ArthropodBorne Viruses Part II Dr Amita Arya Assistant

Arthropod-Borne Viruses (Part II) Dr. Amita Arya Assistant Proffessor. Microbiology.

DENGUE FEVER 2

Man-Arthropod-Man Cycle

History - Dengue • This disease was first described 1780, and the virus was isolated by Sabin 1944. Dengue virus infection is the most common arthropod-borne disease worldwide with an increasing incidence in the tropical regions of Asia, Africa, and Central and South America.

Over view of Dengue • With more than one-third of the world’s population living in areas at risk for transmission, dengue infection is a leading cause of illness and death in the tropics and subtropics. As many as 100 million people are infected yearly. Dengue is caused by any one of four related viruses transmitted by mosquitoes

Dengue • Dengue is the biggest Arbovirus problem in the world today with over 2 million cases per year. Dengue is found in SE Asia, Africa and the Caribbean and S America. • Flavivirus, 4 serotypes, transmitted by Aedes mosquitoes which reside in water-filled containers. • Human infections arise from a human-mosquitoehuman cycle • .

Current Trends • In the 1980 s, DHF began a second expansion into Asia when Sri Lanka, India, and the Maldives Islands had their first major DHF epidemics; Pakistan first reported an epidemic of dengue fever in 1994. .

Dengue Infection and Implications • Dengue virus (DENV) infects 50 million (WHO) to 100 million (NIH) people annually. Forty per cent of the world’s population, predominately in the tropics and sub-tropics, is at risk for contracting dengue virus. DENV infection cause dengue fever, dengue haemorrhagic fever, dengue shock syndrome, and death.
Dengue Mosquito transmitted Viral Infection

What causes Dengue • Dengue (DF) and dengue haemorrhagic fever (DHF) are caused by one of four closely related, but antigenic ally distinct, virus serotypes (DEN-1, DEN-2, DEN-3, and DEN-4), of the genus Flavivirus. Infection with one of these serotypes provides immunity to only that serotype for life,

Aedes aegypti – Vector • Aedes aegypti, a domestic, day-biting mosquito that prefers to feed on humans, is the most common Aedes species. Infections produce a spectrum of clinical illness ranging from a nonspecific viral syndrome to severe and fatal haemorrhagic disease. Other species of Aedes can also transmit.

Dengue Virus – A Flavivirus • Flavivirus are spherical and 40 - 60 mm in diameter. Genome – Positive sense, single sense RNA, 11 kb in size Genome – RNA infectious Enveloped virus Three structural polypeptides two are glycosylated Replication in cytoplasm

How Mosquitos spread the infection • The disease starts during the rainy season, when vector Mosquito Aedes aegypti is abundant • The Aedes breeds in the tropical or semitropical climates in water holding receptacles or in plants close to human dwellings • A female Aedes acquires the infection feeding upon a viremic human. • After a period of 8 – 14 days mosquitoes are infective and remain infective for life. ( 1 - 3 ) months.

Pathogenesis • Presence of existing Dengue antibody, associated with fresh viral infection with new serotype complexes and forms within few days of the second dengue infection. • Non neutralizing enhancing antibodies promote infection of higher number of Mononuclear cells.

Immunology Dengue • Four serotypes exist distinguished by Molecular basis and Nt tests • Infection confers life long immunity • But cross protection between serotypes is of short duration. • Reinfection with different serotype after primary attack is more dangerous causes Dengue hemorrhagic fever.

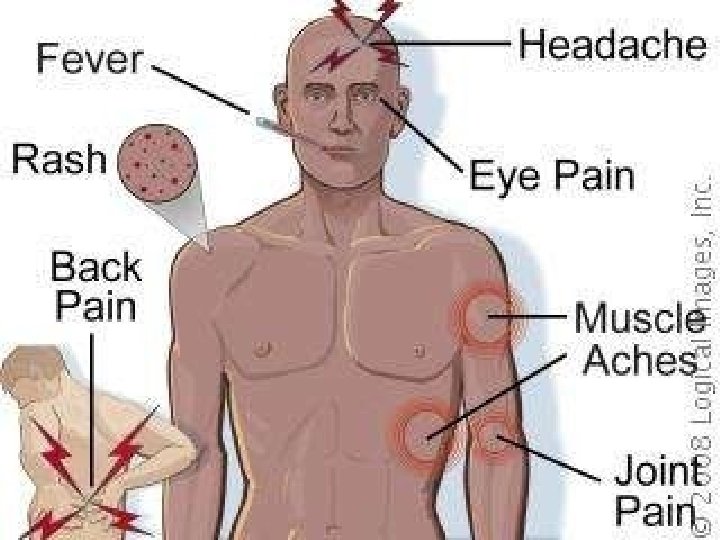
Clinical Manifestations • Any or few of the following events can occur. • Fever, • Severe head ache • Muscle and joint pains • Nausea, vomiting, • Eye pain

