 Weekly carfilzomib dose escalations 45− 88 mg/m 2 + dexamethasone N = 116 • RRMM • 1– 3 prior lines A. R. R. O. W 2, 3 N = 478 • RRMM • 2– 3 prior lines R 1: 1 ENDEAVOR 2, 4 N = 929 • RRMM • 1– 3 prior lines 21 R 1: 1 Phase 2 weekly Kd 70 † (n = 104)‡ Weekly carfilzomib 70 mg/m 2 + dexamethasone Weekly Kd 70 (n = 240) Weekly carfilzomib 70 mg/m 2 + dexamethasone Kd 27 (n = 238) Twice weekly carfilzomib 27 mg/m 2 + dexamethasone Twice-weekly Kd 56 (n = 464) Twice-weekly carfilzomib 56 mg/m 2 + dexamethasone Vd (n = 465) Bortezomib + dexamethasone * Only the phase 2 portion was included in the post-hoc analysis † Cycle 1, day 1: carfilzomib dose is 20 mg/m 2. MTD of 70 mg/m 2 given thereafter ‡ 15 patients included from phase 1 dose level 70 mg/m 2. Kd 27, carfilzomib (27 mg/m 2), dexamethasone; Kd 56, carfilzomib (56 mg/m 2), dexamethasone; Kd 70, carfilzomib (70 mg/m 2), dexamethasone PD, progressive disease; RRMM, relapsed and/or refractory multiple myeloma; Vd, bortezomib, dexamethasone. 1. Berenson JR et al. Blood. 2016; 127: 3360 -3368. 2. Moreau P, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 612. 3. Dimopoulos MA, et al. Presented at: ASH Annual Meeting; December 1– 4, 2018; San Diego, CA, USA. Abstract 3277. 4. Dimopoulos MA, et al. Lancet Oncol. 2016; 17: 27 -38. Do not copy or distribute. © 2019 Amgen. All rights reserved
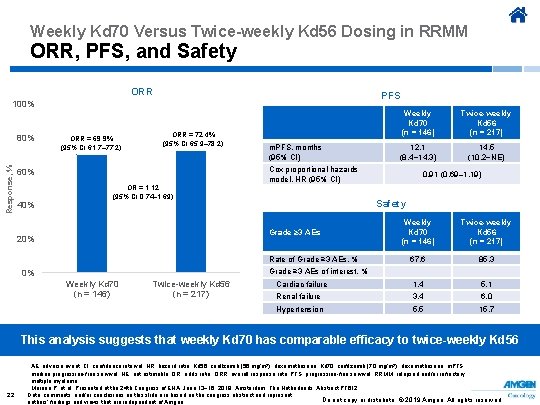
Weekly Kd 70 Versus Twice-weekly Kd 56 Dosing in RRMM ORR, PFS, and Safety ORR PFS 100% Response, % 80% ORR = 69. 9% (95% CI 61. 7– 77. 2) ORR = 72. 4% (95% CI 65. 9– 78. 2) Weekly Kd 70 (n = 146) m. PFS, months (95% CI) 12. 1 (8. 4− 14. 3) Cox proportional hazards model, HR (95% CI) 60% OR = 1. 12 (95% CI 0. 74– 1. 69) 14. 5 (10. 2−NE) 0. 91 (0. 69– 1. 19) Safety 40% Weekly Kd 70 (n = 146) Twice-weekly Kd 56 (n = 217) 67. 6 85. 3 Cardiac failure 1. 4 5. 1 Renal failure 3. 4 6. 0 Hypertension 5. 5 15. 7 Grade ≥ 3 AEs 20% Twice-weekly Kd 56 (n = 217) Rate of Grade ≥ 3 AEs, % Grade ≥ 3 AEs of interest, % 0% Weekly Kd 70 (n = 146) Twice-weekly Kd 56 (n = 217) This analysis suggests that weekly Kd 70 has comparable efficacy to twice-weekly Kd 56 22 AE, adverse event; CI, confidence interval; HR, hazard ratio; Kd 56, carfilzomib (56 mg/m 2), dexamethasone; Kd 70, carfilzomib (70 mg/m 2), dexamethasone; m. PFS, median progression-free survival; NE, not estimable; OR, o dds ratio; ORR, overall response rate; PFS, progression-free survival; RRMM, relapsed and/or refractory multiple myeloma. Moreau P, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 612. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

Non–Amgen-Sponsored Abstracts 23 Do not copy or distribute. © 2019 Amgen. All rights reserved

Curative Strategy (GEM-CESAR) for High-Risk Smoldering Myeloma: KRd as Induction Followed by HDTASCT, Consolidation With KRd and Maintenance With Rd Mateos MV, et al. Oral Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract S 871. HDT-ASCT, high dose therapy-autologous stem cell transplant; KRd, carfilzomib, lenalidomide, dexamethasone; Rd, lenalidomide, dexamethasone. 24 Do not copy or distribute. © 2019 Amgen. All rights reserved

Phase 2 GEM-CESAR: KRd + ASCT in High-Risk SMM Study Design 1, 2 • This phase 2, single-arm trial was designed to evaluate KRd-ASCT-KRd + Rd maintenance as curative therapy, defined as sustained MRD-negativity 5 years after transplant, in patients with high-risk SMM Induction 6 x 28 -day cycles KRd† Carfilzomib 36 mg/m 2 twice weekly + lenalidomide 25 mg, + dexamethasone 40 mg weekly ASCT N = 90 Key inclusion criteria: • SMM • >50% risk of progression at 2 yrs* • ASCT-eligible • <70 years Consolidation‡ 2 x 28 -day cycles KRd Maintenance up to 2 yrs Rd Lenalidomide 10 mg + dexamethasone 20 mg weekly Primary Endpoint: MRD-negative rate after induction and ASCT Secondary Endpoints: ORR, § PFS, OS, safety * High risk of progression was defined as both ≥PC 10% and MC ≥ 3 g/d. L (Mayo) or as one of the two criteria being present + >95% of aberrant PCs within the total PCs. BM compartment by immunophenotyping + immunoparesis (Spanish). † After third cycle of KRd, all patients were mobilized with G-CSF to collect peripheral blood stem cells for ASCT. ‡ Patients received consolidation treatment approximately 3 months after ASCT. § ORR after induction, ASCT, consolidation, and maintenance. ASCT, autologous stem cell transplant; CR, complete response; ECOG, Eastern Cooperative Oncology Group; G-CSF, granulocyte colony-stimulating factor; HDT-ASCT, high-dose therapy, followed by ASCT; KRd, carfilzomib, lenalidomide, dexamethasone; MC, monoclonal protein level; O RR, overall response rate; PC, plasma cell; PCs. BM, bone marrow PCs; Rd, lenalidomide, dexamethasone; MRD, minimal residual disease; OS, overall survival; PFS, progression-free survival; Rd, lenalidomide, dexamethasone; SMM, smoldering multiple myeloma. 1. Clinicaltrials. gov identifier: NCT 02415413. Accessed May 17, 2019. 2. Mateos MV, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 871. 25 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
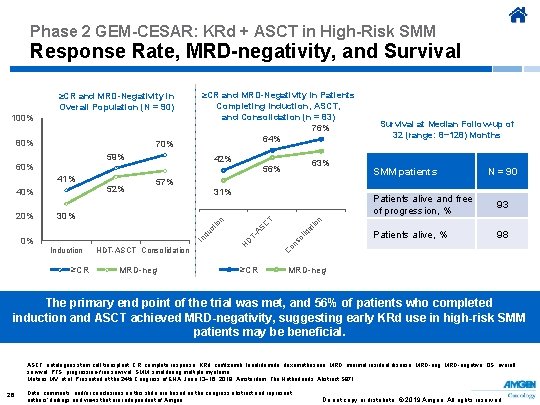
Phase 2 GEM-CESAR: KRd + ASCT in High-Risk SMM Response Rate, MRD-negativity, and Survival 59% 42% 60% 40% 31% n 30% ≥CR HDT-ASCT Consolidation MRD-neg SMM patients N = 90 Patients alive and free of progression, % 93 Patients alive, % 98 C AS T- C Induction D 0% H In du ct io 20% 52% 57% n 41% 63% 56% at io 70% T 80% ol id 100% ≥CR and MRD-Negativity in Patients Completing Induction, ASCT, and Consolidation (n = 83) Survival at Median Follow-up of 76% 32 (range: 8− 128) Months 64% on s ≥CR and MRD-Negativity in Overall Population (N = 90) ≥CR MRD-neg The primary end point of the trial was met, and 56% of patients who completed induction and ASCT achieved MRD-negativity, suggesting early KRd use in high-risk SMM patients may be beneficial. ASCT, autologous stem cell transplant; CR, complete response; KRd, carfilzomib, lenalidomide, dexamethasone; MRD, minimal residual disease; MRD-neg, MRD-negative; OS, overall survival; PFS, progression-free survival; SMM, smoldering multiple myeloma. Mateos MV, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 871. 26 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
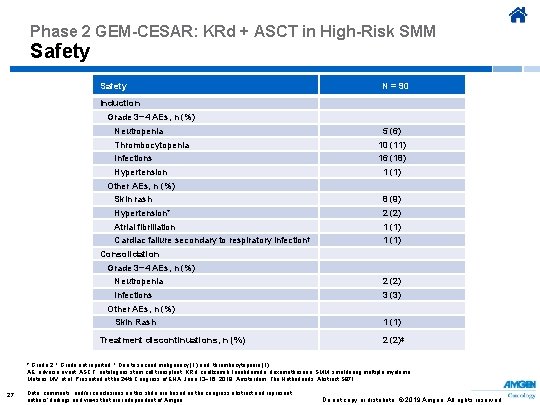
Phase 2 GEM-CESAR: KRd + ASCT in High-Risk SMM Safety N = 90 Induction Grade 3− 4 AEs, n (%) Neutropenia 5 (6) Thrombocytopenia 10 (11) Infections 16 (18) Hypertension 1 (1) Other AEs, n (%) Skin rash 8 (9) Hypertension* 2 (2) Atrial fibrillation 1 (1) Cardiac failure secondary to respiratory infection† 1 (1) Consolidation Grade 3− 4 AEs, n (%) Neutropenia 2 (2) Infections 3 (3) Other AEs, n (%) Skin Rash Treatment discontinuations, n (%) 1 (1) 2 (2)‡ * Grade 2. † Grade not reported. ‡ Due to second malignancy (1) and thrombocytopenia (1) AE, adverse event; ASCT, autologous stem cell transplant; KRd, carfilzomib, lenalidomide, dexamethasone; SMM, smoldering multiple myeloma. Mateos MV, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 871. 27 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved

