ARV Pharmacy Refill Adherence Robert Grossberg MD Montefiore

ARV Pharmacy Refill Adherence • Robert Grossberg, MD • Montefiore Medical Center • Albert Einstein College of Medicine 1

Objectives • Understand the importance of antiretroviral adherence in HIV • Evaluate various adherence measurement methods • Review the use of pharmacy refill adherence methodology in HIV 2
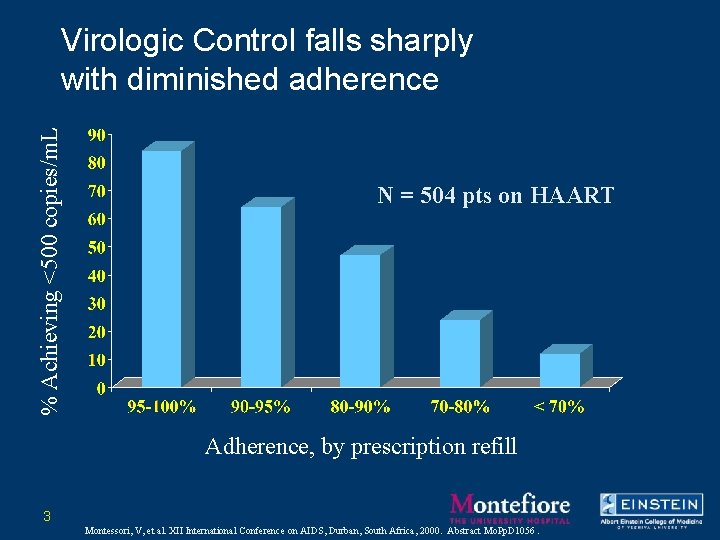
% Achieving <500 copies/m. L Virologic Control falls sharply with diminished adherence N = 504 pts on HAART Adherence, by prescription refill 3 Montessori, V, et al. XII International Conference on AIDS, Durban, South Africa, 2000. Abstract Mo. Pp. D 1056.
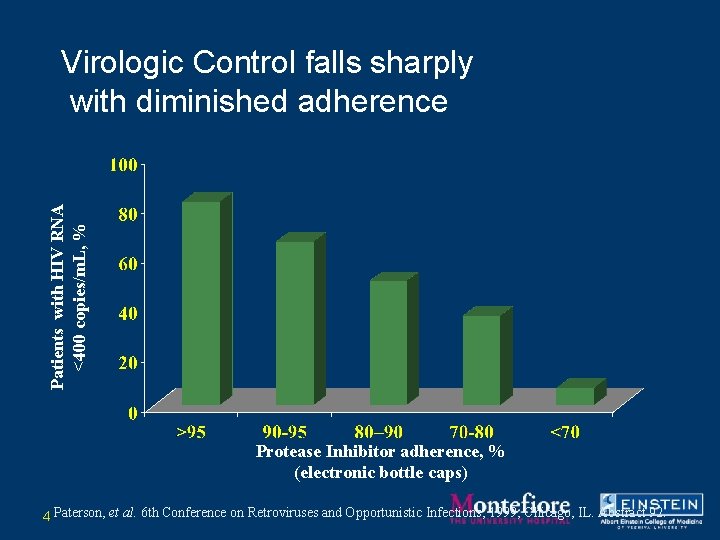
Patients with HIV RNA <400 copies/m. L, % Virologic Control falls sharply with diminished adherence Protease Inhibitor adherence, % (electronic bottle caps) 4 Paterson, et al. 6 th Conference on Retroviruses and Opportunistic Infections; 1999; Chicago, IL. Abstract 92.
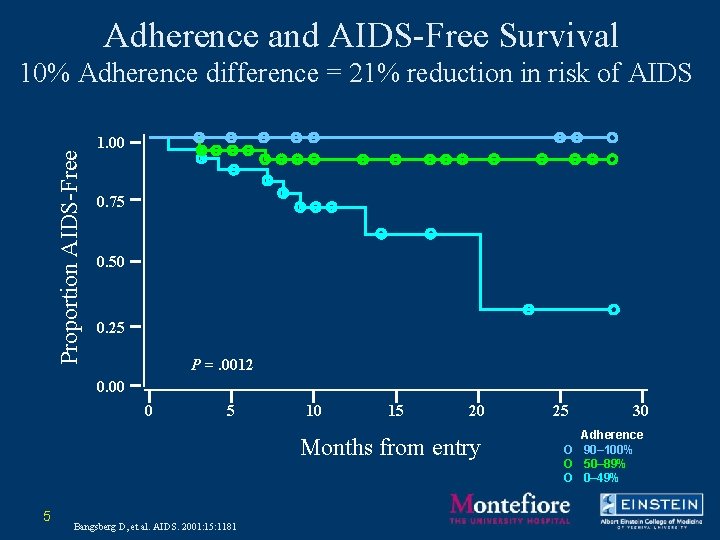
Adherence and AIDS-Free Survival Proportion AIDS-Free 10% Adherence difference = 21% reduction in risk of AIDS 1. 00 0. 75 0. 50 0. 25 P =. 0012 0. 00 0 5 10 15 20 Months from entry 5 Bangsberg D, et al. AIDS. 2001: 15: 1181 25 30 Adherence O 90– 100% O 50– 89% O 0– 49%

