As temperature increases rate increases because WHY does
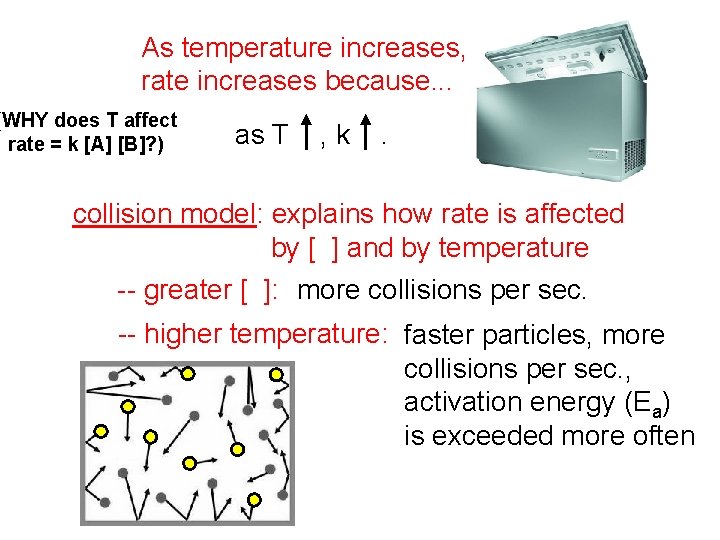
As temperature increases, rate increases because. . . (WHY does T affect rate = k [A] [B]? ) as T , k . collision model: explains how rate is affected by [ ] and by temperature -- greater [ ]: more collisions per sec. -- higher temperature: faster particles, more collisions per sec. , activation energy (Ea) is exceeded more often
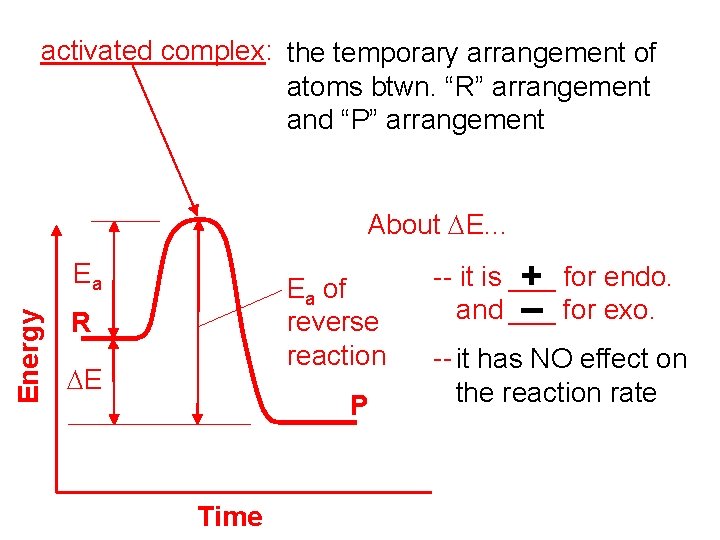
activated complex: the temporary arrangement of atoms btwn. “R” arrangement and “P” arrangement About DE. . . Energy Ea Ea of reverse reaction R DE P Time -- it is ___ + for endo. and ___ – for exo. -- it has NO effect on the reaction rate

For the reaction to occur, collisions must take place with particles oriented in a certain way. e. g. , Cl + NOCl Cl 2 + NO Cl N OCl l C l OC N “NOPE. ” “YEP. ” For a given reaction, the Ea relates rate constants at two different temps WITH those temps. k 1 Ea ln = k 2 R ( 1 1 – T 2 T 1 ) Ea in J/mol R = 8. 314 J/mol-K T = absolute temp.

Using the following information, find. . . (1). . . Ea (2). . . k at 430. 0 K (1) k 1 Ea ln = k 2 R T (o. C) 189. 7 251. 2 ( 1 1 – T 2 T 1 k (s– 1) 2. 52 x 10– 5 3. 16 x 10– 3 ) Ea = 1. 59 x 105 J/mol = 159 k. J/mol

Using the following information, find. . . (1). . . Ea (2). . . k at 430. 0 K T (o. C) 189. 7 251. 2 k (s– 1) 2. 52 x 10– 5 3. 16 x 10– 3 (2) (take “e-to-the-both-sides”…) k 430 = 1. 07 x 10– 6 s– 1 (Using T = 524. 4 K and k = 3. 16 x 10– 3 gives k 430 = 1. 05 x 10– 6 s– 1)
- Slides: 5