Efficacy of Carfilzomib Lenalidomide Dexamethasone (KRd) With or Without Transplantation in NDMM According to Risk Status: Results From the FORTE Trial Gay F, et al. Poster Presentation, ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8002. Gay F, et al. Oral Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract S 872. 28 NDMM, newly diagnosed multiple myeloma. Do not copy or distribute. © 2019 Amgen. All rights reserved
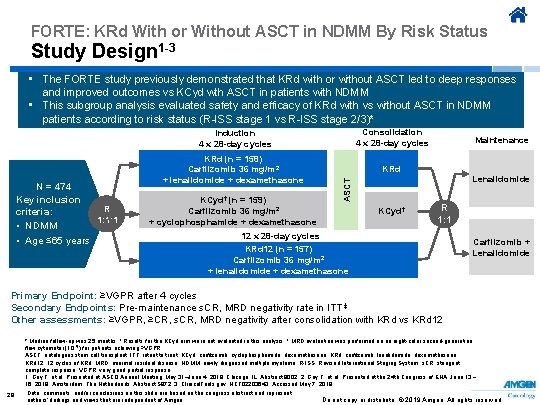
FORTE: KRd With or Without ASCT in NDMM By Risk Status Study Design 1 -3 • The FORTE study previously demonstrated that KRd with or without ASCT led to deep responses and improved outcomes vs KCyd wth ASCT in patients with NDMM • This subgroup analysis evaluated safety and efficacy of KRd with vs without ASCT in NDMM patients according to risk status (R-ISS stage 1 vs R-ISS stage 2/3)* Consolidation 4 x 28 -day cycles KRd (n = 158) Carfilzomib 36 mg/m 2 + lenalidomide + dexamethasone KRd KCyd† (n = 159) Maintenance Lenalidomide ASCT N = 474 Key inclusion R criteria: 1: 1: 1 • NDMM • Age ≤ 65 years Induction 4 x 28 -day cycles Carfilzomib 36 mg/m 2 + cyclophosphamide + dexamethasone KCyd† R 1: 1 12 x 28 -day cycles KRd 12 (n = 157) Carfilzomib 36 mg/m 2 + lenalidomide + dexamethasone Carfilzomib + Lenalidomide Primary Endpoint: ≥VGPR after 4 cycles Secondary Endpoints: Pre-maintenance s. CR, MRD negativity rate in ITT ‡ Other assessments: ≥VGPR, ≥CR, s. CR, MRD negativity after consolidation with KRd vs KRd 12 29 * Median follow-up was 25 months. † Results for the KCyd arm were not evaluated in this analysis. ‡ MRD evaluation was performed on an eight-color second-generation flow cytometer (10– 5) for patients achieving ≥VGPR. ASCT, autologous stem cell transplant; ITT, intent to treat; KCyd, carfilzomib, cyclophosphamide, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; KRd 12, 12 cycles of KRd; MRD, minimal residual disease; NDMM, newly diagnosed multiple myeloma; R-ISS, Revised International Staging System; s. CR, stringent complete response; VGPR, very good partial response. 1. Gay F, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8002. 2. Gay F, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract S 872. 3. Clinical. Trials. gov. NCT 02203643. Accessed May 7, 2019. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
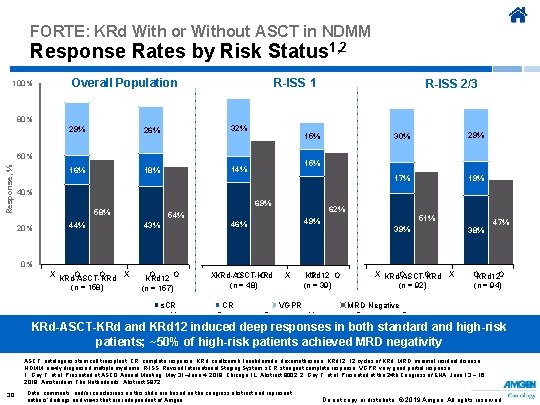
FORTE: KRd With or Without ASCT in NDMM Response Rates by Risk Status 1, 2 100% Overall Population 80% R-ISS 1 29% 26% 32% 16% 18% 14% 15% 60% Response, % R-ISS 2/3 30% 29% 17% 19% 15% 40% 69% 58% 20% 44% 54% 43% 62% 49% 46% 51% 39% 38% 47% 0% X KRd-ASCT-KRd O O X (n = 158) O O KRd 12 (n = 157) s. CR X XKRd-ASCT-KRd O O (n = 48) 30% 39% 17% CR O X KRd 12 O O (n = 39) 51% VGPR O X X KRd-ASCT-KRd O O X (n = 92) OKRd 12 O (n = 94) 38% 19% 29% 47% MRD Negative O O KRd-ASCT-KRd and KRd 12 induced deep responses in both standard and high-risk patients; ~50% of high-risk patients achieved MRD negativity ASCT, autologous stem cell transplant; CR, complete response; KRd, carfilzomib, lenalidomide, dexamethasone; KRd 12, 12 cycles of KRd; MRD, minimal residual disease; NDMM, newly diagnosed multiple myeloma; R-ISS, Revised International Staging System; s. CR, stringent complete response; VGPR, very good partial response. 1. Gay F, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8002. 2. Gay F, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract S 872. 30 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
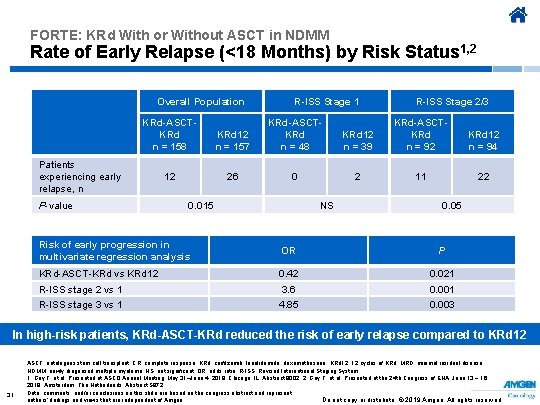
FORTE: KRd With or Without ASCT in NDMM Rate of Early Relapse (<18 Months) by Risk Status 1, 2 Overall Population R-ISS Stage 1 R-ISS Stage 2/3 KRd-ASCTKRd n = 158 KRd 12 n = 157 KRd-ASCTKRd n = 48 KRd 12 n = 39 KRd-ASCTKRd n = 92 KRd 12 n = 94 12 26 0 2 11 22 Patients experiencing early relapse, n P-value 0. 015 NS 0. 05 Risk of early progression in multivariate regression analysis OR P KRd-ASCT-KRd vs KRd 12 0. 42 0. 021 R-ISS stage 2 vs 1 3. 6 0. 001 R-ISS stage 3 vs 1 4. 85 0. 003 In high-risk patients, KRd-ASCT-KRd reduced the risk of early relapse compared to KRd 12 31 ASCT, autologous stem cell transplant; CR, complete response; KRd, carfilzomib, lenalidomide, dexamethasone; KRd 12, 12 cycles of KRd; MRD, minimal residual disease; NDMM, newly diagnosed multiple myeloma; NS, not significant; OR, odds ratio; R-ISS, Revised International Staging System. 1. Gay F, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8002. 2. Gay F, et al. Presented at the 24 th Congress of EHA; June 13 − 16, 2019; Amsterdam, The Netherlands. Abstract S 872. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

