ARAB DIABETES FORUM 2017 Insulin Rx of Diabetes

ARAB DIABETES FORUM 2017 Insulin Rx of Diabetes in Patients with chronic Kidney &liver diseases Ahmed AKL, MD. MRCP endo. Consultant Endocrinologist Centre for Diabetes AFHSR KSA

DIABETES AND CKD • The kidney is a target organ for damage by macro and micro vascular diabetic complications. • Occurs in 20– 40% of patients with diabetes and is the leading cause of end stage renal disease (ESRD). • Diabetes Rx & outcome is affected by CKD. • The kidney is now a main target for DM Treatment.

CKD WITH DIABETES • Chronic kidney disease (CKD) with Diabetes is diagnosed by the presence of elevated urinary albumin excretion (albuminuria) and or low estimated glomerular filtration rate (e. GFR), in the absence of signs or symptoms of other primary causes of kidney damage.

Screening for diabetic Nephropathy • T 1 DM • T 2 DM • Kidney function should be assessed by: Albumin/Creatinine Ratio (ACR) and estimated glomerular filtration rate (e. GFR). • Normal ACR is less than 30 mg/gm • (e. GFR) e. GFR should be calculated from serum Cr using a validated formula. The Chronic Kidney Disease Epidemiology Collaboration (CKD EPI) equation is generally preferred
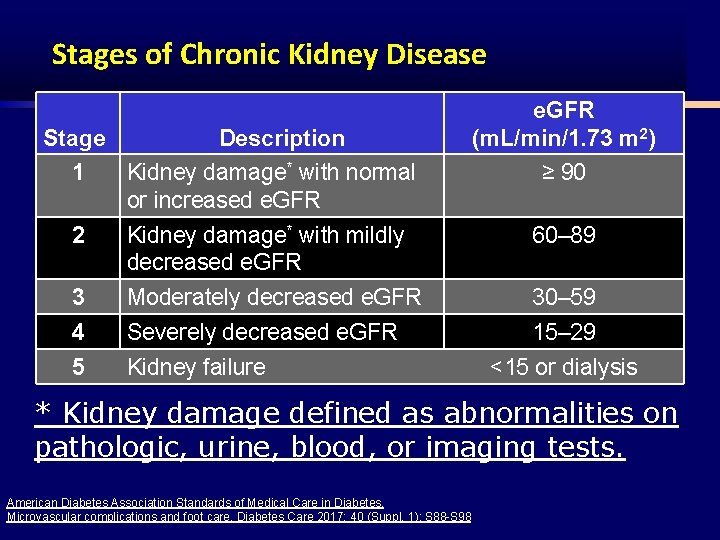
Stages of Chronic Kidney Disease Stage Description e. GFR (m. L/min/1. 73 m 2) 1 Kidney damage* with normal or increased e. GFR ≥ 90 2 Kidney damage* with mildly decreased e. GFR 60– 89 3 Moderately decreased e. GFR 30– 59 4 Severely decreased e. GFR 15– 29 5 Kidney failure <15 or dialysis * Kidney damage defined as abnormalities on pathologic, urine, blood, or imaging tests. American Diabetes Association Standards of Medical Care in Diabetes. Microvascular complications and foot care. Diabetes Care 2017; 40 (Suppl. 1): S 88 -S 98
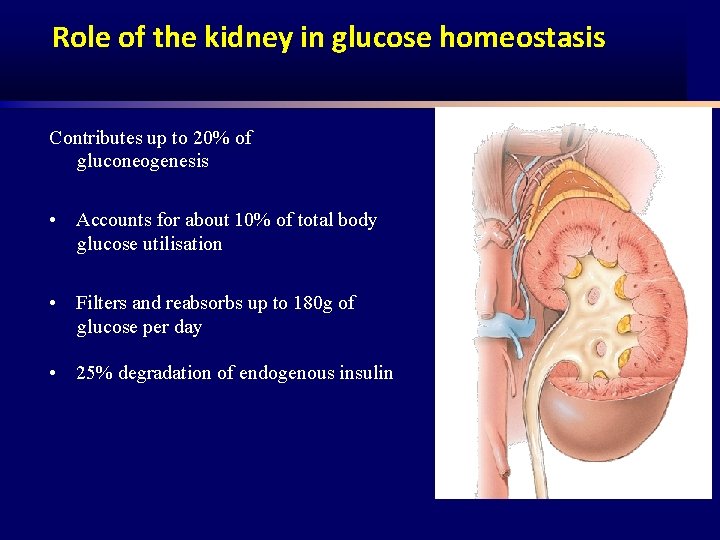
Role of the kidney in glucose homeostasis Contributes up to 20% of gluconeogenesis • Accounts for about 10% of total body glucose utilisation • Filters and reabsorbs up to 180 g of glucose per day • 25% degradation of endogenous insulin
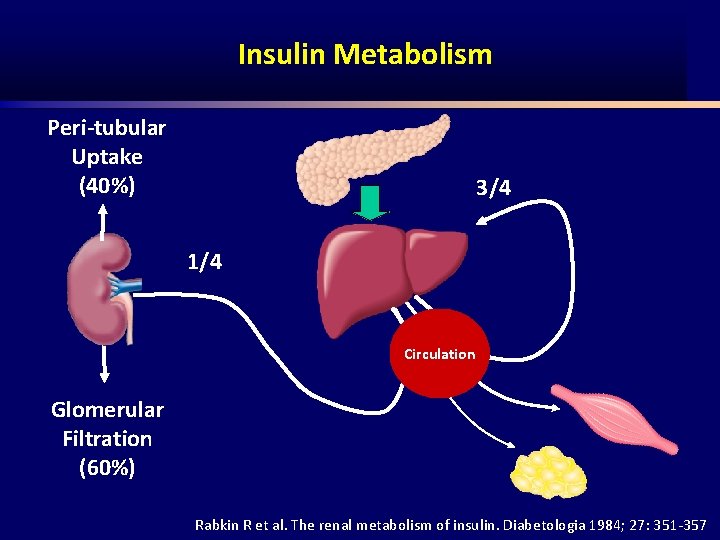
Insulin Metabolism Peri-tubular Uptake (40%) 3/4 1/4 Circulation Glomerular Filtration (60%) Rabkin R et al. The renal metabolism of insulin. Diabetologia 1984; 27: 351 357