Sub-Optimal Adherence Predisposes to Resistance • Sub-optimal adherence ==> sub-therapeutic drug levels ==> incomplete viral suppression ==> generation of resistant HIV strains by selection for mutant viruses • Association between poor adherence and antiretroviral resistance is well-documented 1, 2 6 1. Vanhove G, et al. JAMA. 1996; 276: 1955 -1956. 2. Montaner JS, et al. JAMA. 1998; 279: 930 -937.
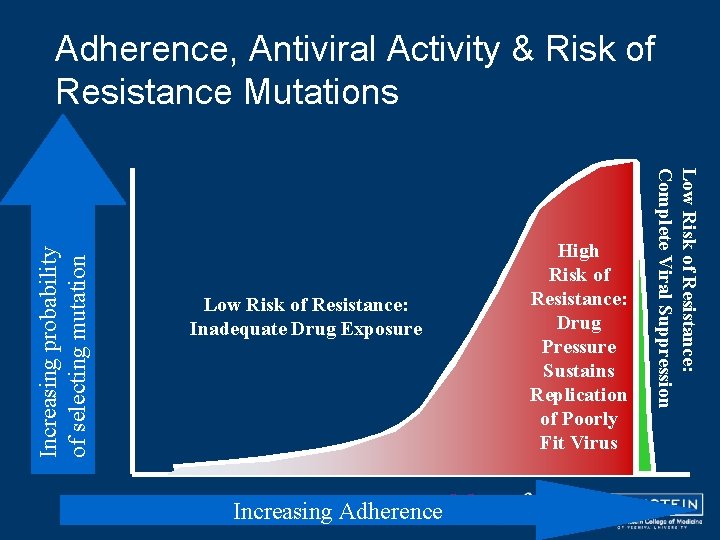
Low Risk of Resistance: Inadequate Drug Exposure Increasing Adherence High Risk of Resistance: Drug Pressure Sustains Replication of Poorly Fit Virus Low Risk of Resistance: Complete Viral Suppression Increasing probability of selecting mutation Adherence, Antiviral Activity & Risk of Resistance Mutations

How do we Measure Adherence? • • 8 Provider Estimates Patient self-report Diaries Pill Count Laboratory Markers Electronic Devices Prescription refill data

Measuring Adherence: Patient Self. Report • patients tend to report what they think the provider wants to hear 1 • patients are unlikely to misrepresent low levels of adherence 3 - hence, patient-reported poor adherence is specific but not sensitive • patient-reported adherence tends to exceed adherence by more objective measurements (such as pill count or electronic monitoring) 2 • Nevertheless, studies have documented an association between patient-reported adherence and viral outcome 4 -6 • Patient-reported adherence may be a useful tool to evaluate adherence at a group level but not so much on an individual level 1. Di. Matteo MR, Di. Nicola DD, eds. Achieving Patient Compliance. New York: Pergamon Press; 1982: 1 -28. 2. Golin C et al. 6 th Conference on Retroviruses and Opportunistic Infections; 1999; Chicago. Abstract 95. 9 3. Bond W, Hussar DA, Am J Public Health 1991; 81: 1978 -1988. 4 Bangsberg DR, et al. 6 th Conference on Retroviruses and Opportunistic Infections; 1999; Chicago. Abstract 93. 5. Duong M, et al. 39 th ICAAC; 1999; San Francisco. Abstract 2069 6. Demasi R, et al. 6 th Conference on Retroviruses and Opportunistic Infections; 1999; Chicago. Abstract 94.