Efficacy of Quadruplet KCy. Rd Induction for Newly Diagnosed Myeloma Patients: Analysis of the Myeloma XI Study by Molecular Risk Pawlyn C, et al. Oral Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract S 873. KCy. Rd, carfilzomib, cyclophosphamide, lenalidomide, dexamethasone. 32 Do not copy or distribute. © 2019 Amgen. All rights reserved
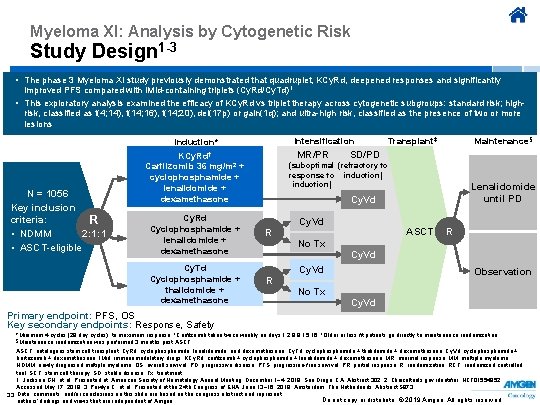
Myeloma XI: Analysis by Cytogenetic Risk Study Design 1 -3 • The phase 3 Myeloma XI study previously demonstrated that quadruplet, KCy. Rd, deepened responses and significantly improved PFS compared with IMid-containing triplets (Cy. Rd/Cy. Td)1 • This exploratory analysis examined the efficacy of KCy. Rd vs triplet therapy across cytogenetic subgroups: standard risk; highrisk, classified as t(4; 14), t(14; 16), t(14; 20), del(17 p) or gain(1 q); and ultra-high risk, classified as the presence of two or more lesions Intensification Induction* MR/PR KCy. Rd† N = 1056 Key inclusion criteria: R 2: 1: 1 • NDMM • ASCT-eligible Transplant‡ Maintenance§ SD/PD (suboptimal (refractory to response to induction) Carfilzomib 36 mg/m 2 + cyclophosphamide + lenalidomide + dexamethasone Lenalidomide until PD Cy. Vd Cy. Rd Cyclophosphamide + lenalidomide + dexamethasone R Cy. Td Cyclophosphamide + thalidomide + dexamethasone R Cy. Vd ASCT R No Tx Cy. Vd No Tx Observation Cy. Vd Primary endpoint: PFS, OS Key secondary endpoints: Response, Safety *Minimum 4 cycles (28 day cycles), to maximum response. †Carfilzomib taken twice weekly on days 1, 2, 8, 9, 15, 16. ‡Older or less fit patients go directly to maintenance randomization. §Maintenance randomization was performed 3 months post ASCT, autologous stem cell transplant; Cy. Rd, cyclophosphamide, lenalidomide, and dexamethasone; Cy. Td, cyclophosphamide + thalidomide + dexamethasone; Cy. Vd, cyclophosphamide + bortezomib + dexamethasone; IMid, immunomodulatory drugs; KCy. Rd, carfilzomib + cyclophosphamide + lenalidomide + dexamethasone; MR, minimal response; MM, multiple myeloma; NDMM, newly diagnosed multiple myeloma; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; R, randomization; RCT, randomized controlled trial; SCT, stem cell therapy; SD, stable disease; Tx, treatment. 1. Jackson GH, et al. Presented at: American Society of Hematology Annual Meeting, December 1− 4, 2018; San Diego, CA. Abstract 302. 2. Clinicaltrials. gov identifier: NCT 01554852. Accessed May 17, 2019. 3. Pawlyn C, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 873. 33 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
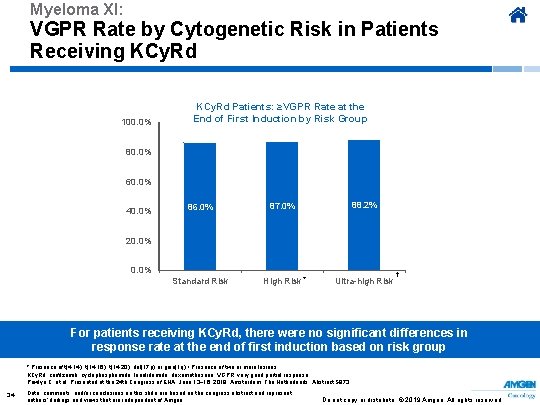
Myeloma XI: VGPR Rate by Cytogenetic Risk in Patients Receiving KCy. Rd 100. 0% KCy. Rd Patients: ≥VGPR Rate at the End of First Induction by Risk Group 80. 0% 60. 0% 40. 0% 86. 0% 87. 0% 88. 2% Standard Risk High Risk * Ultra-high Risk 20. 0% † For patients receiving KCy. Rd, there were no significant differences in response rate at the end of first induction based on risk group * Presence of t(4; 14), t(14; 16), t(14; 20), del(17 p) or gain(1 q) † Presence of two or more lesions KCy. Rd, carfilzomib, cyclophosphamide, lenalidomide, dexamethasone; VGPR, very good partial response. Pawlyn C, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 873. 34 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
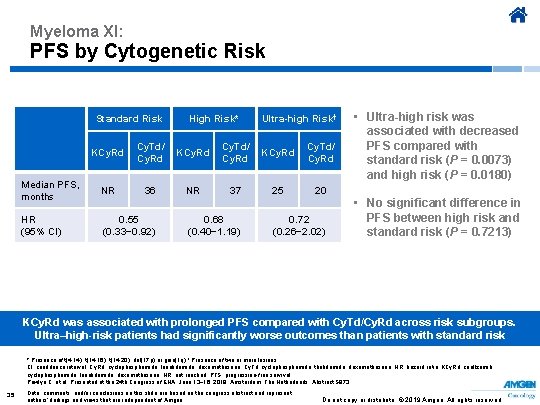
Myeloma XI: PFS by Cytogenetic Risk Standard Risk KCy. Rd High Risk* Cy. Td/ KCy. Rd Median PFS, months NR HR (95% CI) 0. 55 (0. 33− 0. 92) 36 NR Ultra-high Risk† Cy. Td/ KCy. Rd 37 0. 68 (0. 40− 1. 19) 25 Cy. Td/ Cy. Rd • Ultra-high risk was associated with decreased PFS compared with standard risk (P = 0. 0073) and high risk (P = 0. 0180) 20 0. 72 (0. 26− 2. 02) • No significant difference in PFS between high risk and standard risk (P = 0. 7213) KCy. Rd was associated with prolonged PFS compared with Cy. Td/Cy. Rd across risk subgroups. Ultra–high-risk patients had significantly worse outcomes than patients with standard risk * Presence of t(4; 14), t(14; 16), t(14; 20), del(17 p) or gain(1 q) † Presence of two or more lesions CI, confidence interval; Cy. Rd, cyclophosphamide, lenalidomide, dexamethasone; Cy. Td, cyclophosphamide, thalidomide, dexamethasone; HR, hazard ratio; KCy. Rd, carfilzomib, cyclophosphamide, lenalidomide, dexamethasone; NR, not reached; PFS, progression-free survival. Pawlyn C, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 873. 35 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved

Maintenance With Weekly Carfilzomib in Elderly Newly Diagnosed Multiple Myeloma (IFM 2012 -03) Bobin A, et al. Oral Presentation, ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8029. IFM, Intergroupe Francophone du Myélome. 36 Do not copy or distribute. © 2019 Amgen. All rights reserved
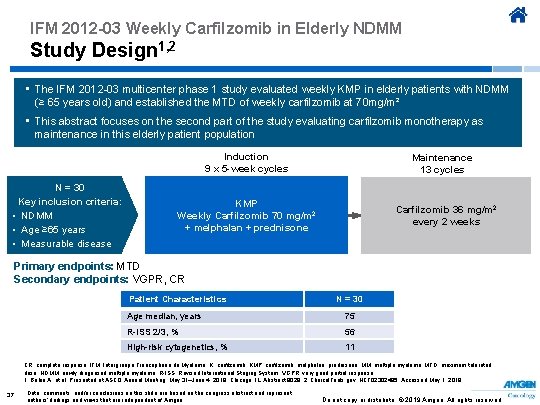
IFM 2012 -03 Weekly Carfilzomib in Elderly NDMM Study Design 1, 2 • The IFM 2012 -03 multicenter phase 1 study evaluated weekly KMP in elderly patients with NDMM (≥ 65 years old) and established the MTD of weekly carfilzomib at 70 mg/m² • This abstract focuses on the second part of the study evaluating carfilzomib monotherapy as maintenance in this elderly patient population Induction 9 x 5 -week cycles N = 30 Key inclusion criteria: • NDMM • Age ≥ 65 years • Measurable disease Maintenance 13 cycles KMP Weekly Carfilzomib 70 mg/m 2 + melphalan + prednisone Carfilzomib 36 mg/m 2 every 2 weeks Primary endpoints: MTD Secondary endpoints: VGPR, CR Patient Characteristics N = 30 Age median, years 75 R-ISS 2/3, % 56 High-risk cytogenetics, % 11 CR, complete response; IFM, Intergroupe Francophone du Myélome; K, carfilzomib; KMP, carfilzomib, melphalan, prednisone; MM, multiple myeloma; MTD, maximum tolerated dose; NDMM, newly diagnosed multiple myeloma; R-ISS, Revised International Staging System; VGPR, very good partial response. 1. Bobin A, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8029. 2. Clinical. Trials. gov. NCT 02302495. Accessed May 1, 2019. 37 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
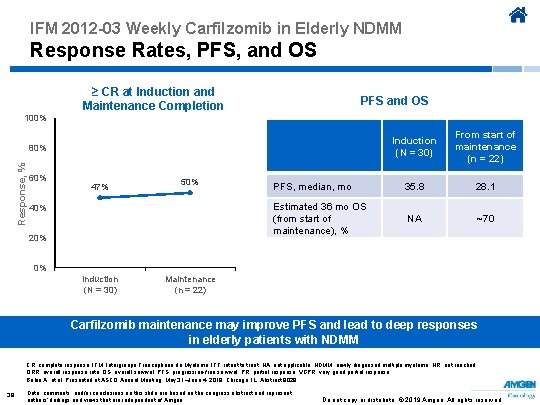
IFM 2012 -03 Weekly Carfilzomib in Elderly NDMM Response Rates, PFS, and OS ≥ CR at Induction and Maintenance Completion PFS and OS 100% Induction (N = 30) From start of maintenance (n = 22) PFS, median, mo 35. 8 28. 1 Estimated 36 mo OS (from start of maintenance), % NA ~70 Response, % 80% 60% 47% 50% 40% 20% 0% Induction (N = 30) Maintenance (n = 22) Carfilzomib maintenance may improve PFS and lead to deep responses in elderly patients with NDMM CR, complete response; IFM, Intergroupe Francophone du Myélome; ITT, intent to treat; NA, not applicable; NDMM, newly diagnosed multiple myeloma; NR, not reached; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PR, partial response; VGPR, very good partial response. Bobin A, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8029. 38 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
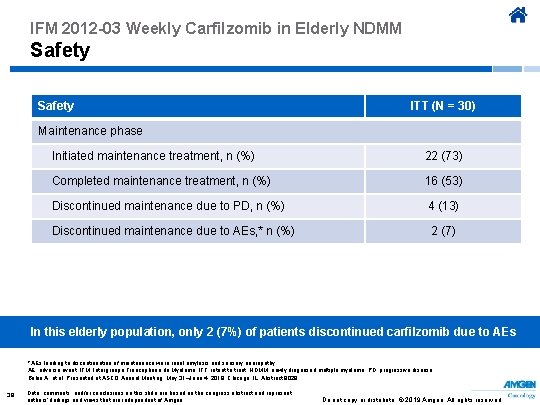
IFM 2012 -03 Weekly Carfilzomib in Elderly NDMM Safety ITT (N = 30) Maintenance phase Initiated maintenance treatment, n (%) 22 (73) Completed maintenance treatment, n (%) 16 (53) Discontinued maintenance due to PD, n (%) 4 (13) Discontinued maintenance due to AEs, * n (%) 2 (7) In this elderly population, only 2 (7%) of patients discontinued carfilzomib due to AEs * AEs leading to discontinuation of maintenance were renal amylosis and sensory neuropathy. AE, adverse event; IFM, Intergroupe Francophone du Myélome; ITT, intent to treat; NDMM, newly diagnosed multiple myeloma; PD, progressive disease. Bobin A, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8029. 39 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved

