Artery White blood cells Platelets Red blood cells
Artery White blood cells Platelets Red blood cells

Major function is to transport nutrients, gases and hormones to the cells and pick up wastes from cells to transport them to areas of body where they are excreted

Functions Of Blood • Transportation - the blood transports dissolved gases, nutrients, hormones and metabolic wastes. • Protection - the blood restricts fluid losses through damaged vessels. Platelets in the blood and clotting proteins minimize blood loss when a blood vessel is damaged. • Phagocytosis and antibody production • Regulation – Blood regulates the p. H and electrolyte composition of the interstitial fluids. – Blood regulates body temperature.
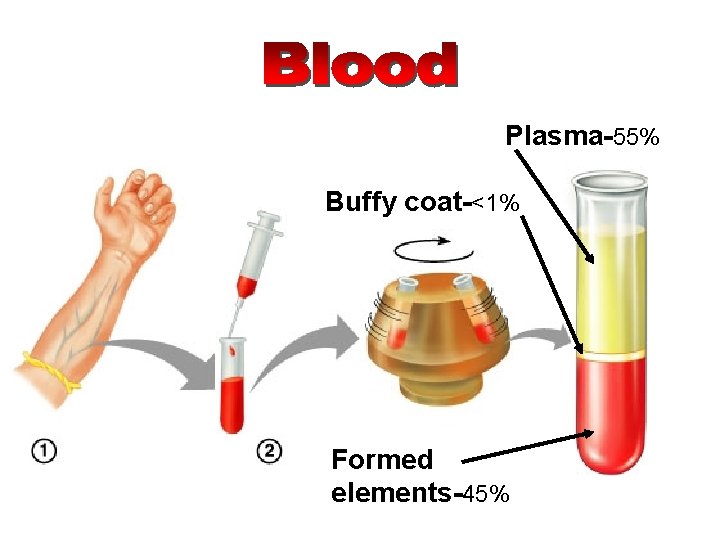
Plasma-55% Buffy coat-<1% Formed elements-45%

Blood Plasma • Straw-colored, sticky fluid portion of blood • Approximately 90% water • Contains: – Ions – Na+ and Cl– Nutrients – sugars, amino acids, lipids, cholesterol, vitamins and trace elements – Three main proteins - Albumin (60%), globulin (35%), fibrinogen (4%) – Dissolved Gasses – including O 2 and CO 2 – Waste Products – other protein wastes such as urea and bilirubin
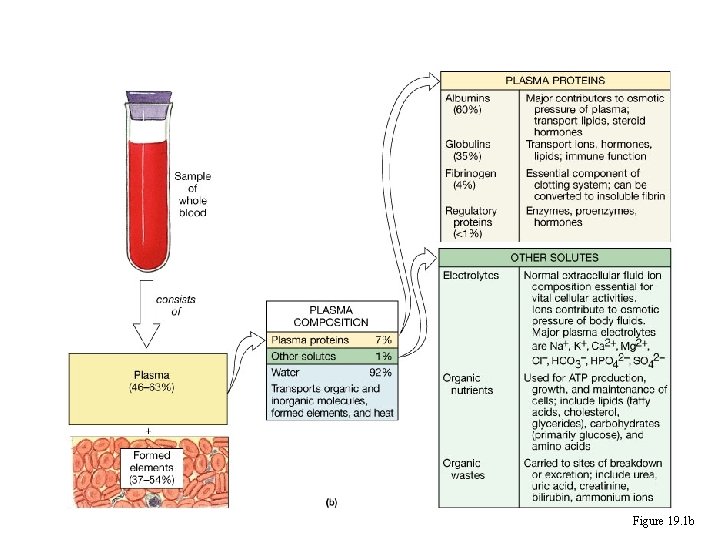
Composition of Whole Blood Figure 19. 1 b

• Platelets • Leukocytes

• Erythrocytes (red blood cells) • Leukocytes (white blood cells) • Platelets (thrombocytes)
Erythrocytes

Erythrocytes – Red Blood Cells (RBCs) • Human blood contain about 30 trillion RBCs • Oxygen-transporting cells • 7. 5 µm in diameter (diameter of capillary 8 – 10µm) • Most numerous of the formed blood cells – Females: 4. 3 – 5. 2 million cells/cubic millimeter – Males: 5. 2 – 6 million cells/cubic millimeter • Made in the red bone marrow in long bones, cranial bones, ribs, sternum, and vertebrae
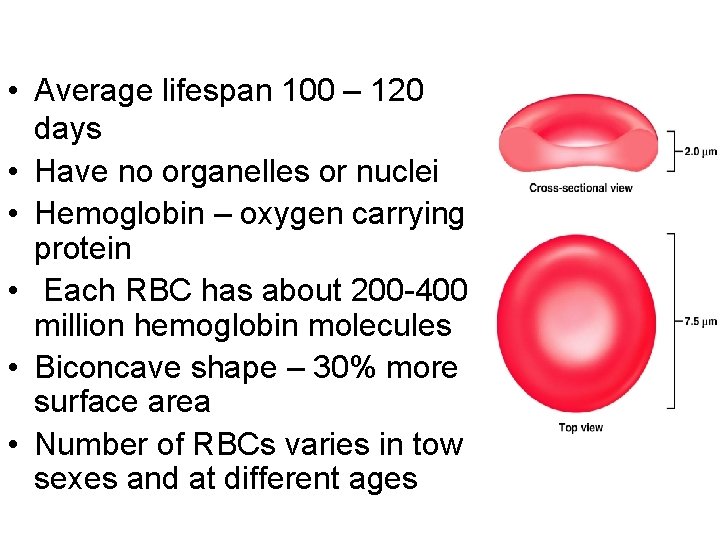
• Average lifespan 100 – 120 days • Have no organelles or nuclei • Hemoglobin – oxygen carrying protein • Each RBC has about 200 -400 million hemoglobin molecules • Biconcave shape – 30% more surface area • Number of RBCs varies in tow sexes and at different ages