Measuring Adherence: Electronic Bottle Caps MEMScaps, Aardex Corp. 10
Quick. Read software, for use with MEMScaps system 11 http: //www. aardex. ch/QRChronology. htm

Measuring Adherence: Electronic Bottle Caps • Advantages – more difficult for patients to exaggerate their adherence – reveals patterns of nonadherence (i. e. , what time of day pills are taken) – studies using these devices have documented relationship between adherence & dosing 12 • Disadvantages – too expensive for routine use outside of research studies – cannot be used for patients who use pillboxes

Pharmacy Refill Data • Advantages – only choice for retrospective studies – can assess short or long-term behavior • Disadvantages – less intra-interval variability – further removed from actual drug taking – may not capture (legitimate) prescriptions from other sources – if automatic refills, data are useless 13

Sources of Refill Data • Automated database – Medicaid – VA System Pharmacies – Pharmacy Benefit Managers • Ad hoc data collection – Call pharmacies – HIPAA barriers? 14

Examples of Refill Data • Antihypertensives –Taken chronically • Disease process over years/decades • Drugs infrequently changed –Metric: number of refills obtained over year • Ratio of number of refills/12 15

Examples of Refill Data • Antiretrovirals – Taken chronically • Disease process over months/years • Drugs frequently changed – Metric: number of days to obtain 4 refills (3 months) • Ratio of 90 days supply/# of days to obtain supply • Time to event approach • Allows for more variability over shorter interval 16

Prior Work using Refills in HIV • Low-Beer et al. (Vancouver) – 886 subjects – Median cd 4 count 290 cells/cm 3 (IQR 130 -440) – Median viral load 130 K (47 K-310 K) – Follow up-median 19 mo (IQR 13 -24 mo) – Adherence defined as • # refills obtained/# months on therapy over 1 year – Outcome-viral load <500 c/ml 17
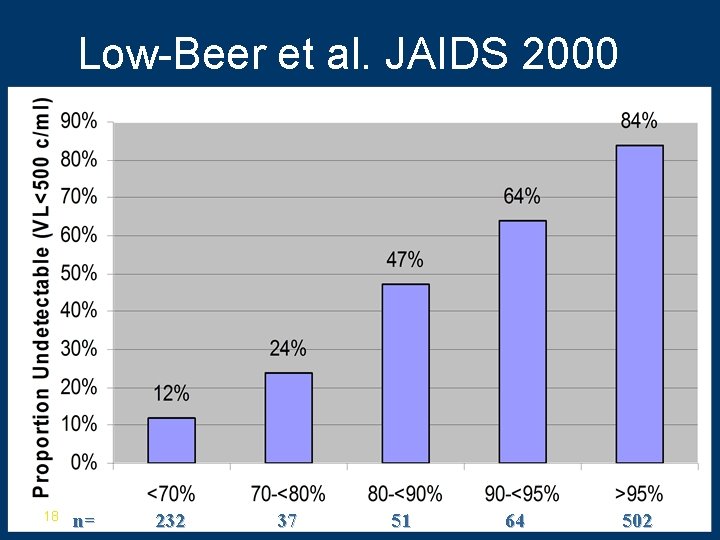
Low-Beer et al. JAIDS 2000 18 n= 232 37 51 64 502

Issues with Refill Data • Variety of other approaches possible – Assessment of time to refill – Assessment of duration of gaps – Others • Limitations – Unclear how they will operate on short term – For example, 3 months of follow-up allows only for 2, 3, or 4 fills using Low-Beer method 19

Choice of Pharmacy Metric –Metric: number of days to obtain 4 refills (3 months) • 90 days supply/# of days to obtain supply • Time to event approach • Allows for more variability over shorter (clinically relevant) interval 20
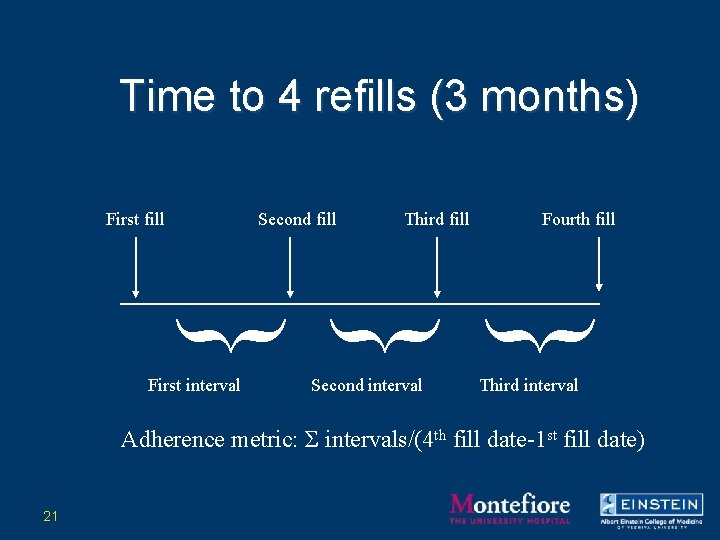
Time to 4 refills (3 months) First fill Second fill Third fill Fourth fill } } } First interval Second interval Third interval Adherence metric: Σ intervals/(4 th fill date-1 st fill date) 21