Elotuzumab in Combination With KRd in Patients With Newly Diagnosed Multiple Myeloma: A Phase 2 MMRC Trial Jasielec J, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1371. KRd, carfilzomib, lenalidomide, dexamethasone; MMRC, Multiple Myeloma Research Consortium. 40 Do not copy or distribute. © 2019 Amgen. All rights reserved
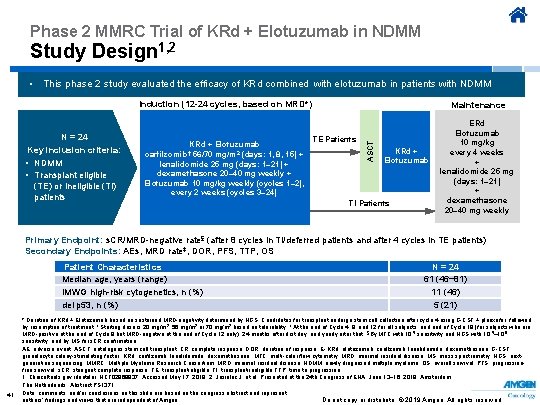
Phase 2 MMRC Trial of KRd + Elotuzumab in NDMM Study Design 1, 2 • This phase 2 study evaluated the efficacy of KRd combined with elotuzumab in patients with NDMM Induction (12 -24 cycles, based on MRD*) KRd + Elotuzumab carfilzomib† 56/70 mg/m 2 (days: 1, 8, 15) + lenalidomide 25 mg (days: 1– 21) + dexamethasone 20– 40 mg weekly + Elotuzumab 10 mg/kg weekly (cycles 1– 2), every 2 weeks (cycles 3– 24) TE Patients ASCT N = 24 Key inclusion criteria: • NDMM • Transplant eligible (TE) or ineligible (TI) patients Maintenance KRd + Elotuzumab TI Patients ERd Elotuzumab 10 mg/kg every 4 weeks + lenalidomide 25 mg (days: 1– 21) + dexamethasone 20– 40 mg weekly Primary Endpoint: s. CR/MRD-negative rate§ (after 8 cycles in TI/deferred patients and after 4 cycles in TE patients) Secondary Endpoints: AEs, MRD rate‡, DOR, PFS, TTP, OS Patient Characteristics Median age, years (range) IMWG high-risk cytogenetics, n (%) del p 53, n (%) 41 N = 24 61 (46− 81) 11 (46) 5 (21) * Duration of KRd + Elotuzumab based on sustained MRD-negativity determined by NGS. Candidates for transplant undergo stem cell collection after cycle 4 using G-CSF + plerixafor, followed by resumption of treatment. † Starting dose is 20 mg/m 2; 56 mg/m 2 or 70 mg/m 2 based on tolerability. ‡ At the end of Cycle 4, 8, and 12 for all subjects, and end of Cycle 18 (for subjects who are MRD-positive at the end of Cycle 8 but MRD-negative at the end of Cycle 12 only), 24 months after first day, and yearly after that. §By MFC with 10 -5 sensitivity and NGS with 10 -5– 10 -6 sensitivity, and by MS for s. CR confirmation AE, adverse event; ASCT, autologous stem cell transplant; CR, complete response; DOR, duration of response; E- KRd, elotuzumab, carfilzomib, lenalidomide, dexamethasone; G-CSF, granulocyte colony-stimulating factor; KRd, carfilzomib, lenalidomide, dexamethasone; MFC, multi-color flow cytometry; MRD, minimal residual disease; MS, mass spectrometry; NGS, nextgeneration sequencing; MMRC, Multiple Myeloma Research Consortium; MRD, minimal residual disease; NDMM, newly diagnosed multiple myeloma; OS, overall survival; PFS, progressionfree survival; s. CR, stringent complete response; TE, transplant-eligible; TI, transplant-ineligible TTP, time to progression. 1. Clinicaltrials. gov identifier: NCT 02969837. Accessed May 17, 2019. 2. Jasielec J, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PS 1371. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
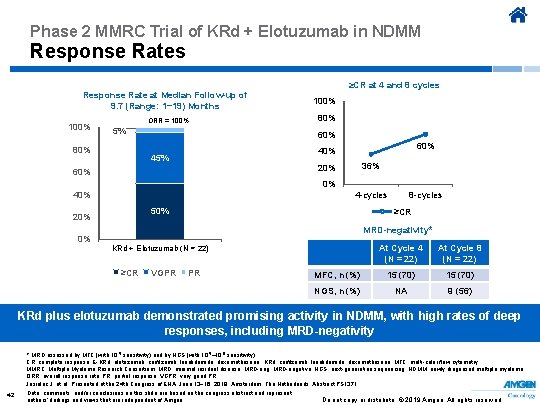
Phase 2 MMRC Trial of KRd + Elotuzumab in NDMM Response Rates ≥CR at 4 and 8 cycles Response Rate at Median Follow-up of 9. 7 (Range: 1− 19) Months 100% ORR = 100% 5% 80% 100% 80% 60% 45% 36% 20% 60% 0% 4 -cycles 50% 20% 0% 8 -cycles ≥CR MRD-negativity* At Cycle 4 (N = 22) At Cycle 8 (N = 22) MFC, n (%) 15 (70) NGS, n (%) NA 9 (56) KRd + Elotuzumab (N = 22) E-KRd (n = 22) ≥CR VGPR PR KRd plus elotuzumab demonstrated promising activity in NDMM, with high rates of deep responses, including MRD-negativity * MRD assessed by MFC (with 10 -5 sensitivity) and by NGS (with 10 -5– 10 -6 sensitivity). CR, complete response; E-KRd, elotuzumab, carfilzomib, lenalidomide, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; MFC, multi-color flow cytometry; MMRC, Multiple Myeloma Research Consortium; MRD, minimal residual disease; MRD-neg, MRD-negative; NGS, next-generation sequencing; NDMM, newly diagnosed multiple myeloma; ORR, overall response rate; PR, partial response; VGPR, very good PR. Jasielec J, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PS 1371. 42 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
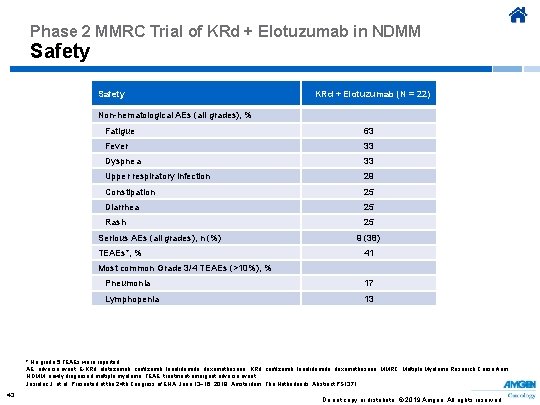
Phase 2 MMRC Trial of KRd + Elotuzumab in NDMM Safety KRd + Elotuzumab (N = 22) Non-hematological AEs (all grades), % Fatigue 63 Fever 33 Dyspnea 33 Upper respiratory infection 29 Constipation 25 Diarrhea 25 Rash 25 Serious AEs (all grades), n (%) TEAEs*, % 9 (38) 41 Most common Grade 3/4 TEAEs (>10%), % Pneumonia 17 Lymphopenia 13 * No grade 5 TEAEs were reported. AE, adverse event; E-KRd, elotuzumab, carfilzomib, lenalidomide, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; MMRC, Multiple Myeloma Research Consortium; NDMM, newly diagnosed multiple myeloma; TEAE, treatment-emergent adverse event. Jasielec J, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PS 1371. 43 Do not copy or distribute. © 2019 Amgen. All rights reserved

Carfilzomib and Dexamethasone Maintenance Prolong Time to Progression Following Salvage ASCT in Multiple Myeloma: A Randomized Phase 2 Trial by the Nordic Myeloma Study Group Gregersen H, et al. Oral Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract S 1602. ASCT, autologous stem cell transplant. 44 Do not copy or distribute. © 2019 Amgen. All rights reserved

