Arrivederci Ruth Mc Aleer Antimicrobial Pharmacist Cardiff and

Arrivederci ………. . Ruth Mc. Aleer Antimicrobial Pharmacist Cardiff and Vale UHB

Co-amoxiclav n Restricted in adults in CAV secondary care n n Removed from empirical treatment guidelines n n Primary care and paediatrics unaffected…for now Animal bites, obstetrics, some ENT, some surgical prophylaxis Removed from ward stock

Why? ! What? ! How? !
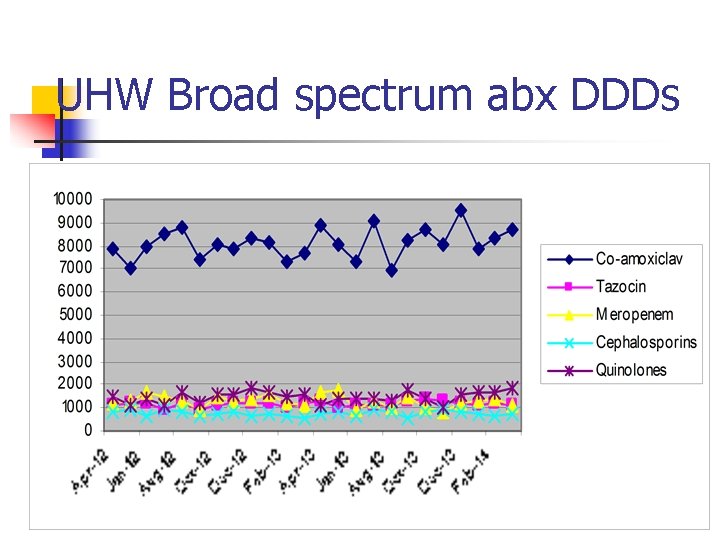
UHW Broad spectrum abx DDDs
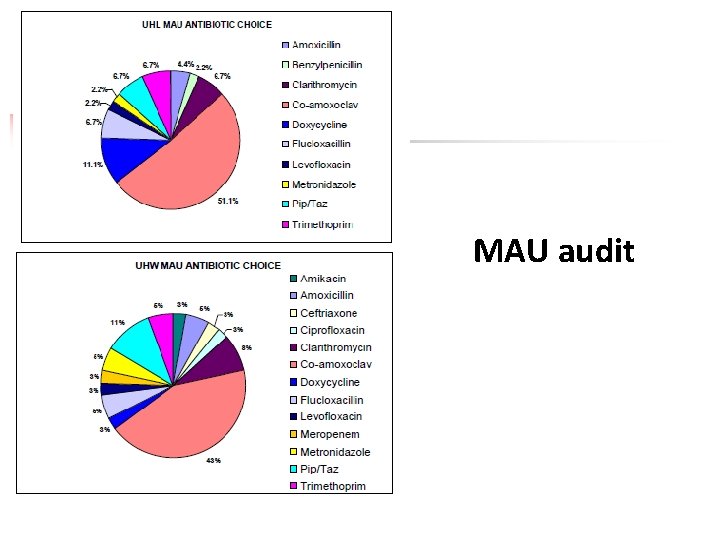
MAU audit

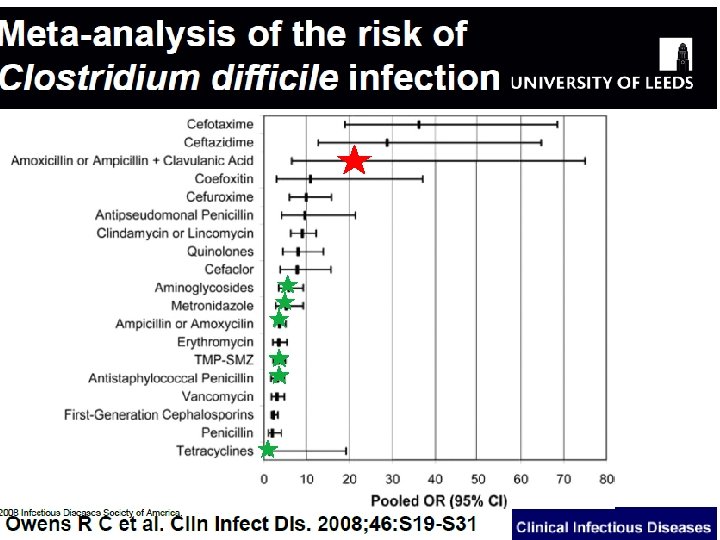
Change in CAVUHB antibiotic guidelines Reduce the use of Co-amoxiclav (Augmentin®) The main aims of this change are n to reduce the current number of c. difficile cases,
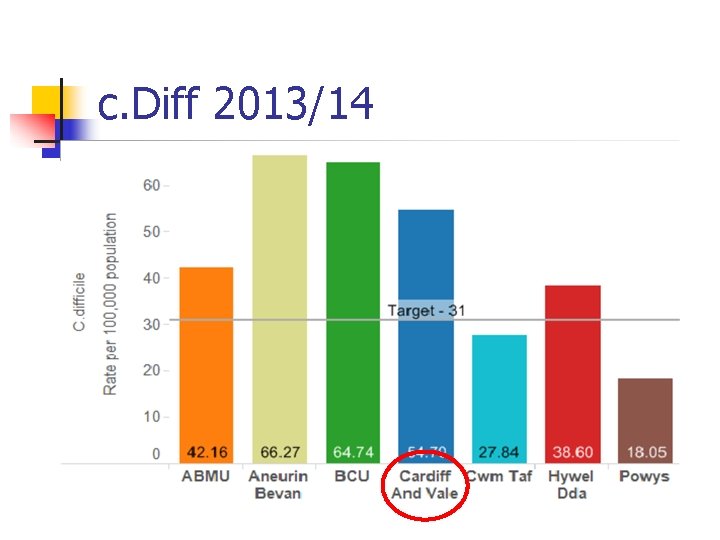
c. Diff 2013/14