How Dengue Infection starts and manifests • Incubation period 4 – 7 days ( 3 – 14 days) • Fever may start with, Malise, chills, head ache • Soon leads to severe back ache, joint pains, muscular pain, pain in the eye ball. • Temperature may persist for 3 -5 days. • On some occasions once again raises in about 5 – 8 days ( Saddle back fever ) • Myalgia may be severe with deep bone pain ( Break bone fever ) characteristic of the Disease On majority of the occasions a self limited condition, Subside on its own Death is a rare event.
Dr. T. V. Rao MD 21
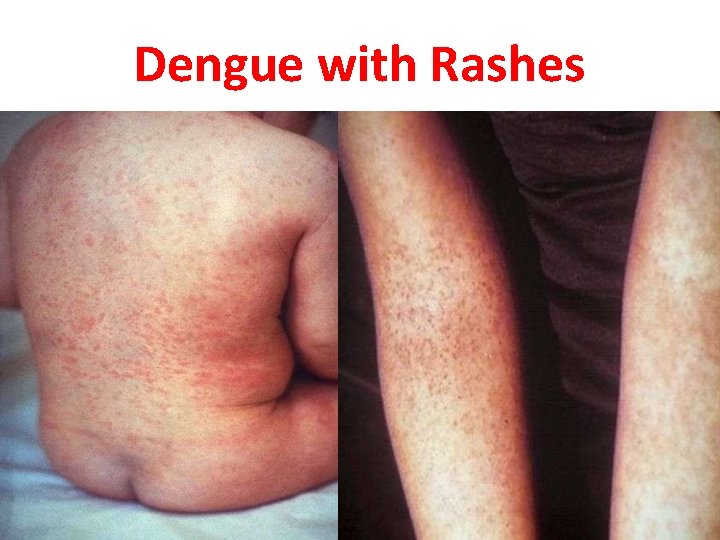
Dengue with Rashes

Dengue Hemorrhagic Fever • DHF was first recognized in the 1950 s during the dengue epidemics in the Philippines and Thailand. Today emerging DHF cases are causing increased dengue epidemics in the Americas, and in Asia, where all four dengue viruses are endemic, DHF has become a leading cause of hospitalization and death among children in several countries. ( WHO )

Dengue Hemorrhagic Fever • Common in children. • In children passively acquired contributed by the maternal antibodies transferred to the fetus. • In other ( Adults ) the presence of antibodies due to previous infection with different serotype • Initially presents like classical Dengue infection • But patients condition abruptly worsens, an important cause of morbidity and mortality in Dengue

Basic Understanding of Dengue Hemorrhagic Fever • Dengue Hemorrhagic Fever is a probable case of dengue and • hemorrhagic tendency evidenced by one or more of the following: • Ø Positive tourniquet test • Ø Petechial, ecchymosis or purpura • Ø Bleeding from mucosa (mostly epistaxis or bleeding from • gums), injection sites or other sites • Ø Haematemesis or melena
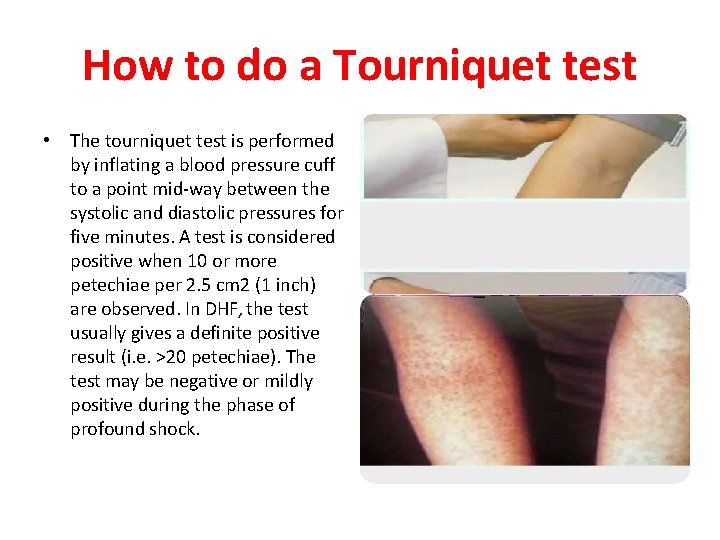
How to do a Tourniquet test • The tourniquet test is performed by inflating a blood pressure cuff to a point mid-way between the systolic and diastolic pressures for five minutes. A test is considered positive when 10 or more petechiae per 2. 5 cm 2 (1 inch) are observed. In DHF, the test usually gives a definite positive result (i. e. >20 petechiae). The test may be negative or mildly positive during the phase of profound shock.

