ARCI Tutorial using RD function as an example




- Slides: 21

ARCI Tutorial using R&D function as an example February 10, 2009 LDN Partners LLC

Contents r ARCI basics u u r Understanding the context and integration of an example ARCI (using an R&D function) with an overall (all functions) Innovation Process (IP) u u u r What does ARCI mean? How to read an ARCI chart Tasks and decisions on ARCI follow Stage Gate steps on an IP process flow Specific R&D function ARCI is integrated with IP overall (all functions) ARCI Overall IP ARCI for all functions must be synchronized with product development project R&D team ARCIs Understanding that individual ARCI charts may have to be customized down to the channel and brand level in some cases u u Example Pharma IP organization breakdown Depicting how R&D PD teams may need custom ARCI charts down to brand levels 2

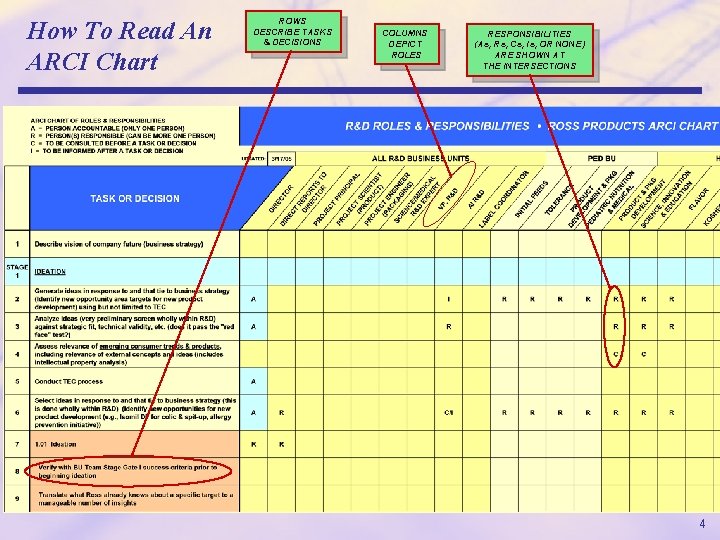
What Does “ARCI” Mean? ARCI CHART OF ROLES & RESPONSIBILITIES A = PERSON ACCOUNTABLE (ONLY ONE PERSON) R = PERSON(S) RESPONSIBLE (CAN BE MORE ONE PERSON) C = TO BE CONSULTED BEFORE A TASK OR DECISION I = TO BE INFORMED AFTER A TASK OR DECISION ARCI Charts • Often called roles and responsibility charting. • It means determining for each task or decision who is going to accountable (“A”), who will be responsible (“R”), who needs to be consulted before (“C”), and who needs to be informed after (“I”). • Only one person can be accountable for any task or decision (“the buck stops here”), whereas one or more people may be responsible. We often say someone has the “A” for something, meaning it is the person who will be held accountable. • The “A” role (accountable role) also means that person has the authority to get the task done or to make the decision. • The person with the “A” role is obligated to use that authority to get the job done. 3

How To Read An ARCI Chart ROWS DESCRIBE TASKS & DECISIONS COLUMNS DEPICT ROLES RESPONSIBILITIES (As, Rs, Cs, Is, OR NONE) ARE SHOWN AT THE INTERSECTIONS 4

