Artificial Skin Vi Tran BME 281 Vi Tran
Artificial Skin Vi Tran BME 281 Vi Tran Section 1 October 23, 2013
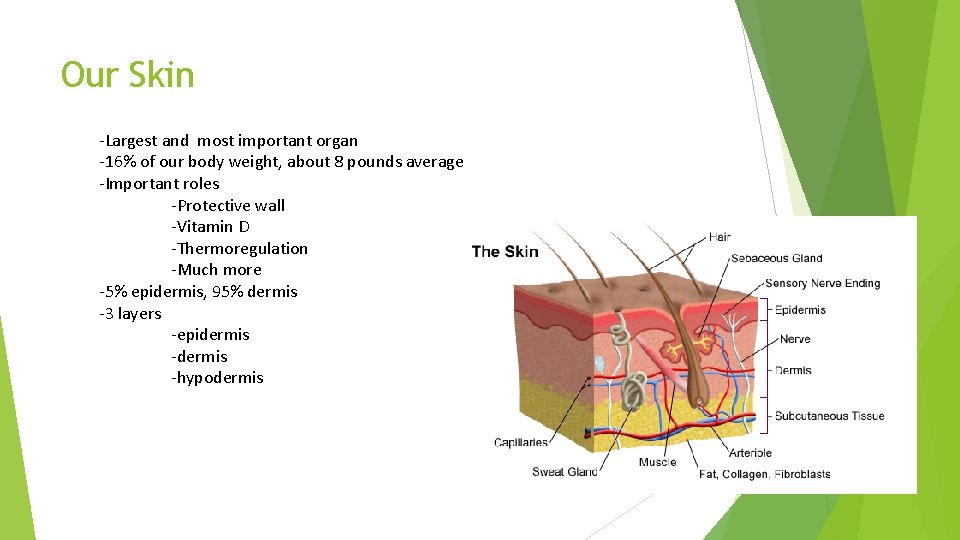
Our Skin -Largest and most important organ -16% of our body weight, about 8 pounds average -Important roles -Protective wall -Vitamin D -Thermoregulation -Much more -5% epidermis, 95% dermis -3 layers -epidermis -hypodermis

Burns -According to American Burn Association -450, 000 people require professional care -45, 000 people require hospitalization for treatment of burns -3, 000 people die from burns -In 2010, every 169 minutes one person dies from a fire -Dangers of Burns -Dehydration -Infection
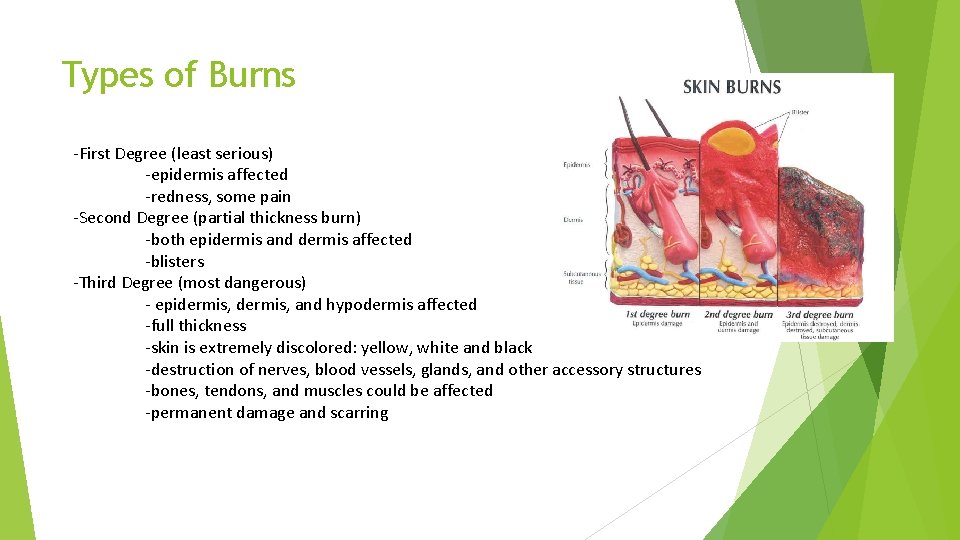
Types of Burns -First Degree (least serious) -epidermis affected -redness, some pain -Second Degree (partial thickness burn) -both epidermis and dermis affected -blisters -Third Degree (most dangerous) - epidermis, and hypodermis affected -full thickness -skin is extremely discolored: yellow, white and black -destruction of nerves, blood vessels, glands, and other accessory structures -bones, tendons, and muscles could be affected -permanent damage and scarring