The shape and size of erythrocytes (RBCs) • Biconcave disc shape, which is suited for gas exchange. • The shape is flexible so that RBCs can pass though the smallest blood vessels, i. e. , capillaries. • RBC is a bag that can be deformed into any shape.
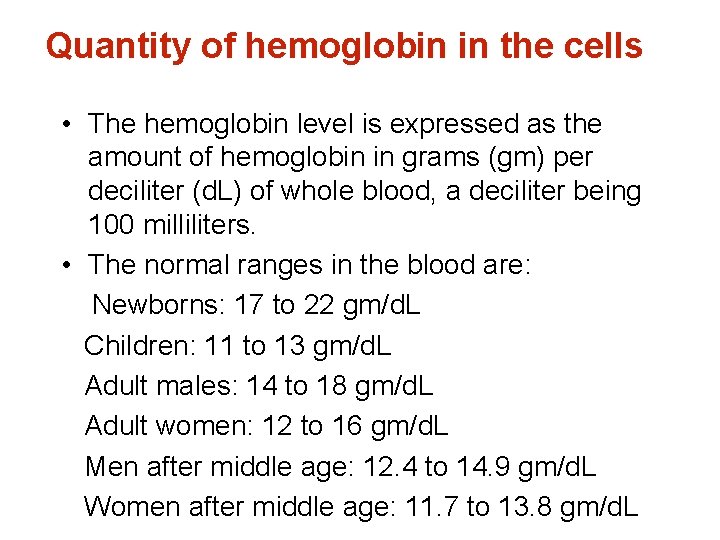
Quantity of hemoglobin in the cells • The hemoglobin level is expressed as the amount of hemoglobin in grams (gm) per deciliter (d. L) of whole blood, a deciliter being 100 milliliters. • The normal ranges in the blood are: Newborns: 17 to 22 gm/d. L Children: 11 to 13 gm/d. L Adult males: 14 to 18 gm/d. L Adult women: 12 to 16 gm/d. L Men after middle age: 12. 4 to 14. 9 gm/d. L Women after middle age: 11. 7 to 13. 8 gm/d. L

A low hemoglobin level • is referred to as anemia or low red blood count. • loss of blood • bone marrow problems • suppression by red blood cell synthesis
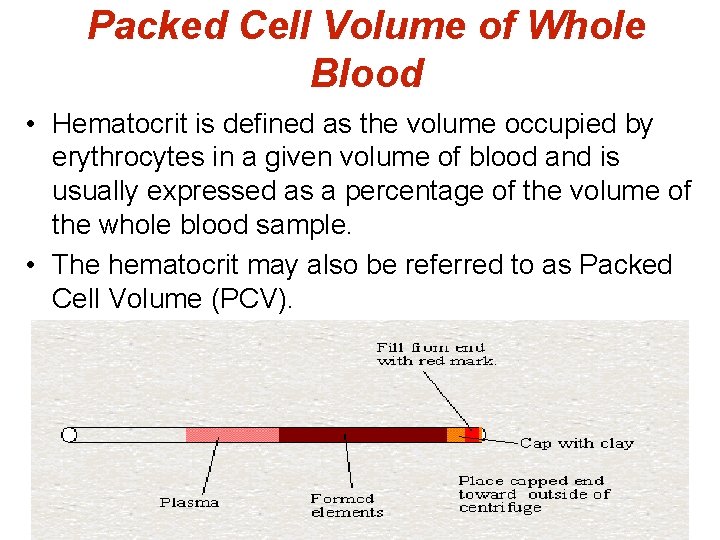
Packed Cell Volume of Whole Blood • Hematocrit is defined as the volume occupied by erythrocytes in a given volume of blood and is usually expressed as a percentage of the volume of the whole blood sample. • The hematocrit may also be referred to as Packed Cell Volume (PCV).
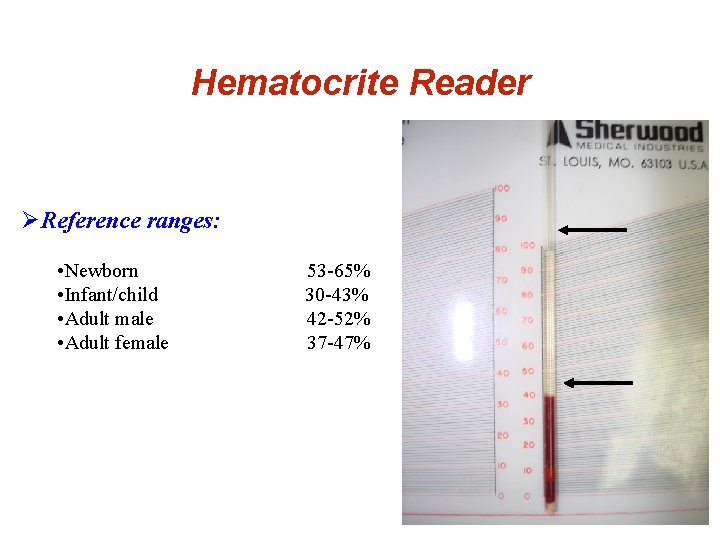
Hematocrite Reader ØReference ranges: • Newborn • Infant/child • Adult male • Adult female 53 -65% 30 -43% 42 -52% 37 -47%