Renal Handling of Insulin • 6 8 units of insulin are degraded by the kidney/day (25% of the daily endogenous insulin production). • Renal metabolism is enhanced in DM on exogenous insulin. (injected insulin enters the systemic circulation directly without first passing through the liver).
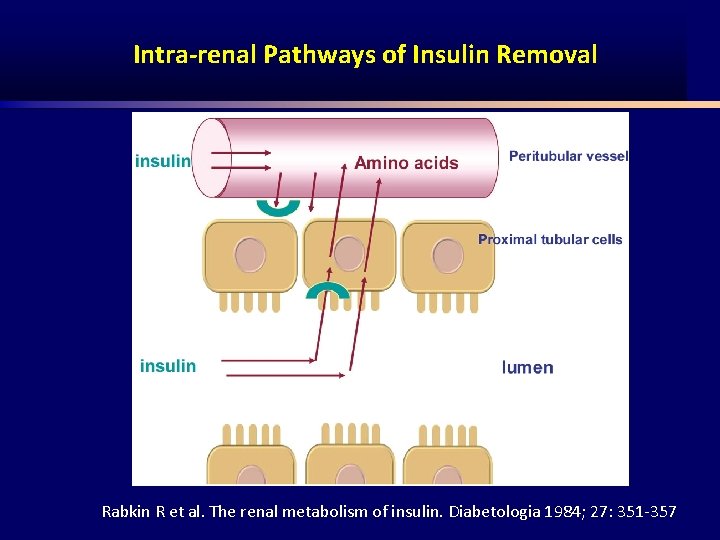
Intra-renal Pathways of Insulin Removal Rabkin R et al. The renal metabolism of insulin. Diabetologia 1984; 27: 351 357

Insulin requirements in CKD. • In DM+CKD, insulin requirements show a biphasic course: A. As renal function deteriorates in both DM types: Glucose control commonly deteriorates (insulin resistance) B. In ESRD; the fall in insulin clearance results in improvement in glucose tolerance judged by: • Lower insulin requirements • Lower OHA dose • cessation of insulin therapy • Spontaneous hypoglycemia (multi-factorial).

