Arthropodborne Viruses Arthropodborne viruses arboviruses are viruses that

Arthropod-borne Viruses Arthropod-borne viruses (arboviruses) are viruses that can be transmitted to man by arthropod vectors. The WHO definition is as follows “Viruses maintained in nature principally, or to an important extent through biological transmission between susceptible vertebrate hosts by haematophagus arthropods or through transovarian and possibly venereal transmission in arthropods. ”

General properties Haemagglutination with fowl RBC’s n Growth in – Chick embryo fibroblasts, vero cells , mosquito cell lines n Inactivated by lipid solvents & Room Temp n All species are capable of producing encephalitis in suckling mice after intracerebral inoculation n

Current taxonomic status of some arboviruses Ш Togaviridae Genus Alphavirus Ш Flaviviridae Genus Flavivirus Ш Bunyaviridae Genus Bunyavirus Ш Reoviridae Genus Orbivirus Ш Rhabdoviridae Genus Vesiculovirus Approximately 80 arboviruses known to cause human Ш Arenaviridae Genus disease Arenavirus

Arthropod Vectors Mosquitoes Japanese encephalitis, dengue, yellow fever, St. Louis encephalitis, EEE, WEE, VEE etc. Ticks Crimean-Congo haemorrhagic fever, various tickborne encephalitides etc. Sandflies Sicilian sandfly fever, Rift valley fever.

Examples of Arthropod Vectors Aedes Aegyti Culex Mosquito Assorted Ticks Phlebotmine Sandfly

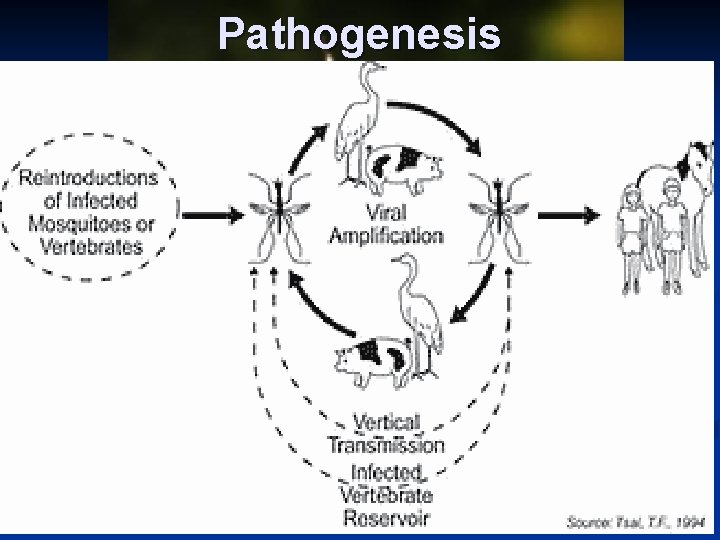
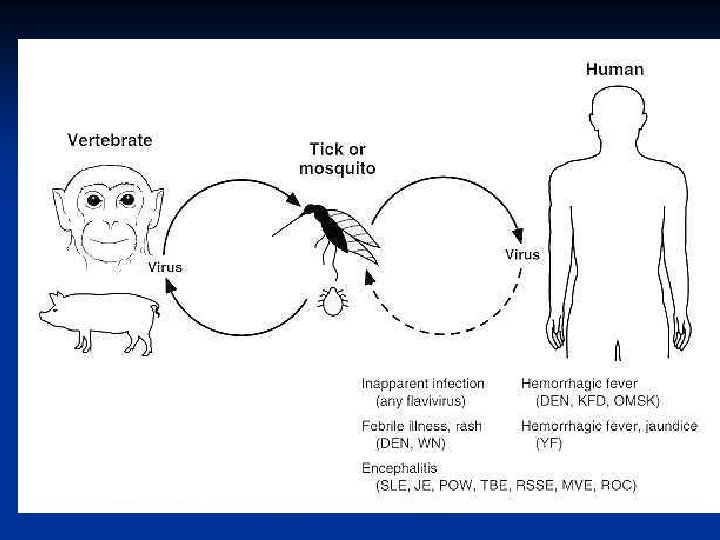
Transmission Cycles n n n Man - arthropod - man n e. g. dengue, urban yellow fever. n Reservoir may be in either man or arthropod vector. n In the latter transovarial transmission may take place. Animal - arthropod vector - man n e. g. Japanese encephalitis, EEE, WEE, jungle yellow fever. n The reservoir is in an animal. n The virus is maintained in nature in a transmission cycle involving the arthropod vector and animal. Man becomes infected incidentally. Both cycles may be seen with some arboviruses such as yellow fever.

