Aryl Halides Nuclear Substitution Products Obtained by replacing
Aryl Halides

Nuclear Substitution Products: Obtained by replacing one or more H atoms from Benzene nucleus by halogen atoms

• Aryl Halides Ar-X • Organic compounds with a halogen atom attached to an aromatic carbon • Aliphatic compounds readily undergo nucleophilic substitution and elimination reactions • Aromatic compounds resist nucleophilic substitution, only reacting under severe conditions or when strongly electron withdrawing groups are present ortho/para to the halogen.

Commercial methods of preparation: Raschig Process Chlorobenzene can be obtained by passing a mixture of benzene vapours , air and HCl over heated cupric chloride.
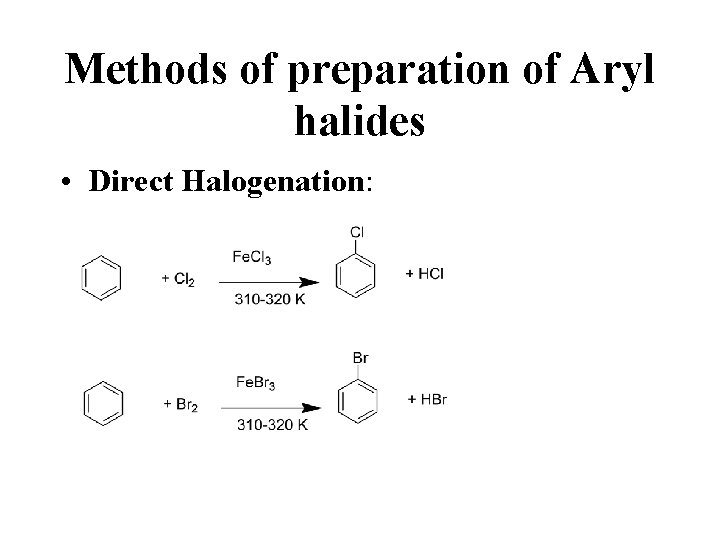
Methods of preparation of Aryl halides • Direct Halogenation:

