Arboviruses ArthropodBorne Viruses The term ARBO is an
Arboviruses Arthropod-Borne Viruses

The term ARBO is an abbreviation of Arthropod BOrne. Group of viruses that are transmitted to vertebrates (people and mammals) by blood-feeding Infected insect bites (arthropods) and takes a blood meal

WHO (1967) Viruses which are maintained in nature principally, or to an important extent through biological transmission between susceptible vertebrate hosts by Hematophagous (feeding on blood) arthropods. • Infected Arthropods • Virus multiply in the tissues of arthropods • Extrinsic incubation • Bites new vertebrates

Classification • Historically important is Yellow fever, Dengue & Sandfly fever in 1951 to 40+ in 1975, > 500 at present. • Many unclassified But 11 major groups, 18 smaller groups named after the place of discovery. • Only a small number capable of infecting human & much smaller capable of producing disease.
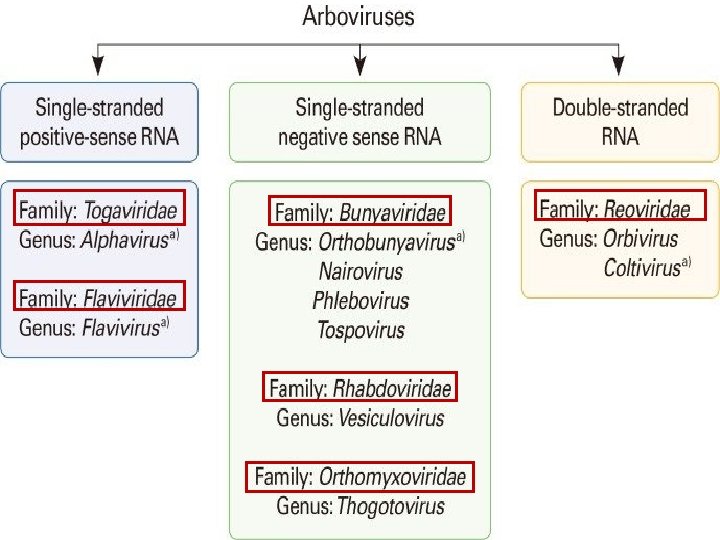

Based on clinical syndrome Febrile Group- Sindbis, Chikungunya, Dengue viruses Hemorrhagic Fevers- Dengue, Chikungunya, Kyasanur Forest Disease Encephalitidis- Japanese Encephalitis

Prevalent Viruses in Indian subcontinent Umbre, Dhori, Sathuperi, Kalsodi
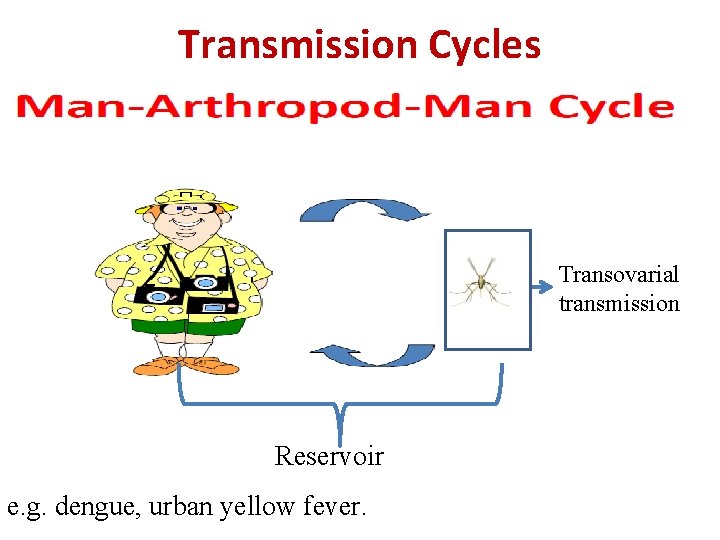
Transmission Cycles Transovarial transmission Reservoir e. g. dengue, urban yellow fever.
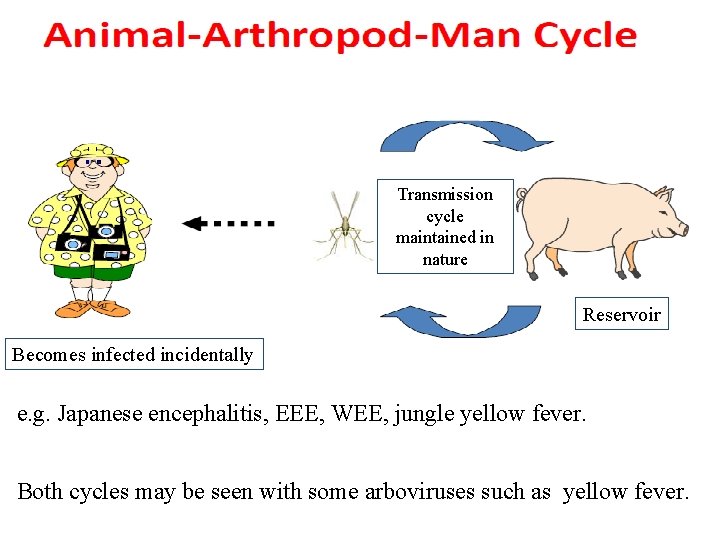
Transmission cycle maintained in nature Reservoir Becomes infected incidentally e. g. Japanese encephalitis, EEE, WEE, jungle yellow fever. Both cycles may be seen with some arboviruses such as yellow fever.