The Challenges of management of DM with CKD: • Target ? HA 1 C • Follow up and ? assessment tool of control • Fluctuation of bl. Glucose • Disturbance of metabolism in ESRD • Medication (OAD Vs INSULIN) • Effect of dialys

The Challenges of management of DM with CKD Target ? HA 1 C • Higher levels of Hb. A 1 c were associated with higher death rates in patients with DM and CKD after adjusting for markers of inflammation and malnutrition. • No agreement regarding the A 1 C target that is associated with the best outcome in (CKD). • K/DOQI and KDIGO 2012 • Individualized based on achieving a balance of best outcome and avoiding hypoglycemia • A 1 C >7 % for those prone to hypoglycemia

The Challenges of management of DM with CKD Follow up and ? assessment of control • Real time: – SMBG – CGM • Long term: – Hb. A 1 c – Fructosamine – Glycated albumin

MONITORING GLYCEMIC CONTROL • Glycated hemoglobin (A 1 C) may not be as accurate among ESRD patients: Ø carbamylated hemoglobin (false ↑). Ø reduced red blood cell life span (false ↓) Ø Recent transfusion (false ↓) Ø iron deficiency (false ↓) Ø accelerated erythropoiesis erythropoietin (false ↓) Ø metabolic acidosis

Monitoring of Glycemic Control III. Alternatives • Fructosamine: short term • Glycated albumin may be a better indicator than Hb. A 1 c in hemodialysis patients with diabetes (Inaba, et al, 2007. • No change in the clinical practice.

MONITORING GLYCEMIC CONTROL • Self monitoring of blood glucose: § More frequent especially for those in insulin therapy and those who are prone to hypos. § False higher values in patient on peritoneal dialysis coz of interference with non glucose sugars *(icodextrin)

Fluctuation of blood glucose and monitoring of glycemic control in CKD • Sever and ESRD significantly alters glycemic control coz Various and opposing effects of ESRD and dialysis can make BG levels fluctuate widely, placing patients at risk of hypoglycemia and present a challenge for physicians.

Management of Diabetes in Chronic Kidney Disease: The Challenges Glucose fluctuations • Increased risk of hyperglycemia – Increased insulin resistance – Impaired glucose disposal – insufficient exercise • Increased risk of hypoglycemia – – Impaired renal gluconeogenesis Decreased insulin clearance Decreased clearance of oral hypoglycemic agents poor food intake (uraemia induced anorexia)

TREATMENT • non pharmacologic o dietary modification o Exercise o weight reduction • Pharmacologic therapies insulin and oral agents.

Factors Influencing Management Strategy of Glycaemia in Chronic Kidney Disease Factors: General Factors: • Severity of kidney disease • Dialysis and type • • • Diabetes Factors: • • Type of diabetes Severity and duration Previous treatment Hypoglycaemia (+/ unawareness) Age Expected survival Resource availability. Cost Family/professional support Patient Choice: • Pt. motives • Willingness to manage

Pharmacologic therapies • According to severity of CKD and DM control Ø Mild CKD e. GFR ≥ 60 Management usually with standard protocols Ø moderate CKD e. GFR ≥ 59 30 Management usually with standard protocols with caution regarding some medication e. g Metformin Ø Sever CKD and pt. on Dialysis Insulin is the best Rx coz of DM – More efficient in control of DM Short acting SU could be used cautiously.