VA Pharmacy Refill Study • Specific aim –To compare validity of self-reported measure and pharmacy refill measure of adherence to antiretroviral therapy in HIV 22

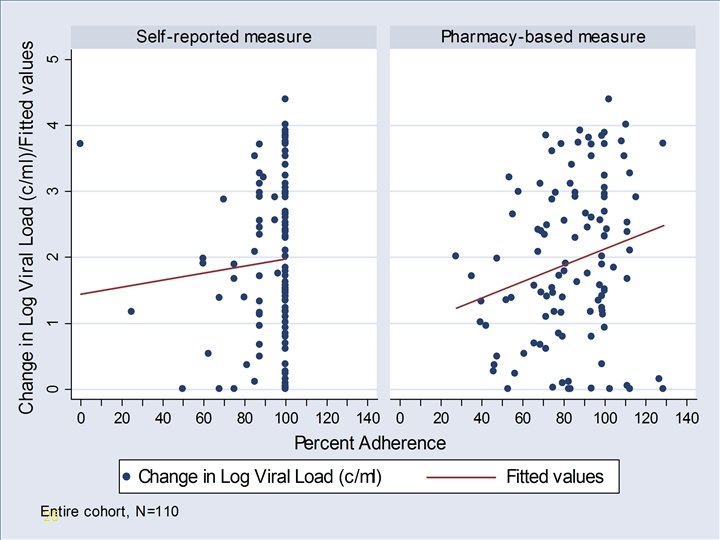
VA Refill Study Design • Observational Study (n=110) conducted in the Philadelphia VA HIV Clinic • Outcomes – Change in HIV viral load from baseline to study date – HIV viral load undetectable or not (dichotomized) • Exposures – Adherence measured via self-report (ACTG measure) – Adherence measured using refill data (time to obtaining 90 days supply) 23

Setting/Study Patients • Subjects on therapy at least 3 months • Philadelphia VA Medical Center – Veterans obtain all HIV Rx here – Electronic pharmacy records – Mailed medications require telephone call 24
25

VA Pharmacy Study Results • Spearman correlation coefficient (95% CI) • Adherence and change in viral load ØPharmacy refill = 0. 22 (0. 01 to 0. 40) ØSelf-report = 0. 10 (-0. 08 to 0. 32) 26

VA Pharmacy Study Results Change in plasma viral load Rank sum test Method Study group Adherence >85% Adherence <85% Pharmacy Entire cohort N=57 2. 4 log c/ml (IQR 1. 4 - 3. 2) N=53 1. 5 log c/ml (IQR 0. 7 - 2. 4) 0. 005 N=96 2. 1 log c/ml (IQR 1. 1 - 3. 0) N=14 1. 4 log c/ml (IQR 0. 4 -1. 9) 0. 04 N=44 2. 4 log c/ml (IQR 1. 4 - 3. 4) N=30 1. 5 log c/ml ( IQR 0. 8 - 2. 4) 0. 03 Self-report Pharmacy 27 Entire cohort 100% by selfreport p value

