ARBOROVIRUSES Mohammed ElKhateeb 2 nd April 2015 Overview

ARBOROVIRUSES Mohammed El-Khateeb 2 nd April 2015

Overview Ø Etiology Ø Epidemiology and history Ø Pathogenesis and Pathology Ø Clinical Manifestation Ø Diagnosis Ø Treatment Ø Prevention and Control 2

Arthropod-Borne Viruses arbov Arthropod-borne viruses ( iruses) are More Than 600 Different viruses that can be transmitted to man by arthropod vectors. The WHO definition is as follows: “Viruses maintained in nature principally, or to an important extent, through biological transmission between susceptible vertebrate hosts by hematophagus arthropods or through transovarial and possibly venereal transmission in arthropods. ”

Arthropod-Borne Viruses Arboviruses belong to three families 1. Togaviruses e. g. EEE, WEE, and VEE 2. Bunyaviruses e. g. Sand fly Fever, Rift Valley Fever, Crimean- Congo Hemorrhagic Fever 3. Flaviviruses e. g. Yellow Fever, dengue, Japanese Encephalitis

Arthropod-Borne Viruses • Togaviridae • Flaviviridae a) Sindbis b) Semliki Forest c) Venezuelan equine encephalitis d) Eastern equine encephalitis e) Western equine encephalitis f) Chikungunya a) Dengue b) Yellow fever c) Japanese encephalitis d) West Nile encephalitis e) St. Louis encephalitis f) Russian spring-summer encephalitis g) Powassan encephalitis

Negatively Stained Virions of Semliki Forest Virus Togaviridae, genus Alphavirus

Arthropod-Borne Viruses FAMILY ENVELOPE SYMMETRY GENOME Icosahedral 40 -75 nm 40 -45 nm ss. RNA (+ve) Yes, Heat, detergent labile Helical ss. RNA (-ve) segmented no Icosahedral ds. RNA, segmented Yes. Heat, detergent labile

VIRAL STRUCTURE It has three proteins • Envelope protein • Core protein • Membrane protein

General Antigenic Properties of Flaviviral Proteins - 1 Ø E-glycoprotein § Most important flaviviral antigen § Very immunogenic § Most serological assays detect reactivity with this protein Ø Capsid § Elicits primarily group-reactive antibody Ø M-protein § Very small (75 a. a. ) § Embedded in the virion envelope membrane and not highly immunogenic
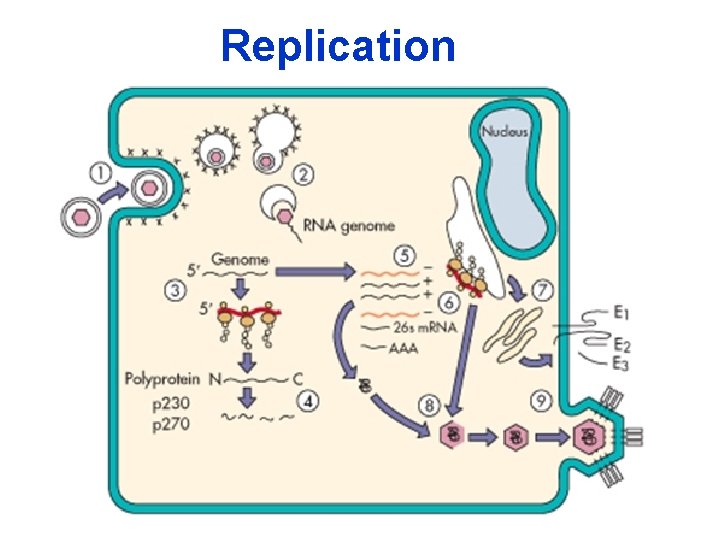
Replication 10

Arthropod Vectors Mosquitoes Japanese encephalitis, dengue, yellow fever, St. Louis encephalitis, EEE, WEE, VEE etc. Ticks Crimean-Congo haemorrhagic fever, various tick-borne encephalitides etc. Sandflies Sicilian sandfly fever, Rift valley fever.

Examples of Arthropod Vectors Aedes Aegyti Culex Mosquito Assorted Ticks Phlebotmine Sandfly

Animal Reservoirs In many cases, the actual reservoir is not known. The following animals are implicated as reservoirs Birds Japanese encephalitis, St Louis encephalitis, EEE, WEE Pigs Japanese encephalitis Monkeys Yellow Fever Rodents VEE, Russian Spring-Summer encephalitis

Transmission • The most common route of infection is • bite of infectious mosquito Other transmission modes where revealed in 2002 such as § § § Blood Transfusion Organ Transplantation Intrauterine Percutaneous exposure (occ. exposure) Breastmilk (probable)

Transmission Cycles Man - arthropod -man e. g. dengue, urban yellow fever. Reservoir may be in either man or arthropod vector. In the latter transovarial transmission may take place. Animal - arthropod vector - man e. g. Japanese encephalitis, EEE, WEE, jungle yellow fever. The reservoir is in an animal. The virus is maintained in nature in a transmission cycle involving the arthropod vector and animal. Man becomes infected incidentally. Both cycles may be seen with some arboviruses such as yellow fever.

