Archimedes Archimedes and the Golden Crown In 247





























- Slides: 36


Archimedes

Archimedes and the Golden Crown In 247 BC Archimedes was ordered by King Hieron of Syracuse to determine if his new crown was pure gold

The king suspected that the goldsmith stole some of the gold and replaced it with silver when he made the crown.

The king told Archimedes not to damage the crown in the process just in case it was pure gold.

Archimedes discovered how to test the crown while bathing.

He ran naked through the streets of Syracuse to the king’s palace yelling “Eureka”. Which in greek means “I have found it”.

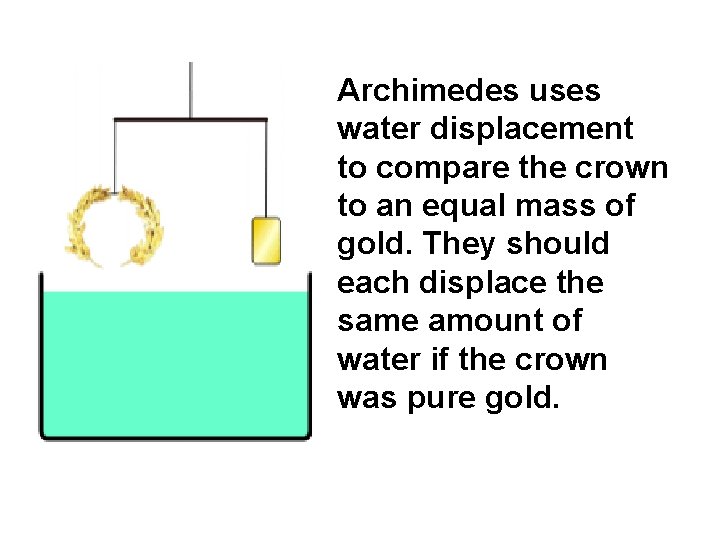
Archimedes uses water displacement to compare the crown to an equal mass of gold. They should each displace the same amount of water if the crown was pure gold.

The crown displaced more water than an equal mass of gold. Therefore the crown was not pure gold.

The unfortunate goldsmith was executed.

Density

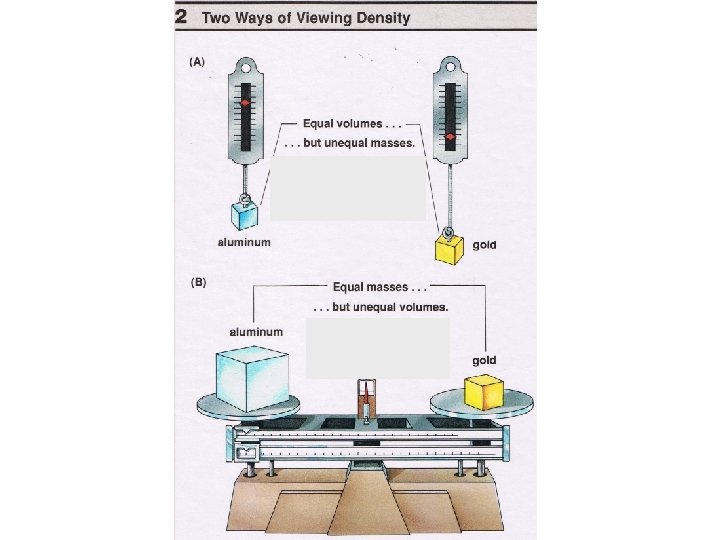
Density • Density is how compact matter is. • More mass shoved into a certain amount of space makes something more dense.

Density Considers Two Things • How much mass is present. • How much volume does it take up. • Make sure to consider both these factors when discussing density.