What Is Artificial Skin? -Skin substitute used to restore epidermal and dermal layers -Can be completely man made or made from using patients cells (biological or synthetic) -Different from skin grafting -Many uses -Most common: serious burns -Some skin diseases -Cosmetic uses

History of Artificial Skin -Before artificial skin, skin grafting performed -Successful with some setbacks -Infection, rejection , dehydration, not enough skin to cover, etc -First successful artificial skin produced by Professor Ioannis Yannis of MIT and Doctor John Burke of Harvard in 1979 -Developed Integra and is FDA approved
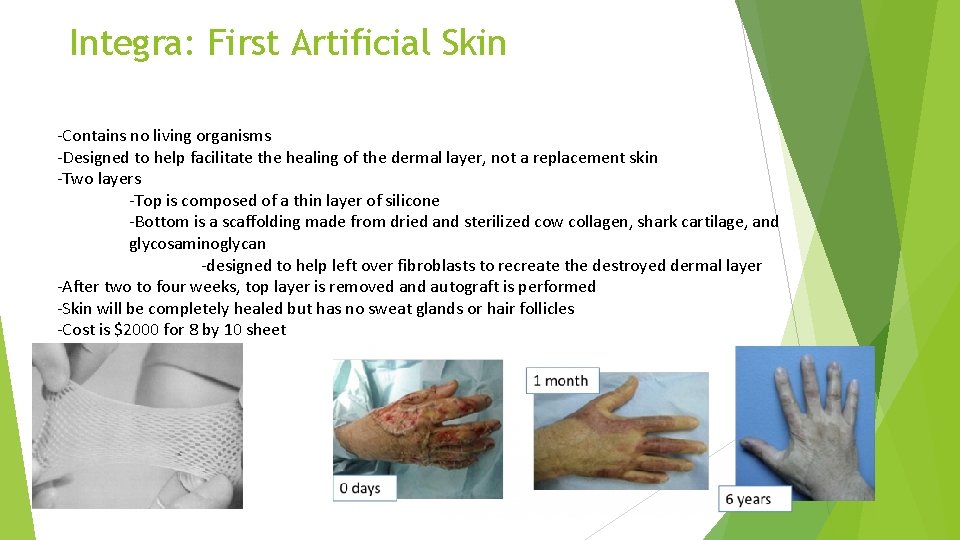
Integra: First Artificial Skin -Contains no living organisms -Designed to help facilitate the healing of the dermal layer, not a replacement skin -Two layers -Top is composed of a thin layer of silicone -Bottom is a scaffolding made from dried and sterilized cow collagen, shark cartilage, and glycosaminoglycan -designed to help left over fibroblasts to recreate the destroyed dermal layer -After two to four weeks, top layer is removed and autograft is performed -Skin will be completely healed but has no sweat glands or hair follicles -Cost is $2000 for 8 by 10 sheet

Dermagraft -Artificial skin can be used to treat -Skin loss -Non-healing wounds -Dermagraft -Bioengineered skin substitute used in treatment of full thickness skin ulcers caused by diabetes -FDA approved - Requirements -Used when ulcers last more than 6 weeks, -do not affect any surrounding structures such as the tendon, muscles, ligaments, and bones

Dermagraft (Continued) -Bioengineered scaffold containing living human fibroblasts -Fibroblasts come donated newborn foreskin tissue -Fibroblasts secrete substances -Designed to help heal dermal layer -Apply 8 times over 12 week period -Sheet of 2 by 3 inches -Costs $1500 per application
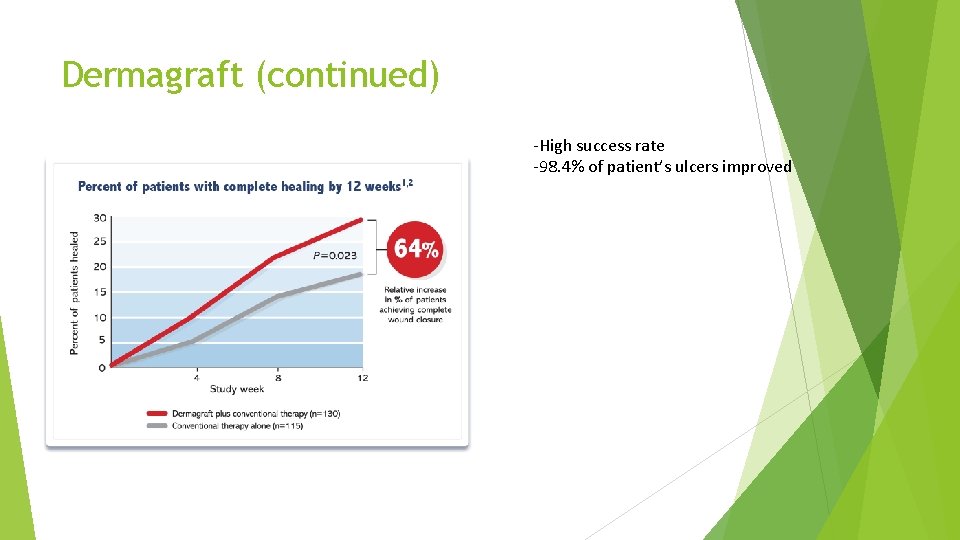
Dermagraft (continued) -High success rate -98. 4% of patient’s ulcers improved

