Aristotle 384 BC322 BC Aristotle believed that four


Aristotle (384 BC-322 BC) 希臘 Aristotle believed that four elements make up everything under the moon (the terrestrial): earth, air, fire and water. Different substances are made from different proportions of these elements. Democritus (460 BC-370 BC) 希臘 The theory of Democritus and Leucippus held that everything is composed of "atoms", which are indivisible; that between atoms lies empty space; that atoms are indestructible; have always been, and always will be, in motion; that there an infinite number of atoms, and kinds of atoms, which differ in shape, and size.
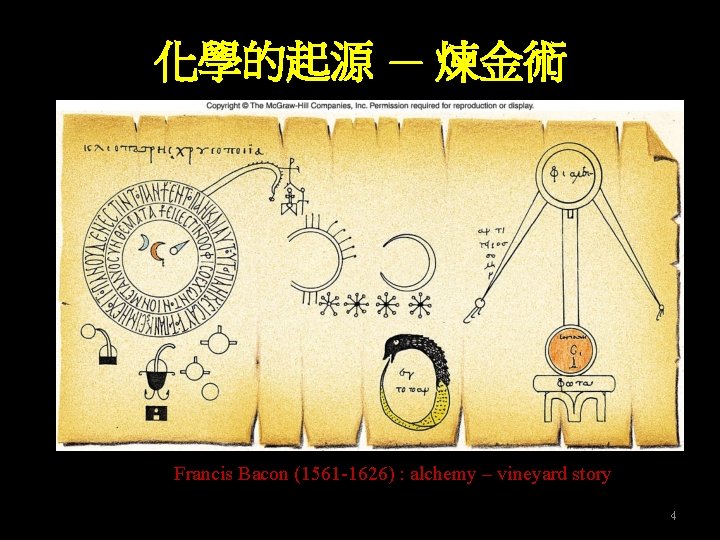
化學的起源 — 煉金術 Francis Bacon (1561 -1626) : alchemy – vineyard story 4

Robert Boyle (1627 -1691) 英國 元素的概念及氣體定律 (PV = constant) 1. Boyle is largely regarded today as the first modern chemist, and therefore one of the founders of modern chemistry, and one of the pioneers of modern experimental scientific method. 2. He endorsed the view of elements as the undecomposable constituents of material bodies; and made the distinction between mixtures and compounds.

Antoine Lavoisier (1743 -1794) 法國 拉瓦錫 – 現代化學之父 Lavoisier: Conservation of Mass Although matter might change its form during a chemical reaction, it cannot be created or destroyed – mass, and therefore matter, is conserved. Lagrange: "It took them only an instant to cut off his head, but France may not produce another such head in a century. ")

John Dalton (1766 -1844) 英國 成熟的原子學說 1. Elements are made of extremely small particles called atoms. 2. Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties. 3. Atoms cannot be subdivided, created, or destroyed. 4. Atoms of different elements combine in simple whole-number ratios to form chemical compounds. 5. In chemical reactions, atoms are combined, separated, or rearranged.

Amedeo Avogadro (1766 -1844) 亞佛加厥 義大利 正確的分子學說 Amedeo Avogadro hypothesized in 1811 that two given samples of an ideal gas, at the same temperature, pressure and volume, contain the same number of molecules. Thus, the number of molecules or atoms in a specific volume of gas is independent of their size or the molar mass of the gas. He believed that gases are composed of molecules, and these molecules are composed of atoms. NA = 6. 02214179(30)× 1023 mol− 1 Turin, Italy

Dmitri Mendeleev (1834 -1907) 門德烈夫 俄國 週期表的創始人 (also Julius Lothar Meyer) 54 known elements in 1864 元素之間的關聯? 元素發現的年代

門德烈夫對週期表德說明 On 6 March 1869, Mendeleev made a formal presentation to the Russian Chemical Society, entitled The Dependence between the Properties of the Atomic Weights of the Elements 1. The elements, if arranged according to their atomic weight, exhibit an apparent periodicity of properties. 2. Elements which are similar in regards to their chemical properties have atomic weights which are either of nearly the same value (e. g. , Pt, Ir, Os) or which increase regularly (e. g. , K, Rb, Cs). 3. The arrangement of the elements in groups of elements in the order of their atomic weights corresponds to their so-called valencies, as well as, to some extent, to their distinctive chemical properties; as is apparent among other series in that of Li, Be, B, C, N, O, and F. 4. The magnitude of the atomic weight determines the character of the element, just as the magnitude of the molecule determines the character of a compound body. 5. We must expect the discovery of many yet unknown elements–for example, two elements, analogous to aluminium and silicon, whose atomic weights would be between 65 and 75. (Ga: 70, Ge: 73) 6. Certain characteristic properties of elements can be foretold from their atomic weights. 10