Hematopoiesis • Hematopoiesis (hemopoiesis): blood cell formation – Occurs in red bone marrow of axial skeleton, girdles and proximal epiphyses of humerus and femur

Hematopoiesis • Early few weeks of embryo nucleated RBCs are formed in yolk sac. • Middle trimester mainly in liver & spleen & lymph nodes. • Last months RBCs are formed in bone marrow of all bones • Bone marrow of flat bone continue to produce RBC into adult life • Shaft of long bone stop to produce RBC at puberty while epiphysis continued • Beyond 20 age the marrow of membranous bones such as the sternum, ribs, vertebra and pelvis Dr Sitelbanat 20

Genesis of erythrocytes • RBCs are derived from cells known as hemocytoblasts (hematopoietic stem cells) are cells that can divide, through mitosis and differentiate into specialized cell types and that can self-renew to produce more stem cells

Erythropoiesis • Erythropoiesis: red blood cell production – A hemocytoblast is transformed into a proerythroblast – Proerythroblasts develop into early erythroblasts

Erythropoiesis – Phases in development 1. Ribosome synthesis 2. Hemoglobin accumulation 3. Ejection of the nucleus and formation of reticulocytes – Reticulocytes (contain small amount of basophilic endoplasmic reticulum interspersed among the HG in the cytoplasm) then become mature erythrocytes
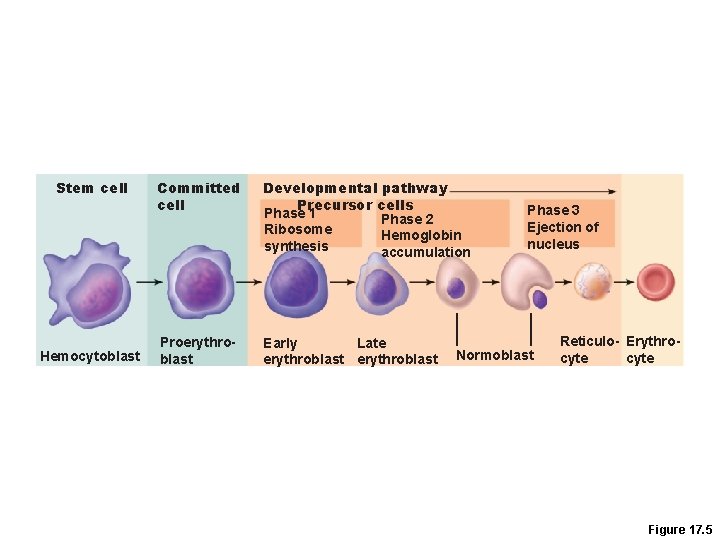
Stem cell Hemocytoblast Committed cell Developmental pathway Precursor cells Phase 1 Phase 2 Ribosome Hemoglobin synthesis accumulation Proerythroblast Early Late erythroblast Phase 3 Ejection of nucleus Normoblast Reticulo- Erythrocyte Figure 17. 5

Regulation of Erythropoiesis • Too few RBCs leads to tissue hypoxia • Too many RBCs increases blood viscosity • Balance between RBC production and destruction depends on – Hormonal controls – Adequate supplies of iron, amino acids, folic acid and B 12 vitamin

Regulation of Erythropoiesis • Importance • Factors affecting Erythropoiesis – Tissue Oxygenation • Anemia, High Altitudes, Cardiac failure, Resp Problem • ERYTHROPOITEN – Vitamins – Metals – Proteins – Hormones – Other factors/Conditions

Normal RBC Count • Importance: • Must Not fall • To supply oxygen from lungs to tissues • Must Not rise • Blood viscosity may increase • May impede blood flow

1. Tissue Oxygenation – Most Important Regulator of Erythropoiesis • ANY CONDITION Decrease Tissue Oxygenation Increase Erythropoiesis • Anemia Immediately • Increase RBCs production Bone marrow destruction: Hyperplasia of remaining cells Increase production of RBCs • Hemorrhage or increased RBC destruction reduces RBC numbers • Insufficient hemoglobin (e. g. , iron deficiency

• High altitudes: – Partial pressure of oxygen in air less – Decrease in oxygen transport to tissues – Tissue hypoxia – Result? • Cardiac Failure – Inefficient pumping by heart – Decreased blood flow to peripheral vessels – Tissue hypoxia – Result?