Insulin Therapy Recommendations • Consider insulins as: – Short/Rapid – Intermeditae – Long or Basal • Analogues have less risk of hypoglycaemia and could be dosed post meal • Premix insulin may be used within the context of meal pattern • Adjust insulin doses as follows: • Start with the lowest dose and titrate up based on regular SMBG • If established on insulin: • Reduce by 25% GFR<50 • Reduce by 50% GFR<10 • Adjust for changing Mode of dialysis O’Toole et al. 2012
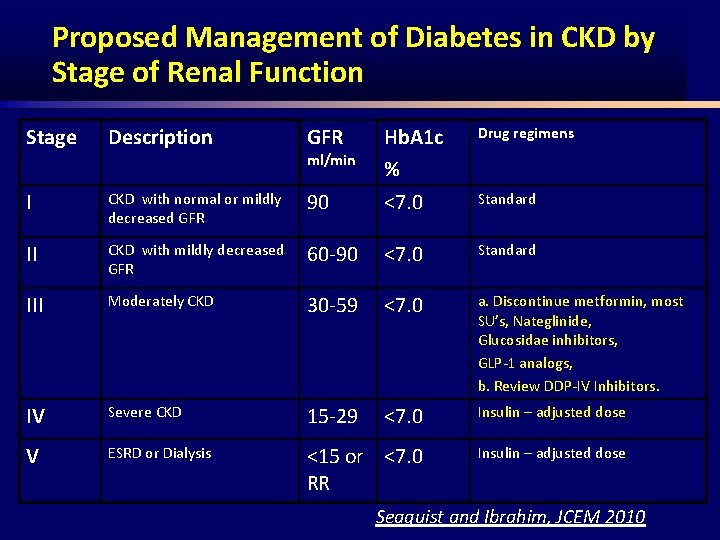
Proposed Management of Diabetes in CKD by Stage of Renal Function Stage Description GFR Drug regimens ml/min Hb. A 1 c % I CKD with normal or mildly decreased GFR 90 <7. 0 Standard II CKD with mildly decreased GFR 60 90 <7. 0 Standard III Moderately CKD 30 59 <7. 0 a. Discontinue metformin, most SU’s, Nateglinide, Glucosidae inhibitors, GLP 1 analogs, b. Review DDP IV Inhibitors. IV Severe CKD 15 29 <7. 0 Insulin – adjusted dose V ESRD or Dialysis <15 or <7. 0 RR Insulin – adjusted dose Seaquist and Ibrahim, JCEM 2010
Management of Diabetes in Peritoneal Dialysis

Intra-peritoneal insulin in peritoneal dialysis patients PROS • More physiologic absorption (less fluctuation of BG). • Continuous insulin infusion. • Avoids injections • Less hyper insulinemia • Avoids antibody formation. • Improved Hb. A 1 c CONS • High insulin doses • Higher cost • Insulin losses in effluent • Lipid effects • Specific dialysis complications: e. g. excess glucose absorption. (Quellhorst, 2002; Grodstein et al, 1981)

THANK YOU

ARAB DIABETES FORUM 2017 Insulin Rx of Diabetes in Patients with liver disease Ahmed AKL, MD. MRCP endo. Consultant Endocrinologist Centre for Diabetes AFHSR KSA

Diabetes & Liver disease q. Diabetes is a frequent comorbidity in patients with CLD. q. DM ↔liver disease [bi directional]. Both problems have an important interaction considering etiology & the presence of any problem of each affect the management and outcome of the other.

Diabetes & Liver disease • Liver disease associated with DM can be divided into three groups : • 1. Liver disease occurring as a consequence of diabetes • ↑ liver enzymes • Glycogen deposition • Steatosis and NASH • Fibrosis and cirrhosis • Biliary disease, cholelithiasis and cholecystitis • Complications of therapy of DM (cholestatic and necroinflammatory)

2. DM occurring as a complication of liver disease • Hepatitis • Cirrhosis (hepatogenous DM 30%) • HCC • Fulminant hepatic failure • Post liver transplantation 3. Liver disease occurring coincidentally with DM • Hemochromatosis • Glycogen storage diseases • Chronic active autoimmune hepatitis • Autoimmune biliary disease