Direct halogenation • Lewis acids Fe. Cl 3 acts as catalysts • Carry halogen to the benzene ring • Also called halogen carriers Electrophilic substitution reaction • The reaction is done in the absence of light to prevent the formation of addition disubstituted products
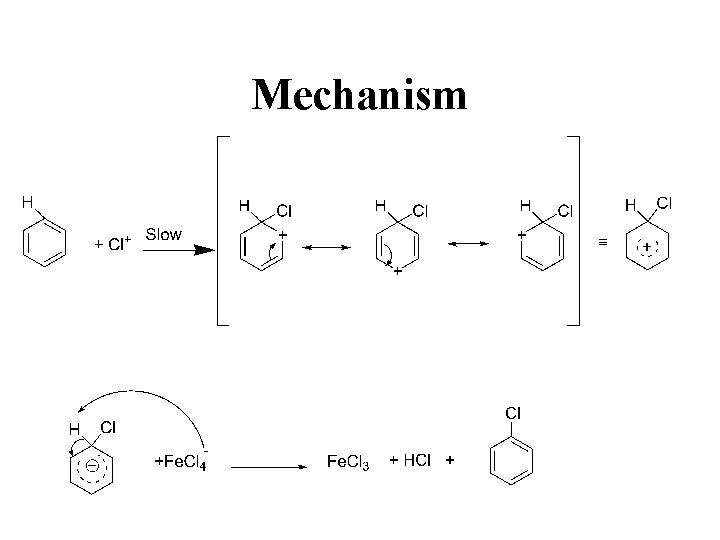
Mechanism
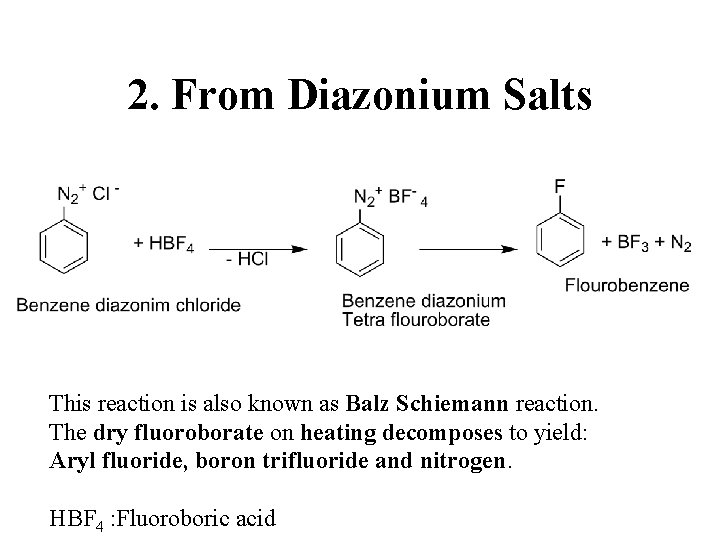
2. From Diazonium Salts This reaction is also known as Balz Schiemann reaction. The dry fluoroborate on heating decomposes to yield: Aryl fluoride, boron trifluoride and nitrogen. HBF 4 : Fluoroboric acid
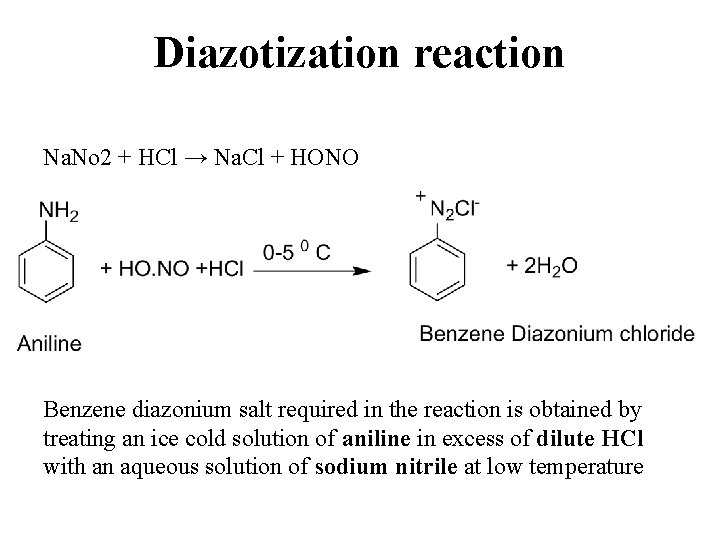
Diazotization reaction Na. No 2 + HCl → Na. Cl + HONO Benzene diazonium salt required in the reaction is obtained by treating an ice cold solution of aniline in excess of dilute HCl with an aqueous solution of sodium nitrile at low temperature
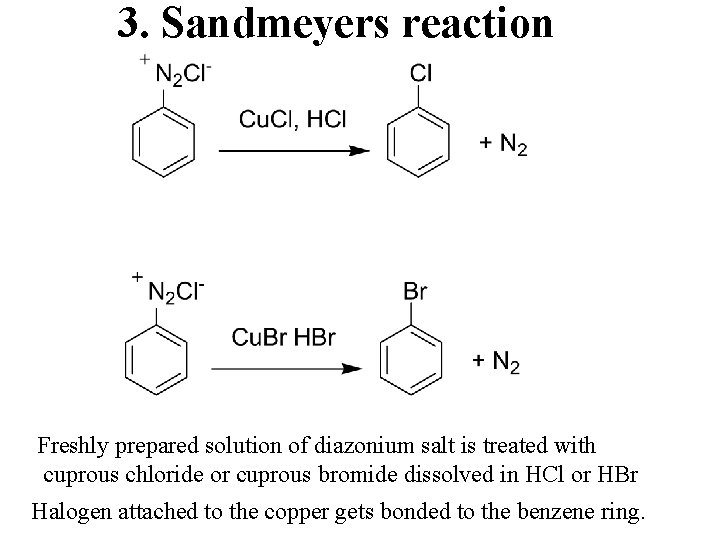
3. Sandmeyers reaction Freshly prepared solution of diazonium salt is treated with cuprous chloride or cuprous bromide dissolved in HCl or HBr Halogen attached to the copper gets bonded to the benzene ring.
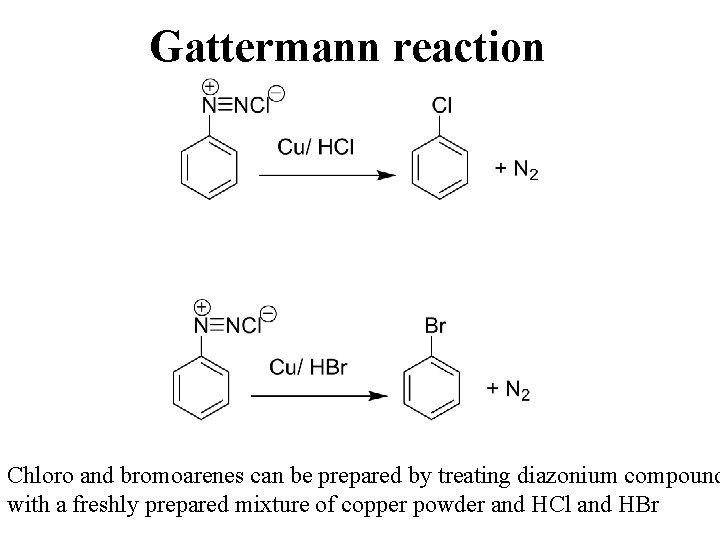
Gattermann reaction Chloro and bromoarenes can be prepared by treating diazonium compound with a freshly prepared mixture of copper powder and HCl and HBr
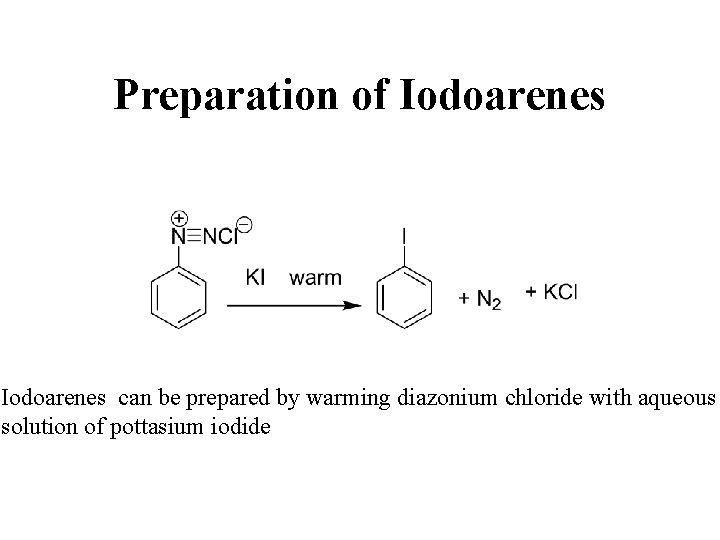
Preparation of Iodoarenes can be prepared by warming diazonium chloride with aqueous solution of pottasium iodide