• Lung diseases: – Failure of oxygen absorption in Lungs – Blood carries less Oxygen – Tissue hypoxia – Result? • All conditions have one common problem • HYPOXIA ERYTHROPOIETIN

Hormonal Control of Erythropoiesis • Erythropoietin or erythropoietic stimulating factor (EPO) – Direct stimulus for erythropoiesis – Released by the kidneys in response to hypoxia

Erythropoietin • Glycoprotein • Mol weight between 39000 -70000 • Sites of production – Kidneys – Liver • Synthesized by peritubular cells of kidney in response to hypoxemia • Liver secretes 10% of endogenous erythropoietin. • Present in minute amounts in urine • Half life of 6 -9 hrs. in anemic patient

Role of kidneys in the Erythropoiten production § Decreased oxygen supply (hypoxia) to tissues stimulates secretion of erythropoietin (EP) hormone. § Hypoxia stimulates kidney to release renal erythropoietic factor (REF). § Hypoxia stimulates liver to produce a special type of globulin. § Both REF & globulin unite in plasma and form EP. § EP then stimulates bone marrow to produce RBCs. § Erythropoietin accelerates nearly all stages of RBCs formation

Stimulants of Erythropoiten production: • RENAL MECHANISM – Anemic blood to kidney – Less oxygen supplied – Renal tubular cells - High O 2 consuming – Stimulates production of Erythropoiten • NON RENAL MECHANISM – Hypoxia in other parts – Non renal sensor – Nor epinephrine, prostaglandins • In Renal failure?

Effect of Erythropoietin on Erythrogenesis • Erythropoiten - increase hemocytoblast division – Stimulates Proerythroblasts formation – Causes cells to pass through various stages of Erythropoiesis rapidly

Effect of Erythropoietin on Erythrogenesis • Person in low oxygen atmosphere (new. RBCs appear after day in circulation) • Person remove from low oxygen atmosphere (the rate of Erythropoietin formation decreased)

When the bone marrow produced RBCs at very rapid many of immature erythrocytes released in circulating blood. • Reticulocyte rise from 1% to 3050% • Normoblasts may appears in large number from 5% to 20% • erythroblast may appears in large number

Factors affecting erythropoiesis Dietary factors: i-Proteins: Proteins of high biological value are needed in the formation of RBCs. ii-Metal ions: Ø Iron Fe: is essential for RBCs formation because it enters in the formation of the hem part. Ø Copper Cu: It is carried & transported by plasma protein ceruloplasmin. It catalyses the oxidation of Fe++ to Fe+++, a reaction that must occur before transferrin can combine and transport iron. Ø Cobalt Co: It stimulates EP release from kidney. So, excess Co may produce polycythaemia.

Factors affecting erythropoiesis Dietary factors: iii-Vitamins: ü Both vitamins B 12 & folic acid are essential for final maturation of RBCs because they are needed in DNA synthesis. ü Deficiency of either B 12 or folic acid results in failure of nuclear maturation and causing maturation failure anemia. ü Vitamin C is a strong reducing agent which is important in reducing the ferric form of iron to ferrous to facilitate its absorption and transport.
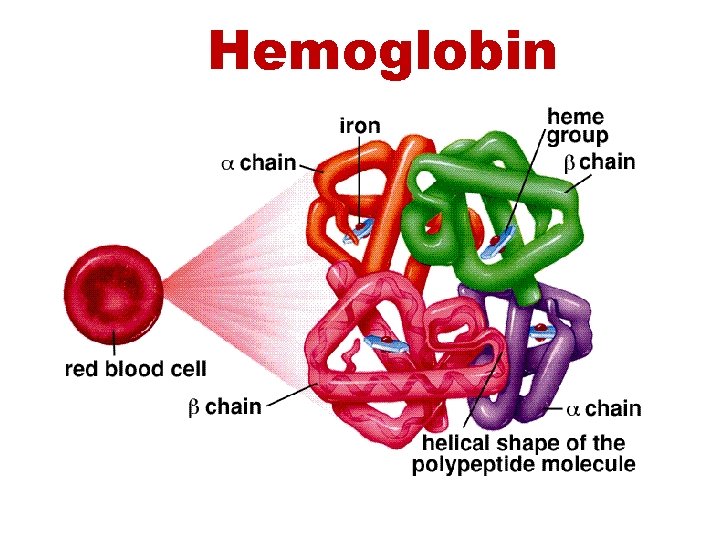
Hemoglobin

Hemoglobin (Hb) Hb is found in RBCs its main function is to transport O 2 to tissues. Structure: 2 parts : heme + globin Globin: four chains. Heme: porphyrin ring with central iron. Iron is the site of attachment with O 2. There are 4 heme groups each attached to on globin chain. So one Hb molecule can carry up to 4 O 2 molecules. According to sequence of amino acids in the primary structure of each chain, there are four types of chains; α, β, γ and δ.