What Happens in Dengue Hemorrhagic Fever • Thrombocytopenia (platelets 100, 000/cu. mm or less) and Ø Evidence of plasma leakage due to increased capillary permeability manifested by one or more of the following: • – A >20% rise in hematocrit for age and sex • – A >20% drop in hematocrit following treatment with • fluids as compared to baseline • – Signs of plasma leakage (pleural effusion, ascites or • hypoproteinaemia).

Risk factor for DHF • Important risk factors for DHF include the strain of the infecting virus, as well as the age, and especially the prior dengue infection history of the patient

Dengue Hemorrhagic Syndrome • Chateresied by shock and hemoconcentration • Contributed by circumstantial evidence suggests secondary infection with Dengue type 2 following type 1 infection in the past. Dr. T. V. Rao MD 29

Dengue hemorraghigic Syndrome • DHS is caused due to release of, 1 Release of cytokines 2 Vasoactive mediators. 3 Procoagulants Manifest with disseminated intravascular coagulation

Diagnosis In resource rich establishments 1 Reverse transcriptase polymerase chain reaction methods help rapid identification 2 Isolation of virus is difficult 3 The current favored approach is inoculation of mosquito cell line with patient serum coupled with nucleic acid assay to identify a recovered virus.

Dengue Serology • The serology is limited with cross reactivity of Ig. G antibodies to heterologous Flavivirus antigens • Most commonly used methods are Viral protein specific capture Ig. M or Ig. G by ELISA Ig. M antibodies develop within few days of illness Neutralizing anti Haemagglutination inhibiting antibodies appear within a week after onset of Dengue fever

Importance of paired sample testing in Serology • Testing one sample for serum and reporting a negative test is fallacious • Analysis of paired acute and convalescent sera to show significant rise in antibody titer is the most reliable evidence of an active dengue infection.
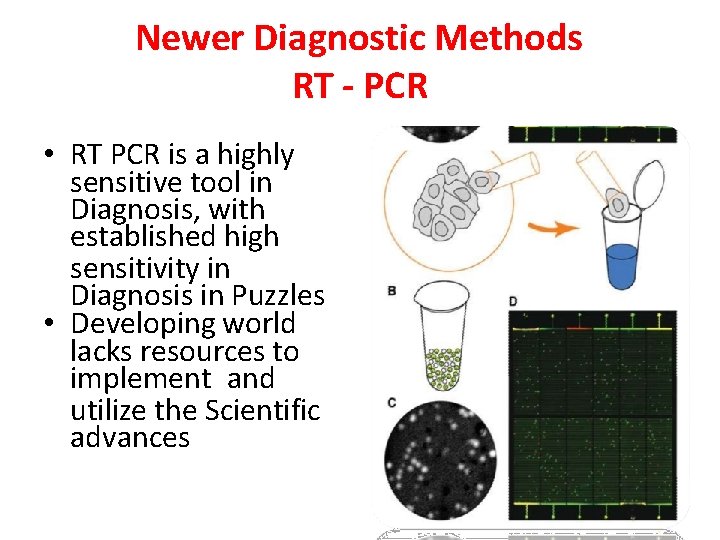
Newer Diagnostic Methods RT - PCR • RT PCR is a highly sensitive tool in Diagnosis, with established high sensitivity in Diagnosis in Puzzles • Developing world lacks resources to implement and utilize the Scientific advances

Treatment • No Anti viral therapy available • Symptomatic management in Majority of cases • Dengue Hemorrhagic fever to be treated with suitable fluid replacement • No Vaccine available, difficult in view of four serotypes.

