Arterial Blood Gas Interpretation Michael Ushay M D

Arterial Blood Gas Interpretation Michael Ushay, M. D. Pediatric Critical Care Medicine

7. 40 40 100 98% BE n n n 7. 40 = p. H 40 = PCO 2, mm Hg, dissolved CO 2 100 = PO 2, mm Hg, dissolved O 2 98% = Oxyhemoglobin saturation BE = Base excess

Hypoxemia - assessment n n ABG Pa. O 2 ~ 100 (50) A-a gradient Fi. O 2(760 -47) -Pa. CO 2/R - Pa. O 2 (. 21)(713) - 40/. 8 - 97 = 3 (368) Pa. O 2/Fi. O 2 100/. 21 = 476 (77) Oxygenation index (OI) (MAP)(Fi. O 2)(100) ÷ Pa. O 2 (28)

Oxygen n n PO 2 is not the whole answer Ca. O 2 = (Sa. O 2)(Hb g/dl)(1. 34 ml. O 2/g Hb) + (0. 003)(Pa. O 2) DO 2 = (Q)(Ca. O 2) VO 2 = (Ca. O 2 – Cmv. O 2)(Q) Q = VO 2/(Ca. O 2 – Cmv. O 2)

Oxygen Content - Example n Room air, Hb 15, normal lungs, 1 atm n n Ca. O 2 = (. 98)(15 g/dl)(1. 34 ml/g Hb) + (. 003)(100) = 19. 7 + 0. 03 = 19. 73 (20) Hb concentration, not PO 2, affects O 2 content and hence O 2 delivery the most!

Ca. O 2 – hyperbaric conditions n n n 15 gm Hgb, 3 atm, Fi. O 2 1. 0 PAO 2 = (3)(760 -47)(1. 0) – 120/0. 8 PAO 2 = 2139 – 150 = 1989 mm. Hg If lungs normal, A-a 10 Ca. O 2 = (15)(1)(1. 34) + (1979)(0. 003) Ca. O 2 = 20 + 6 = 26 ml O 2/dl blood

Hemoglobin – O 2 Saturation Curve

Changes in Hb – O 2 Sat Curve
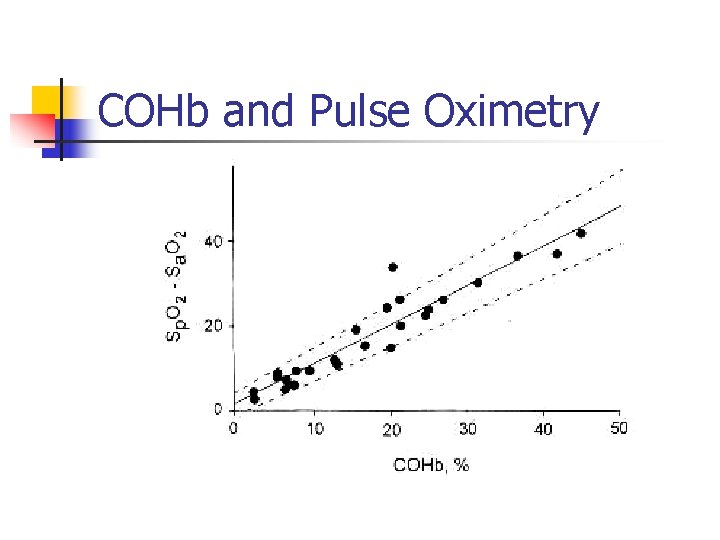
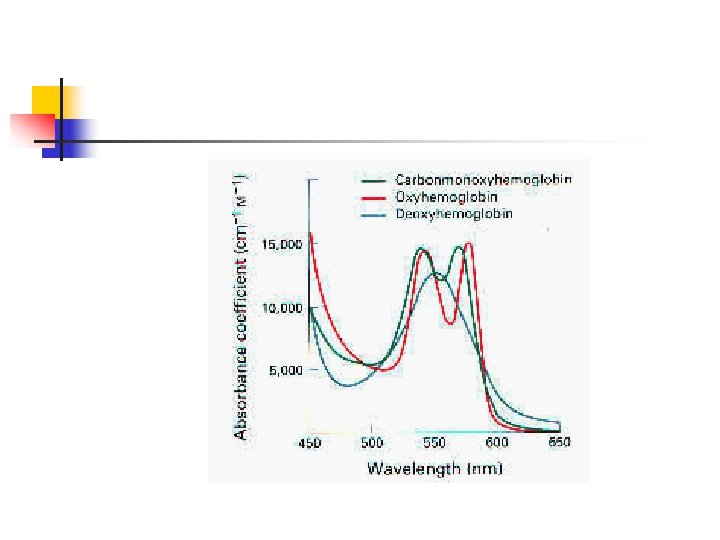
COHb and Pulse Oximetry

Hyperbaric physiology… n n Ca. O 2 (3 atm) = 20 ml O 2/dl blood + (1989 mm Hg x 0. 003 ml O 2/mm Hg/dl blood) = 20 + 6 = 26 ml. O 2/dl blood In general, each additional atmosphere of pressure O 2 dissolution in plasma by 2 ml O 2/dl (vol %)

What is the approximate Pa. O 2 of a subject with normal lung function in Denver, CO; Atmospheric pressure = 630 mm Hg?