EFFECT OF CHRONIC LIVER DISEASE ON DIABETES. • Increase morbidity and mortality outcome. • Difficult and challenging management of Diabetes ü ↓ capacity for gluconeogenesis and reduced hepatic breakdown of insulin. ü ↑ IR ü Nausea and↓ appetite ü Number of pharmacotherapeutic options is limited in decompensated liver disorders

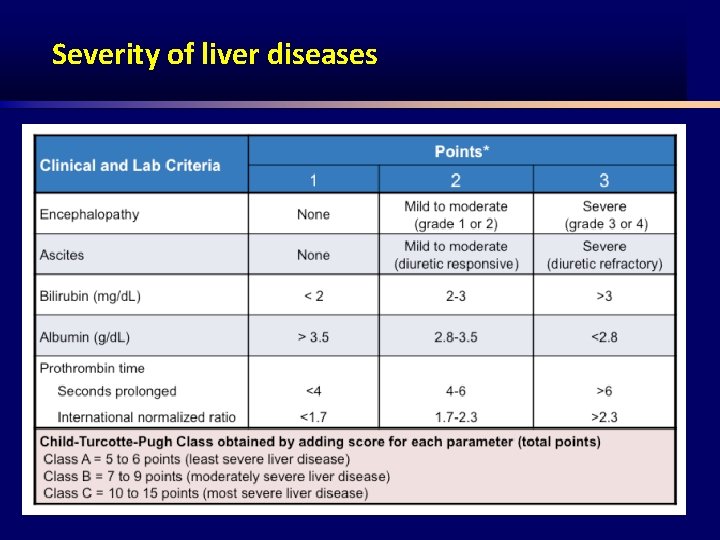
Severity of liver diseases • 1 - child-A. • 2 - child-B. • 3 - child-C.


The Challenges of management of DM with CLD: • Target ? HA 1 C • Follow up and ? assessment tool of control • Fluctuation of bl. Glucose • Disturbance of metabolism decompensated liver diseases • Medication (OAD Vs INSULIN) • Encphalopathy

Important notes in management of DM in CLD Ø The treatment of DM of cirrhotic patients has particular characteristics that make it different from type 2 DM without liver disease: 1 - About 1/2 the patients have malnutrition. 2 - When clinical DM is diagnosed, the patient has advanced liver disease. 3 - Most of the oral hypoglycemic agents are metabolized in the liver. 4 - Patients often have episodes of hypoglycemia. Diabetes Care. 2007; 30: 734– 743

Important notes in management of DM in CLD Ø In patients with compensated cirrhosis requirements may be greater compared to patients with decompensated cirrhosis, Ø since insulin resistance predominates in the former while in the latter liver metabolism of insulin is greatly reduced and impaired glycogenlysis and Gluconeogensis. Ø Therefore, close monitoring of blood glucose levels is recommended to avoid hypoglycemia. Diabetes Care. 2007; 30: 734– 743

Ø insulin secretagogues, despite the fact that they are safe drugs in patients with liver disease, probably are not useful, since they do not modify insulin resistance and exhibit burden on the liver cells Ø Insulin therapy is probably the safest and most effective therapy in patients with liver dysfunction, with the limitation that increased risk of hypoglycemia that can be ameliorated by use of insulin analogues, proper nutrition, frequent dose adjustment and SMBG Single-dose pharmacokinetics of nateglinide in subjects with hepatic cirrhosis. JClin Pharmacol. 2000; 40: 634– 640

Ø insulin analogs may offer equivalent or improved glycemic control compared to standard insulin while being associated with a lower risk for hypoglycemia, particularly nocturnal and severe hypoglycemia. and associated with superior renal and cardiac outcomes compared to human insulin.


Ø In patients with hepatic encephalopathy who require high-carbohydrate diets, resulting in postprandial hyperglycemia, rapid-acting insulin analogs such as insulin lispro, aspart, or glulisine may be particularly useful.