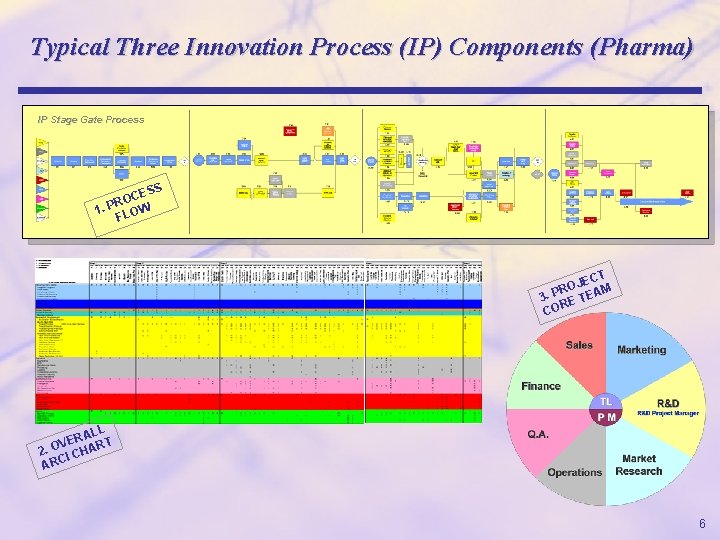
Pharma Example Explained r The following slides demonstrate how a typical Pharma product development project might be executed through a stage gate process, including development and integration of ARCI charts at the appropriate levels. r There is no significance to either the functional example (an innovation process) or the industry (pharma); ARCI charting is used universally in all industries and functional processes. r These slides exemplify how custom ARCI charts must be developed in detail, in some cases down to the brand level. r However, there is no boilerplate rule for how many ARCI charts are needed; ARCIs must be developed for each business process down to the appropriate level; each client circumstance and environment will require a custom solution; what is appropriate for one situation may differ from another. 5

Typical Three Innovation Process (IP) Components (Pharma) IP Stage Gate Process S CES O R 1. P LOW F T JEC O M R 3. P E TEA R CO ALL R E T V 2. O I CHAR ARC 6

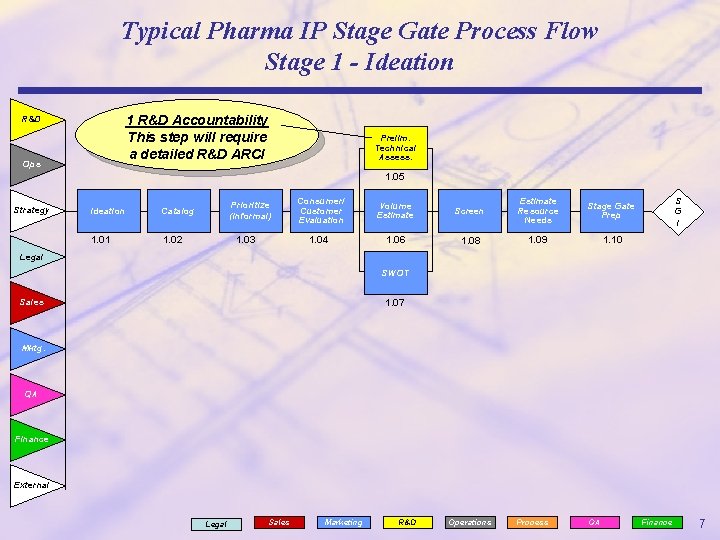
Typical Pharma IP Stage Gate Process Flow Stage 1 - Ideation 1 R&D Accountability This step will require a detailed R&D ARCI R&D Ops Prelim. Technical Assess. 1. 05 Strategy Ideation Catalog 1. 01 1. 02 Prioritize (Informal) 1. 03 Consumer/ Customer Evaluation 1. 04 Volume Estimate Screen Estimate Resource Needs Stage Gate Prep 1. 06 1. 08 1. 09 1. 10 S G I Legal SWOT Sales 1. 07 Mktg. QA Finance External Legal Sales Marketing R&D Operations Process QA Finance 7