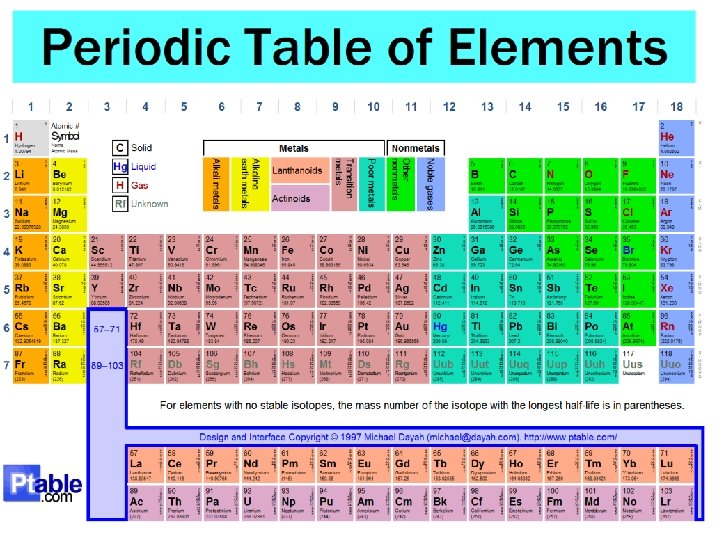
12
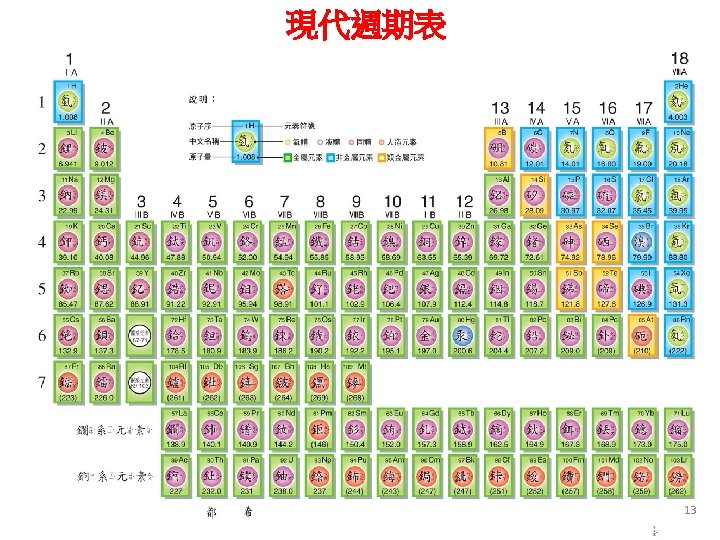

Wilhelm C. Roentgen (1845 -1923) Discovery of X-Ray Nov. 8, 1895 Nobel Prize for Physics (1901) 14