Control of Dengue • Control of Mosquito breeding places. • Anti mosquito measures • Use of Insecticides. • Screened windows and doors can reduce exposure to vectors.

Epidemiology - Dengue • Dengue virus are distributed world wide in tropical regions. • Where the Aedes vectors exist, are endemic areas • Changing and increasing incidences are associated with rapid urban population growth, over crowding and lax mosquito control measures
Viral Hemorrhagic Fevers

Viral Haemorrhagic Fevers • Acute infection: fever, myalgia, malaise; progression to prostration • Small vessel involvement: increased permeability, cellular damage • Multisystem compromise (varies with pathogen) • Hemorrhage may be small in volume (indicates small vessel involvement, thrombocytopenia) • Poor prognosis associated with: shock, encephalopathy, extensive hemorrhage

Viral Hemorrhagic Fevers • Diverse group of illnesses caused by RNA viruses from 4 families: – Arenaviridae, Bunyaviridae, Filoviridae, Flaviridae – Differ by geographic occurrence and vector/reservoir – Share certain clinical and pathogenic features • Potential for aerosol dissemination, with human infection via respiratory route (except dengue) • Target organ: vascular bed • Mortality 0. 5 - 90%, depending on agent

Viral Hemorrhagic Fever viruses • Filoviruses Ebola Hemorrhagic fever (EHF) Marburg virus • Arenaviruses Lassa fever “New World Arenaviruses” • Bunyaviruses Rift Valley fever (RVF) Crimean Congo Hemorrhagic fever (CCHF)
Viral Hemorrhagic Fevers • Category A agents – Filoviruses – Arenaviruses • Category C agents – Hantaviruses – Tick-borne hemorrhagic fever viruses – Yellow fever

Viral Hemorrhagic Fevers Transmission • Zoonotic diseases – Rodents and arthropods main reservoir – Humans infected via bite of infected arthropod, inhalation of rodent excreta, or contact with infected animal carcasses • Person-to-person transmission possible with several agents – Primarily via blood or bodily fluid exposure – Rare instances of airborne transmission with arenaviruses and filoviruses • Rift Valley fever has potential to infect domestic animals following a biological attack

Viral Hemorrhagic Fevers Clinical Presentation • Clinical manifestations nonspecific, vary by agent • Incubation period 2 -21 days, depending on agent • Onset typically abrupt with filoviruses, flaviviruses, and Rift Valley fever • Onset more insidious with arenaviruses

Viral Hemorrhagic Fevers Initial Symptoms Prodromal illness lasting < 1 week may include: – – – High fever Headache Malaise Weakness Exhaustion – – – Dizziness Muscle aches Joint pain Nausea Non-bloody diarrhea

VHF Surveillance: Clinical Identification of Suspected Cases • Clinical criteria: – Temperature 101 F(38. 3 C) for <3 weeks – Severe illness and no predisposing factors for hemorrhagic manifestations – 2 or more of the following: • • Hemorrhagic or purple rash Epistaxis Hematemesis Hemoptysis Blood in stools Other hemorrhagic symptoms No established alternative diagnosis Dr. T. V. Rao MD JAMA 2002; 287 Adapted from WHO 46

Viral Hemorrhagic Fevers Treatment • Supportive care • Correct coagulopathies as needed • No antiplatelet drugs or IM injections • Investigational treatments, available under protocol: – Ribavirin x 10 days for arenaviridae and bunyaviridae – Convalescent plasma w/in 8 d of onset for AHF

Viral Hemorrhagic Fevers Management of Exposed Persons • Medical surveillance for all potentially exposed persons, close contacts, and high-risk contacts (i. e. , mucous membrane or percutaneous exposure) x 21 days – Report hemorrhagic symptoms (slide 47) – Record fever 2 x/day • Report temperatures 101 F(38. 3 C) � Initiate presumptive ribavirin therapy • Percutaneous/mucocutaneous exposure to blood or body fluids of infected: – Wash thoroughly with soap and water, irrigate mucous membranes with water or saline