Animal Reservoirs In many cases, the actual reservoir is not known. The following animals are implicated as reservoirs n. Birds Japanese encephalitis, St Louis encephalitis, EEE, WEE n. Pigs Japanese encephalitis n. Monkeys Yellow Fever n. Rodents VEE, Russian Spring. Summer encephalitis
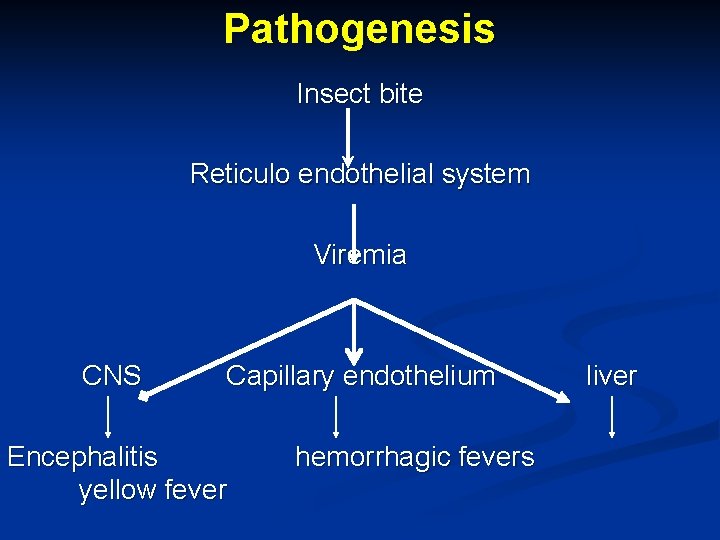
Pathogenesis Insect bite Reticulo endothelial system Viremia CNS Capillary endothelium Encephalitis yellow fever hemorrhagic fevers liver

TOGAVIRUSES Spherical enveloped viruses n 50 – 70 nm diameter n ss RNA n Alpha virus n Rubivirus – rubella virus n
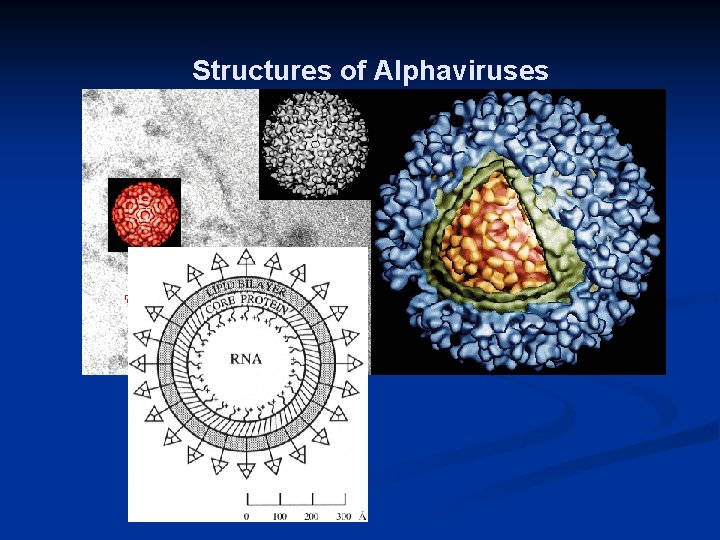
Structures of Alphaviruses

Principal medically important alphaviruses Virus Antigenic Clinical Syndrome Vector Host Distribution Eastern Encephalitis equine (EEE) encephalitis Mosquito Birds Americas Western equine encephalitis Encephalitis (WEE) Mosquito Birds North America Venezuelan Febrile equine illness, encephalitis (VEE) Mosquito Rodents, horses Americas
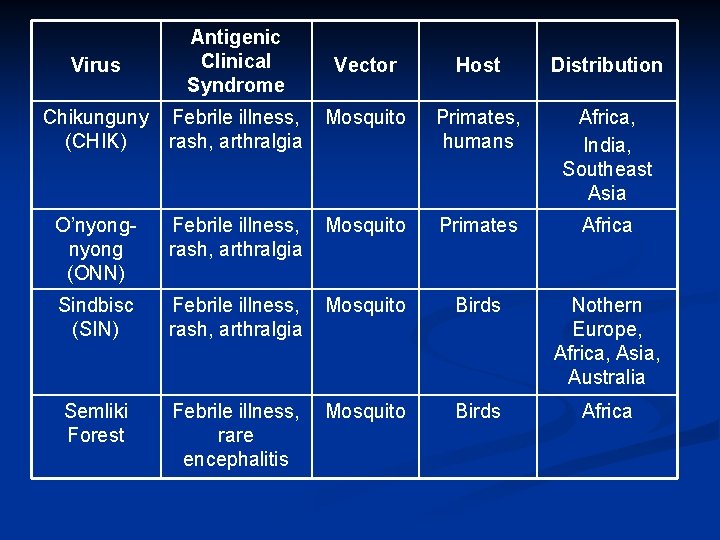
Virus Antigenic Clinical Syndrome Vector Chikunguny Febrile illness, Mosquito (CHIK) rash, arthralgia Host Distribution Primates, humans Africa, India, Southeast Asia O’nyong (ONN) Febrile illness, Mosquito rash, arthralgia Primates Africa Sindbisc (SIN) Febrile illness, Mosquito rash, arthralgia Birds Nothern Europe, Africa, Asia, Australia Semliki Forest Febrile illness, Mosquito rare encephalitis Birds Africa
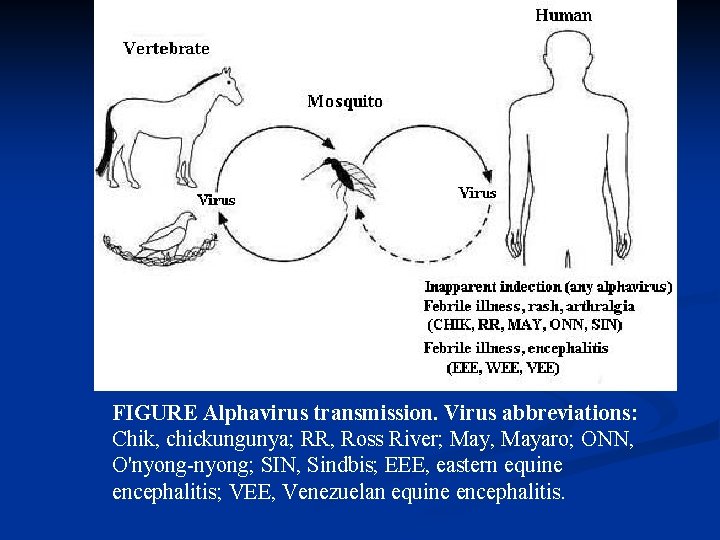
FIGURE Alphavirus transmission. Virus abbreviations: Chik, chickungunya; RR, Ross River; May, Mayaro; ONN, O'nyong-nyong; SIN, Sindbis; EEE, eastern equine encephalitis; VEE, Venezuelan equine encephalitis.