Advanatges of preparing Aryl halides with Diazonium salts • Product is not contaminated with ortho or para isomers or disubstitutes products • Flouride and iodide are not prepared by direct halogenation hence can be prepared with diazonium salts.

Physical Properties • Colorless oily liquids • Characteristic aromatic odour • Insoluble in water: Absense of Hydrogen bonding • Soluble in organic solvents • B. p, M. p. rise from Flouro to Iodo
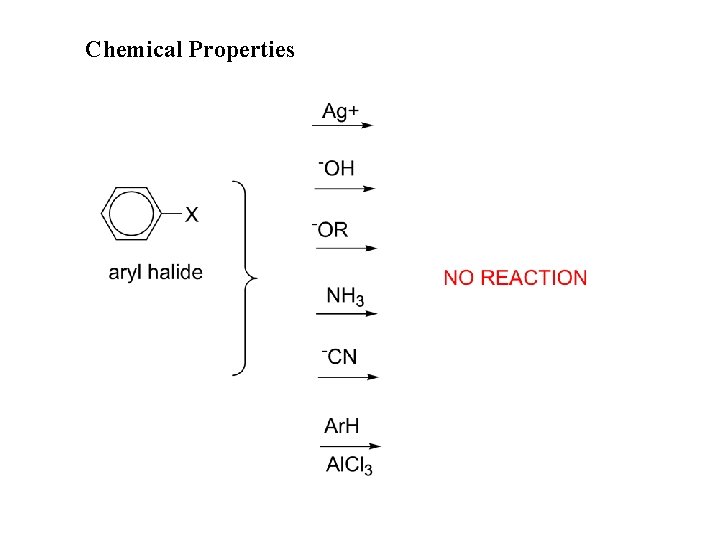
Chemical Properties