Animal Reservoirs Animals implicated as reservoirs Birds Disease Japanese encephalitis, St Louis encephalitis, EEE, WEE Pigs Monkeys Rodent Japanese encephalitis Yellow Fever VEE, Russian Spring-Summer encephalitis In many cases, the actual reservoir is not known.

Arthropod Vectors Aedes Aegyti Japanese encephalitis, dengue, yellow fever, St. Louis encephalitis, EEE, WEE, VEE etc. Exoskeleton, Segmented Bodies. . Jointed Appendages. , Bilateral Symmetry. Open Circulatory System. Assorted Ticks Crimean-Congo hemorrhagic fever, various tickborne encephalitis's etc. Culex Mosquito Phlebotmine Sandfly Sicilian sandfly fever, Rift valley fever.

Diseases Caused Three clinical syndromes: 1. Fevers of an undifferentiated type with or without a maculopapular rash , usually benign; 2. Encephalitis (inflammation of the brain), often with a high case-fatality rate; and 3. Hemorrhagic fevers, also frequently severe and fatal. Some arboviruses may be associated with more than one syndrome, eg, dengue.

Viral hemorrhagic fevers

Definition Severe multisystem syndrome. Characteristically, the overall vascular system is damaged. Often accompanied by hemorrhage (bleeding); Hemorrhagic fever viruses can cause relatively mild illnesses, BUT many of these viruses cause severe, life-threatening disease.

Agents • Viruses of four distinct families – Arenaviruses – Filoviruses – Bunyaviruses – Flaviviruses Arenaviridae Filoviridae Bunyaviridae Junin Ebola Crimean- Congo H. F. Kyasanur Forest Disease Machupo Marburg Hantavirus Omsk H. F. Rift Valley fever Yellow Fever Sabia Guanarito Lassa Flaviviridae Dengue

General features • RNA viruses, covered, or enveloped, in a fatty (lipid) coating. • Survival is dependent on an animal or insect host, (natural reservoir). • Geographically restricted to the areas where their host species live. • Humans are not the natural reservoir for any of these viruses.

• Humans infection occurs through contact with infected hosts , after the accidental transmission from the host, humans can transmit the virus to one another. • With a few noteworthy exceptions, there is no cure or established drug treatment for VHFs. • Rodents and arthropods are the main reservoirs and vectors for viruses causing VHFs.

• However, the hosts of some viruses remain unknown -Ebola and Marburg viruses are well-known examples. • Role in bioterrorism (biological weapons due to high morbidity and mortality and due to aerosol transmission of most of them except dengue fever virus) Atigenic structure: 3 antigens : haemagglutinins, complement fixing and neutralising antigens. Important in serological studies. Cross reaction occur among arboviruses.