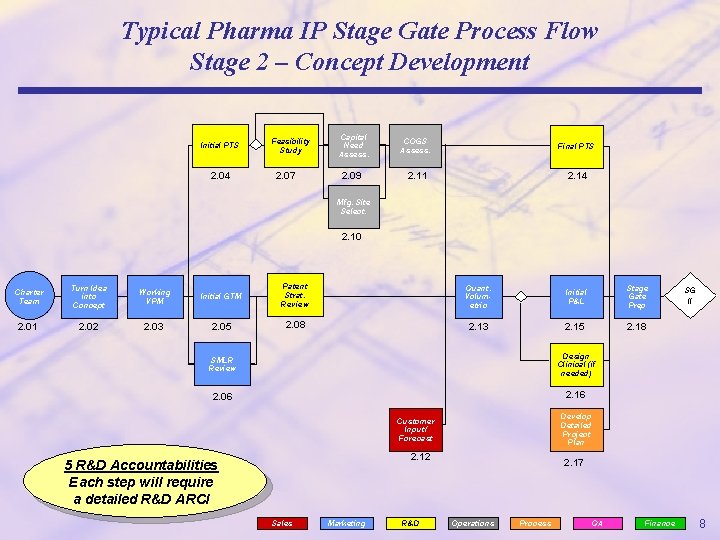
Typical Pharma IP Stage Gate Process Flow Stage 2 – Concept Development Initial PTS 2. 04 Feasibility Study 2. 07 Capital Need Assess. COGS Assess. Final PTS 2. 09 2. 11 2. 14 Mfg. Site Select. 2. 10 Charter Team Turn Idea into Concept Working VPM Initial GTM Patent Strat. Review Quant. Volumetric Initial P&L Stage Gate Prep 2. 01 2. 02 2. 03 2. 05 2. 08 2. 13 2. 15 2. 18 SMLR Review Design Clinical (if needed) 2. 06 2. 16 Develop Detailed Project Plan Customer Input/ Forecast 2. 12 5 R&D Accountabilities Each step will require a detailed R&D ARCI Sales Marketing R&D SG II 2. 17 Operations Process QA Finance 8

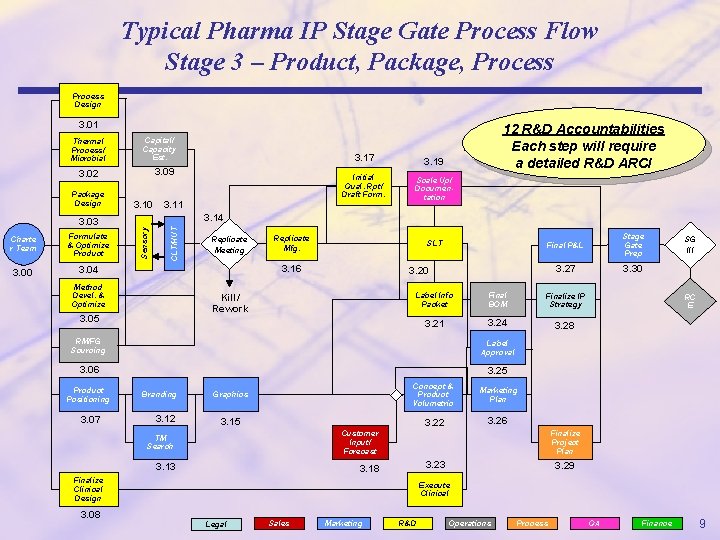
Typical Pharma IP Stage Gate Process Flow Stage 3 – Product, Package, Process Design 3. 01 Capital/ Capacity Est. 3. 09 3. 02 Package Design 3. 10 3. 04 Sensory Formulate & Optimize Product 12 R&D Accountabilities Each step will require a detailed R&D ARCI 3. 17 3. 19 Initial Qual. Rpt/ Draft Form. Scale Up/ Documentation 3. 14 3. 03 Charte r Team 3. 11 CLT/HUT Thermal Process/ Microbial Replicate Meeting Replicate Mfg. SLT 3. 16 Method Devel. && Optimize Finalize IP Strategy 3. 21 3. 24 3. 28 3. 06 3. 25 3. 07 3. 12 3. 30 Final BOM Label Approval Branding 3. 27 Label Info Packet RM/FG Sourcing Product Positioning Stage Gate Prep 3. 20 Kill/ Rework 3. 05 Final P&L Graphics Concept & Product Volumetric Marketing Plan 3. 22 3. 26 3. 15 TM Search Customer Input/ Forecast 3. 13 3. 18 SG III RC E Finalize Project Plan 3. 23 Finalize Clinical Design 3. 29 Execute Clinical 3. 08 Legal Sales Marketing R&D Operations Process QA Finance 9