Aryl halides, reactions: 1. Formation of Grignard reagent 2. Electrophilic aromatic substituion 3. Nucleophilic aromatic substitution (bimolecular displacement) (Ar must contain strongly electron withdrawing groups ortho and/or para to X) 4. Nucleophilic aromatic substitution (elimination-addition) (Ring not activated to bimolecular displacement)
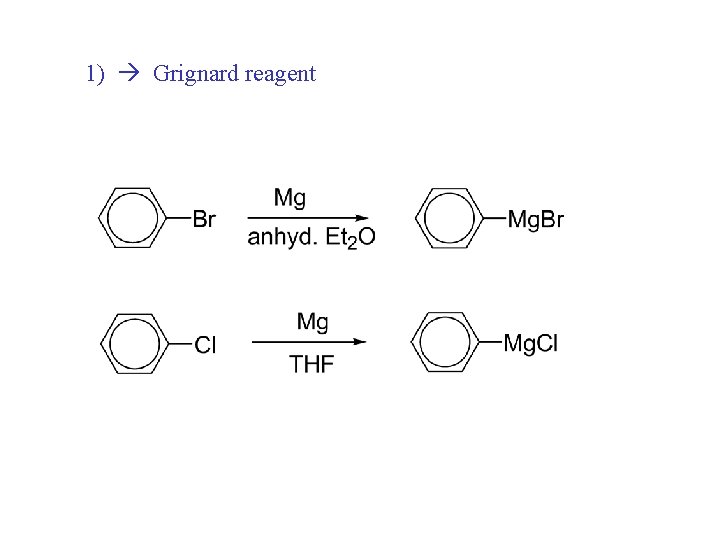
1) Grignard reagent
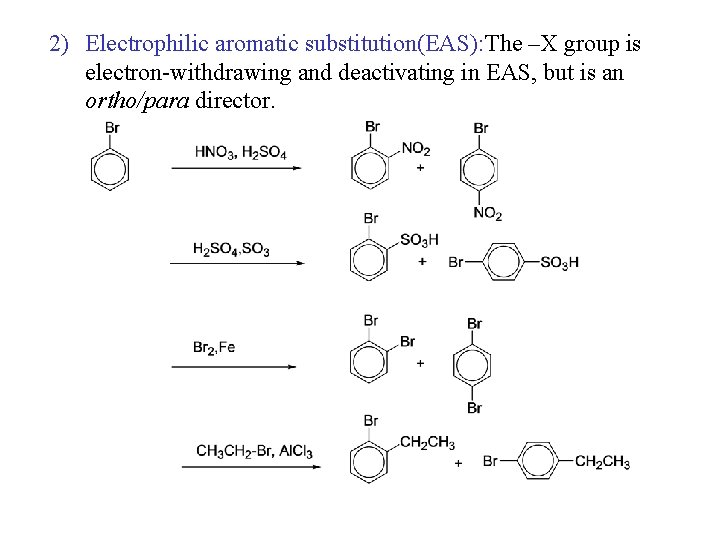
2) Electrophilic aromatic substitution(EAS): The –X group is electron-withdrawing and deactivating in EAS, but is an ortho/para director.
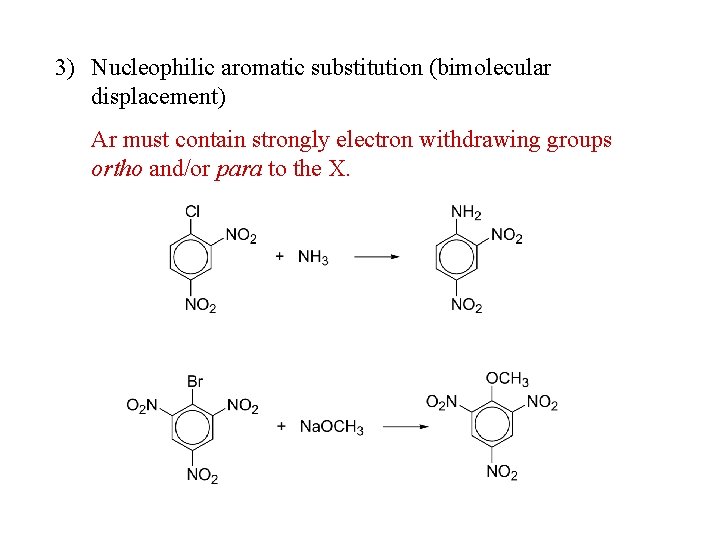
3) Nucleophilic aromatic substitution (bimolecular displacement) Ar must contain strongly electron withdrawing groups ortho and/or para to the X.