Phase 2 CARFI: Kd Maintenance Every Two Weeks After Salvage ASCT Study Design 1, 2 • This open, randomized phase 2 trial was designed to evaluate the efficacy and safety of KCyd induction and conditioning in salvage ASCT followed by Kd maintenance or observation N = 200 Key inclusion criteria: • First relapse after upfront ASCT • >18 yrs Maintenance Conditioning KCyd Carfilzomib* 36 mg/m 2 Carfilzomib (days: 1, 2, 8, 9, 15, 16) 27 mg/m 2 + cyclophosphamide (days 1, 2) + 300 mg/m 2 Melphalan (days: 1, 8, 15) 200 mg/m 2 + (day 2) dexamethasone 20 mg (days: 1, 2, 8, 9, 15, 16) ASCT Induction 4 x 28 -day cycles R 1: 1 Kd (n = 82) Carfilzomib† 56 mg/m 2 every 2 weeks + dexamethasone 20 mg Observation (n = 86) Primary Endpoint: TTP Secondary Endpoints: AEs, ORR, time to marrow regeneration after HDT, OS, Qo. L Patients Characteristics: Median age = 62 years (range: 56 -66). Age, time from myeloma diagnosis, treatment at diagnosis, ECOG performance status, ISS stage, and high-risk cytogenetics were balanced between the two groups 45 * Carfilzomib 20 mg/m 2 is only administered on days 1 and 2 of cycle 1. † Maintenance carfilzomib at 20 mg/m 2 and escalated to 56 mg/m 2 after 4 weeks provided acceptable side effects. AE, adverse event; ASCT, autologous stem cell transplant; ECOG, European cooperative oncology group; HDT, high-dose therapy; IV, intravenous; KCyd, carfilzomib, cyclophosphamide, dexamethasone; Kd, carfilzomib, dexamethasone; NDMM, newly diagnosed multiple myeloma; ORR, overall response rate; OS, overall survival; Qo. L, quality of life; TTP, time to progression. 1. Clinicaltrials. gov identifier: NCT 02572492. Accessed May 17, 2019. 2. Gregersen H, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 1602. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
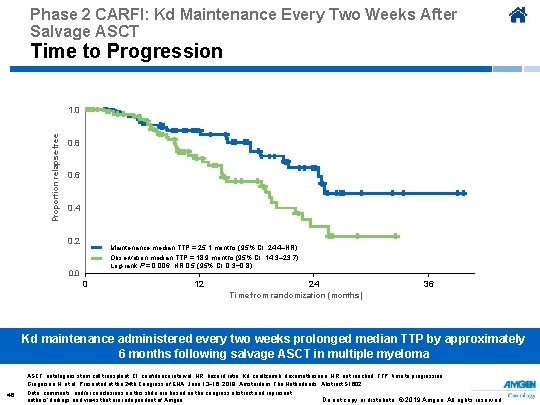
Phase 2 CARFI: Kd Maintenance Every Two Weeks After Salvage ASCT Time to Progression Proportion relapse-free 1. 0 0. 8 0. 6 0. 4 0. 2 Maintenance median TTP = 25. 1 months (95% CI: 24. 4–NR) Observation median TTP = 18. 9 months (95% CI: 14. 3– 23. 7) Log-rank P = 0. 006; HR 0. 5 (95% CI: 0. 3− 0. 8) 0. 0 0 12 24 36 Time from randomization (months) Kd maintenance administered every two weeks prolonged median TTP by approximately 6 months following salvage ASCT in multiple myeloma ASCT, autologous stem cell transplant; CI, confidence interval; HR, hazard ratio; Kd, carfilzomib, dexamethasone; NR, not reached; TTP, time to progression. Gregersen H, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 1602. 46 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
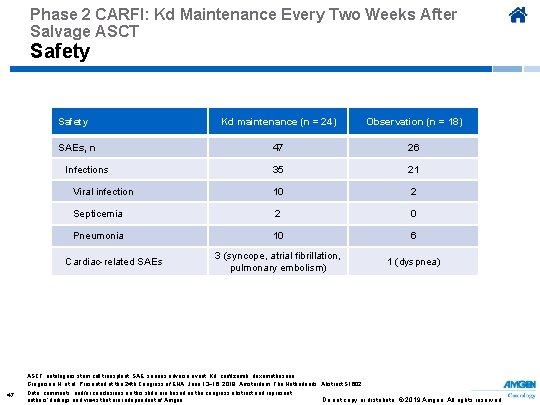
Phase 2 CARFI: Kd Maintenance Every Two Weeks After Salvage ASCT Safety Kd maintenance (n = 24) Observation (n = 18) 47 26 35 21 Viral infection 10 2 Septicemia 2 0 Pneumonia 10 6 3 (syncope, atrial fibrillation, pulmonary embolism) 1 (dyspnea) SAEs, n Infections Cardiac-related SAEs ASCT, autologous stem cell transplant; SAE, serious adverse event; Kd, carfilzomib, dexamethasone. Gregersen H, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 1602. 47 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved

Weekly 20/56 mg/m² Carfilzomib, Lenalidomide, and Dexamethasone Until Progression in Early Relapsed Refractory Multiple Myeloma Gruchet C, et al. Poster Presentation, ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8041. 48 Do not copy or distribute. © 2019 Amgen. All rights reserved

w. KRd 56 Until Progression in RRMM Study Design • In the phase 3 ASPIRE study evaluating twice-weekly KRd, carfilzomib was stopped after 18 cycles 1 • This cohort study evaluated weekly KRd 56 until progression in patients with RRMM, aimed for a duration beyond 18 cycles 2 28 -day cycles until progression 2 N = 282 Key inclusion criteria: • RRMM 1, 2 • ≥ 1 prior therapy 1 w. KRd 56 Carfilzomib* (56 mg/m 2; days 1, 8, 15) + Lenalidomide (25 mg/d; days 1– 21) + Dexamethasone (weekly) Objective: Evaluate the efficacy and safety of w. KRd 562 49 Patient Characteristics 2 N = 28 Age ≥ 65 years, n (%) 5 (18) 1 prior therapy, n (%) 24 (86) *Carfilzomib 20 mg/m 2 was administered on day 1 of cycle 1. 3 KRd, carfilzomib, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma; w. KRd 56, weekly carfilzomib 56 mg/m 2, lenalidomide, dexamethasone. 1. Stewart AK, et al. N Engl J Med. 2015; 372(2): 142 -152. 2. Gruchet C, et al. Presented at ASCO Annual Meeting; May 31 –June 4, 2019; Chicago, IL. Abstract 8041. 3. Richez V, et al. Am J Hematol. 2019; 94(1): E 17 -E 20. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

w. KRd 56 Until Progression in RRMM Efficacy Results Response Rate Duration of Response, % ORR = 85. 7% (CBR = 89. 3%) N = 28 Median DOR, mo 13 18 -month DOR, % 43 PFS, EFS, and OS N = 28 w. KRd 56 (N = 28) Median, mo 30 -Month Expected, % PFS 29 45 EFS 29 45 OS NR 56 Weekly KRd 56 until progression demonstrated promising efficacy in RRMM * 10– 6 and normalized PET-CT. CBR, clinical benefit rate; CR, complete response; DOR, duration of response; EFS, event-free survival; MRD, minimal residual disease; NR, not reached; ORR, overall response rate; OS, overall survival; PET-CT, positron emission tomography-computed tomography; PFS, progression-free survival; PR, partial response; RRMM, relapsed and/or refractory multiple myeloma; SD, stable disease; VGPR, very good partial response; w. KRd 56, weekly carfilzomib 56 mg/m 2, lenalidomide, dexamethasone. Gruchet C, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8041. 50 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
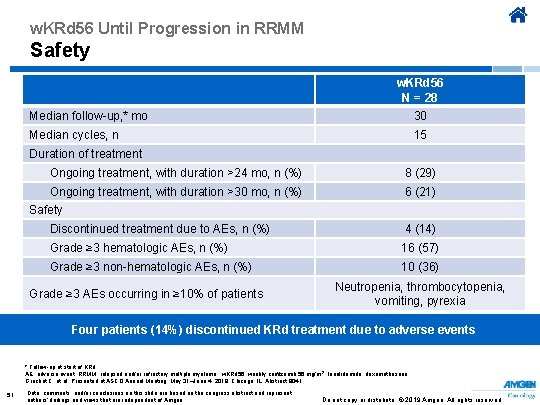
w. KRd 56 Until Progression in RRMM Safety w. KRd 56 N = 28 Median follow-up, * mo 30 Median cycles, n 15 Duration of treatment Ongoing treatment, with duration >24 mo, n (%) 8 (29) Ongoing treatment, with duration >30 mo, n (%) 6 (21) Safety Discontinued treatment due to AEs, n (%) 4 (14) Grade ≥ 3 hematologic AEs, n (%) 16 (57) Grade ≥ 3 non-hematologic AEs, n (%) 10 (36) Grade ≥ 3 AEs occurring in ≥ 10% of patients Neutropenia, thrombocytopenia, vomiting, pyrexia Four patients (14%) discontinued KRd treatment due to adverse events * Follow-up at start of KRd. AE, adverse event; RRMM, relapsed and/or refractory multiple myeloma ; w. KRd 56, weekly carfilzomib 56 mg/m 2, lenalidomide, dexamethasone. Gruchet C, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8041. 51 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
Twice-Weekly Carfilzomib is an Accepted Treatment by Patients With Multiple Myeloma who Have Received at Least One Prior Therapy: Interim Results of the CARO Study Knauf W, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1407. 52 Do not copy or distribute. © 2019 Amgen. All rights reserved

Interim Results of the CARO Study in RRMM Study Design 1 -3 • The objective of this prospective, non-interventional study was to collect real-world data on the implementation of the KRd and Kd regimens in routine clinical practice in Germany, with a special focus on treatment adherence 1 28 -day cycles until PD or unacceptable toxicity N = 102* • RRMM • ≥ 1 prior line of therapy R KRd (n = 68) Carfilzomib† 27 mg/m 2 twice-weekly + lenalidomide 25 mg daily (days: 1− 21) + dexamethasone 40 mg weekly Treatment with K longer than 18 cycles based on benefit/risk assessment Kd (n = 32) Carfilzomib‡ 56 mg/m 2 twice-weekly + dexamethasone 20 mg twice-weekly Primary Objective: Patient adherence to carfilzomib as prescribed by the treating physician Secondary Objectives: Patient adherence to lenalidomide and dexamethasone and evaluation of implementation of the recommended dosing regimens Exploratory Objectives: Efficacy, safety, and HRQo. L 53 * Patients enrolled October 2016–October 2017. Study was designed to collect data on 300 patients. † 20 mg/m 2 on days 1– 2, cycle 1 only. Cycles 13+ of carfilzomib dosed at 27 mg/m 2 on days: 1, 2, 15, 16. ‡ 20 mg/m 2 on days 1– 2, cycle 1 only. HRQo. L, health-related quality of life; Kd, carfilzomib, dexamethasone; KRd, carfilzomib, lenalidomide, dexamethasone; MM, multiple myeloma; PD, progressive disease; Sm. PC, Summary of Product Characteristics. 1. Knauf W, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PS 1407. 2. Clinical. Trials. gov. NCT 02970747. Accessed May 19, 2019. 3. Kyprolis. [Sm. PC]. Breda, The Netherlands: Amgen Europe B. V. 2015. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
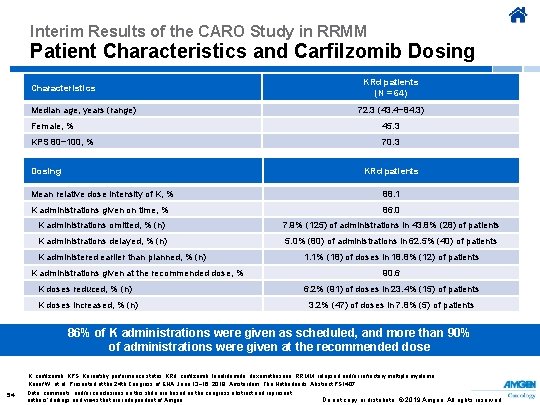
Interim Results of the CARO Study in RRMM Patient Characteristics and Carfilzomib Dosing KRd patients (N = 64) Characteristics Median age, years (range) 72. 3 (43. 4− 84. 3) Female, % 45. 3 KPS 80− 100, % 70. 3 Dosing KRd patients Mean relative dose intensity of K, % 88. 1 K administrations given on time, % 86. 0 K administrations omitted, % (n) 7. 9% (125) of administrations in 43. 8% (28) of patients K administrations delayed, % (n) 5. 0% (80) of administrations in 62. 5% (40) of patients K administered earlier than planned, % (n) K administrations given at the recommended dose, % K doses reduced, % (n) K doses increased, % (n) 1. 1% (18) of doses in 18. 8% (12) of patients 90. 6 6. 2% (91) of doses in 23. 4% (15) of patients 3. 2% (47) of doses in 7. 8% (5) of patients 86% of K administrations were given as scheduled, and more than 90% of administrations were given at the recommended dose K, carfilzomib; KPS, Karnofsky performance status; KRd, carfilzomib, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma. Knauf W, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PS 1407. 54 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
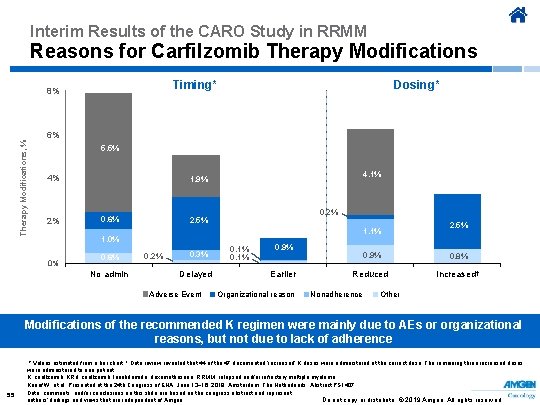
Interim Results of the CARO Study in RRMM Reasons for Carfilzomib Therapy Modifications, % Dosing* Timing* 8% 6% 5. 5% 4% 2% 4. 1% 1. 9% 0. 6% 0. 2% 2. 5% 1. 1% 1. 0% 0% 0. 6% No admin 0. 2% 0. 3% Delayed Adverse Event 0. 1% 0. 9% Earlier Organizational reason 2. 5% 0. 9% 0. 8% Reduced Increased† Nonadherence Other Modifications of the recommended K regimen were mainly due to AEs or organizational reasons, but not due to lack of adherence 55 * Values estimated from a bar chart. † Data review revealed that 44 of the 47 documented ‘increased’ K doses were administered at the correct dose. The remaining three increased doses were administered to one patient. K, carfilzomib; KRd, carfilzomib, lenalidomide, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma. Knauf W, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PS 1407. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