Pathogenesis of alphaviruses

Symptoms : EEE n Most people have no symptoms n Central Nervous system symptoms develop 4 -10 days after being bitten n Sudden onset of fever, muscle aches, headache n May progress to more severe symptoms such as seizure and coma (encephalitis) n 30 to 50% of patients with

CHIKUNGUNYA Aedes aegypti n Fever, Sever joint pains, lymphadenopathy, conjunctivitis, rash n Remission after 1 -6 days n Hemorrhagic manifestations in few n No animal reservoir, no vaccine n

FLAVIVIRIDAE Spherical, 40 -50 nm n Single stranded RNA ENCEPHALITIS VIRUSES n St. Louis encephalitis virus n Ilheus virus n West Nile virus n Murry valley encephalitis virus n Japanese B encephalitis virus n
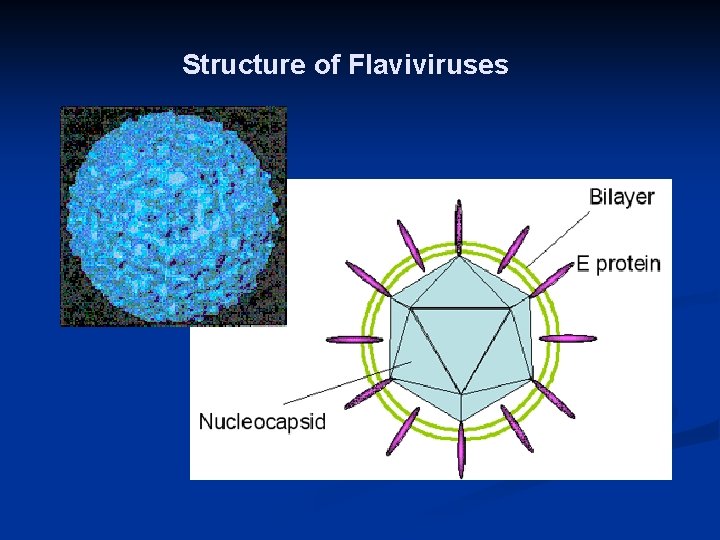
Structure of Flaviviruses
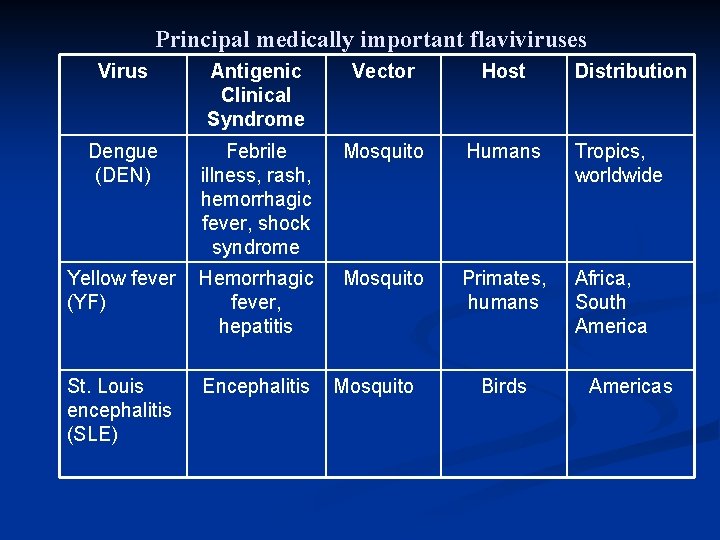
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Dengue (DEN) Febrile illness, rash, hemorrhagic fever, shock syndrome Mosquito Humans Tropics, worldwide Yellow fever Hemorrhagic (YF) fever, hepatitis Mosquito Primates, humans Africa, South America St. Louis encephalitis (SLE) Encephalitis Mosquito Birds Distribution Americas
