AS Chemistry Water of Crystallisation Hydrated Copper Carbonate

AS Chemistry Water of Crystallisation Hydrated Copper Carbonate

Water of Crystallisation When crystallised from aqueous solution, some substances incorporate a fixed number of molecules of water into the lattice structure. This is called "water of crystallisation". The red dots represent the oxygen atoms of the 261 water molecules incorporated into this lipase crystal

Hydrated Substances A substance is described as "hydrated" if it contains water of crystallisation. eg Cu. SO 4. 5 H 2 O is hydrated copper sulphate which are blue crystals. Na 2 CO 3. 10 H 2 O is hydrated sodium carbonate which are colourless crystals.

Anhydrous Substances A substance is described as "anhydrous" if it does not contain water of crystallisation eg Cu. SO 4 is anhydrous copper sulphate which is a white powder. Na 2 CO 3 is anhydrous sodium carbonate which is also a white powder.
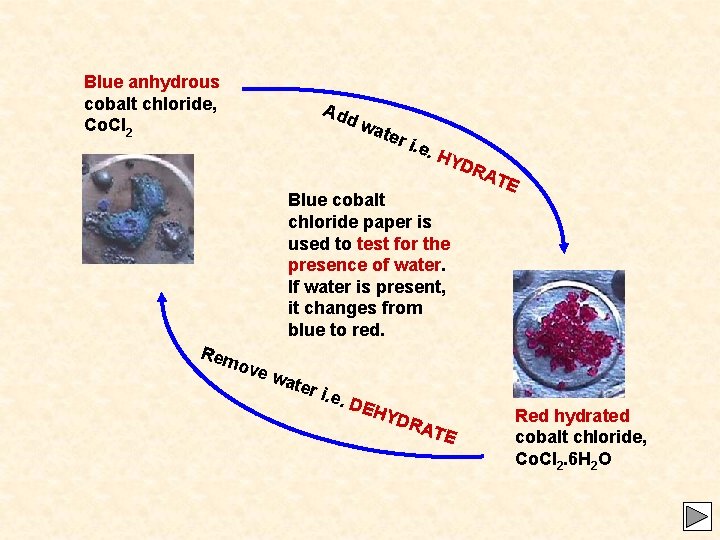
Blue anhydrous cobalt chloride, Co. Cl 2 Add wa ter i. e. HY DR Blue cobalt chloride paper is used to test for the presence of water. If water is present, it changes from blue to red. Rem ATE ove wa ter i. e. DEH YDR A TE Red hydrated cobalt chloride, Co. Cl 2. 6 H 2 O

e. g. 2. 528 g of a hydrate of calcium sulphate, Ca. SO 4. x. H 20, is heated to constant mass. 1. 999 g of anhydrous Ca. SO 4 is produced. Calculate the empirical formula of the hydrate. 1 Formula Ca. SO 4 H 2 O 2 Grams 1. 999 g 2. 528 – 1. 999 = 0. 529 g 3 Moles 1. 999/136 0. 529/18 = 0. 0147 = 0. 0294 4 Ratio 0. 0147/0. 0147 0. 0294/0. 0147 = 1. 00 = 2. 00 5 Formula Ca. SO 4. 2 H 20
- Slides: 6