Conclusions of Refill Study • Time to refill is a valid adherence measure – may perform better than self-report • Generalizability outside of VA? • Unclear function over shorter intervals (e. g. , 1 or 2 months) 28
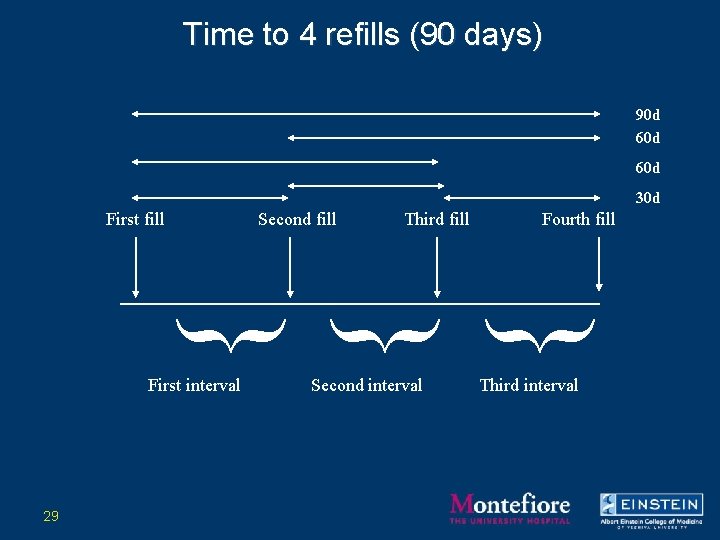
Time to 4 refills (90 days) 90 d 60 d 30 d First fill Second fill Third fill Fourth fill } } } First interval 29 Second interval Third interval
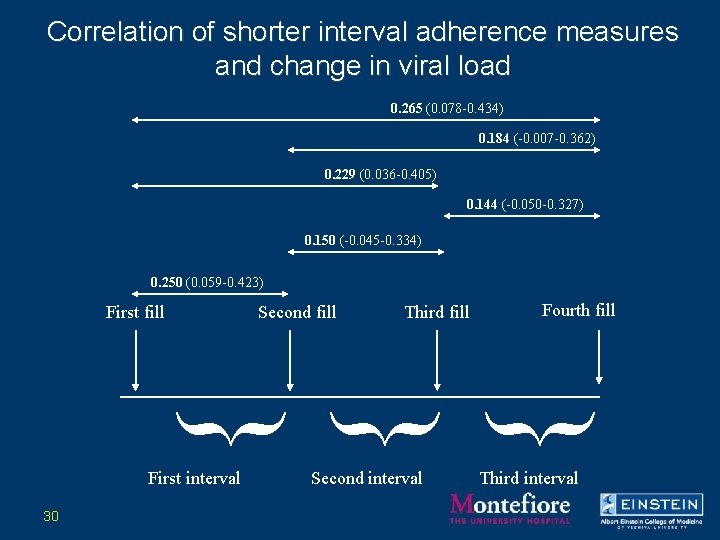
Correlation of shorter interval adherence measures and change in viral load 0. 265 (0. 078 -0. 434) 0. 184 (-0. 007 -0. 362) 0. 229 (0. 036 -0. 405) 0. 144 (-0. 050 -0. 327) 0. 150 (-0. 045 -0. 334) 0. 250 (0. 059 -0. 423) First fill Second fill Third fill Fourth fill } } } First interval 30 Second interval Third interval
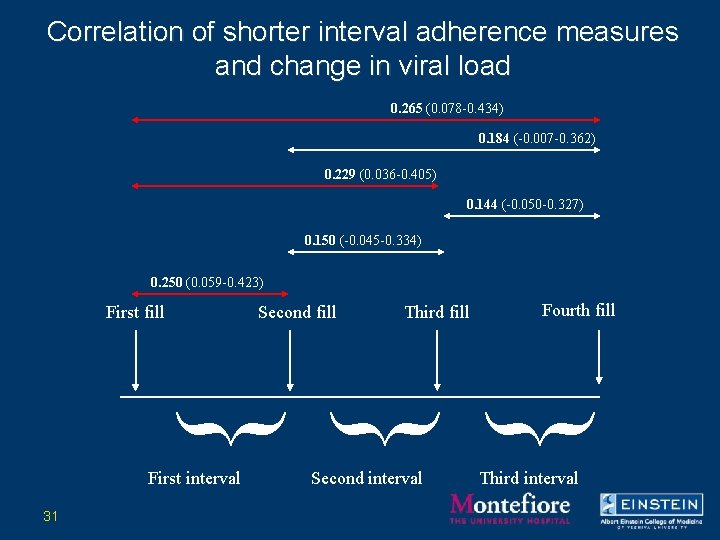
Correlation of shorter interval adherence measures and change in viral load 0. 265 (0. 078 -0. 434) 0. 184 (-0. 007 -0. 362) 0. 229 (0. 036 -0. 405) 0. 144 (-0. 050 -0. 327) 0. 150 (-0. 045 -0. 334) 0. 250 (0. 059 -0. 423) First fill Second fill Third fill Fourth fill } } } First interval 31 Second interval Third interval

Conclusions regarding shorter interval measurements of refill adherence • Shorter interval measurements of refill adherence are associated with virologic outcome. • The “upstream” interval is the best predictor of outcome. 32

Summary • Refill adherence is a valid method for measuring adherence. • Refill adherence correlates with outcome. • Short interval measurements of refill adherence are valid, but only if measured 60 -90 days in advance of the point of interest. • Clinical use of refill data to inform providers about medication adherence is evolving. 33
- Slides: 33