Change in antibiotic guidelines Reduce the use of Co-amoxiclav (Augmentin®) The main aims of this change are n to reduce the current number of c. difficile cases, n to reduce resistance towards co-amoxiclav and

Co-amoxiclav Resistance of E. coli isolated from blood cultures: UHW: 37% UHL: 58% All Wales: 42%

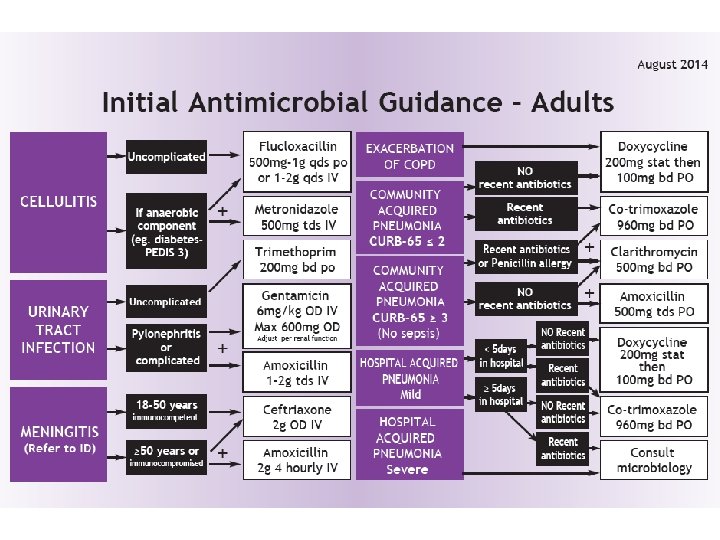
Change in antibiotic guidelines Reduce the use of Co-amoxiclav (Augmentin®) The main aims of this change are n to reduce the current number of c. difficile cases, n to reduce resistance towards co-amoxiclav and n to improve overall antimicrobial stewardship n -removing a default “one size fits all” antibiotic option New guidelines will recommend replacing co-amoxiclav with different antibiotics, depending on the indication n Doxycycline n Amoxicillin n Metronidazole n Gentamicin n Co-trimoxazole
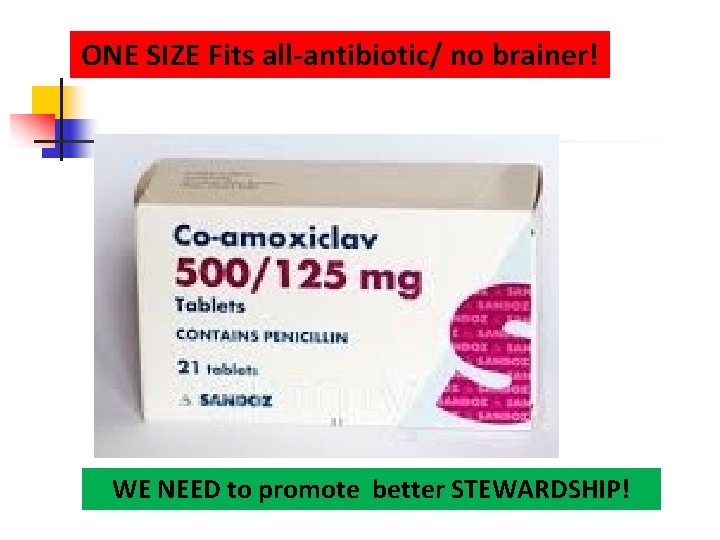
ONE SIZE Fits all-antibiotic/ no brainer! WE NEED to promote better STEWARDSHIP!