TRANSMISSION A. Natural infection of humans (mode of transmission): 1. Bite of infected arthropod (ticks or mosquitoes). 2. Aerosol from infected rodent excreta. 3. Direct contact with infected animals/carcasses or fomities.

B- Human to Human & Nosocomial Transmission Possible for most HFVs - Direct contact with infected blood & body fluids (Hospital acquired infections). - Mucous membrane contact, - Aerosolized, (airborne in Ebola, Lassa, Junín, & may be yellow fever), - Semen, - vomitus – Sweat.

PATHOGENESIS Incubation period ranging from 2 to 21 days for all of them. Transmission • Multipliciation • viraemia • The target organ is the vascular bed (hemorrhage). • The replication of virus is intracellularly. • Cytokine release leads to shock and hypotension. • Affects platelet functions and numbers (thrombocytopenia). • Affects bone marrow and clotting factors.

Clinical Presentation • Clinical manifestations nonspecific, vary by agent. • Incubation period 2 -21 days, depending on agent. • Onset typically abrupt with filoviruses, flaviviruses, and Rift Valley fever. • Onset more insidious with arenaviruses.

Prodromal illness lasting < 1 week may include – High fever – Dizziness – Headache – Muscle aches – Malaise – Joint pain – Weakness – Nausea – Exhaustion – Non-bloody diarrhea

Clinical Signs • Flushing, conjunctival injection (“red eye”) • Pharyngitis • Rash • Edema • Hypotension • Shock • Mucous membrane bleeding

Clinical Identification of Suspected Cases • Clinical criteria: – Temperature 101 F(38. 3 C) for <3 weeks – Severe illness and no predisposing factors for hemorrhagic manifestations – 2 or more of the following: - Hemorrhagic or purple rash - Epistaxis - Hematemesis - Hemoptysis - Blood in stools - Other hemorrhagic symptoms - No established alternative diagnosis

Clinical Description Common Clinical Features of VHFs
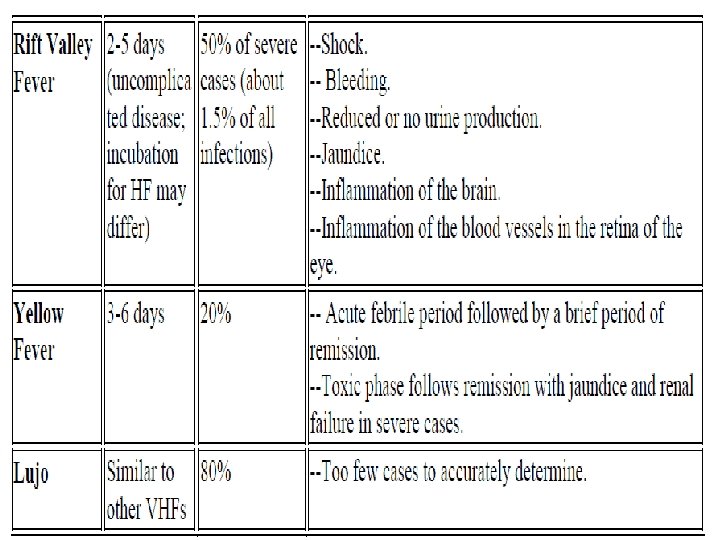