Man-Arthropod-Man Cycle Man - arthropod -man e. g. dengue, urban yellow fever. Reservoir may be in either man or arthropod vector. In the latter transovarial transmission may take place.
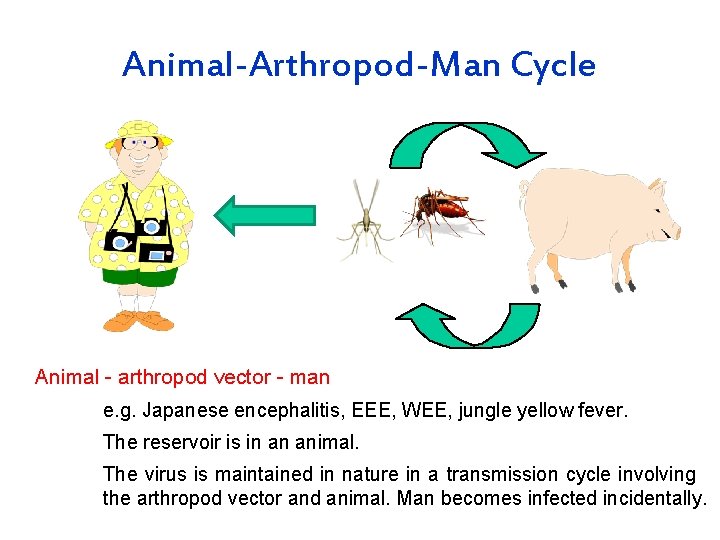
Animal-Arthropod-Man Cycle Animal - arthropod vector - man e. g. Japanese encephalitis, EEE, WEE, jungle yellow fever. The reservoir is in an animal. The virus is maintained in nature in a transmission cycle involving the arthropod vector and animal. Man becomes infected incidentally.

Epidemiologic Feature Ø The major outbreaks coincided with the heavy rainfall or floods. Ø Seasonal: more common in summer, July to October Ø Infection provides life long immunity. Ø Worldwide distribution Ø More than 530 species, 150 pathogen to man 18

Pathogenesis The nature of flavivirus disease is determined primarily by • The specific tropisms of the individual virus type • The concentration of infecting virus • Individual host response to the infection 19
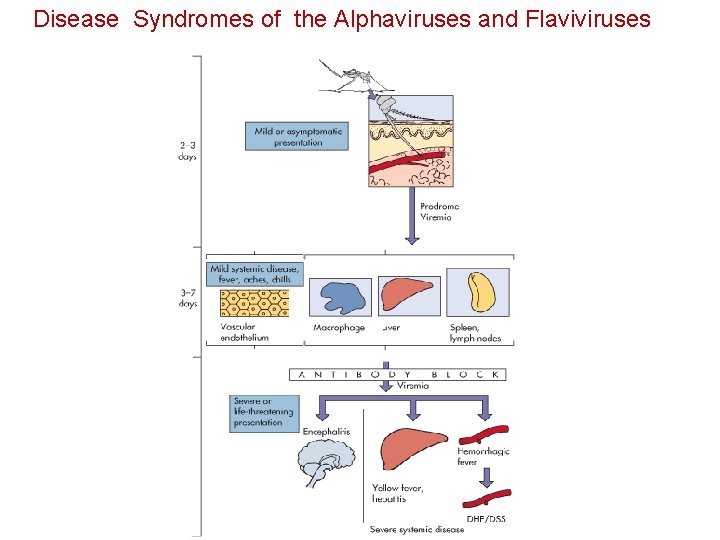
Disease Syndromes of the Alphaviruses and Flaviviruses

Pathogenesis Virus Mononuclear Phagocyte Blood Circulation Viremia Adequate Immunological Response Subclinical or mild Systemic disease Weak Immunological Response Invades the CNS induce mortality 21
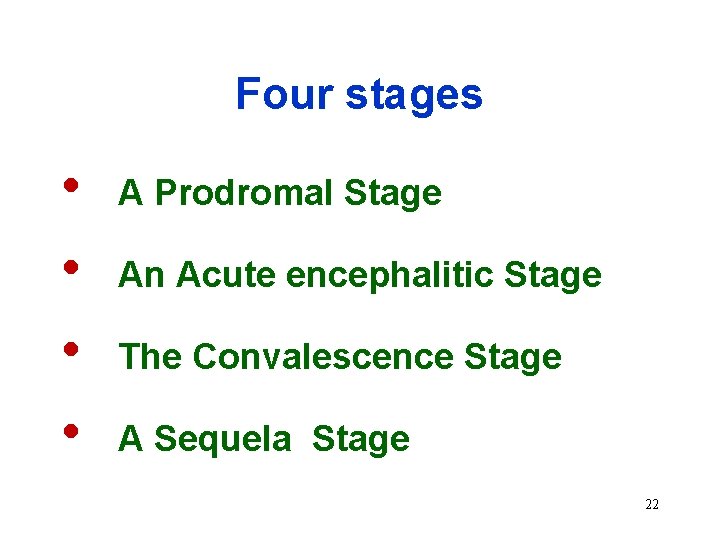
Four stages • • A Prodromal Stage An Acute encephalitic Stage The Convalescence Stage A Sequela Stage 22

Diseases Caused • Fever and rash - this is usually a non-specific illness resembling a number of other viral illnesses such as influenza, rubella, and enterovirus infections. The patients may go on to develop encephalitis or haemorrhagic fever. • Encephalitis - e. g. EEE, WEE, St Louis encephalitis, Japanese encephalitis. • Haemorrhagic fever - e. g. yellow fever, dengue, Crimean-Congo haemorrhagic fever.