Take home messages • Kidney and liver play a crucial role in glucose homeostasis. • Management of diabetes in CKD and CLD is challenging task and require individualization depending on severity and other comorbidity • Insulin is the Best therapy in both conditions especially in advanced stages and insulin analogues has better profile in such conditions • Dose adjustment of insulin is needed to avoid hypoglycemia and fluctuations • Special consideration should be taken in mind when interpret the methods of follow up and control of HA 1 C


Insulin therapy recommendation in CLD • The major site of the metabolism for circulating insulin is liver. Approximately 40%– 50% of the endogenous insulin produced by the pancreas is metabolized by the liver.

• Insulin is the first‑line agent to treat diabetes in CLD such as cirrhosis or chronic hepatitis. • Insulin therapy can be used at any stage of HI • Short‑acting insulin's are preferred because the duration of action may vary in such situations • insulin analogs may offer equivalent or improved glycemic control compared to standard insulin while being associated with a lower risk for hypoglycemia, particularly nocturnal and severe hypoglycemia.

• The PKs and PDs of rapid‑acting insulin analogs suggest that they can be given just after meals. This is of benefit to many patients with advanced CLD as they may have nauseaand reduced appetite • insulin requirement may vary in decompensated liver disease patients. due to two opposing factors: • ↓ capacity for gluconeogenesis and reduced hepatic breakdown of insulin. • ↑ IR

Management of diabetes in patients with concomitant liver disease • In patients with decompensated liver disease, the requirement may be decreased due to reduced capacity for gluconeogenesis and reduced hepatic breakdown of insulin. • However, patients with impaired hepatic function may have an increased need for insulin due to insulin resistance. • In patients with hepatic encephalopathy who require high carbohydrate diets, resulting in postprandial hyperglycemia, rapid acting insulin analogs may be particularly useful.
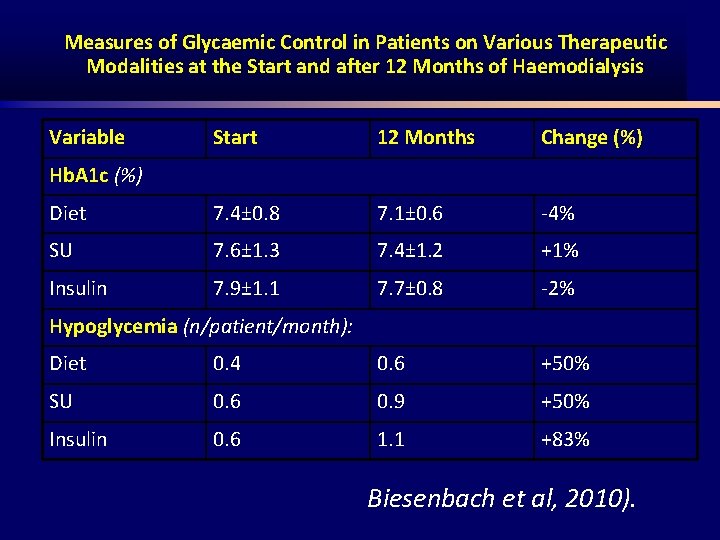
Measures of Glycaemic Control in Patients on Various Therapeutic Modalities at the Start and after 12 Months of Haemodialysis Variable Start 12 Months Change (%) Diet 7. 4± 0. 8 7. 1± 0. 6 4% SU 7. 6± 1. 3 7. 4± 1. 2 +1% Insulin 7. 9± 1. 1 7. 7± 0. 8 2% Hb. A 1 c (%) Hypoglycemia (n/patient/month): Diet 0. 4 0. 6 +50% SU 0. 6 0. 9 +50% Insulin 0. 6 1. 1 +83% Biesenbach et al, 2010).
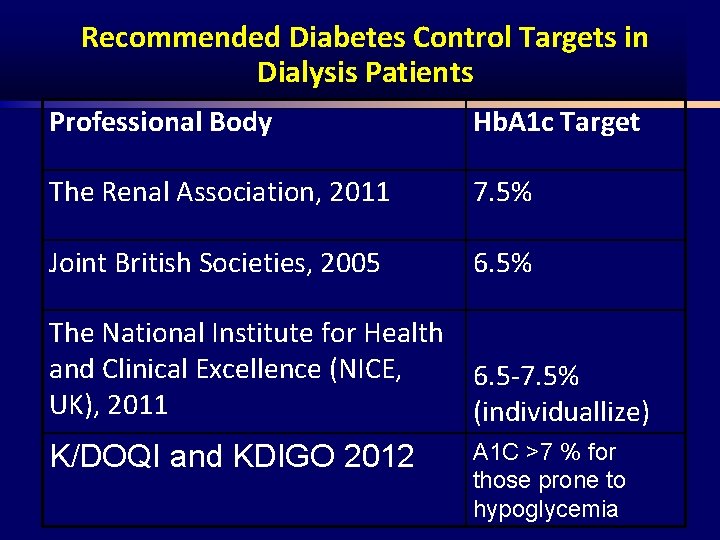
Recommended Diabetes Control Targets in Dialysis Patients Professional Body Hb. A 1 c Target The Renal Association, 2011 7. 5% Joint British Societies, 2005 6. 5% The National Institute for Health and Clinical Excellence (NICE, 6. 5 7. 5% UK), 2011 (individuallize) K/DOQI and KDIGO 2012 A 1 C >7 % for those prone to hypoglycemia