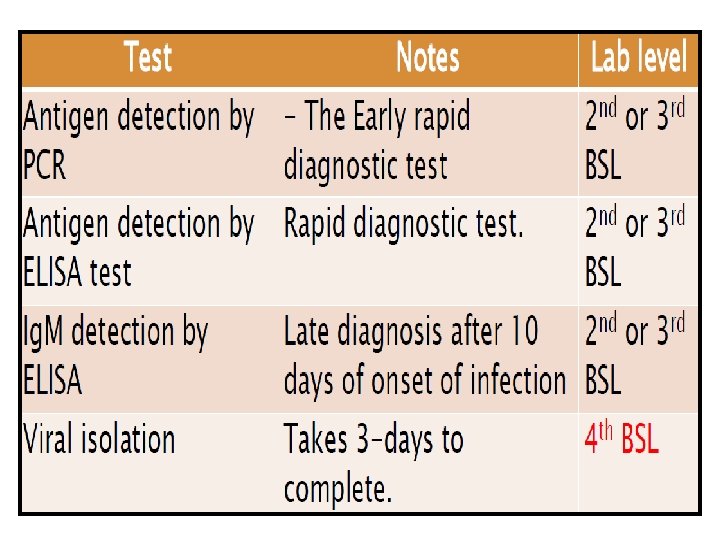
Laboratory criteria for diagnosis Laboratory diagnosis may be made by any of the following procedures: • Isolation of the virus by cell culture. • Detection of viral RNA by use of PCR. • Detection of viral antigens by use of antigen-capture EIA. • Detection of Ig. M , Ig. G antibodies to the virus.

Samples required Whole blood, Serum or plasma , clot Tissues Liver biopsy Skin biopsy (any site) Tissue biopsy from fatal cases (spleen, lung, heart, kidney) Do not centrifuge suspected VHF specimens because this increases risk to the lab worker. Place specimens in plastic tubes for shipping and storage and be sure that the tubes are sealed and properly labeled.

• VIRAL ISOLATION = GOLD STANDARD technique Filtration of the inoculum on 0. 22 µm Inoculation of cell cultures Visualization of cytopathogenic effect. Chick embryo fibroblast, verro cell lines, insect tissue, Mosquito cell line, yolk sac, chorioallantoic membrane, suckling mice.

Non specific Lab Abnormalities • • • Leukopenia Anemia Hemoconcentration Thrombocytopenia Elevated LFTs Azotemia • Coagulation abnormalities (Prolonged bleeding time, PTT) • Urine analysis Hematuria, proteinuria, oliguria
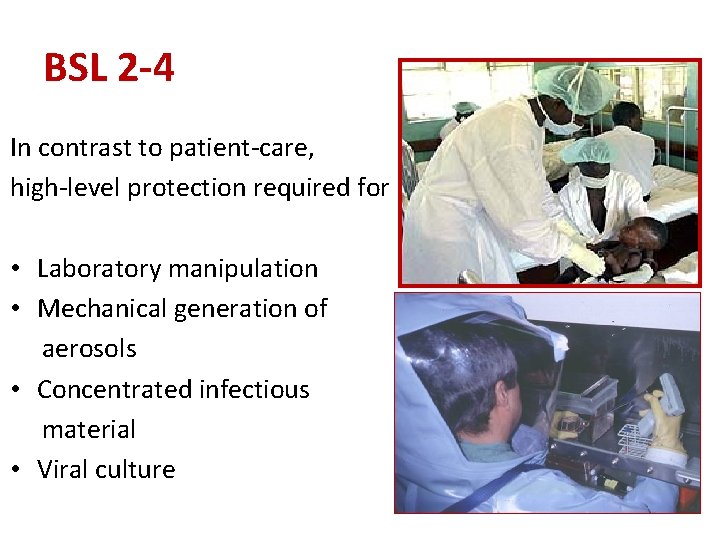
BSL 2 -4 In contrast to patient-care, high-level protection required for • Laboratory manipulation • Mechanical generation of aerosols • Concentrated infectious material • Viral culture

Treatment • Supportive treatment • Correct coagulopathies as needed • Ribavirin – Not approved by FDA – Effective in some individuals – Arenaviridae and Bunyaviridae only • Convalescent-phase plasma w/in 8 d of onset for AHF – Argentine HF, Bolivian HF and Ebola • Strict isolation of affected patients is required • Report to health authorities

Prevention and control • Avoid contact with host species – Rodents and Insects • Vaccine available for Yellow fever • Experimental vaccines under study – Argentine HF, Rift Valley Fever, – Hantavirus and Dengue HF • If human case occurs – Decrease person-to-person transmission – Isolation of infected individuals