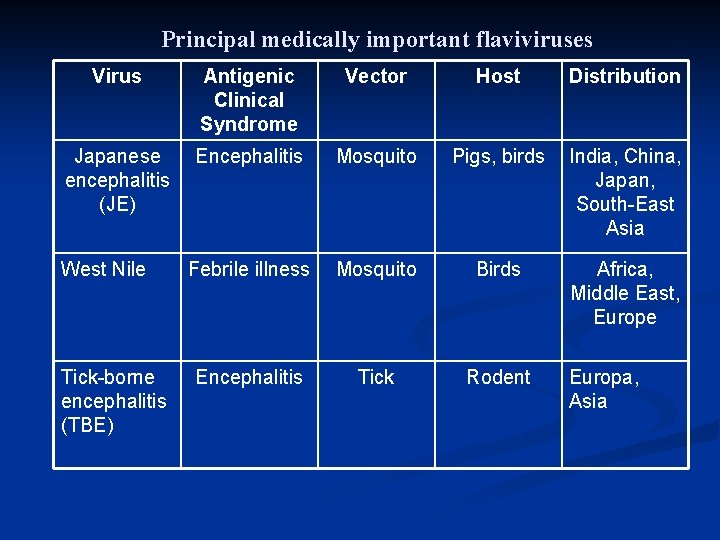
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Distribution Japanese Encephalitis encephalitis (JE) Mosquito Pigs, birds India, China, Japan, South-East Asia West Nile Febrile illness Mosquito Birds Africa, Middle East, Europe Encephalitis Tick Rodent Tick-borne encephalitis (TBE) Europa, Asia
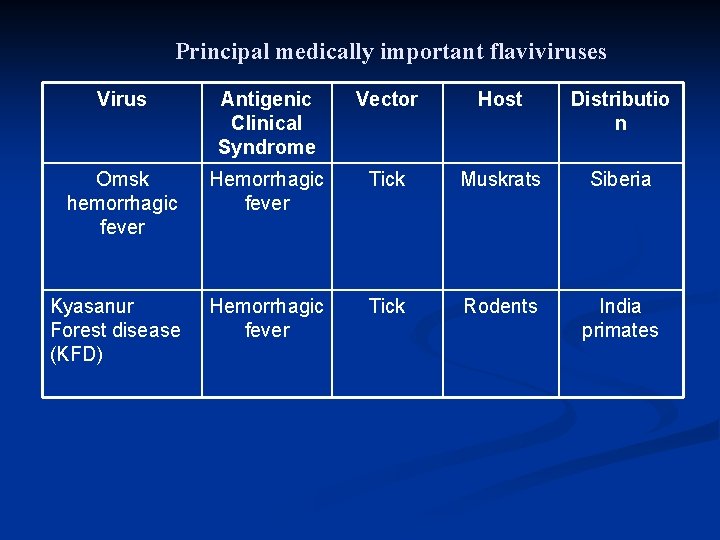
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Distributio n Omsk hemorrhagic fever Hemorrhagic fever Tick Muskrats Siberia Kyasanur Forest disease (KFD) Hemorrhagic fever Tick Rodents India primates

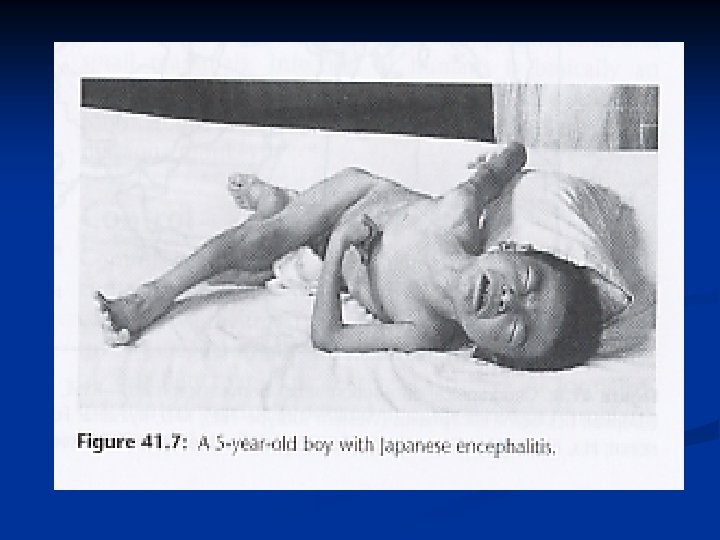
Japanese B Encephalitis n First discovered and originally restricted to Japan. Now large scale epidemics occur in China, India and other parts of Asia. n Transmitted by culex mosquitoes. n The virus is maintained in nature in a transmission cycle involving mosquitoes, birds and pigs. n Most human infections are subclinical: the inapparent to clinical cases is 300: 1
Pathogenesis Culex mosquito biting human.