Pathogenesis and immunity Ø Besides viral receptor, virus may attach to Fc receptor (macrophages and monocytes) via Ab to result in an increase of virus infection. Ø Antibody is produced to block infection. However, non-neutralizing Ab may have antibody dependent enhancement (ADE) effect to enhance virus replication by hundred folds.

Arthropod-Borne Viruses a) Dengue b) Yellow fever c) Chikungunya

Fever/Rash/Arthritis 1. Triad of fever/rash/arthritis is characteristic of Chikungunya, o’nyong-nyong, Ross River, Mayaro, and Sindbis viruses 2. Symptoms generally appear after 2 -3 days incubation a) fever, chills, myalgia b) polyarthralgia mainly affecting small joints c) maculopapular rash 3. Arthritis generally resolves in a few weeks, but may persist for months, or years in some cases.

Arthropod-Borne Viruses Encephalitis • • West Nile encephalitis Japanese encephalitis St. Louis encephalitis Russian spring-summer encephalitis Powassan encephalitis Venezuelan equine encephalitis Eastern equine encephalitis Western equine encephalitis

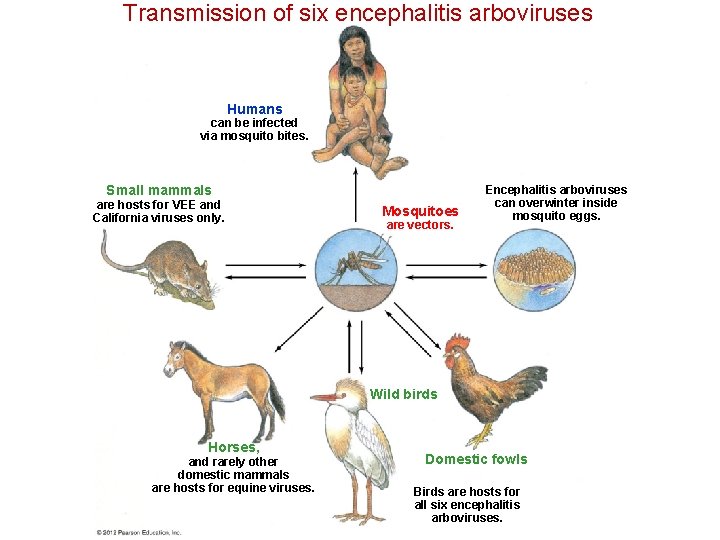
Transmission of six encephalitis arboviruses Humans can be infected via mosquito bites. Small mammals are hosts for VEE and California viruses only. Mosquitoes are vectors. Encephalitis arboviruses can overwinter inside mosquito eggs. Wild birds Horses, and rarely other domestic mammals are hosts for equine viruses. Domestic fowls Birds are hosts for all six encephalitis arboviruses.
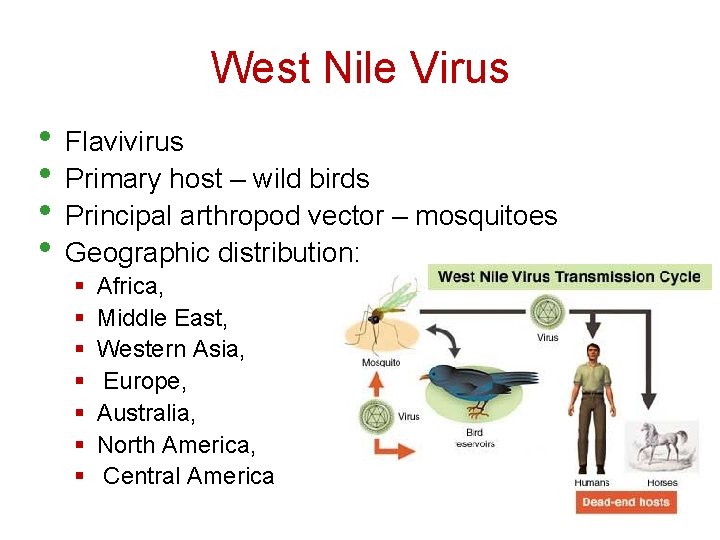
West Nile Virus • Flavivirus • Primary host – wild birds • Principal arthropod vector – mosquitoes • Geographic distribution: § § § § Africa, Middle East, Western Asia, Europe, Australia, North America, Central America

Encephalitis 1. Small proportion of individuals infected (a few days after the onset of fever) may develop drowsiness, neck rigidity, progressing to confusion, paralysis, convulsions and coma. 1. Case-fatality rates average 10 – 20 % (higher in elderly). 3. Survivors may be left with permanent neurologic sequelae such as mental retardation, epilepsy, paralysis, deafness, and blindness.