Hemoglobin Synthesis • Synthesis – Occurs in the mitochondria of developing red cells as they mature in the bone marrow – Processes necessary for normal synthesis • Adequate iron supply & delivery • Adequate synthesis of protoporphyrins • Adequate globin synthesis
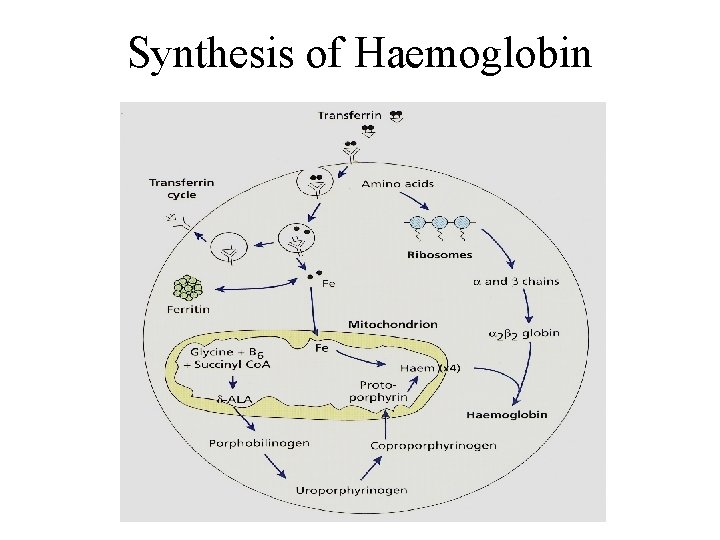
Synthesis of Haemoglobin
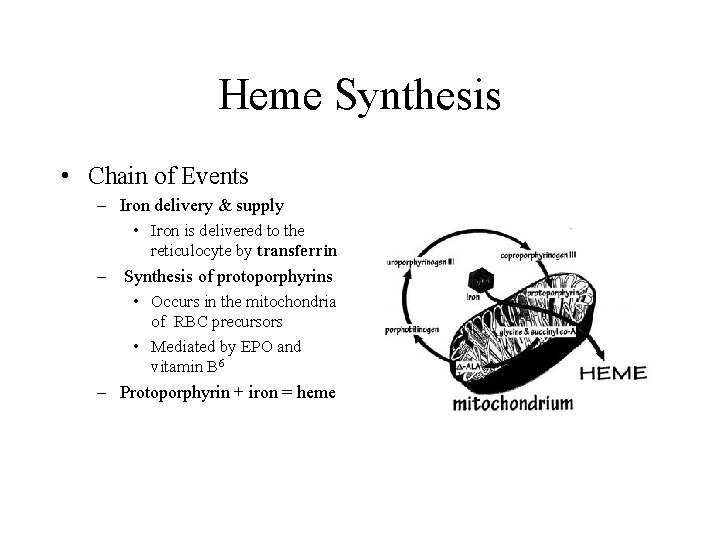
Heme Synthesis • Chain of Events – Iron delivery & supply • Iron is delivered to the reticulocyte by transferrin – Synthesis of protoporphyrins • Occurs in the mitochondria of RBC precursors • Mediated by EPO and vitamin B 6 – Protoporphyrin + iron = heme
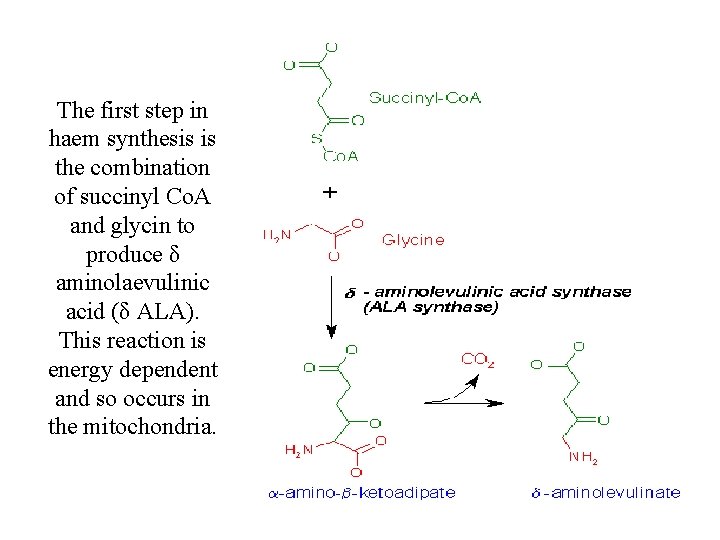
The first step in haem synthesis is the combination of succinyl Co. A and glycin to produce δ aminolaevulinic acid (δ ALA). This reaction is energy dependent and so occurs in the mitochondria.