Pathogenesis: n Natural infections in birds (Herons) – Reservoirs n Pigs – amplifier hosts n Other birds, cattle & buffaloes - reservoirs Epidemiology: n China, Japan & south east asia n India – Eastern & southern parts n Karnataka - Kolar

AREA OF HIGH OCCURRENCE IN INDIA The three southern states of Tamil Nadu (TN), Andra Pradesh, Karnataka were reporting higher incidence. n JE is emerging as a public health problem in Kerala n In a few villages of Cuddalore district of Tamil Nadu, a known JE-endemic area (Chidambaram, Virudhachalam, Thittakudi) n


JE clinical features n A Prodromal Stage n An Acute encephalitic Stage n and a late stage

Prodromal Stage Fever n Rigors n Headache n Nausea n and Vomiting n The Prodromal stage usually lasts for 1 to 6 days. It can be as short as less than 24 hours or as long as 14 days n

An Acute Encephalitic Stage: 3 rd – 5 th day Convulsions n Altered sensorium, unconsciousness, coma n Mask like face n Stiff Neck n Muscular Rigidity n Tremors in fingers, tongue, eyelids and eyes. n Abnormal movements of limbs n Speech impairment n

Late Stage: n n n the persistence of signs of CNS injury such as, Mental impairment. Increased deep Tendon reflexes Paresis either of the upper or lower motor neuron type. speech impairment Epilepsy, Abnormal movements, Behaviour abnormalities.

Differential Diagnosis Cerebral Malaria n Meningitis n Febrile Convulsions n Rey’s Syndrome n Rabies n Toxic Encephalopathy n

LAB DIAGNOSIS Peripheral blood picture shows moderate peripheral leucocytosis with neurophilia and mild anemia. n CSF: (cerbro spinal fluid) analysis: Neutrophils may predominate in early CSF samples but a lymphocytic pleocytosis is typical. n CSF protein is moderately elevated in about 50% of cases. n

Serological tests These are to detect antibodies to viral antigens, which include the plaque reduction virus neutralization test, hemagglutination inhibition, and complement fixation. . n A significant rise in titer should be seen with paired samples from the acute and convalescent stages. n

Culture The virus is isolated from CSF by inoculating into 2 -4 day old mice and the virus is identified by haemagglutination inhibition. n Japanese encephalitis virus may also be identified by infection of cell cultures (chicken embryo or hamster kidney cells, or the mosquito cell line C 3/36) and by IFA. n

Prophylaxis Formalin inactivated mouse brain vaccine n Nakayama strain n Two doses with 2 weeks interval and 1 booster after 12 months n

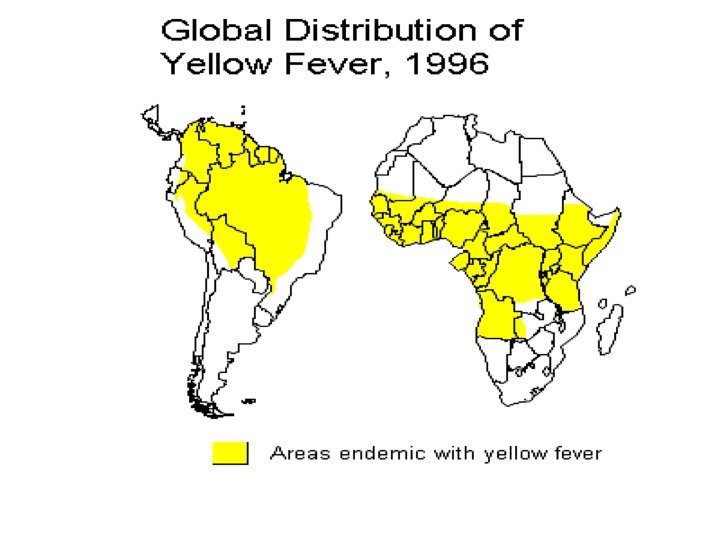
Yellow Fever n Flavivirus, mainly found in West Africa and S America n Yellow fever occurs in 2 major forms: urban and jungle (sylvatic) yellow fever. Jungle YF is the natural reservoir of the disease in a cycle involving nonhuman primates and forest mosquitoes. Man may become incidentally infected on venturing into jungle areas.

n The urban form is transmitted between humans by the Aedes aegypti mosquito n Classically Yellow Fever presents with chills, fever, and headache. Generalized myalgias and GI complaints n Some patients may experience an asymptomatic infection or a mild undifferentiated febrile illness.

Yellow Fever n After a period of 3 to 4 days, the more severely ill patients with a classical YF course will develop bradycardia (Faget's sign), jaundice, and haemorrhagic manifestations. n 50% of patients with frank YF will develop fatal disease characterized by severe haemorrhagic manifestations, oliguria and hypotension.

n Diagnosis is usually made by serology n There is no specific antiviral treatment n An effective live attenuated vaccine is available against yellow fever and is used for persons living in or traveling to endemic areas.