Diagnosis Ø Materials of epidemiology Ø Clinical Ø Laboratory Tests – Tentative diagnosis § Antibody titer: HI, IF, CF, ELISA § JE-specific Ig. M in serum or CSF – Definitive diagnosis § Virus isolation: Blood, CSF sample, brain 32

Diagnosis § Serology - usually used to make a diagnosis of arbovirus infections. Antibody titer: HI, IF, CF, ELISA • Culture - a number of cell lines may be used, including mosquito cell lines. However, it is rarely carried out since many of the pathogens are group 3 or 4 pathogens. (Blood, CSF sample, brain) • Direct detection tests - e. g detection of antigen and nucleic acids are available but again there are safety issues.

Prevention • Surveillance - of disease and vector populations • Control of vector - pesticides, elimination of breeding grounds • Personal protection - screening of houses, bed nets, insect repellants • Vaccination - available for a number of arboviral infections e. g. Yellow fever, Japanese encephalitis, Russian tick-borne encephalitis

Treatment/Vaccines/Control measures A. Encephalitis 1. Vaccines exist for a number of these viruses, but are used mainly for horses, at risk lab workers, and some fowl known to be intermediate hosts 2. Control of mosquitoes is major countermeasure. B. Yellow Fever 1. Live attenuated virus vaccine. Used when going to endemic areas
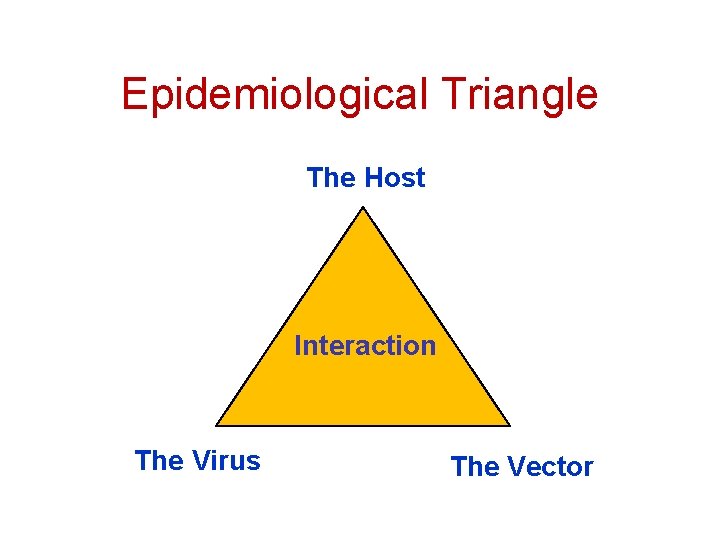
Epidemiological Triangle The Host Interaction The Virus The Vector

Prevention Vector (Mosquito) control – Eliminate mosquito breeding areas: Chemical larvicides, Biolarvicides, Environmental management – Adult and larval control: Anti-larval treatment Vaccination Personal protective measures – Avoid prime mosquito hours: from dusk to dawn – Indoor spray and fogging: Use of Insecticide 37

Repellent Guidance • Skin Ø DEET still “gold standard” • Both new additions good for shorter term protection Ø Picaridin • Roughly equivalent to DEET at same concentration • Only a 7% product currently sold in US Ø Oil of lemon eucalyptus • Plant based • 30% product similar to low concentration DEET • Not for kids <3 years old • Clothing Ø Permethrin