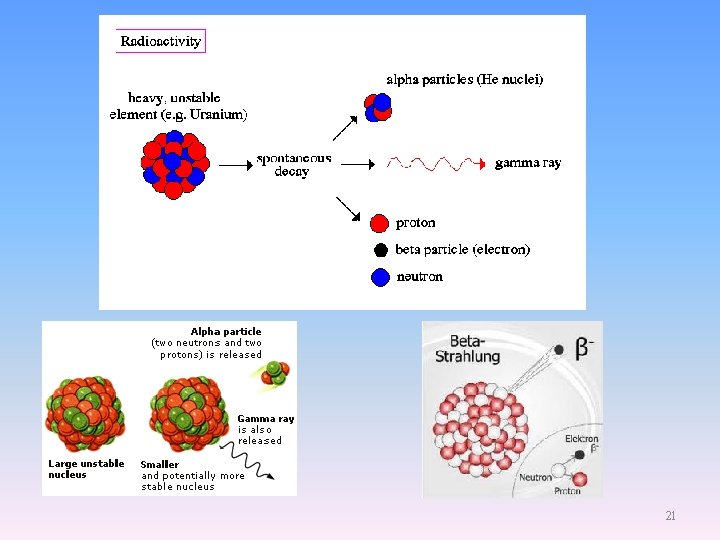
Henri Becquerel Discovery of Radioactivity Feb. 26, 1896 K 2 UO 2(SO 4)2 15
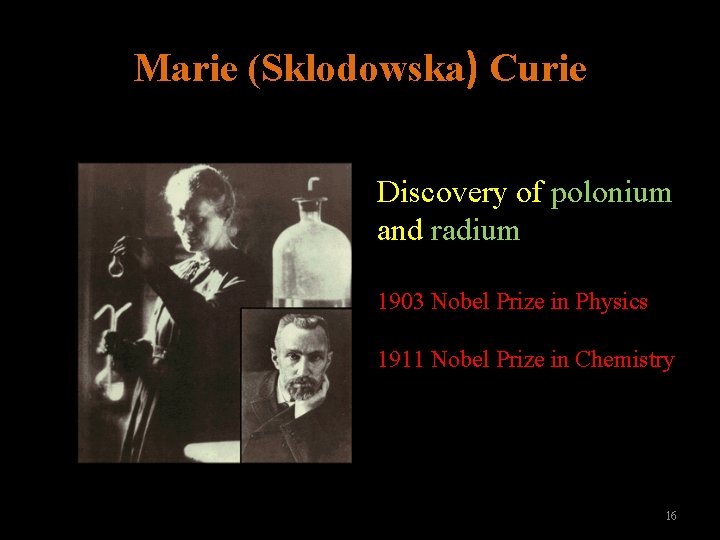
Marie (Sklodowska) Curie Discovery of polonium and radium 1903 Nobel Prize in Physics 1911 Nobel Prize in Chemistry 16

J. J. Thomson (1856 -1940) 英國 1. His experiments suggested not only that cathode rays were over 1000 times lighter than the hydrogen atom, but also that their mass was the same whatever type of atom they came from. 2. He concluded that the rays were composed of very light, negatively charged particles which were a universal building block of atoms. 3. He determined the charge-to-mass ratio of an electron. e/m = -1. 76 x 108(C/g) 4. The atom must also contain positive particles that balance exactly the negative charge carried by particles that we now call electrons. 17

Robert Millikan (1868 -1953) 美國 1. 2. 3. Performed experiments involving charged oil drops. Determined the magnitude of the charge on a single electron. Calculated the mass of the electron. e = -1. 6 x 10 -19 (C) m = 9. 1 x 10 -28 (g) 18
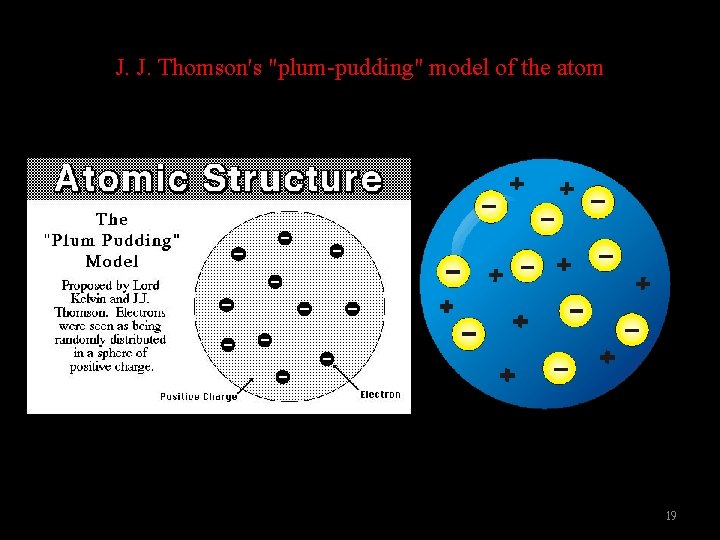
J. J. Thomson's "plum-pudding" model of the atom 19