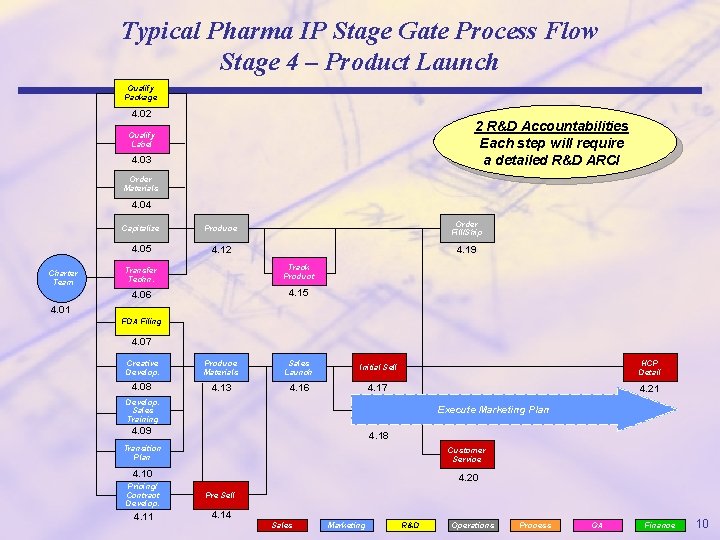
Typical Pharma IP Stage Gate Process Flow Stage 4 – Product Launch Qualify Package 4. 02 2 R&D Accountabilities Each step will require a detailed R&D ARCI Qualify Label 4. 03 Order Materials 4. 04 Charter Team Capitalize Produce Order Fill/Ship 4. 05 4. 12 4. 19 Transfer Techn. Track Product 4. 06 4. 15 4. 01 FDA Filing 4. 07 Creative Develop. Produce Materials Sales Launch Initial Sell HCP Detail 4. 08 4. 13 4. 16 4. 17 4. 21 Develop. Sales Training Execute Marketing Plan 4. 09 4. 18 Transition Plan Customer Service 4. 10 4. 20 Pricing/ Contract Develop. Pre Sell 4. 11 4. 14 Sales Marketing R&D Operations Process QA Finance 10

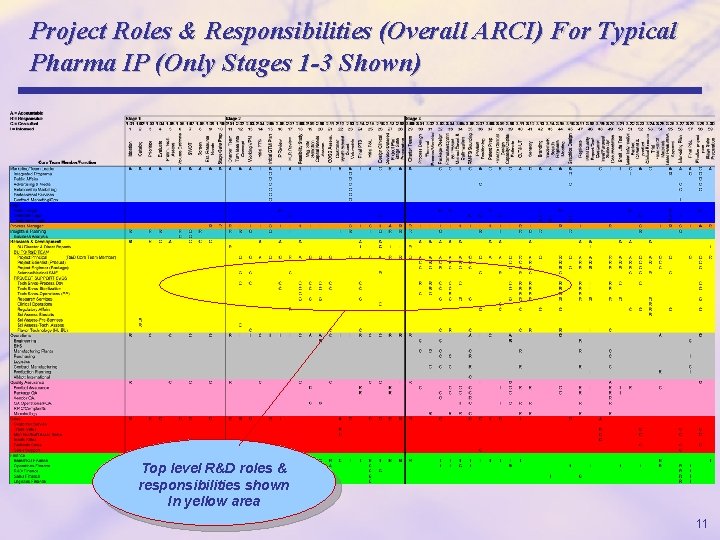
Project Roles & Responsibilities (Overall ARCI) For Typical Pharma IP (Only Stages 1 -3 Shown) Top level R&D roles & responsibilities shown In yellow area 11