A A

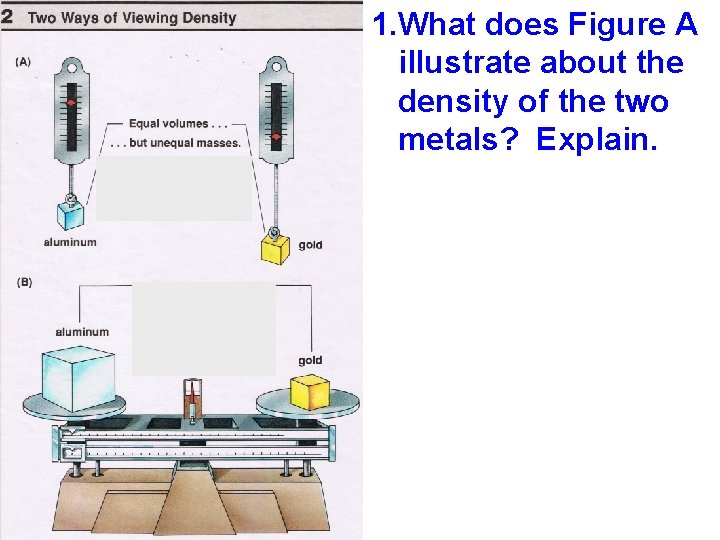
1. What does Figure A illustrate about the density of the two metals? Explain. A A

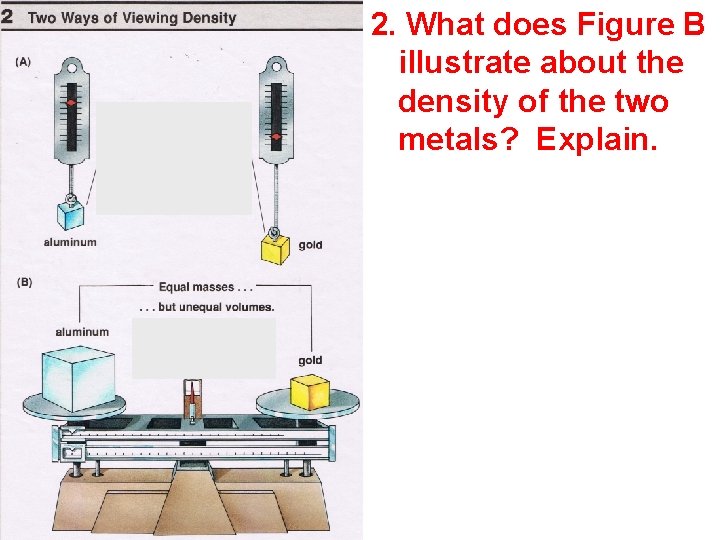
A A 2. What does Figure B illustrate about the density of the two metals? Explain.

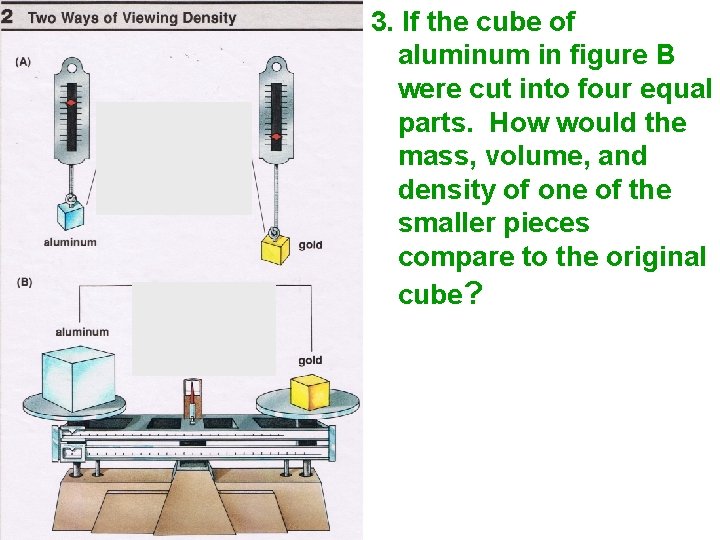
A A 3. If the cube of aluminum in figure B were cut into four equal parts. How would the mass, volume, and density of one of the smaller pieces compare to the original cube?

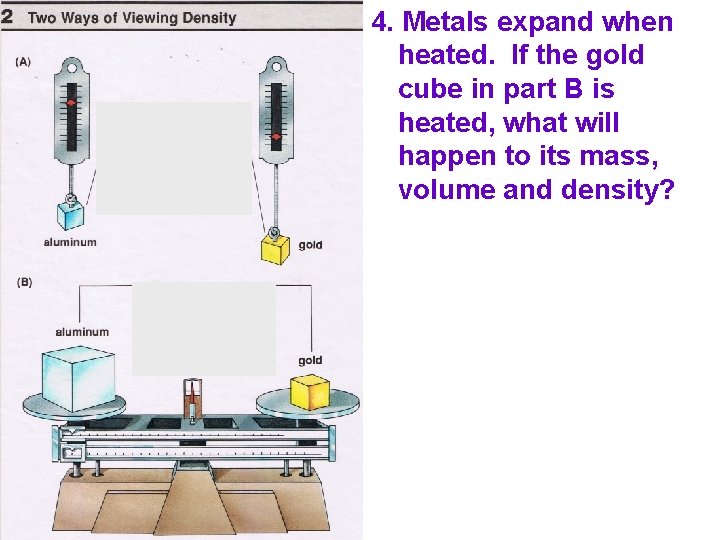
A A 4. Metals expand when heated. If the gold cube in part B is heated, what will happen to its mass, volume and density?