NR: No reaction

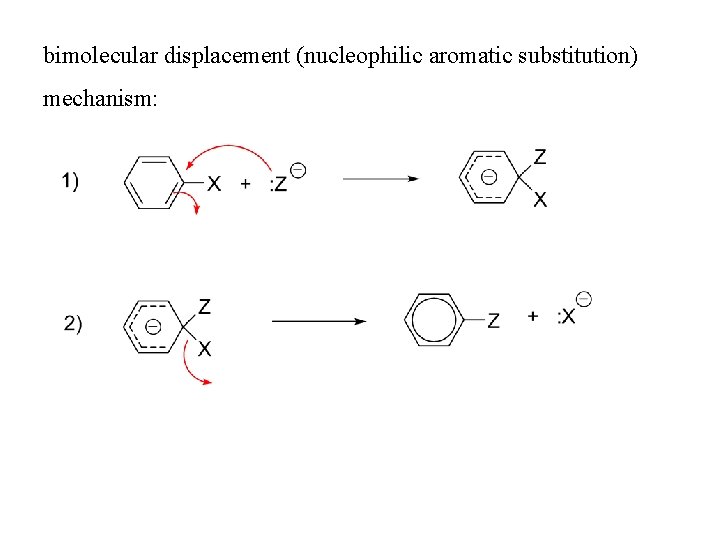
bimolecular displacement (nucleophilic aromatic substitution) mechanism:

Bimolecular displacement mechanism • Involves two steps • First Step: Slow: Rate determining step • Nucleophile attacks Carbon carrying halogen atom to form an intermediate(carbanion) • The intermediate is resonance stabilized • Aromatic character of benzene is destroyed as the hybridization state changes from sp 2 to sp 3 • Second step: Fast: doesn’t affect the rate of reaction • Loss of halide ion • Product formed has aromatic character

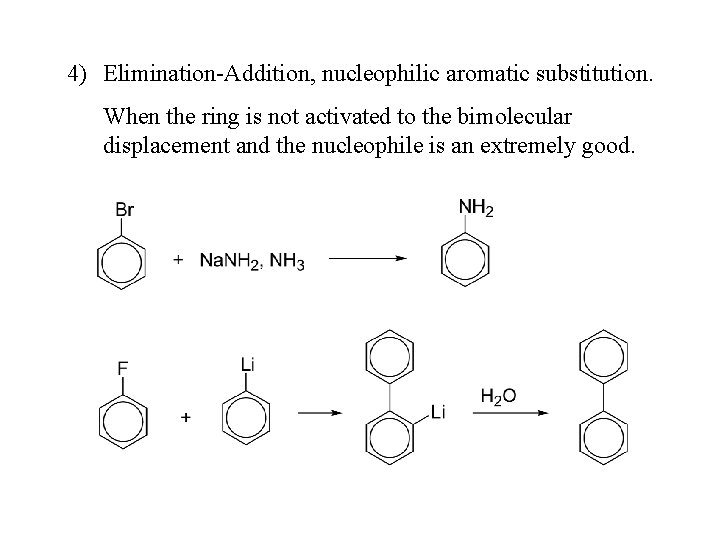
4) Elimination-Addition, nucleophilic aromatic substitution. When the ring is not activated to the bimolecular displacement and the nucleophile is an extremely good.

Elimination-Addition mechanism (nucleophilic aromatic substitution)

Ellimination-Addition mechanism / Benzyne Mechanism • Halogen atom on benzene ring is: ortho and para directing • Electron withdrawing groups deactivate the ring • Attack of nucleophile is facilitated • Strong nucleophile like amide ion(: NH 2) can undergo nucleophilic substitution

Benzyne • It is also called dehydrobenzene or Aryne • Additional bond is formed between two carbon atoms which are holding the halogen atom and hydrogen atom by sideways overlapping of sp 2 orbitals • Sideways overlapping is weak and hence weak bond is formed • Benzyne is highly reactive and hasn’t been isolated
- Slides: 28