• Protective clothing – Disposable gowns, gloves, masks and shoe covers, protective eyewear when splashing might occur, or if patient is disoriented or uncooperative. • WHO and CDC developed manual – “Infection Control for Viral Hemorrhagic Fevers ”
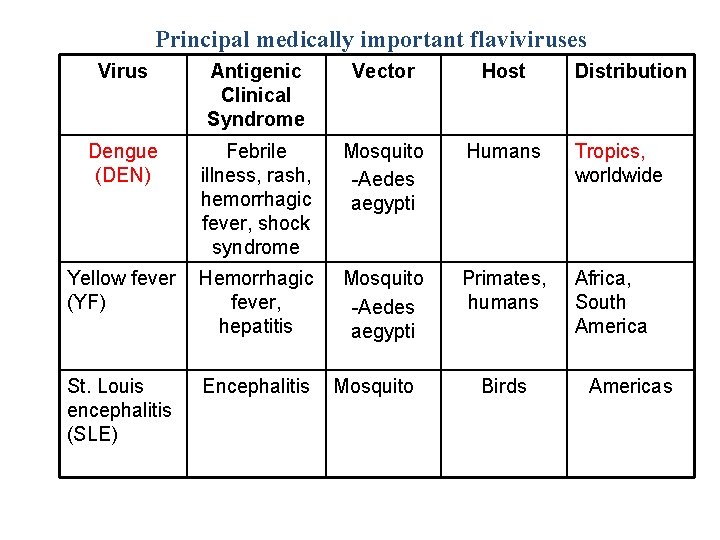
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Dengue (DEN) Febrile illness, rash, hemorrhagic fever, shock syndrome Mosquito -Aedes aegypti Humans Tropics, worldwide Yellow fever (YF) Hemorrhagic fever, hepatitis Mosquito -Aedes aegypti Primates, humans Africa, South America St. Louis encephalitis (SLE) Encephalitis Mosquito Birds Distribution Americas
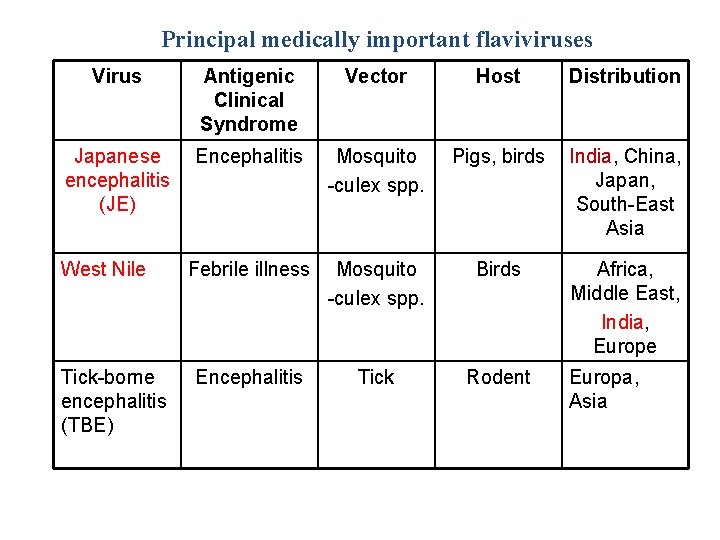
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Distribution Japanese encephalitis (JE) Encephalitis Mosquito -culex spp. Pigs, birds India, China, Japan, South-East Asia Febrile illness Mosquito -culex spp. Birds Africa, Middle East, India, Europe Encephalitis Tick Rodent West Nile Tick-borne encephalitis (TBE) Europa, Asia