Answer n (0. 21)(630 -47) – 40/. 8 = 72

What is the Pa. O 2 of a scuba diver swimming at a depth of 33 feet over Molokini Island, HI?

Answer n (0. 21)(1520 – 94) – 80/. 8 = 299 – 100 = 200

Question n A 1 ml sample of arterial blood is drawn from a healthy subject breathing RA at 1 atm pressure. The sample is mixed with 1 ml of heparin which had been sitting in room air and then sent to the lab for analysis. What would you expect the result to show?

Answer n n n p. H = 7. 4 PCO 2 = (40 + 0) / 2 = 20 mm Hg Pa. O 2 = (100 + 150) / 2 = 125 mm Hg

Question n Under what circumstance will an air bubble increase the Pa. O 2 of an ABG? Decrease the Pa. O 2? What will an air bubble do to the Pa. CO 2?

Answer n n n If the true Pa. O 2 is < 150, the new Pa. O 2 will be increased. If the true Pa. O 2 is > 150, the new Pa. O 2 will be decreased The Pa. CO 2 will be decreased.
![Acid Base Balance n n p. H = p. K + log [base]/[acid] p. Acid Base Balance n n p. H = p. K + log [base]/[acid] p.](http://slidetodoc.com/presentation_image_h2/a5af317b46654fa2aaf2bb9128153e93/image-20.jpg)
Acid Base Balance n n p. H = p. K + log [base]/[acid] p. H = p. K + log [HCO 3 -]/[H 2 CO 3] CO 2 + H 2 O = H 2 CO 3 p. H base/acid 20/1 n n The body works to maintain this ratio! Physiologic compensation mechanisms never overcorrect!

Rules of Acid-Base Analysis n Acute change in Pa. CO 2 of 1 mm Hg results in p. H of 0. 008 units n n The O. 08 per 10 rule p. H change of 0. 01 units results from HCO 3 - change of 0. 67 m. EQ/L n n If p. H by 0. 15 units, there is a 10 m. EQ/L in [HCO 3 -] p. H 0. 15 units 10 m. EQ/L [HCO 3 -]

Acid - Base n A patient has an acute rise in PCO 2 from 40 to 60 mm Hg. What is the change in the arterial p. H? Does the p. H go up or down? (20)(0. 008) = 0. 16 Down, CO 2 + H 2 O = H 2 CO 3, 7. 4 -. 16 = 7. 24

Acid - Base n The p. H of a patient whose PCO 2 is fixed at 40 mm Hg, acutely rises to 7. 7. What is the change in serum [HCO 3 -]? (7. 7 – 7. 4)/0. 01 = 30 (30)(0. 67) = 20 Or, (0. 3)/0. 15 = (2)(10) = 20 m. EQ/L

Base Excess (or deficit) n n n BE or BD calculated by multiplying difference between measured p. H and p. H predicted by Pa. CO 2 by 67. Predicted p. H subtracted from actual p. H Pa. CO 2 = 50, p. H = 7. 26 n n n 50 – 40 = 10, 10 x 0. 008 = 0. 08, 7. 32 7. 26 -7. 32= -0. 06 x 67 = -4 BE
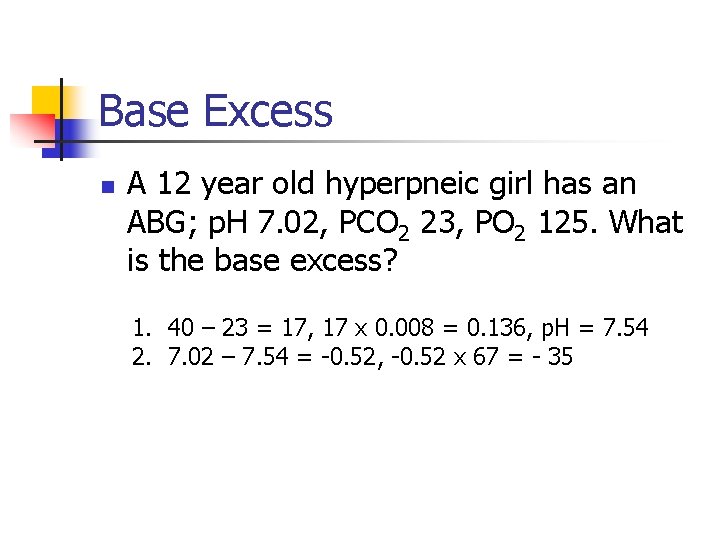
Base Excess n A 12 year old hyperpneic girl has an ABG; p. H 7. 02, PCO 2 23, PO 2 125. What is the base excess? 1. 40 – 23 = 17, 17 x 0. 008 = 0. 136, p. H = 7. 54 2. 7. 02 – 7. 54 = -0. 52, -0. 52 x 67 = - 35

Total body bicarb deficit n n Base deficit (m. EQ/L) x wt in kg x 0. 3 35 x 50 x 0. 3 = 525 m. EQ

Blood gas question n n An unconscious person is found while breathing room air to have the following ABG: p. H 7. 1, PCO 2 80, PO 2 40, Sa. O 2 79% What is the mechanism for the hypoxemia?
- Slides: 27