Viral Hemorrhagic Fevers Infection Control • Airborne & contact precautions for health care, environmental, and laboratory workers • Negative pressure room, if available – 6 -12 air changes/hour – Exhausted outdoors or through HEPA filter • Personal protective equipment – Double gloves – Impermeable gowns, leg and shoe coverings – Face shields and eye protection – N-95 mask or PAPR

Tick Borne Hemorrhagic Fevers • Kyasanur Forest Disease, ( Karnataka India ) • Like Russian Spring Summer Encephalitis, • Present with Fever, Headache, Conjunctivitis, Myalgia, Severe prostration,

Viral Hemorrhagic Fevers Infection Control • Dedicated medical equipment for patients • If available, point-of-care analyzers for routine laboratory analyses – – If unavailable, pretreat serum w/Triton X-100 Lab samples double-bagged & hand-carried to lab • Prompt burial or cremation of deceased with minimal handling – Autopsies performed only by trained personnel with PPE

Viral Hemorrhagic Fevers Summary of Key Points • A thorough travel and exposure history is key to distinguishing naturally occurring from intentional viral hemorrhagic fever cases. • Viral hemorrhagic fevers can be transmitted via exposure to blood and bodily fluids.

Pathogenesis. • Enters through the bite of Insect vector, • Multiply in RES. • Target the organ CNS Encephalitis, Liver Yellow fever, Capillary endothelium in Hemorrhagic fevers.

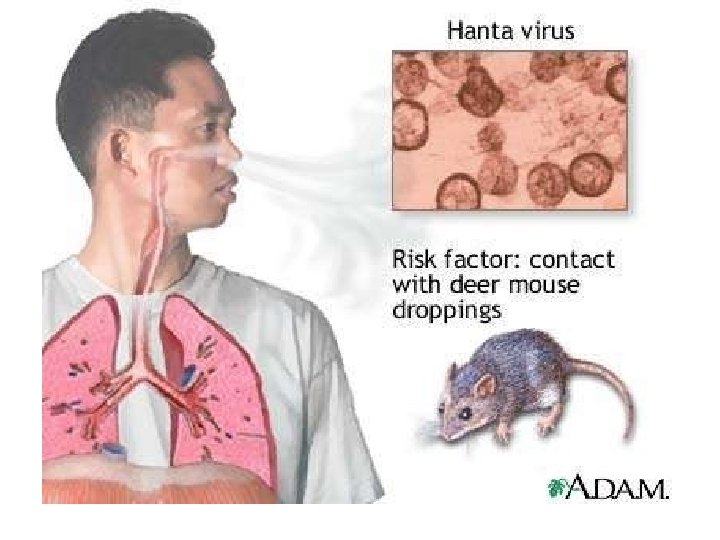
Rodent Borne Hemorrhagic Fevers, • Hanta Virus, Produces pulmonary infections in USA • Belong to Bunya Virus – Hanta Viruses

Hanta Viruses, • Human disease Hemorrhagic fever with renal syndrome • Hanta virus pulmonary syndrome. • Spread by inhalation of Aerosols of Rodent Excreta, • Renal Involvement and failure • Lead to Hemorrhagic shock, Korea • Spread by Rats carried in ships,

Laboratory Diagnosis • Detection of viral nucleic acid, • Grown in culture lines, • PCR,

Filoviruses, African Hemorrhagic Fevers. • Most important Diseases are Marburg and Ebola. • The nature of Viruses are 80 nm Filamentous threads, • Produce Internal and external Bleeding.

Filoviruses. Marburg • Marburg 1967 African Green Monkey, • Bat – Rodent – Host Human. • East Africa Monkey – Humans.