Haem synthesis • It’s catalyzed by the enzyme δ ALA synthetase. • This step is a first-limiting step for the whole process of haem synthesis. • This represents an important control mechanism of the rate of haem synthesis and it’s coordination with globin synthesis. • Several factors are required for this step, including vitamin B 6, free ferrous and copper ions.
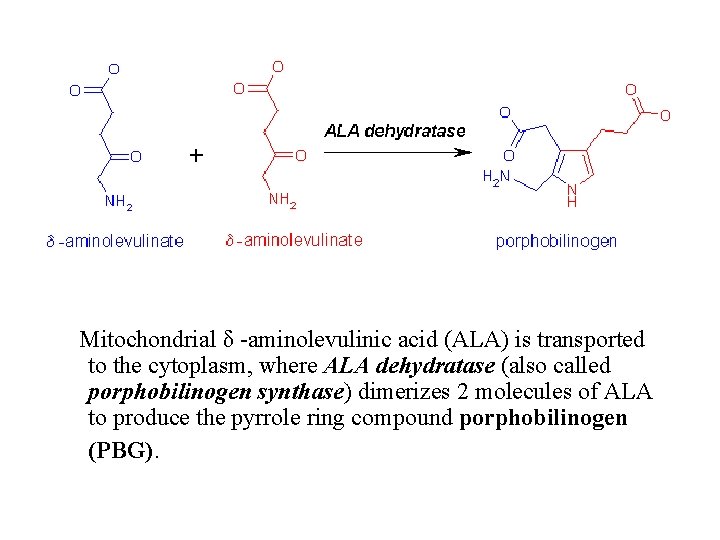
Mitochondrial δ -aminolevulinic acid (ALA) is transported to the cytoplasm, where ALA dehydratase (also called porphobilinogen synthase) dimerizes 2 molecules of ALA to produce the pyrrole ring compound porphobilinogen (PBG).

Haem synthesis • The next step requires the synthesis of porphyrin ring. • The reactions involved are extremely complex but can be summarized as the condensation of four PBG molecules to form the asymmetric cyclic uroporphyrinogen III (UPGIII). • Synthesis of UPGIII requires the presence of two enzymes (uroporphyrinogen I synthetase and uroporphyrinogen III cosynthetase) and involves the formation of several short-lived intermediates.
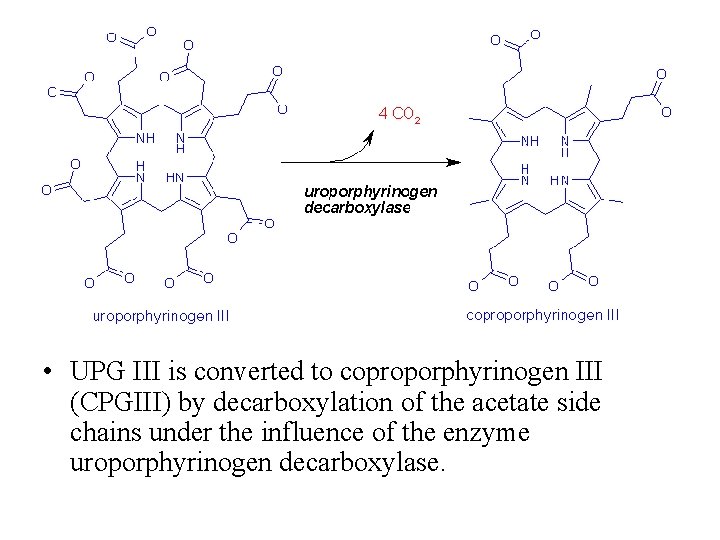
• UPG III is converted to coproporphyrinogen III (CPGIII) by decarboxylation of the acetate side chains under the influence of the enzyme uroporphyrinogen decarboxylase.
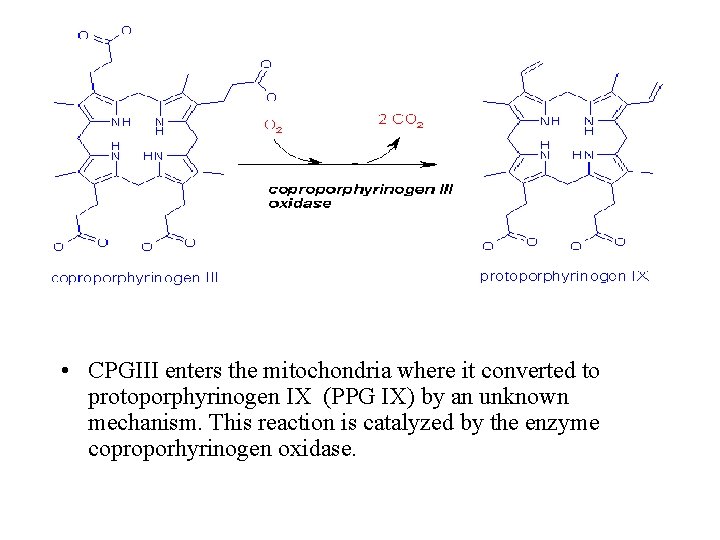
• CPGIII enters the mitochondria where it converted to protoporphyrinogen IX (PPG IX) by an unknown mechanism. This reaction is catalyzed by the enzyme coproporhyrinogen oxidase.
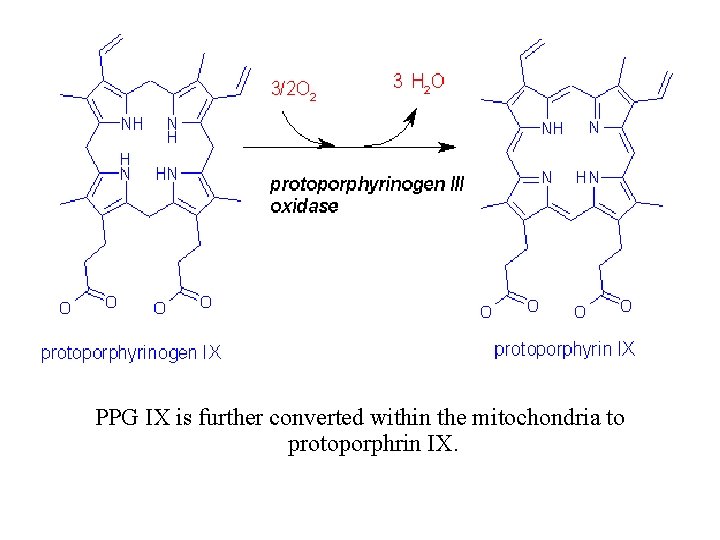
PPG IX is further converted within the mitochondria to protoporphrin IX.
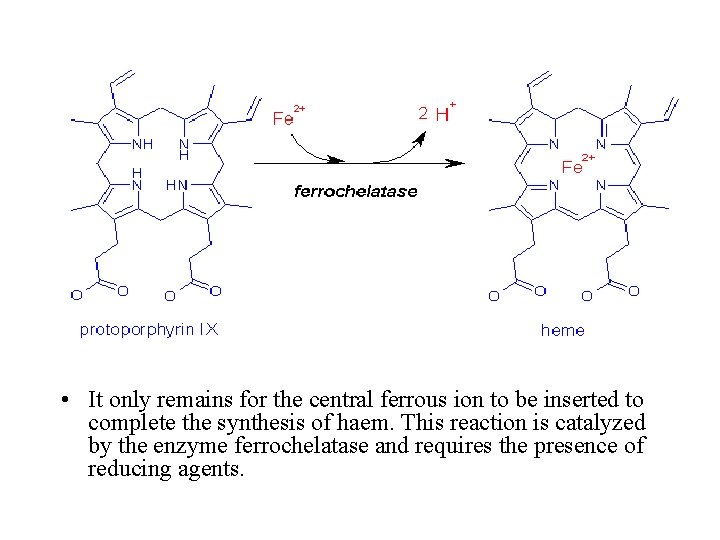
• It only remains for the central ferrous ion to be inserted to complete the synthesis of haem. This reaction is catalyzed by the enzyme ferrochelatase and requires the presence of reducing agents.