Does it Float? • If something floats or rises it is less dense than the material it is floating/rising in. • Objects of greater density will sink.




Density • Density is the mass per unit volume. – How much mass there is in a certain amount of space.

Density • Density is the mass per unit volume. • Density = Mass ÷ Volume

Density • Density is the mass per unit volume. • D = Mass ÷ Volume ÷

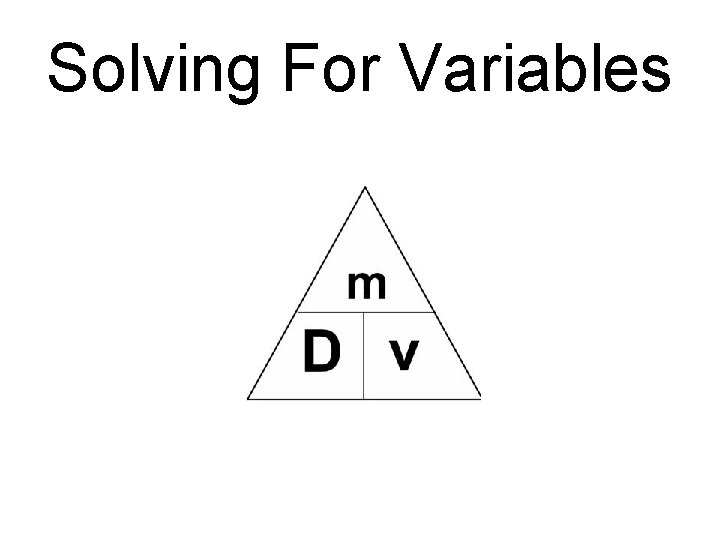
Solving For Variables

Common Volume Units

Mass = 2680 g Volume = 1450 cm 3 Illegal ivory is often detected on the basis of density. What is the density of the elephant tusk?

Mass = 2680 g Volume = 1450 cm 3 D=M÷V = 2680 g ÷ 1450 cm 3 = 1. 8482 g/cm 3 = 1. 85 g/cm 3 Illegal ivory is often detected on the basis of density. What is the density of the elephant tusk?

Calcium chloride is used as a deicer on roads in winter. Its density is 2. 50 g/cm 3. What mass of calcium chloride will the truck hold if its volume is 4560 L?

Calcium chloride is used as a deicer on roads in winter. Its density is 2. 50 g/cm 3. What mass of calcium chloride will the truck hold if its volume is 4560 L? 4560 L = 4, 560, 000 cm 3

Calcium chloride is used as a deicer on roads in winter. Its density is 2. 50 g/cm 3. What mass of calcium chloride will the truck hold if its volume is 4560 L? 4560 L = 4, 560, 000 cm 3 M=Dx. V = 2. 50 g/cm 3 x 4, 560, 000 cm 3 = 11, 400, 000 g

The density of methanol is 0. 788 g/cm 3. What is the minimum volume of a tank that can hold 795 kg of methanol.

The density of methanol is 0. 788 g/cm 3. What is the minimum volume of a tank that can hold 795 kg of methanol. 795 kg = 795, 000 g

The density of methanol is 0. 788 g/cm 3. What is the minimum volume of a tank that can hold 795 kg of methanol. 795 kg = 795, 000 g V=M÷D = 795, 000 g ÷ 0. 788 g/cm 3 = 1, 008, 883 cm 3 = 1, 010, 000 cm 3 How many liters is this?

The density of methanol is 0. 788 g/cm 3. What is the minimum volume of a tank that can hold 795 kg of methanol. 1, 010, 000 cm 3 ÷ 1000 cm 3/L = 1010 L