Filoviruses - Ebola • • Incubation 2 -21 days Carries 80% mortality. Barrier Nursing Most essential. ELISA test Culturing Hazardous. RT-PCR Transporting and carrying Primates is Hazardous
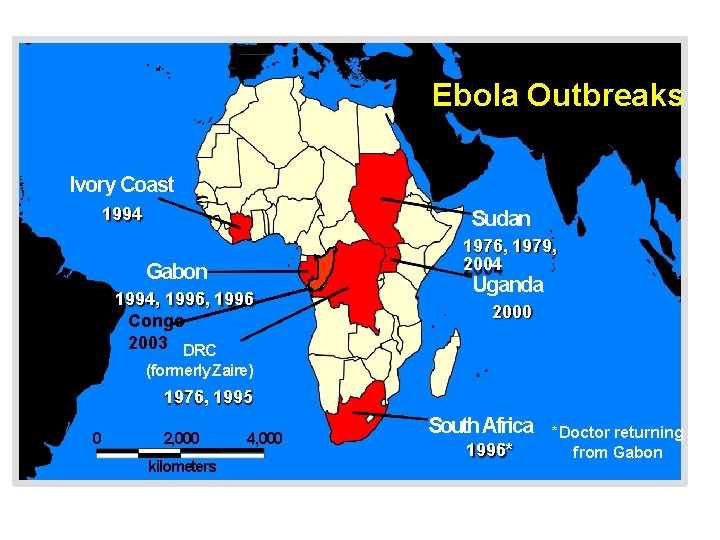
Ebola Outbreaks 1979, 2004 Ivory Coast 1994 Sudan 1976, 1979, 2004 Gabon 1994, 1996 Congo 2003 DRC Uganda 2000 (formerly Zaire) 1976, 1995 0 2, 000 kilometers 4, 000 South Africa 1996* *Doctor returning from Gabon
Bunya viruses • Rift Valley fever • Crimean Congo hemorrhagic fever
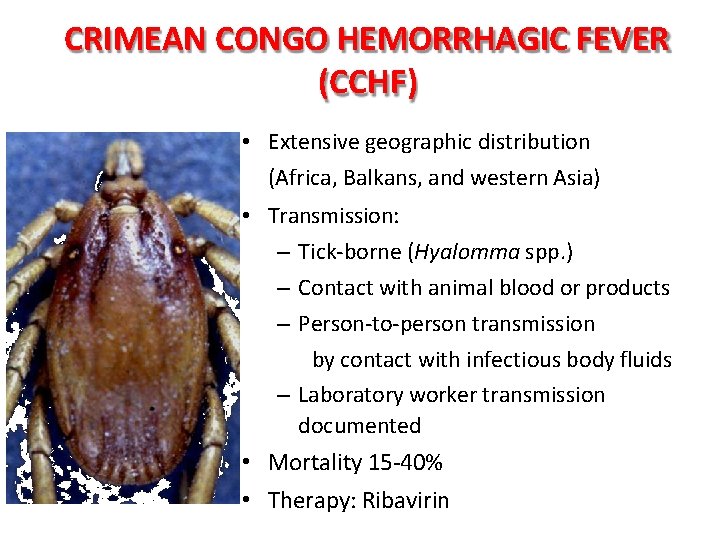
CRIMEAN CONGO HEMORRHAGIC FEVER (CCHF) • Extensive geographic distribution (Africa, Balkans, and western Asia) • Transmission: – Tick-borne (Hyalomma spp. ) – Contact with animal blood or products – Person-to-person transmission by contact with infectious body fluids – Laboratory worker transmission documented • Mortality 15 -40% • Therapy: Ribavirin

CCHF: Pathogenesis • Viremia present throughout disease • IFA becomes positive in patients destined to survive days 4 -6, often simultaneously with viremia • Recovery may be due to CMI or neutralizing antibodies • Patients that die usually still viremic • Virus grows in macrophages and other cells • DIC often present • Poor prognosis signaled by early elevated AST and clotting

CCHF: Clinical features • 4 -12 day incubation after tick exposure • 2 -7 day incubation after direct contact with infected fluids • Abrupt onset fever, chills, myalgia, severe headache • Malaise, GI symptoms, anorexia • Leukopenia, thrombocytopenia, hemoconcentration, proteinuria, elevated AST • Hemorrhages may be profuse (hematomas, ecchymoses)

PREVENTION OF CCHF • DEET repellents for skin • Permethrin repellents for clothing – (0. 5% permethrin should be applied to clothing ONLY) • Check for and remove ticks at least twice daily. • If a tick attaches, do not injure or rupture the tick. Remove ticks by grasping mouthparts at the skin surface using forceps and apply steady traction.

Stay Healthy & Safe. THANK YOU.
- Slides: 65