nd 2 : Globin synthesis • Humans normally carry 8 functional globin genes, arranged in two duplicate gene clusters: • The β-like cluster on the short arm of chromosome 11. • The α-like cluster on the short arm of chromosome 16. • These genes code for 6 different types of globin chains: α, β, γ, δ, ε, ζ, globin.
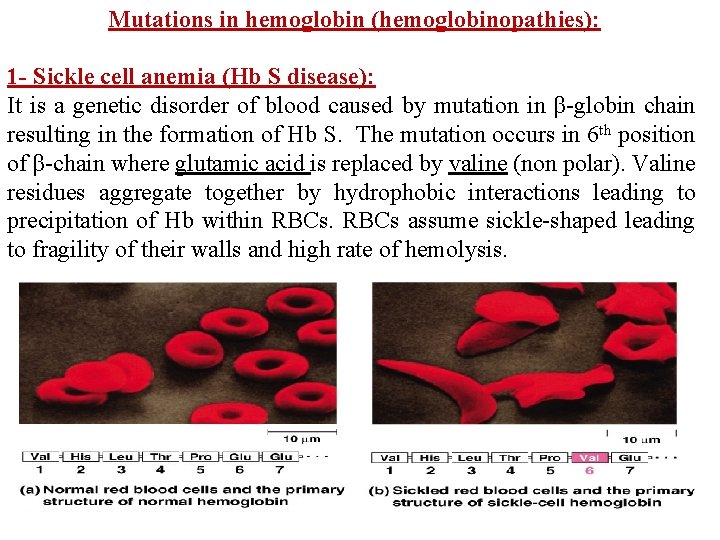
Mutations in hemoglobin (hemoglobinopathies): 1 - Sickle cell anemia (Hb S disease): It is a genetic disorder of blood caused by mutation in β-globin chain resulting in the formation of Hb S. The mutation occurs in 6 th position of β-chain where glutamic acid is replaced by valine (non polar). Valine residues aggregate together by hydrophobic interactions leading to precipitation of Hb within RBCs assume sickle-shaped leading to fragility of their walls and high rate of hemolysis.
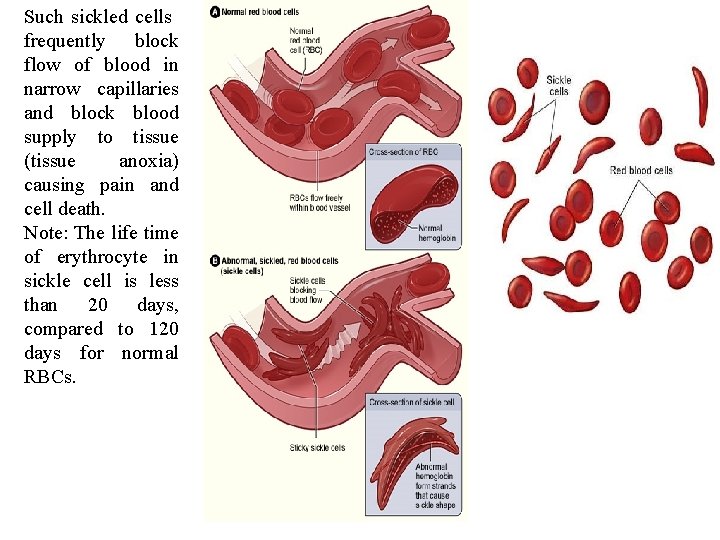
Such sickled cells frequently block flow of blood in narrow capillaries and block blood supply to tissue (tissue anoxia) causing pain and cell death. Note: The life time of erythrocyte in sickle cell is less than 20 days, compared to 120 days for normal RBCs.
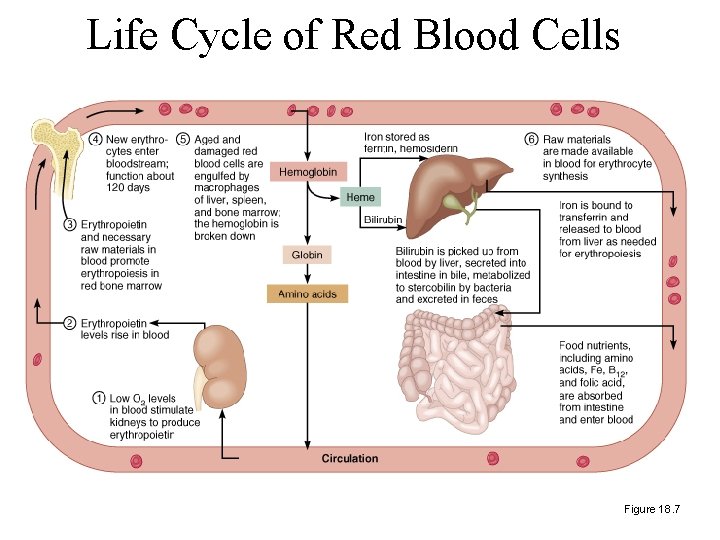
Life Cycle of Red Blood Cells Figure 18. 7