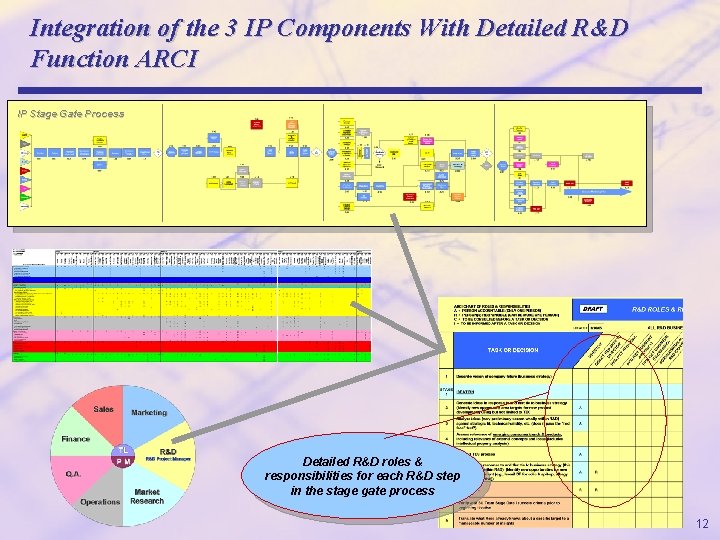
Integration of the 3 IP Components With Detailed R&D Function ARCI IP Stage Gate Process Detailed R&D roles & responsibilities for each R&D step in the stage gate process 12

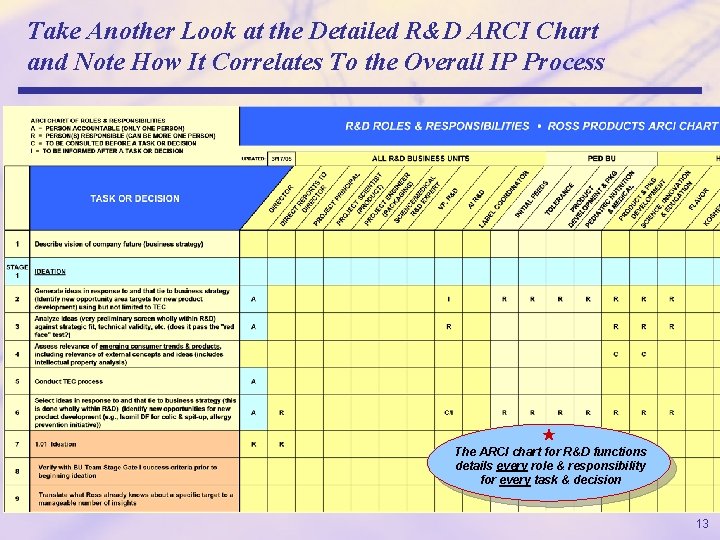
Take Another Look at the Detailed R&D ARCI Chart and Note How It Correlates To the Overall IP Process The ARCI chart for R&D functions details every role & responsibility for every task & decision 13

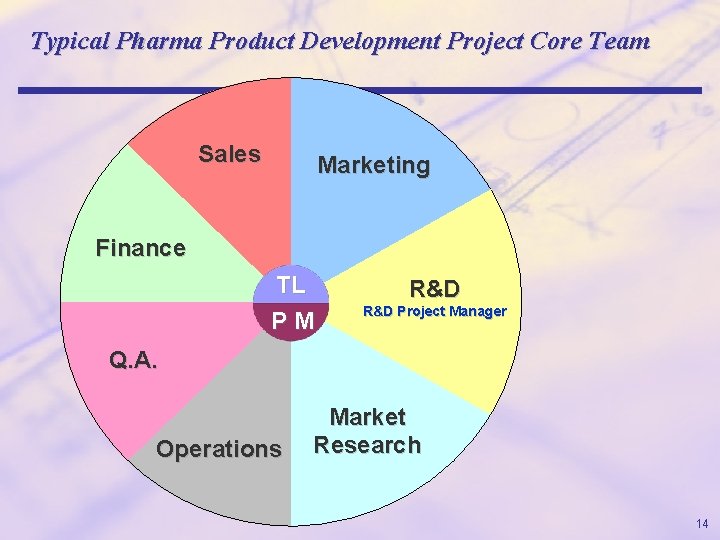
Typical Pharma Product Development Project Core Team Sales Marketing Finance TL PM R&D Project Manager Q. A. Operations Market Research 14