Vaccine, Immunization Schedule, Immunogenicity and Protection n Yellow fever vaccine is a live attenuated vaccine, the two vaccine substrains used for production (17 D-204 and 17 DD) are derived from the 17 D strain n The vaccine is produced in embryonated eggs n Immunization schedule: administration of 1 dose for persons 9 months n Seroconversion rate: 95% n A single dose provides protection for at least 10 years, probably for live

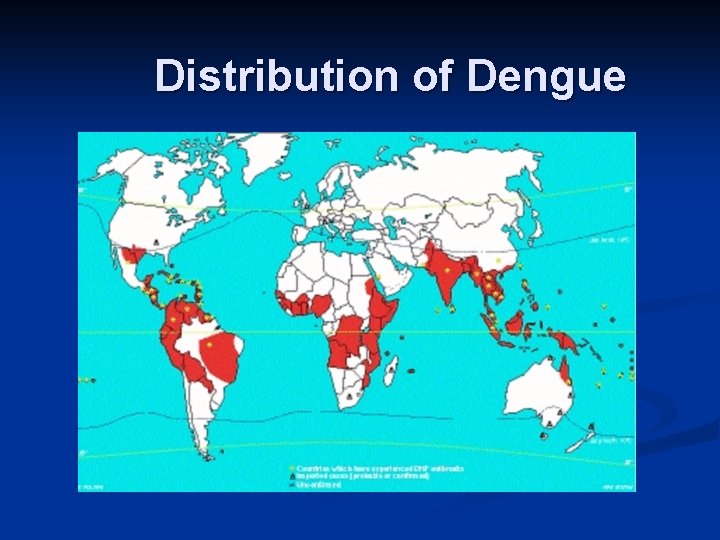
Dengue n Dengue is the biggest arbovirus problem in the world today with over 2 million cases per year. Dengue is found in SE Asia, Africa and the Caribbean and S America. n Flavivirus, 4 serotypes, transmitted by Aedes mosquitoes which reside in water-filled containers. n Human infections arise from a humanmosquitoe-human cycle

n Classically, dengue presents with a high fever, lymphadenopathy, myalgia, bone and joint pains, headache, and a maculopapular rash. n Severe cases may present with haemorrhagic fever and shock with a mortality of 5 -10%. (Dengue haemorrhagic fever or Dengue shock syndrome. )
Distribution of Dengue

Dengue n Dengue haemorrhagic fever and shock syndrome appear most often in patients previously infected by a different serotype of dengue, thus suggesting an immunopathological mechanism. n Diagnosis is made by serology. n No specific antiviral therapy is available.

n Prevention of dengue in endemic areas depends on mosquito eradication. The population should remove all containers from their premises which may serve as vessels for egg deposition. n A live attenuated vaccine is being tried in Thailand with encouraging results.

Transmitting WNV infection

Symptoms : West Nile virus Most people do not develop symptoms n An estimated 20% become ill 3 -15 days after being bitten n. Mild illness: fever, headache, body aches, and sometimes skin rash and swollen glands n An estimated 1 in 150 persons infected develop a more severe form of the disease n. West Nile encephalitis: inflammation of the brain, high fever, stiff neck, stupor, disorientation, coma, tremors, convulsions, muscle weakness, and paralysis; few cases have been fatal n

Russian spring summer encepahlitis (Louping Ill) Central & Eastern Europe, Russia n Vector: Ixodid ticks - Tranovarial transmission n Resrvoir: Wild rodents & birds n Hosts: Sheep, Goats & Humans n Transmitted by Tick bites or ingestion of milk of infected goats n Vaccine: Formalin inactivated vaccine n

KYASANUR FORST DISEASE Tick borne haemorrhagic fever n 1957 – Fatal Epidemic in monkeys & vilagers of Kyasnur forest in Shimoga district, Karnataka n Virus related to RSSE n Vector – Haemaphysalis spinigera; transovarial transmission present n Reservoir – Birds & small mammals n Monkeys – amplifier hosts n

Haemaphysalis spinigera

IP – 3 – 7 days n Fever, headache, vomiting, myalgia, conjunctivitis n Massive internal hemorrhages n Case fatality – 5% n Vaccine – Killed KFD vaccine n Prevention – avoiding tick bites n