Cosmetic Uses of Artificial Skin -Testing of Comestic products -Eliminate need for animal testing -Greiner Bio-One, European biotechnology company -Designs Thin. Cert Cell Culture Inserts -Thin membrane where human skin can grow

Benefits and Disadvantages -For burns -Better than skin grafting -Increases survival after serious third degree burns -Quicken recovery process -Higher success rate than skin grafting -Less scarring -Can be customized to fit patient’s needs and conditions -For Ulcers -Short recovery period -Less Scarring -Guarantee of success -Can be expensive -Allergic reaction to artificial compound

Future -Artificial skin results are promising -More methods and products being designed to eliminate need for skin grafting and full skin restoration

Works Cited "American Burn Association Burn Treatment Facts. " The University of New Mexico, 2013. Web. 19 Oct. 2013. <http: //hospitals. unm. edu/burn/facts. shtml>. "Artificial Skin. " Advameg, Inc. , 2013. Web. 19 Oct. 2013. <http: //www. discoveriesinmedicine. com/Apg-Ban/Artificial-Skin. html>. "Artificial Skin. " N. p. , n. d. Web. 19 Oct. 2013. <http: //www. shrinershospitalboston. org/blog/understanding-different-types-burns>. "Artificial Skin. " Wikipedia, 17 Oct. 2013. Web. 19 Oct. 2013. <http: //en. wikipedia. org/wiki/Artificial_skin>. Baker, Chris. "New "artificial Skin" Product Launched. " William Reed Business Media, 24 July 2013. Web. 19 Oct. 2013. <http: //www. cosmeticsdesign-europe. com/Formulation- Science/New-artificial-skin-product-launched>. DERMAGRAFT® - P 000036. FDA, 13 Sept. 2013. Web. 19 Oct. 2013. <http: //www. fda. gov/Medical. Devices/Productsand. Medical. Procedures/Device. Approvalsand. Clearances/Recently-Approved. Devices/ucm 085085. htm>. Geldhard, Katie. "Artificial Skin. " Open. Wet. Ware, 18 Jan. 2013. Web. 19 Oct. 2013. <http: //openwetware. org/wiki/Artificial_Skin, _by_Katie_Geldart>. "Greiner Bio-One Launches Artificial Skin to Replace Animal Testing. " Zenopa Ltd, 15 July 2013. Web. 19 Oct. 2013. <http: //www. zenopa. com/news/801612581/greiner-bio-one- launches-artificial-skin-to-replace-animal-testing>. Halim, Ahmad S. , Teng L. Khoo, and Shah J. Yussof. Biologic and Synthetic Skin Substitutes: An Overview (n. d. ): n. pag. Indian Journal of Plastic Surgery. Web. 19 Oct. 2013. <http: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 3038402/>. Shingledecker, Leon. N. p. , n. d. Web. 19 Oct. 2013. <http: //www. drshingledecker. com/wound_care. html>. Shire Regenerative Medicine, In, 2013. Web. 19 Oct. 2013. <http: //www. dermagraft. com/>. "Skin Grafts. " Web. MD LLC, 2013. Web. 19 Oct. 2013. <http: //emedicine. medscape. com/article/1295109 -overview#a 1>. "Understanding Differences Types of Burns. " Shriners Hospitals for Children, 2013. Web. 19 Oct. 2013. <http: //www. shrinershospitalboston. org/blog/understanding-different- types-burns>.
- Slides: 14