Arboviruses Structure Positive sense ss. RNA genome, icosahedral nucleocapsid, enveloped • Pathogenesis • Transmitted by bite of insect from host species; sylvan and urban cycles • Replication in cytoplasm; budding • Viremia to target tissue • Influenza-like initial symptoms; different viruses cause encephalitis, hemorrhagic fever, hepatitis, rash, arthritis Diagnosis • Serology and nucleic acid Treatment/prevention • No human vaccines except for Yellow Fever live attenuated vaccine, control of insect population
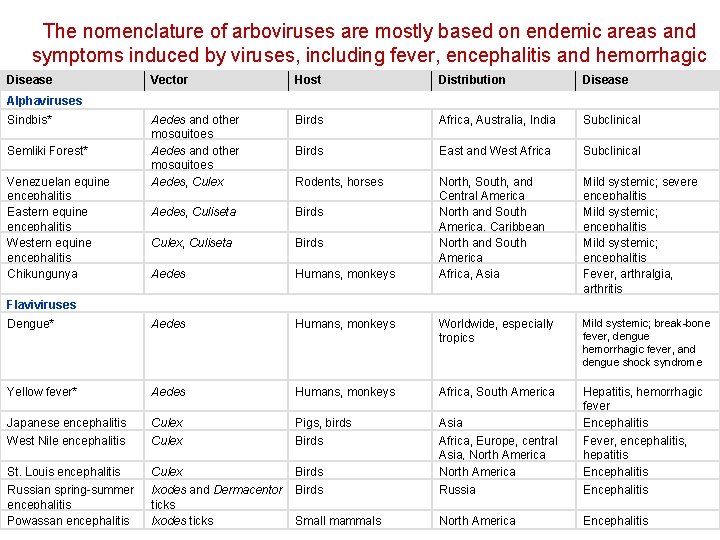
The nomenclature of arboviruses are mostly based on endemic areas and symptoms induced by viruses, including fever, encephalitis and hemorrhagic Disease Vector Host Distribution Disease fever Alphaviruses Sindbis* Aedes and other mosquitoes Aedes, Culex Birds Africa, Australia, India Subclinical Birds East and West Africa Subclinical Rodents, horses Aedes, Culiseta Birds Culex, Culiseta Birds Aedes Humans, monkeys North, South, and Central America North and South America, Caribbean North and South America Africa, Asia Mild systemic; severe encephalitis Mild systemic; encephalitis Fever, arthralgia, arthritis Dengue* Aedes Humans, monkeys Worldwide, especially tropics Mild systemic; break-bone fever, dengue hemorrhagic fever, and dengue shock syndrome Yellow fever* Aedes Humans, monkeys Africa, South America Japanese encephalitis Culex Pigs, birds Asia Hepatitis, hemorrhagic fever Encephalitis West Nile encephalitis Culex Birds St. Louis encephalitis Culex Birds Africa, Europe, central Asia, North America Fever, encephalitis, T 2 hepatitis Encephalitis Russian spring-summer encephalitis Powassan encephalitis lxodes and Dermacentor ticks lxodes ticks Birds Russia Encephalitis Small mammals North America Encephalitis Semliki Forest* Venezuelan equine encephalitis Eastern equine encephalitis Western equine encephalitis Chikungunya Flaviviruses

RETROVIRUSES

Retro. Viruses • RNA Viruses • DNA From RNA by Reverse Transcriptase • Insertion of new DNA into cellular DNA • Hijacks the cell machinery to make VIRUS • The virus only grows on T 4 cells that are proliferating in response to an immune stimulus • Difficult to grow in culture • Robert Gallo : HTLV-3 • Luc Montagnier: LAV • Human Immunodeficiency Virus (HIV) 42

Introduction to Retroviruses I. Overview of retroviruses A. History B. Shared characteristics C. Classification II. Function of different regions of the retroviral genome A. Cis acting elements B. Gag proteins C. Pol proteins D. Env proteins III. Details of life cycle: A. Early stage B. Late stage 43

General Introduction to Retroviruses § Ubiquitous; found in all vertebrates § Large, diverse family § Includes HIV, FIV and Fe. LV Definition and classification of retroviruses § Common features- structure, composition and replication § Distinctive life cycle: RNA-DNA-RNA § Nucleic acid is RNA in virus, and DNA in infected cell Transmission may be either: § Horizontal - by infectious virus (exogenous virus) or vertical- by proviruses integrated in germ cells (endogenous virus) § Can transmit either as free viral particle or (for some retroviruses) through cell contact 44

A Little Retrovirus History 1960 s: Howard Temin: suggested DNA “provirus” was part ofreplication cycle: RNA � DNA � RNA �Protein – Won Nobel prize (with Baltimore) in 1970 after they independently discovered RT activity in infected cells 1980: Human T-cell leukemia virus discovered, the first pathogenic human retrovirus. 1982: Human immunodeficiency virus discovered. 1990: First gene therapy trial involving the use of retroviral-based vectors in patient with a deficiency in adenosine deaminase (ADA). 2006: Xenotropic murine leukemia-related virus discovered. 45

Retroviruses • Strange Viruses ? At time-“central dogma of molecular biology”: DNA� RNA� Protein So. . RNA couldn’t be template for DNA § Unique replication cycle based on reverse transcription. Flow of information from RNA to DNA. (1971 Nobel Prize Temin / Baltimore) § Retroviruses have been isolated from numerous species including chickens (RSV), mice (MLV), monkeys (SIV), and humans (HIV, HTLV) § “Simple Retroviruses” encode only the genes gag, pol, and env (RSV) § “Complex Retroviruses” encode in addition regulatory genes (HIV) • Retroviruses are single-stranded RNA viruses that replicate through a double-stranded DNA intermediate.

• They are association with the development of tumors in their host organisms. • Study of these viruses eventually led to the discovery and development of the oncogene theory of tumorgenesis • Some of the viruses actually contained oncogenes within their genomes, while others interacted with oncogenes in either a direct or indirect way to contribute to tumor formation.

• Historically, because of their pattern of pathogenicity, these viruses were grouped into three subfamilies: 1. The acutely oncogenic retroviruses, or oncoretroviruses (such as those described above) 2. The lentiviruses (associated with “slow” diseases or those with long latent periods) 3. The spumaviruses (“foamy” viruses, named because of the pathogenic changes observed in infected cells).