Carfilzomib-Lenalidomide-Dexamethasone in the Management of Lenalidomide-Refractory Multiple Myeloma Cerchione C, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PF 622. 56 Do not copy or distribute. © 2019 Amgen. All rights reserved

KRd in Lenalidomide-Refractory RRMM Study Design • This retrospective, observational trial evaluated efficacy and safety of KRd as a salvage regimen in patients with RRMM refractory to lenalidomide Median 8 treatment cycles (range, 2 -18) N = 41 Key inclusion criteria: • RRMM • Refractory to lenalidomide KRd Twice-weekly carfilzomib 27 mg/m 2 + lenalidomide + dexamethasone Objectives: Efficacy and safety of KRd in lenalidomide-refractory RRMM Patient Characteristics Median age at start of treatment, years (range) Prior lines, n (range) Prior bortezomib and IMi. Ds, % Refractory to bortezomib and IMi. Ds, % ≥ 1 ASCT, % 57 N = 41 67 (48− 84) 3 (2− 11) 100 61 ASCT, autologous stem cell transplant; IMi. D, immunomodulatory drug; K, carfilzomib; KRd, carfilzomib, lenalidomide, dexamethasone; MM, multiple myeloma; PD, progressive disease; RRMM, relapsed and/or refractory multiple myeloma. Cerchione C, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 622. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

KRd in Lenalidomide-Refractory RRMM Response Rate and OS Response, % Response Rate* Time to Response, OS, Patients undergoing Salvage ASCT ORR = 68. 2% (CBR = 87. 8%) KRd (N = 41) Median time to response, months (range) Bridge to salvage ASCT, n 1. 3 (1– 4) 11 Median OS, months (range) From diagnosis 62 (9– 170) From start of carfilzomib 11 (2– 18) KRd (N = 41) KRd induced deep responses and prolonged OS in lenalidomide-refractory RRMM patients. In addition, KRd treatment may be considered prior to salvage ASCT in this patient population. * Median follow-up, 9 months (range, 2– 18 months). ASCT, autologous stem cell transplant; CBR, clinical benefit rate; CR, complete response; KRd, carfilzomib, lenalidomide, dexamethasone; MM, multiple myeloma; ORR, overall response rate; OS, overall survival; PR, partial response; VGPR, very good partial response. Cerchione C, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 622. 58 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
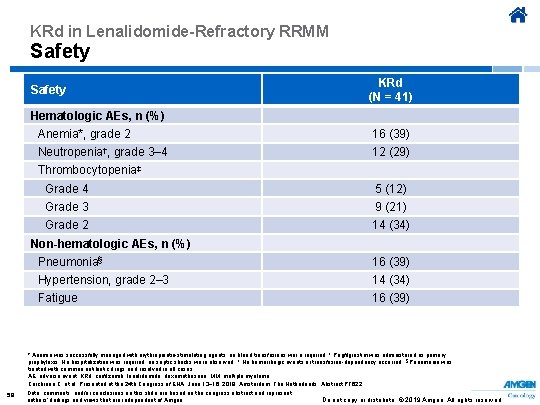
KRd in Lenalidomide-Refractory RRMM Safety KRd (N = 41) Hematologic AEs, n (%) Anemia*, grade 2 16 (39) Neutropenia†, grade 3– 4 12 (29) Thrombocytopenia‡ Grade 4 5 (12) Grade 3 9 (21) Grade 2 14 (34) Non-hematologic AEs, n (%) Pneumonia§ 16 (39) Hypertension, grade 2– 3 14 (34) Fatigue 16 (39) * Anemia was successfully managed with erythropoietin-stimulating agents; no blood transfusions wer e required. † Pegfilgrastim was administered as primary prophylaxis. No hospitalization was required; no septic shocks were observed. ‡ No hemorrhagic events or transfusion-dependency occurred. § Pneumonia was treated with common antibiotic drugs and resolved in all cases. AE, adverse event; KRd, carfilzomib, lenalidomide, dexamethasone; MM, multiple myeloma. Cerchione C, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract PF 622. 59 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved

Updated Safety and Efficacy From a Phase 2 Study of Venetoclax Plus Carfilzomib and Dexamethasone in Patients With RRMM Costa L, et al. Oral Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1375. RRMM, relapsed and/or refractory multiple myeloma. 60 Do not copy or distribute. © 2019 Amgen. All rights reserved
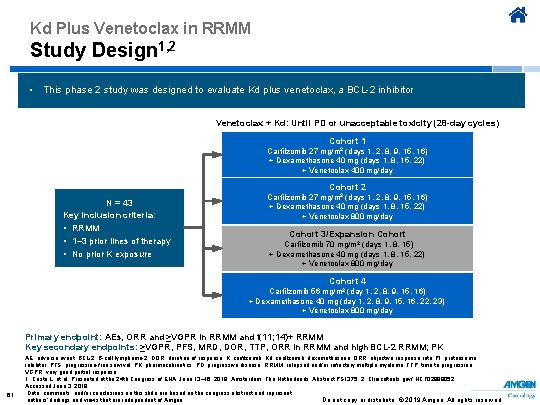
Kd Plus Venetoclax in RRMM Study Design 1, 2 • This phase 2 study was designed to evaluate Kd plus venetoclax, a BCL-2 inhibitor Venetoclax + Kd: Until PD or unacceptable toxicity (28 -day cycles) Cohort 1 Carfilzomib 27 mg/m 2 (days 1, 2, 8, 9, 15, 16) + Dexamethasone 40 mg (days 1, 8, 15, 22) + Venetoclax 400 mg/day Cohort 2 N = 43 Key inclusion criteria: • RRMM • 1– 3 prior lines of therapy • No prior K exposure Carfilzomib 27 mg/m 2 (days 1, 2, 8, 9, 15, 16) + Dexamethasone 40 mg (days 1, 8, 15, 22) + Venetoclax 800 mg/day Cohort 3/Expansion Cohort Carfilzomib 70 mg/m 2 (days 1, 8, 15) + Dexamethasone 40 mg (days 1, 8, 15, 22) + Venetoclax 800 mg/day Cohort 4 Carfilzomib 56 mg/m 2 (day 1, 2, 8, 9, 15, 16) + Dexamethasone 40 mg (day 1, 2, 8, 9, 15, 16, 22, 23) + Venetoclax 800 mg/day Primary endpoint: AEs, ORR and ≥VGPR in RRMM and t(11; 14)+ RRMM Key secondary endpoints: ≥VGPR, PFS, MRD, DOR, TTP, ORR in RRMM and high BCL-2 RRMM; PK 61 AE, adverse event; BCL-2, B-cell lymphoma-2; DOR, duration of response; K, carfilzomib; Kd, carfilzomib, dexamethasone; ORR, objective response rate; PI, proteasome inhibitor; PFS, progression-free survival; PK, pharmacokinetics; PD, progressive disease; RRMM, relapsed and/or refractory multiple myeloma; TTP, time to progression; VGPR, very good partial response. 1. Costa L, et al. Presented at the 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1375. 2. Clinicaltrials. gov/ NCT 02899052. Accessed June 3, 2019. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
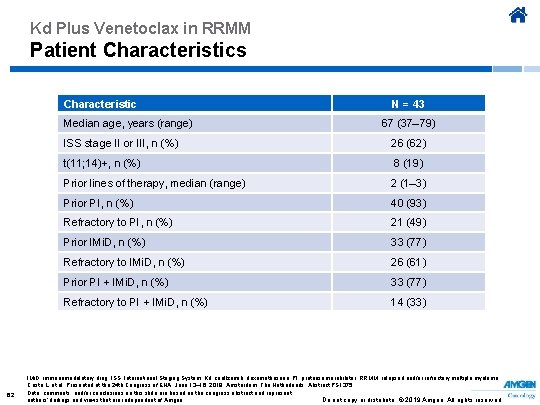
Kd Plus Venetoclax in RRMM Patient Characteristics Characteristic Median age, years (range) 62 N = 43 67 (37– 79) ISS stage II or III, n (%) 26 (62) t(11; 14)+, n (%) 8 (19) Prior lines of therapy, median (range) 2 (1– 3) Prior PI, n (%) 40 (93) Refractory to PI, n (%) 21 (49) Prior IMi. D, n (%) 33 (77) Refractory to IMi. D, n (%) 26 (61) Prior PI + IMi. D, n (%) 33 (77) Refractory to PI + IMi. D, n (%) 14 (33) IMi. D, immunomodulatory drug; ISS, International Staging System; Kd, carfilzomib, dexamethasone; PI, proteasome inhibitor; RRMM, relapsed and/or refractory multiple myeloma. Costa L, et al. Presented at the 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1375. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
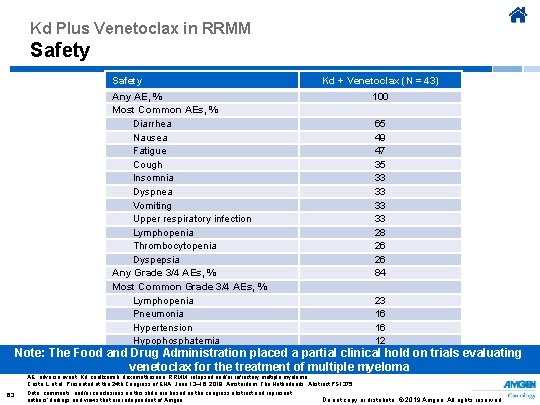
Kd Plus Venetoclax in RRMM Safety Kd + Venetoclax (N = 43) 100 Any AE, % Most Common AEs, % Diarrhea Nausea Fatigue Cough Insomnia Dyspnea Vomiting Upper respiratory infection Lymphopenia Thrombocytopenia Dyspepsia Any Grade 3/4 AEs, % Most Common Grade 3/4 AEs, % Lymphopenia Pneumonia Hypertension Hypophosphatemia 65 49 47 35 33 33 28 26 26 84 23 16 16 12 Note: The Food and Drug Administration placed a partial clinical hold on trials evaluating venetoclax for the treatment of multiple myeloma AE, adverse event; Kd, carfilzomib, dexamethasone; RRMM, relapsed and/or refractory multiple myeloma. Costa L, et al. Presented at the 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1375. 63 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
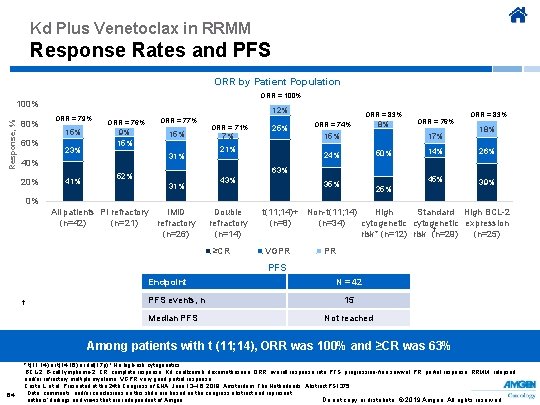
Kd Plus Venetoclax in RRMM Response Rates and PFS ORR by Patient Population ORR = 100% Response, % 100% 80% 60% 12% ORR = 79% 15% 23% ORR = 76% ORR = 77% 9% 15% 31% 40% 20% 41% ORR = 71% 7% 25% 21% ORR = 83% ORR = 74% 8% 17% 15% 50% 24% 63% 52% 31% 43% ORR = 76% 35% ORR = 83% 18% 14% 26% 45% 39% 25% 0% All patients PI refractory IMi. D (n=42) (n=21) refractory (n=26) Double refractory (n=14) ≥CR t(11; 14)+ Non-t(11; 14) High Standard High BCL-2 (n=8) (n=34) cytogenetic expression † risk* (n=12) risk (n=29) (n=25) VGPR PR PFS Endpoint † PFS events, n Median PFS N = 42 15 Not reached Among patients with t (11; 14), ORR was 100% and ≥CR was 63% 64 * t(11; 14) or t(14; 16) or del(17 p) † No high-risk cytogenetics BCL-2, B-cell lymphoma-2; CR, complete response; Kd, carfilzomib, dexamethasone; ORR, overall response rate; PFS, progression-free survival; PR, partial response; RRMM, relapsed and/or refractory multiple myeloma; VGPR, very good partial response. Costa L, et al. Presented at the 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1375. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

A Phase 1 b/2 Study of Selinexor, Carfilzomib, and Dexamethasone in RRMM Gasparetto C, et al. Poster Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1414. RRMM, relapsed and/or refractory multiple myeloma. 65 Do not copy or distribute. © 2019 Amgen. All rights reserved

Kd Plus Selinexor in RRMM Study Design • A previous study had demonstrated that twice-weekly carfilzomib can be safely combined with twice-weekly selinexor 1 • This phase 1 study was initiated to determine the MTD of weekly carfilzomib, selinexor, and dexamethasone (Kd. S) in RRMM 2 N = 9 • RRMM • Prior exposure to a PI were eligible • Carfilzomib-refractory not eligible Kd. S Carfilzomib (56 mg/m 2 or 70 mg/m 2) weekly + Dexamethasone (40 mg) weekly + Selinexor (80 mg or 100 mg) weekly Primary objectives: MTD, recommended phase 2 dose, safety, tolerability, preliminary efficacy Patient Characteristics Age, median years (range) Prior treatment regimens, median (range) Carfilzomib-naïve, n Refractory to prior bortezomib, n Refractory to prior lenalidomide or pomalidomide, n Refractory to prior lenalidomide and pomalidomide, n Refractory to prior daratumumab, n 66 (N = 9) 71 (50 -76) 4 (2 -8) 9 7 8 3 5 Kd, carfilzomib, dexamethasone; MTD, maximum tolerated dose; RRMM, relapsed and/or refractory multiple myeloma; Kd. S, carfilzomib, dexamethasone, selinexor. 1. Jakubowiak A, et al. Presented at the 58 th ASH Annual Meeting; December 3 -6, 2016. Abstract 973. 2. Gasparetto C, et al. Presented at the 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1414. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
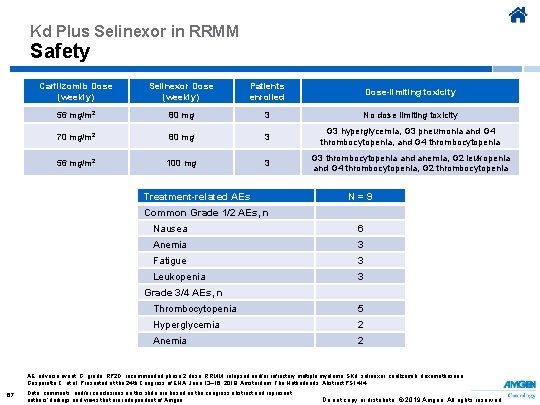
Kd Plus Selinexor in RRMM Safety Carfilzomib Dose (weekly) Selinexor Dose (weekly) Patients enrolled Dose-limiting toxicity 56 mg/m 2 80 mg 3 No dose limiting toxicity 70 mg/m 2 80 mg 3 G 3 hyperglycemia, G 3 pneumonia and G 4 thrombocytopenia, and G 4 thrombocytopenia 56 mg/m 2 100 mg 3 G 3 thrombocytopenia and anemia, G 2 leukopenia and G 4 thrombocytopenia, G 2 thrombocytopenia Treatment-related AEs N = 9 Common Grade 1/2 AEs, n Nausea 6 Anemia 3 Fatigue 3 Leukopenia 3 Grade 3/4 AEs, n Thrombocytopenia 5 Hyperglycemia 2 Anemia 2 AE, adverse event; G, grade; RP 2 D, recommended phase 2 dose; RRMM, relapsed and/or refractory multiple myeloma; SKd, selinexor, carfilzomib, dexamethasone. Gasparetto C, et al. Presented at the 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1414. 67 Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved
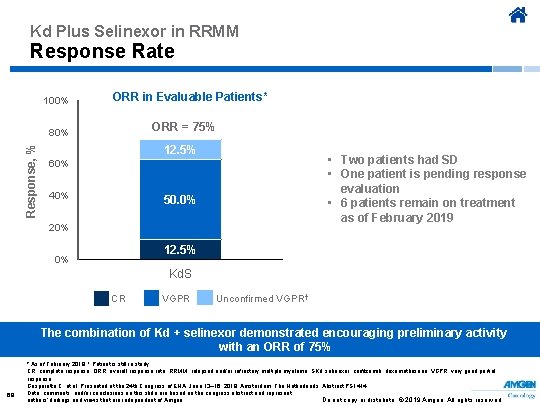
Kd Plus Selinexor in RRMM Response Rate 100% ORR in Evaluable Patients* ORR = 75% Response, % 80% 12. 5% • Two patients had SD • One patient is pending response evaluation • 6 patients remain on treatment as of February 2019 60% 40% 50. 0% 20% 12. 5% 0% Kd. S CR VGPR Unconfirmed VGPR † The combination of Kd + selinexor demonstrated encouraging preliminary activity with an ORR of 75% 68 * As of February 2019. † Patient is still in study. CR, complete response; ORR, overall response rate; RRMM, relapsed and/or refractory multiple myeloma; SKd, selinexor, carfilzomib, dexamethasone; VGPR, very good partial response. Gasparetto C, et al. Presented at the 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract PS 1414. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

Phase II Trial of Carfilzomib and Irinotecan in Relapsed Small Cell Lung Cancer Arnold SM, et al. Poster Presentation, ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8513. 69 Do not copy or distribute. © 2019 Amgen. All rights reserved