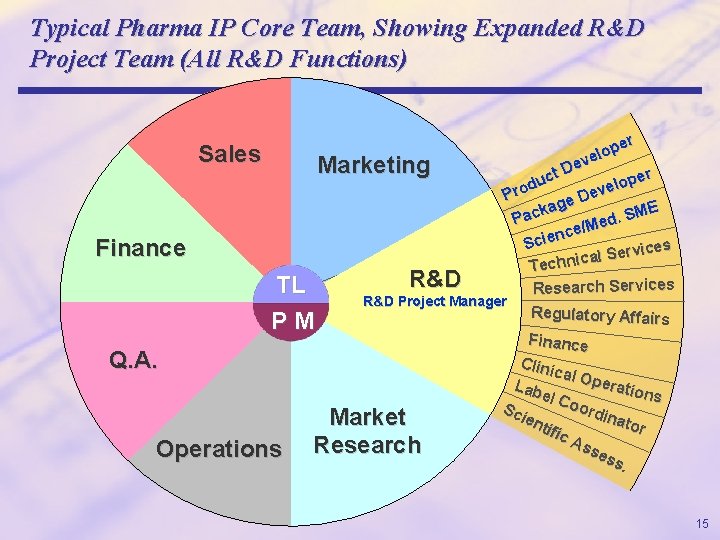
Typical Pharma IP Core Team, Showing Expanded R&D Project Team (All R&D Functions). Sales Marketing Finance TL PM R&D Project Manager Q. A. Operations per o l eve D er ct p u o l d eve Pro D e kag ME c S a. P d e/Me c n e Sci vices r e S l a c Techni Research Services Market Research Regulatory Affairs Finance Clini cal O pera Lab tions el C oor Sc dina ien tor tifi c. A sse ss. 15

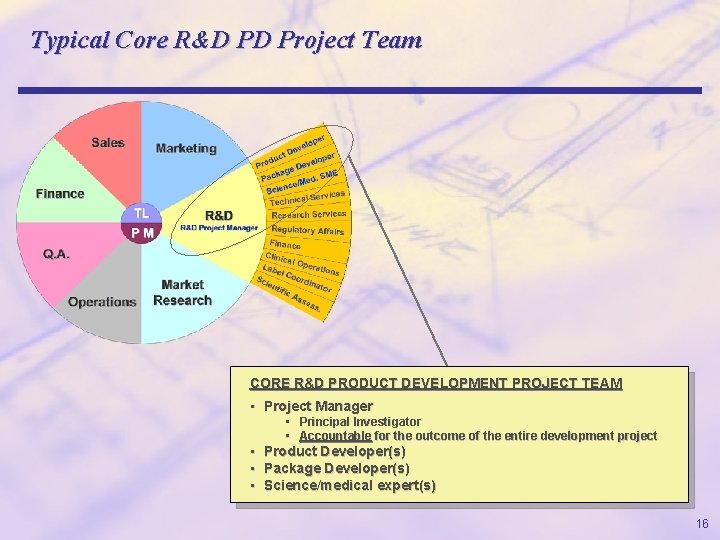
Typical Core R&D PD Project Team CORE R&D PRODUCT DEVELOPMENT PROJECT TEAM • Project Manager • Principal Investigator • Accountable for the outcome of the entire development project • Product Developer(s) • Package Developer(s) • Science/medical expert(s) 16