THE OLD NOMENCLATURE Members: 1. Oncogenic viruses (Oncoviruses) (endogenous) § § § Avian oncoviruses: RSV, AMV, AEV, RAVs [ RAV- 0 ; RAV-1 ]. Murine oncoviruses: e. g. , Mo. MLV, A-Mu. LV. Mammalian oncoviruses: e. g. , Fe. LV, Ha. MSV, SSV. Mouse Mammary Tumor Virus (MMTV); [ the only B- type particle ]. Mason-Pfizer Monkey Virus (MPMV); [ one of the few D-type particles ]. Human T-Cell Lymphotropic Virus ( HTLV-1 & 2 ). 2. Lentiviruses - HIV-1, HIV-2, SIV, FIV, EIAV, CAEV. 3. Spumaviruses – HFVs, SFV. 2 and 3 non endogenous
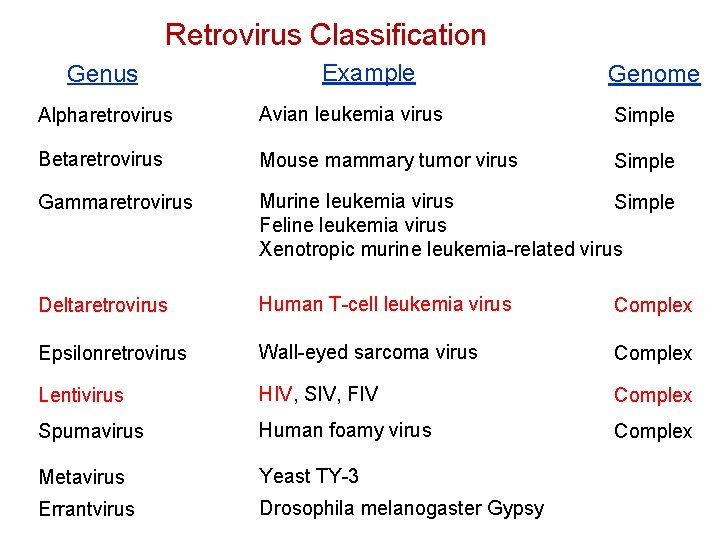
Retrovirus Classification Genus Example Genome Alpharetrovirus Avian leukemia virus Simple Betaretrovirus Mouse mammary tumor virus Simple Gammaretrovirus Murine leukemia virus Simple Feline leukemia virus Xenotropic murine leukemia-related virus Deltaretrovirus Human T-cell leukemia virus Complex Epsilonretrovirus Wall-eyed sarcoma virus Complex Lentivirus HIV, SIV, FIV Complex Spumavirus Human foamy virus Complex Metavirus Yeast TY-3 Errantvirus Drosophila melanogaster Gypsy