Ernest Rutherford 1871 -1937 英國,紐西蘭 1. Discovered the true nature of the radioactive radiation, a, b, and g rays. Invented isotope dating. 2. His a particle scattering experiments (1909) revealed the true nature of the atomic structure. 3. Rutherford became in 1917 the first person to deliberately transmute one element into another, when he converted nitrogen into oxygen through the nuclear reaction 14 N + α → 17 O + proton. 20
21
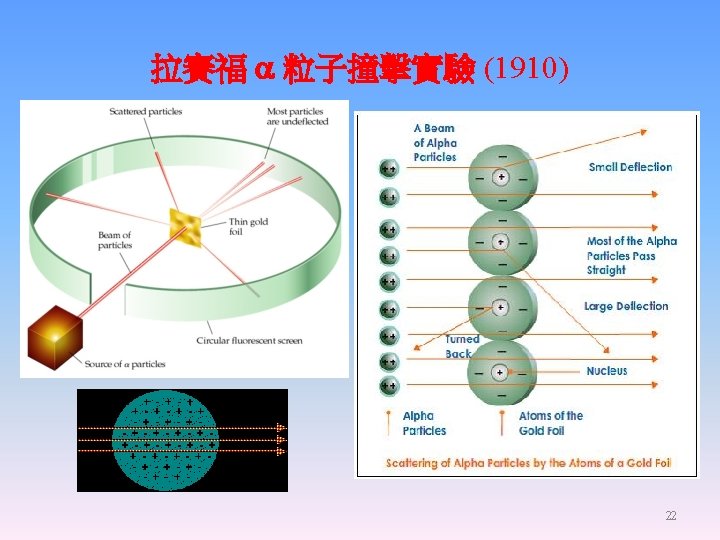

Rutherford’s Gold Foil Experiment 23

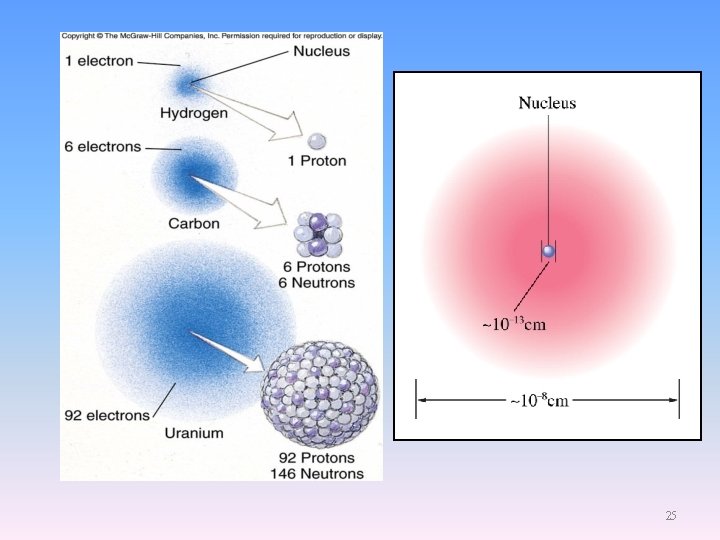
25
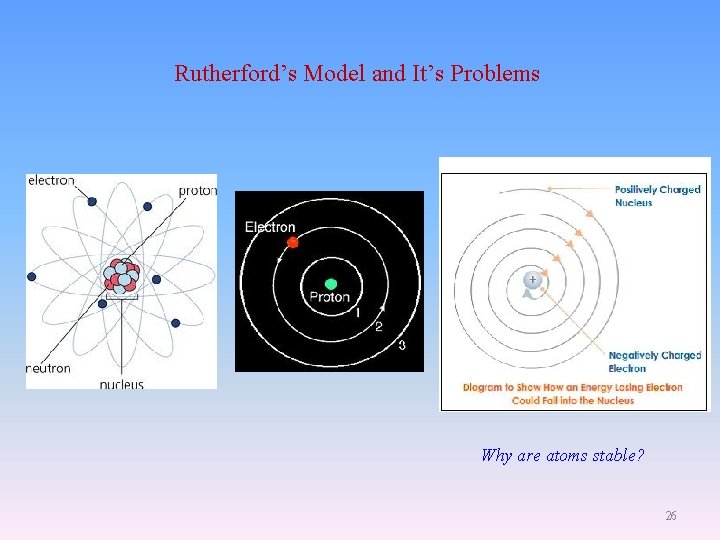
Rutherford’s Model and It’s Problems Why are atoms stable? 26
- Slides: 26