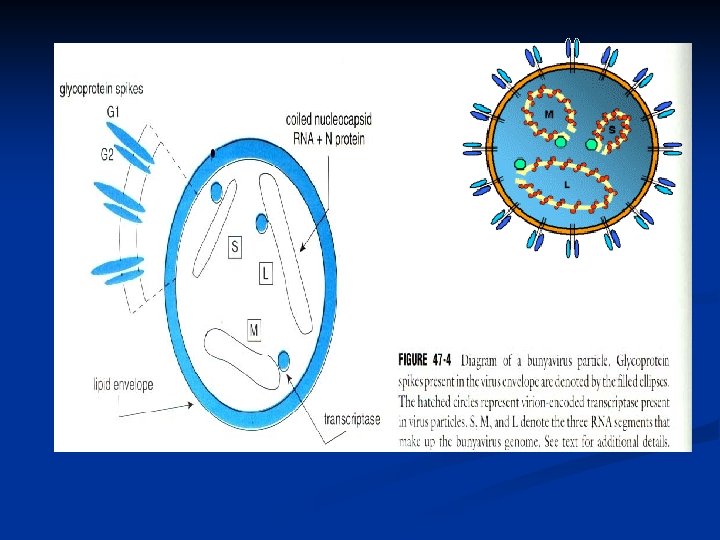
Bunyaviridae • Bunyaviridae is a family of arthropodborne or rodent-borne, spherical, enveloped RNA viruses. • Bunyaviruses are responsible for a number of febrile diseases in humans and other vertebrates. • They have either a rodent host or an arthropod vector and a vertebrate host.

Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distributio n Fever Mosquito Africa Bunyavirus Bunyamwera Bwamba Fever , Rash Mosquito Africa California encephalitis Enceph a-litis Mosquito North America Simbu Shuni Fever Mosquito Africa, Asia

Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Phlebovirus fever Alenquer Fever Unknown South America Naples Fever Sand fly Europe, Asia, Africa Rift Valley Fever, encephalitis, hemorrhagic fever, blindness Mosquito Africa Sicilian Fever Sand fly Europe, Africa, Asia

Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Nairovirus Crimean. Congo Nairobi sheep disease Crimean. Congo hemorrhagic fever Hemorrhagic fever Tick Nairobi sheep disease Fever Tick Africa, Asia
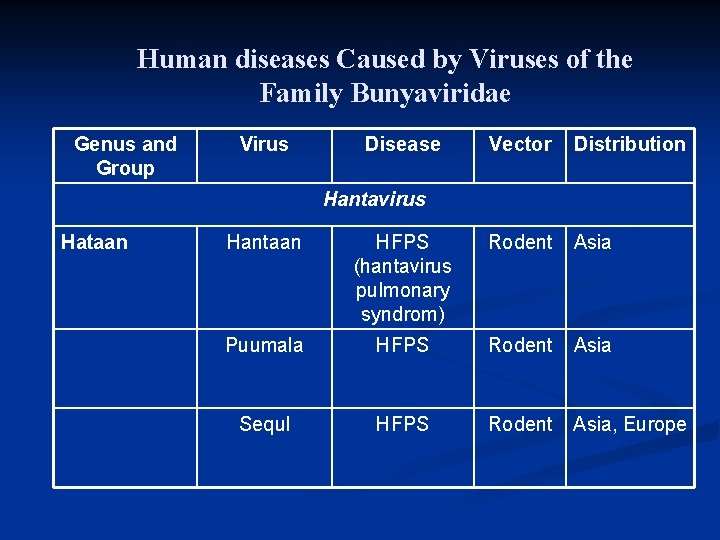
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Hantavirus Hataan Hantaan HFPS (hantavirus pulmonary syndrom) Rodent Asia Puumala HFPS Rodent Asia Sequl HFPS Rodent Asia, Europe

Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distributi on Genus unassigned Bangui Fever, rash Unknown Africa Bhanja Fever, encephalitis Tick Africa, Europa, Asia Issk-kul Fever Tick Asia Kasokero Fever Unknown Africa Nyando Fever Mosquito Africa Tataguine Fever Mosquito Africa Wanowrie Fever, hemorrhage Tick Middle East, Asia
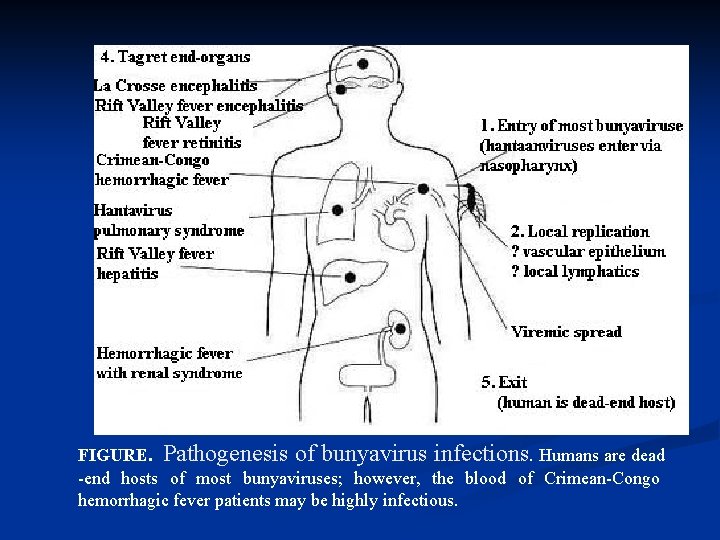
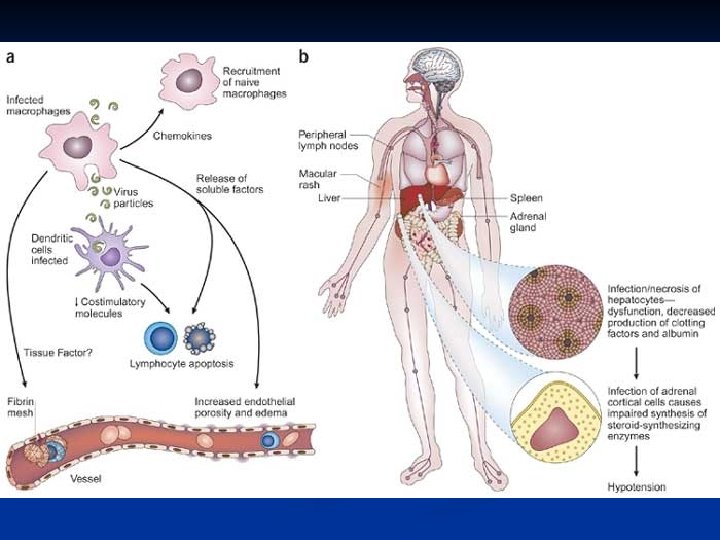
FIGURE. Pathogenesis of bunyavirus infections. Humans are dead -end hosts of most bunyaviruses; however, the blood of Crimean-Congo hemorrhagic fever patients may be highly infectious.
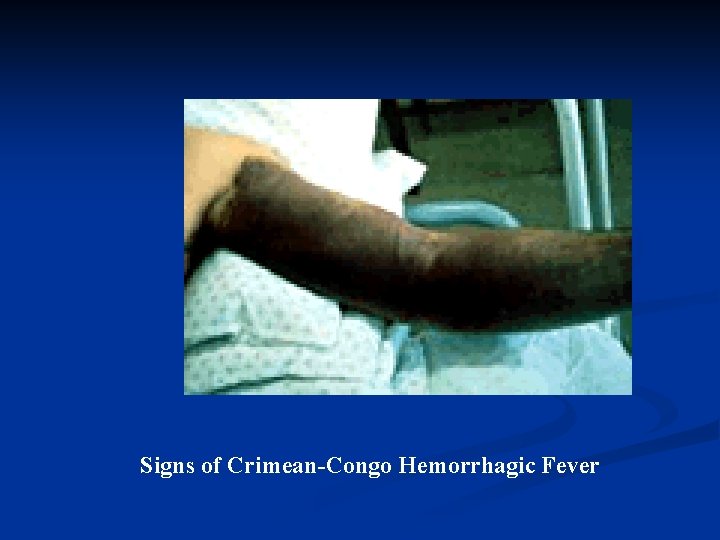
Signs of Crimean-Congo Hemorrhagic Fever
Hantaviruses n n n Forms a separate genus in the Bunyavirus family. Unlike under bunyaviridae, its transmission does not involve an arthropod vector. Enveloped ss. RNA virus. Virions 98 nm in diameter with a characteristic square grid-like structure. Genome consists of three RNA segments: L, M, and S.

Rodent Carriers of Hantaviruses Stripped field mouse (Apodemus agrarius) Bank vole (Clethrionomys glareolus) Deer Mouse (Peromyscus maniculatus) Rat (Rattus)

Clinical Features of Hantavirus Disease n n The multisystem pathology of HVD is characterized by damage to capillaries and small vessel walls, resulting in vasodilation and congestion with hemorrhages. Classically, hantavirus disease consists of 5 distinct phases. These phases may be blurred in moderate or mild cases. n n n Febrile phase Hypotensive phase Oliguric phase Diuretic phase Convalescent phase

Hantavirus Pulmonary Syndrome (HPS) n In common with classical HVD, HPS has a similar febrile phase. n However, the damage to the capillaries occur predominantly in the lungs rather than the kidney. n Shock and cardiac complications may lead to death. n The majority of HPS cases are caused by the Sin Nombre virus. The other cases are associated with a variety of other hantaviruses e. g. New York and Black Creek Canal viruses.

Diagnosis n Serological diagnosis - IF, HAI, ELISAs n Direct detection of antigen - The virus antigen can be demonstrated in the blood or urine. n RT-PCR - found to of great use in diagnosing hantavirus pulmonary syndrome. n Virus isolation - isolation of the virus from urine is successful early in hantavirus disease. Isolation of the virus from the blood is less consistent. Sin Nombre virus has never been isolated from patients with HPS. n Immunohistochemistry - useful in diagnosing HPS.

Treatment and Prevention n Treatment of HVD and HPS depends mainly on supportive measures. n Ribavirin - reported to be useful if given early in the course of hantavirus disease. Its efficacy is uncertain in hantavirus pulmonary syndrome. n Vaccination - an inactivated vaccine is being tried out in China. Other candidate vaccines are being prepared. n Rodent Control - control measures should be aimed at reducing contact between humans and rodents.

REOVIRIDAE Orthoreovirus n Coltivirus n Orbivirus –Colorado tick fever n Rotavirus n Double stranded DNA n

RHABDOVIRIDAE Vesiculovirus - Chandipura virus - Nagpur n Sandflies & Aedes mosquitoes n
- Slides: 72