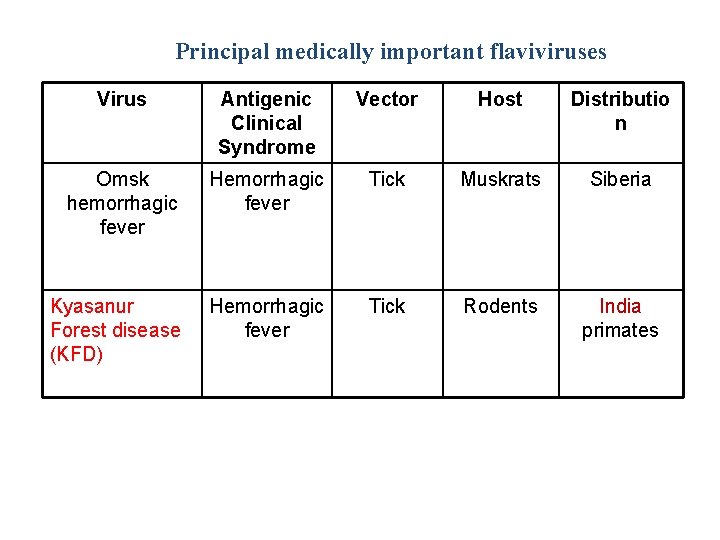
Principal medically important flaviviruses Virus Antigenic Clinical Syndrome Vector Host Distributio n Omsk hemorrhagic fever Hemorrhagic fever Tick Muskrats Siberia Kyasanur Forest disease (KFD) Hemorrhagic fever Tick Rodents India primates
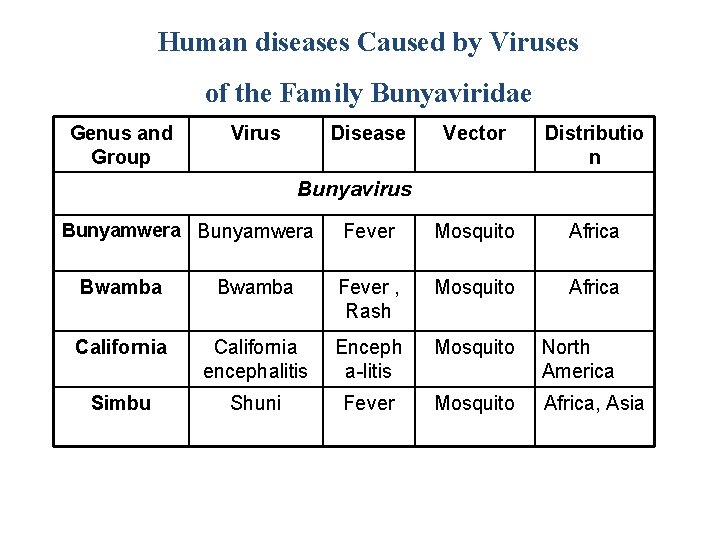
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distributio n Fever Mosquito Africa Bunyavirus Bunyamwera Bwamba Fever , Rash Mosquito Africa California encephalitis Enceph a-litis Mosquito North America Simbu Shuni Fever Mosquito Africa, Asia
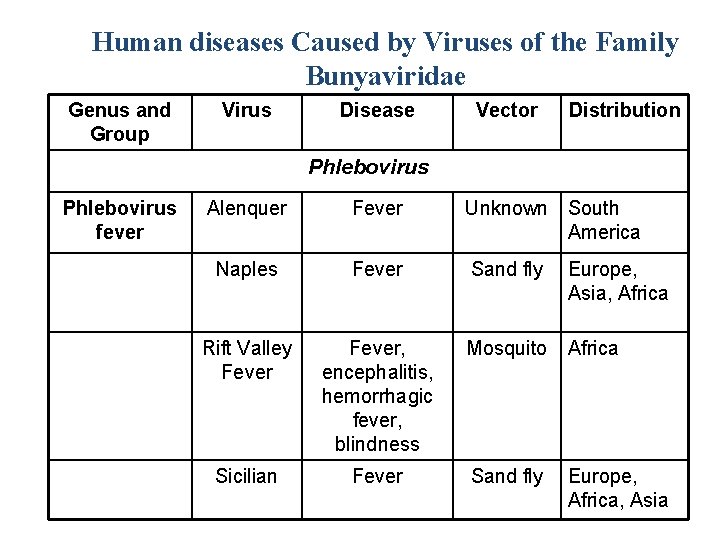
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Phlebovirus fever Alenquer Fever Unknown South America Naples Fever Sand fly Europe, Asia, Africa Rift Valley Fever, encephalitis, hemorrhagic fever, blindness Mosquito Africa Sicilian Fever Sand fly Europe, Africa, Asia

Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Nairovirus Crimean. Congo Nairobi sheep disease Crimean. Congo hemorrhagic fever Hemorrhagic fever Tick Nairobi sheep disease Fever Tick Africa, Asia
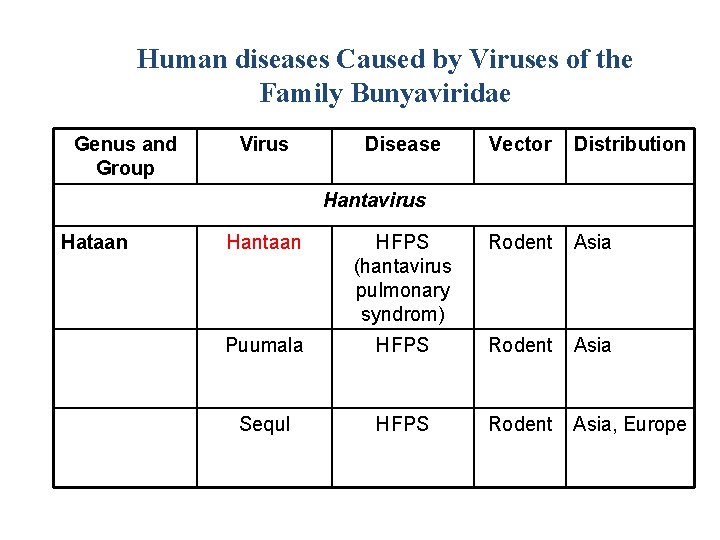
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distribution Hantavirus Hataan Hantaan HFPS (hantavirus pulmonary syndrom) Rodent Asia Puumala HFPS Rodent Asia Sequl HFPS Rodent Asia, Europe
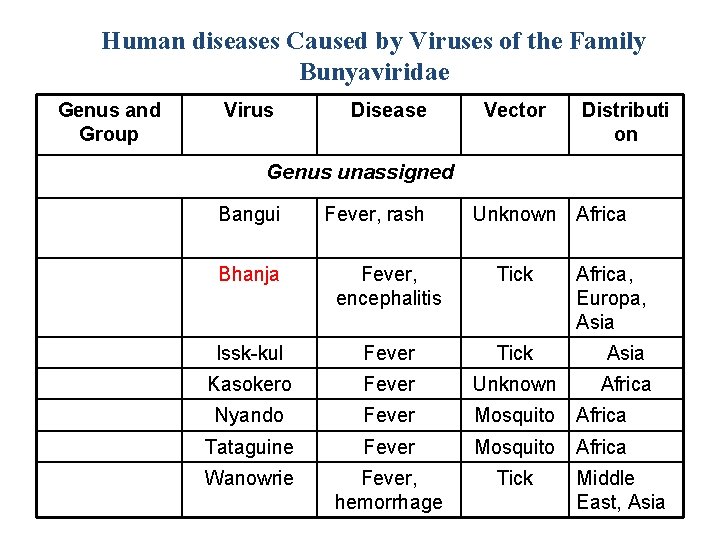
Human diseases Caused by Viruses of the Family Bunyaviridae Genus and Group Virus Disease Vector Distributi on Genus unassigned Bangui Fever, rash Unknown Africa Bhanja Fever, encephalitis Tick Africa, Europa, Asia Issk-kul Fever Tick Asia Kasokero Fever Unknown Africa Nyando Fever Mosquito Africa Tataguine Fever Mosquito Africa Wanowrie Fever, hemorrhage Tick Middle East, Asia
Protective equipment worn by a nurse during Ebola outbreak in Zaire Thank you , 1995
- Slides: 45