Red Blood Cell Metabolism: Summary • Three areas of RBC metabolism are crucial for RBC survival and function. – RBC membrane – Hemoglobin structure and function – RBC metabolic pathways= cellular energy

Erythrocyte Destruction • Breakdown of the RBC – Toward the end of 120 day life span of the RBC, it begins to break down. • The membrane becomes less flexible. • The concentration of cellular hemoglobin increases. • Enzyme activity, especially glycolysis, diminishes • Removal – Aging RBC’s or senescent RBC’s are removed from the circulation by the reticuloendothelial system (RES) which is a system of fixed macrophages. These cells are located all over the body, but those in the spleen are the most efficient at removing old RBC’s.

Hemoglobin Destruction • The RES cells lyses the RBC’s and digest them. Components of the RBC are recycled. – Iron is transported by transferrin to the bone marrow to be recycled into hemoglobin which storage in the form of ferritin. – Amino acids from globin are recycled into new globin chains. – The protoporphyrin ring from heme is broken and converted into biliverdin – Biliverdin is converted to direct bilirubin and carried to the liver by albumin, a plasma protein. – Bilirubin is indirect in the liver and excreted into the intestine, where intestinal flora convert it to urobilinogen. – Most urobilinogen is excreted in the stool, but some is picked up by the blood and excreted in the urine. – Conjugated (indirect) and unconjugated (direct) bilirubin can be used to monitor hemolysis.

Erythrocyte Disorders • Anemia – blood has abnormally low oxygencarrying capacity – Too rabid loss of RBCs – Many abnormality of production of RBCs – Blood oxygen levels cannot support normal metabolism

Anemia: Insufficient Erythrocytes • Hemorrhagic anemia – result of acute or chronic loss of blood • Hemolytic anemia – many abnormality of production of RBCs • Aplastic anemia – destruction or inhibition of red bone marrow

Anemia: Decreased Hemoglobin Content • Iron-deficiency anemia results from: – A secondary result of hemorrhagic anemia – Inadequate intake of iron-containing foods – Impaired iron absorption • mautration failure anemia (Pernicious) results from: – Deficiency of folic acid, vitamin B 6 and B 12 – Often caused by lack of intrinsic factor needed for absorption of B 12

Anemia: Abnormal Hemoglobin This anemia called Hemolytic anemia many different abnormalities of RBCs • RBCs become fragilehe • The life span is so short , some of these types of thia anemia are following: • Thalassemias – absent or faulty globin chain in hemoglobin – Erythrocytes are thin, delicate, and deficient in hemoglobin • Sickle-cell anemia It is a genetic disorder of blood caused by mutation in β-globin chain resulting in the formation of Hb S

Hereditary spherocytosis - RBCs are very small - RBCs shape is spherical not biconcave - Loose the bag – like cell membrane

Polycythemia • Polycythemia – excess RBCs that increase blood viscosity • Tow main polycythemias are: – Polycythemia vera – Secondary polycythemia
- Slides: 65