Retrovirus Structural Overview Enveloped virus with lipid bilayer and viral spike glycoproteins. Have outer matrix protein and inner core capsid containing viral genome. Genome: Two copies of single stranded positive-stranded RNA (8 -10 kb). All retroviruses contain gag, pol and env genes. Simple - only gag, pol, env Complex - additional genes involved in replication. Reverse transcriptase to generate DNA Viral genes are integrated into host genome. Progeny virus produced using host cell transcriptional and translational machinery. 51
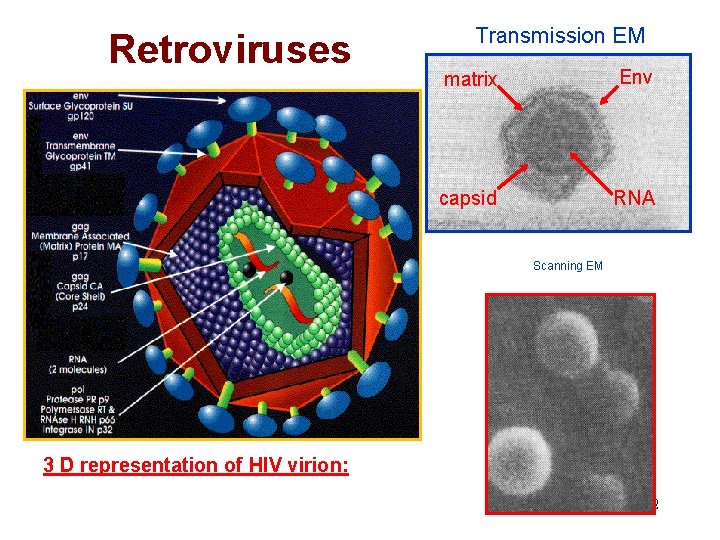
Retroviruses Transmission EM matrix Env capsid RNA Scanning EM 3 D representation of HIV virion: 52
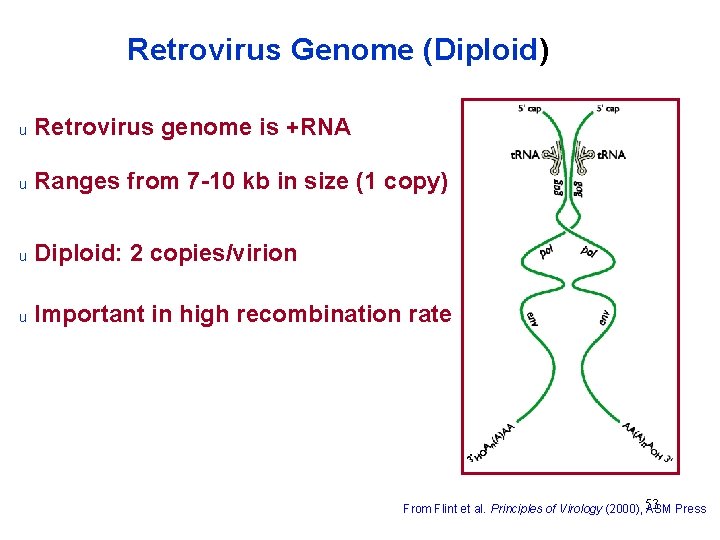
Retrovirus Genome (Diploid) u Retrovirus genome is +RNA u Ranges from 7 -10 kb in size (1 copy) u Diploid: 2 copies/virion u Important in high recombination rate 53 From Flint et al. Principles of Virology (2000), ASM Press
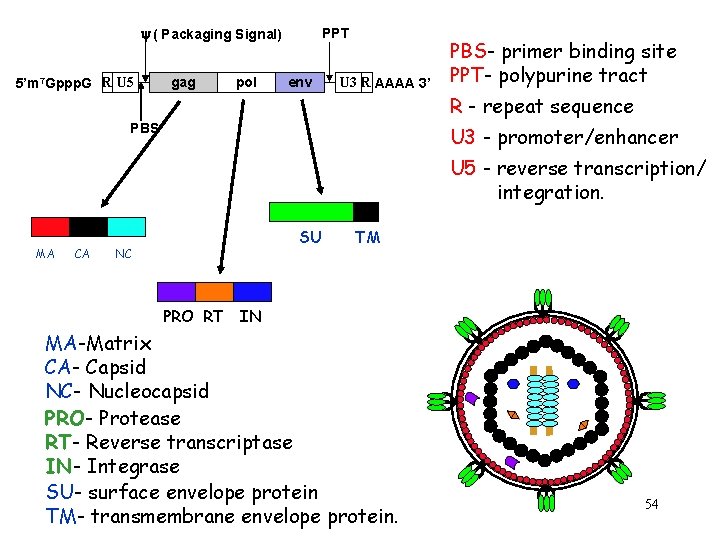
y ( Packaging Signal) 5’m 7 Gppp. G R U 5 gag pol PPT env U 3 R AAAA 3’ PBS- primer binding site PPT- polypurine tract R - repeat sequence PBS U 3 - promoter/enhancer U 5 - reverse transcription/ integration. CA MA CA SU NC TM PRO RT IN MA-Matrix CA- Capsid NC- Nucleocapsid PRO- Protease RT- Reverse transcriptase IN- Integrase SU- surface envelope protein TM- transmembrane envelope protein. 54
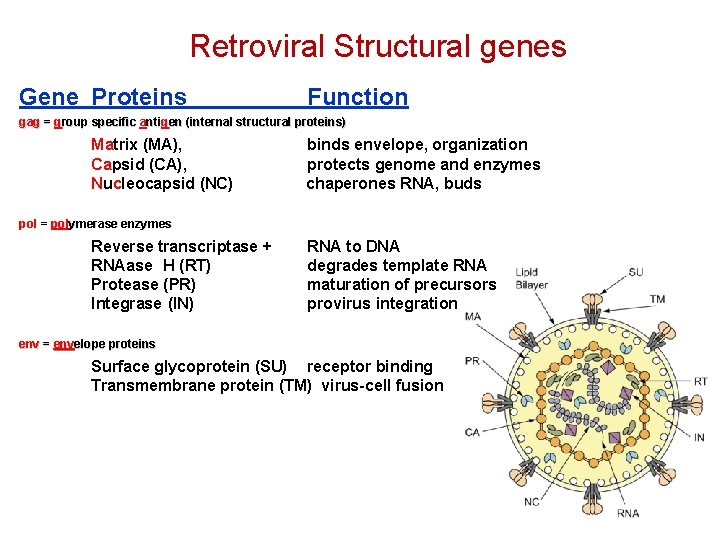
Retroviral Structural genes Gene Proteins Function gag = group specific anti ntig gen (internal structural proteins) Matrix (MA), Capsid (CA), Nucleocapsid (NC) binds envelope, organization protects genome and enzymes chaperones RNA, buds pol = polymerase enzymes Reverse transcriptase + RNAase H (RT) Protease (PR) Integrase (IN) RNA to DNA degrades template RNA maturation of precursors provirus integration env = envelope proteins Surface glycoprotein (SU) receptor binding Transmembrane protein (TM) virus-cell fusion 55

Viral life cycles § Virulent – these viruses lyse (kill) their host cell after infection. § Temperate – these viruses can replicate their genome along with the host cell genome without killing the host cell. § These viruses are also capable of lyzing the host cell