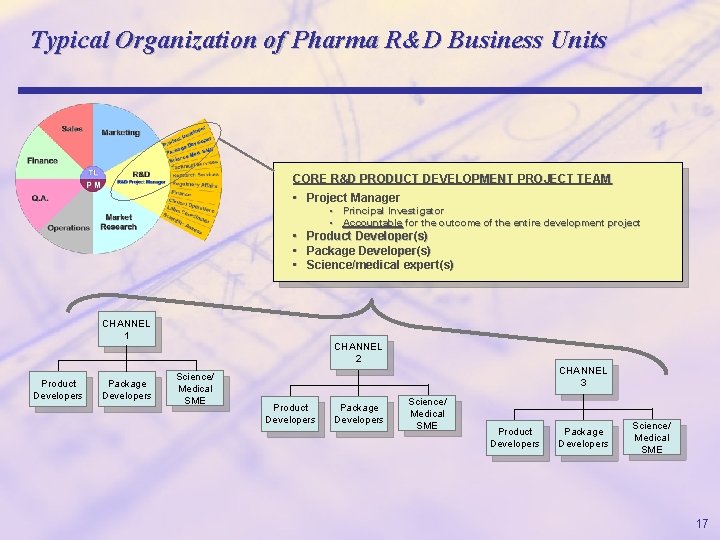
Typical Organization of Pharma R&D Business Units CORE R&D PRODUCT DEVELOPMENT PROJECT TEAM • Project Manager • Principal Investigator • Accountable for the outcome of the entire development project • Product Developer(s) • Package Developer(s) • Science/medical expert(s) CHANNEL 1 Product Developers Package Developers CHANNEL 2 Science/ Medical SME CHANNEL 3 Product Developers Package Developers Science/ Medical SME 17

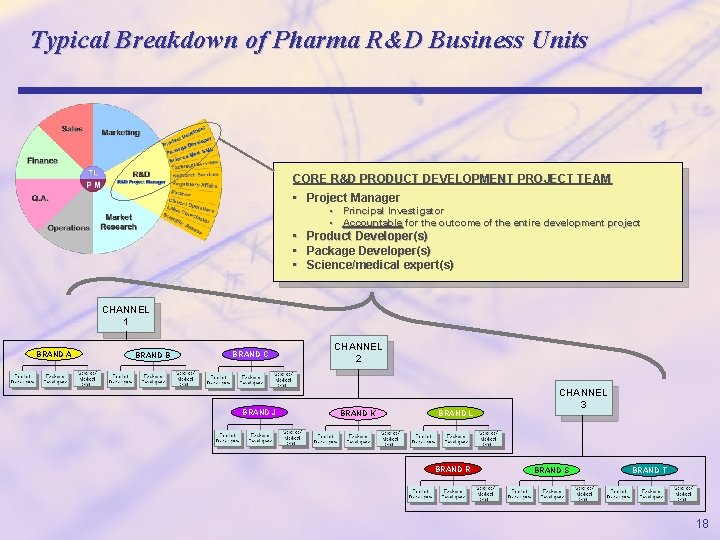
Typical Breakdown of Pharma R&D Business Units CORE R&D PRODUCT DEVELOPMENT PROJECT TEAM • Project Manager • Principal Investigator • Accountable for the outcome of the entire development project • Product Developer(s) • Package Developer(s) • Science/medical expert(s) CHANNEL 1 BRAND A BRAND B BRAND C BRAND J CHANNEL 2 BRAND K BRAND L BRAND R CHANNEL 3 BRAND S BRAND T 18