Carfilzomib and Irinotecan in Relapsed SCLC Study Design • Phase 2 study of carfilzomib + irinotecan in patients with SCLC whose disease progressed on prior platinum-based therapy 6 x 28 -day cycles or until progression, unacceptable toxicity, or treatment delay >3 wk N = 62 • Recurrent/metastatic SCLC • Progression after one platinumcontaining regimen • Stratification factors: previous response to platinum-based therapy (platinum-sensitive vs -refractory) Carfilzomib 36 mg/m 2* IV (over 30 min), Days 1, 2, 8, 9, 15, 16 of each 28 -day cycle + Irinotecan 125 mg/m 2 IV (over 90 min), Days 1, 8, 15 of each 28 -day cycle Primary endpoint: MTD (phase 1 b), 6 -month OS rate (phase 2) Secondary endpoint: Response rate, PFS, safety/tolerability, biomarkers Exploratory endpoint: Proteosome inhibition, topoisomerase-I expression 70 * Starting carfilzomib dose 20 mg/m 2. IV, intravenous; MTD, maximum tolerated dose; OS, overall survival; PFS, progression-free survival; SCLC, small cell lung cancer. 1. Arnold SM, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8513. 2. Clinicaltrials. gov. NCT 01941316. Accessed May 1, 2019. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.
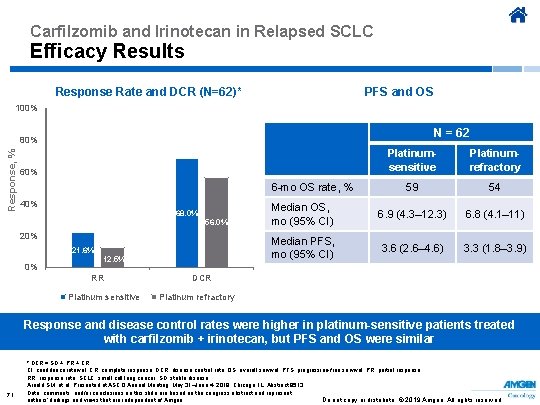
Carfilzomib and Irinotecan in Relapsed SCLC Efficacy Results Response Rate and DCR (N=62)* PFS and OS 100% N = 62 Response, % 80% Platinumsensitive Platinumrefractory 59 54 Median OS, mo (95% CI) 6. 9 (4. 3– 12. 3) 6. 8 (4. 1– 11) Median PFS, mo (95% CI) 3. 6 (2. 6– 4. 6) 3. 3 (1. 8– 3. 9) 60% 6 -mo OS rate, % 40% 68. 0% 56. 0% 21. 6% 0% 12. 5% RR Platinum sensitive DCR Platinum refractory Response and disease control rates were higher in platinum-sensitive patients treated with carfilzomib + irinotecan, but PFS and OS were similar 71 * DCR = SD + PR + CR. CI, confidence interval; CR, complete response; DCR, disease control rate; OS, overall survival; PFS, progression-free survival; PR, partial response; RR, response rate; SCLC, small cell lung cancer; SD, stable disease. Arnold SM, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8513. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent Do not copy or distribute. © 2019 Amgen. All rights reserved authors’ findings and views that are independent of Amgen.

Carfilzomib and Irinotecan in Relapsed SCLC Safety Carfilzomib + irinotecan N = 62 Grade 3 AE, n (%) 29 (47) Grade 4 AE, n (%) 8 (13) Decreased neutrophils, leukocytes, and lymphocytes; diarrhea; vomiting; sepsis; hypokalemia; hypocalcemia; dehydration Treatment-related grade 5 AEs, n (%) 72 3 (5) Myocardial infarction (possible), lung infection (possible), sepsis (probable) AE, adverse event; SCLC, small cell lung cancer. Arnold SM, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8513. Data, comments, and/or conclusions on this slide are based on the congress abstract and represent authors’ findings and views that are independent of Amgen. Do not copy or distribute. © 2019 Amgen. All rights reserved

Evaluation of AMG 420, an Anti-BCMA Bispecific T-cell Engager (Bi. TE®) Immunotherapy, in RRMM Patients: Updated Results of a First-in-Human Phase 1 Dose. Escalation Study Topp MS, et al. Oral Presentation, ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8007. Topp MS, et al. Oral Presentation, 24 th Congress of EHA; June 13– 16, 2019; Amsterdam, The Netherlands. Abstract S 825. BCMA, B-cell maturation antigen; RRMM, relapsed and/or refractory multiple myeloma. 73 Do not copy or distribute. © 2019 Amgen. All rights reserved

AMG 420 FIH Phase 1 Dose-Escalation Study in RRMM Study Design 1 -4 • First-in-human study to assess safety, tolerability, MTD, and antitumor activity of AMG 420 in RRMM patients N = 42* Key inclusion criteria: • RRMM • Progression after ≥ 2 prior treatment lines (including both proteasome inhibitor and IMi. D) Dose escalation AMG 420 by continuous IV infusion 6 -wk cycles (4 wk on, 2 wk off) for up to 10 cycles Single-patient cohorts (0. 2– 1. 6 mcg/d) were followed by cohorts of 3– 6 patients (3. 2– 800 mcg/d) Primary endpoint: DLT, MTD Key secondary endpoints: ORR, DOR, MRD-negative RR, PFS, Safety Patient Characteristics Age, median, years Duration of disease, median, years Prior lines of therapy, median, n Daratumumab refractory, % Duration of treatment, mean (SD), cycles N = 42 65 5. 2 4 21 2. 7 (2. 8) * As of December 10, 2018. DLT, dose-limiting toxicity; DOR, duration of response; FIH, first-in-human; IMi. D, immunomodulatory drug; IV, intravenous; MRD, minimal residual disease; MTD, maximum tolerated dose; ORR, overall response rate; PFS, progression-free survival; RR, response rate; RRMM, relapsed and/or refractory multiple myeloma; SD, standard deviation. 1. Topp MS, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8007. 2. Topp MS, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 825. 3. Clinicaltrials. gov/ NCT 02514239. Accessed May 7, 2019. 4. Topp MS, et al. Blood. 2018; 132: Abstract 1010. 74 Do not copy or distribute. © 2019 Amgen. All rights reserved
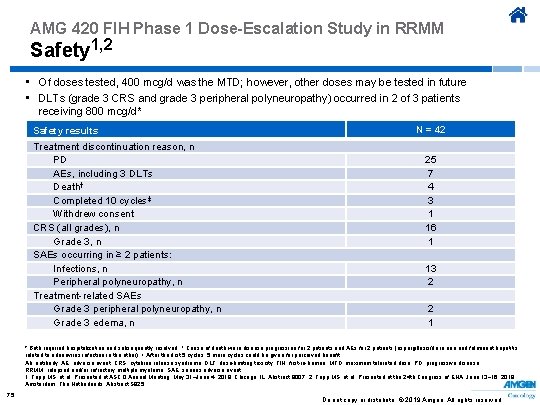
AMG 420 FIH Phase 1 Dose-Escalation Study in RRMM Safety 1, 2 • Of doses tested, 400 mcg/d was the MTD; however, other doses may be tested in future • DLTs (grade 3 CRS and grade 3 peripheral polyneuropathy) occurred in 2 of 3 patients receiving 800 mcg/d* Safety results Treatment discontinuation reason, n PD AEs, including 3 DLTs Death† Completed 10 cycles‡ Withdrew consent CRS (all grades), n Grade 3, n SAEs occurring in ≥ 2 patients: Infections, n Peripheral polyneuropathy, n Treatment-related SAEs Grade 3 peripheral polyneuropathy, n Grade 3 edema, n N = 42 25 7 4 3 1 16 1 13 2 2 1 * Both required hospitalization and subsequently resolved. † Cause of death were disease progression for 2 patients and AEs for 2 patients (aspergillosis/flu in one and fulminant hepatitis related to adenovirus infection in the other). ‡ After the first 5 cycles, 5 more cycles could be given for perceived benefit. Ab, antibody; AE, adverse event; CRS, cytokine release syndrome; DLT, dose-limiting toxicity; FIH, first-in-human; MTD, maximum tolerated dose; PD, progressive disease; RRMM, relapsed and/or refractory multiple myeloma; SAE, serious adverse event. 1. Topp MS, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8007. 2. Topp MS, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 825. 75 Do not copy or distribute. © 2019 Amgen. All rights reserved
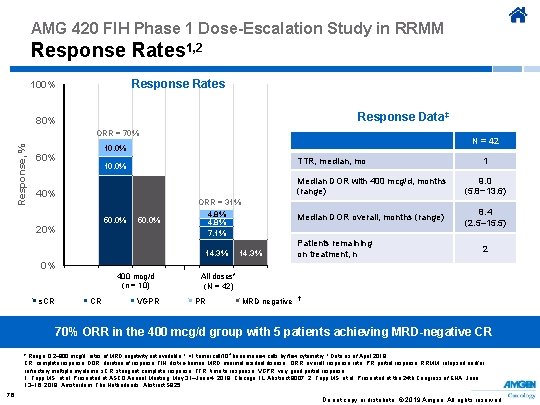
AMG 420 FIH Phase 1 Dose-Escalation Study in RRMM Response Rates 1, 2 Response Rates 100% Response Data‡ 80% Response, % ORR = 70% N = 42 10. 0% 60% TTR, median, mo 10. 0% 40% 50. 0% 20% 50. 0% ORR = 31% 4. 8% 7. 1% 14. 3% 0% s. CR 400 mcg/d (n = 10) CR VGPR 14. 3% 1 Median DOR with 400 mcg/d, months (range) 9. 0 (5. 8− 13. 6) Median DOR overall, months (range) 8. 4 (2. 5– 15. 5) Patients remaining on treatment, n 2 All doses* (N = 42) PR MRD negative † 70% ORR in the 400 mcg/d group with 5 patients achieving MRD-negative CR * Range, 0. 2– 800 mcg/d; rates of MRD negativity not available. † <1 tumor cell/104 bone marrow cells by flow cytometry. ‡ Data as of April 2019 CR, complete response; DOR, duration of response; FIH, first-in-human; MRD, minimal residual disease; ORR, overall response rate; PR, partial response; RRMM, relapsed and/or refractory multiple myeloma; s. CR, stringent complete response; TTR, time to response; VGPR, very good partial response. 1. Topp MS, et al. Presented at ASCO Annual Meeting; May 31–June 4, 2019; Chicago, IL. Abstract 8007. 2. Topp MS, et al. Presented at the 24 th Congress of EHA; June 13− 16, 2019; Amsterdam, The Netherlands. Abstract S 825. 76 Do not copy or distribute. © 2019 Amgen. All rights reserved
- Slides: 76