Distinct Steps in the Retroviral Life Cycle § § § Attachment, Fusion, and Entry Reverse Transcription Integration Transcription Translation Assembly, Budding, and Maturation
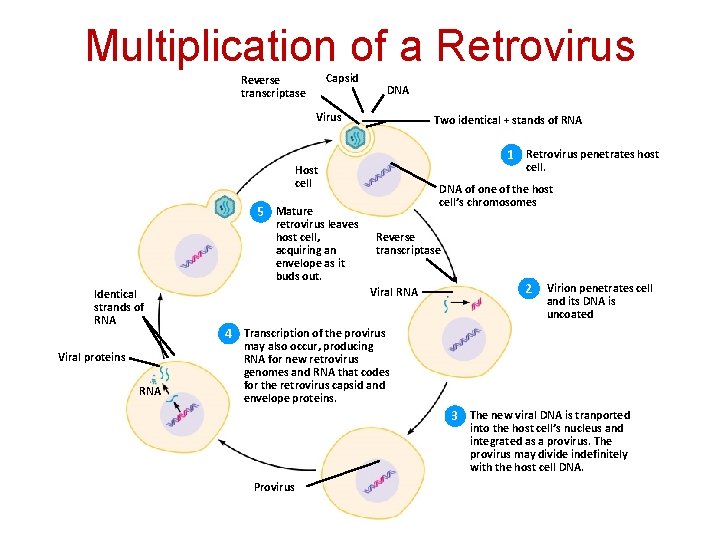
Multiplication of a Retrovirus Capsid Reverse transcriptase DNA Virus Two identical + stands of RNA 1 Retrovirus penetrates host cell. Host cell DNA of one of the host cell’s chromosomes 5 Mature retrovirus leaves host cell, acquiring an envelope as it buds out. Identical strands of RNA Viral proteins RNA Reverse transcriptase Viral RNA 2 Virion penetrates cell and its DNA is uncoated 4 Transcription of the provirus may also occur, producing RNA for new retrovirus genomes and RNA that codes for the retrovirus capsid and envelope proteins. 3 The new viral DNA is tranported into the host cell’s nucleus and integrated as a provirus. The provirus may divide indefinitely with the host cell DNA. Provirus
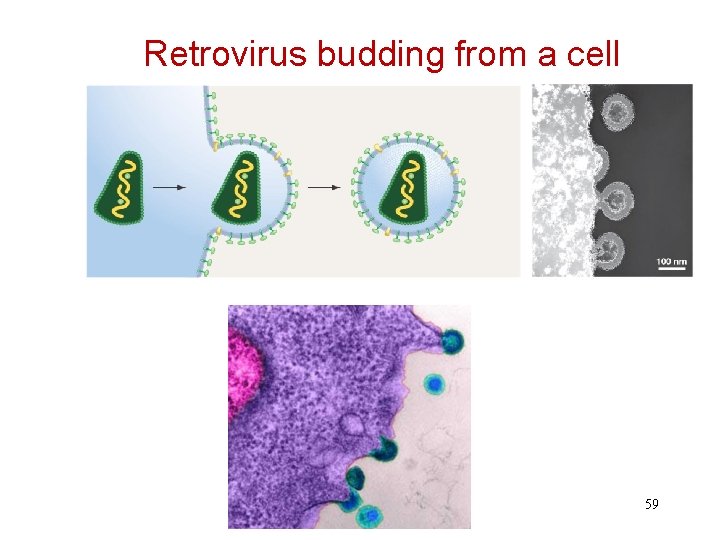
Retrovirus budding from a cell 59

After Budding, Virus Goes from Immature to Mature Form (after budding): -Core becomes more dense -Different retroviruses have different morphology in mature form 60

Retroviruses May Transduce Cellular Sequences l Transforming retroviruses are generated by a recombination event in which a cellular RNA sequence replaces part of the retroviral RNA. l Transducing virus : carries part of the host genome in place of part of its own sequence. The best known examples are retroviruses in eukaryotes and DNA phages in E. coli. l Replication-defective virus : cannot sustain an infective cycle by itself, because some of the necessary genes are absent (replaced by host DNA in a transducing virus) or mutated. It can, however, be perpetuated in the company of a helper virus. l Helper virus : provides missing viral functions to a defective virus, enabling to complete the infective cycle during a mixed infection. l Transformation (oncogenesis) : the ability to transform cultured cells so that the usual regulation of growth is released to allow unrestricted division.

Transmission of Pathogenic Retroviruses Virus Primary modes of transmission Range Preventative measures ALV feces, saliva, skin, contact; mother to offspring via egg worldwide; common removal of infected in untreated dams; breeding commercial flocks resistant strains lacking endogenous viruses that limit infection REV feces, saliva, skin, contact; mother to offspring via egg worldwide; common none taken due to in commercial low incidence of flocks; contaminant disease in Marek's disease vaccine MLV mother to offspring via milk rare; Lake Casitas, CA; La Puente, CA none MMTV mother to offspring via milk most inbred strains none
- Slides: 62