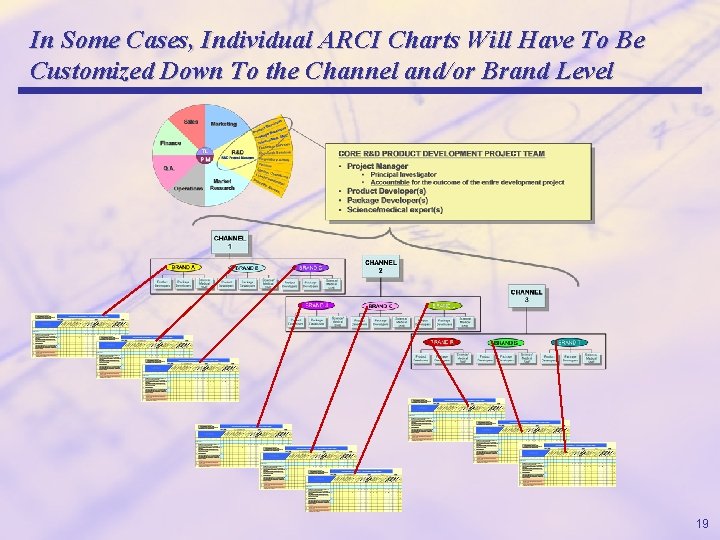
In Some Cases, Individual ARCI Charts Will Have To Be Customized Down To the Channel and/or Brand Level 19

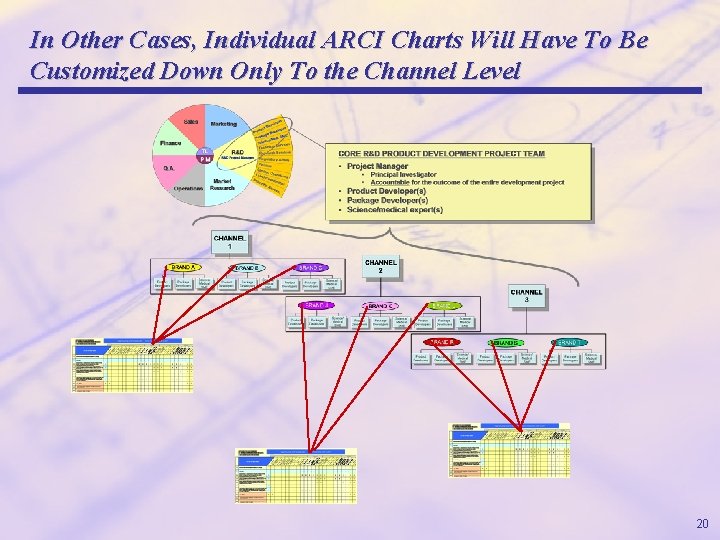
In Other Cases, Individual ARCI Charts Will Have To Be Customized Down Only To the Channel Level 20

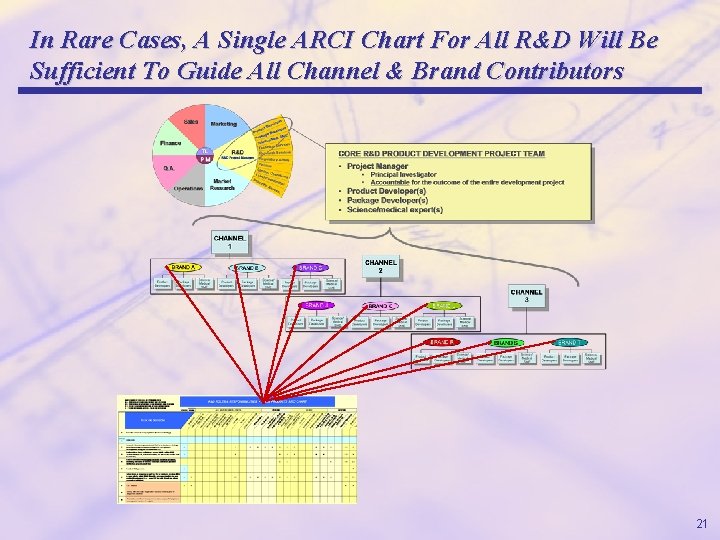
In Rare Cases, A Single ARCI Chart For All R&D Will Be Sufficient To Guide All Channel & Brand Contributors 21