Change in antibiotic guidelines Reduce the use of Co-amoxiclav (Augmentin®) The main aims of this change are n to reduce the current number of c. difficile cases, n to reduce resistance towards co-amoxiclav and n to improve overall antimicrobial stewardship n -removing a default “one size fits all” antibiotic option New guidelines will recommend replacing co-amoxiclav with different antibiotics, depending on the indication n Doxycycline n Amoxicillin n Metronidazole n Gentamicin n Co-trimoxazole
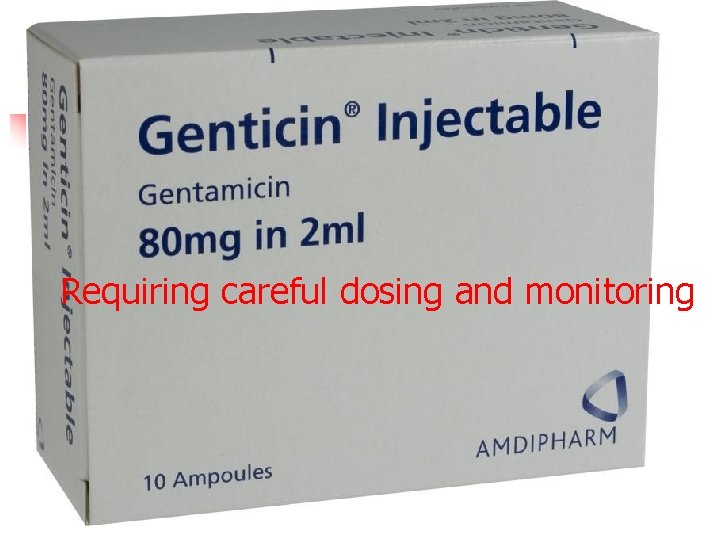
Requiring careful dosing and monitoring
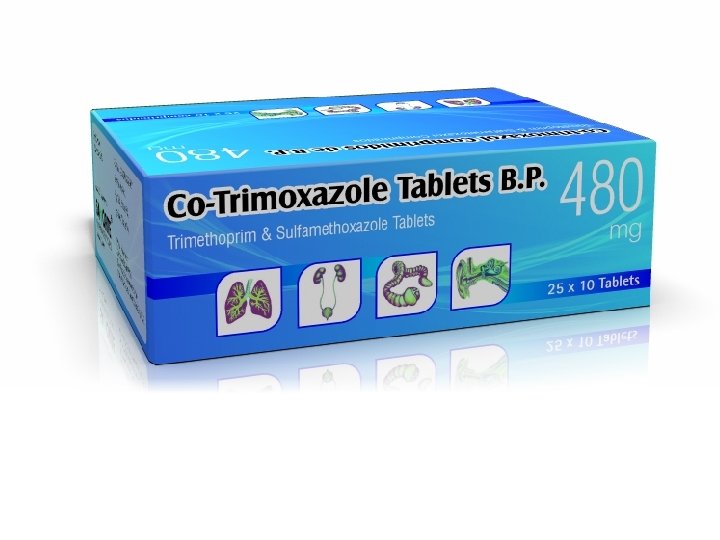

Co-trimoxazole (Septrin®) n n Off licence use Co-trimoxazole 960 mg bd n n Very good oral absorption n n Available in tablets, liquid and IV (IV restricted to those NBM or with sepsis) Interactions n n n (reduced to 480 mg bd in renal impairment egfr<30 m. L/min) Trimethoprim and sulfamethoxazole- BNF lists separately Warfarin, MTX, 6 MP, AZA, ciclosporin, clozapine, amiodarone Potential side effects n n n Nausea, vomiting, diarrhoea Rash (usually minor but rare cases of SJS, TEN so stop if suspect rash 2° Septrin) Reversible small increase in creatinine (monitor U&Es) Blood dyscrasias (watch FBC) Hyperkalaemia (monitor U&Es)

Remember remember the 5 th November
Countdown to the 5 th November



Tools ANTIMICROBIAL (Approved Name) Indication: D/W MICRO Y/N Duration/ review date: 48 hr rv consultant signature: PRESCRIBER’ S SIGNATURE Bleep No. PHARM SUPPLY

Available from Microbiology and Pharmacy

Thank you for listening Any questions?
- Slides: 26