Diabetes in the Dialysis Patient: The Principles • Poor control is associated with extensive morbidity and mortality. • Capillary BG measurements can be overestimated in peritoneal dialysis patients using icodextrin, leading to risk of ‘missed’ hypoglycaemic episodes. • Interpretation of Hb. A 1 c in dialysis populations is complicated. • The therapeutic arsenal to achieve glycaemic control is severely restricted in patients with ESRD. • Management of DM complications centres around risk reduction, early identification, good glycaemic control and a multidisciplinary team
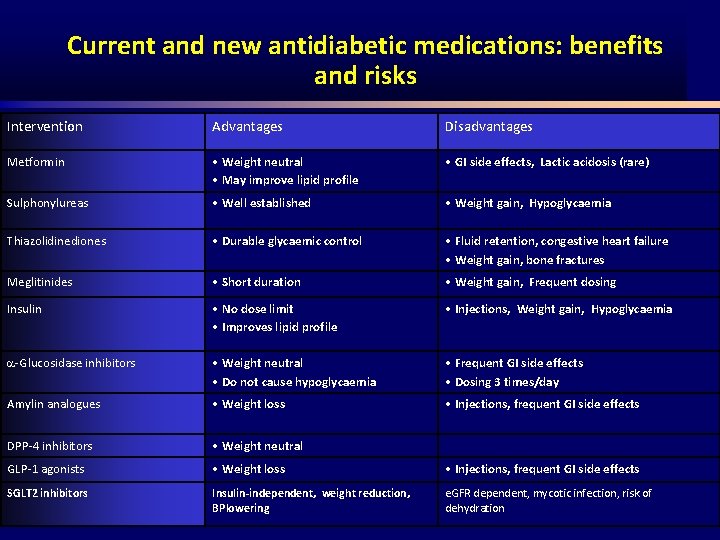
Current and new antidiabetic medications: benefits and risks Intervention Advantages Disadvantages Metformin • Weight neutral • May improve lipid profile • GI side effects, Lactic acidosis (rare) Sulphonylureas • Well established • Weight gain, Hypoglycaemia Thiazolidinediones • Durable glycaemic control • Fluid retention, congestive heart failure • Weight gain, bone fractures Meglitinides • Short duration • Weight gain, Frequent dosing Insulin • No dose limit • Improves lipid profile • Injections, Weight gain, Hypoglycaemia Glucosidase inhibitors • Weight neutral • Do not cause hypoglycaemia • Frequent GI side effects • Dosing 3 times/day Amylin analogues • Weight loss • Injections, frequent GI side effects DPP 4 inhibitors • Weight neutral GLP 1 agonists • Weight loss • Injections, frequent GI side effects SGLT 2 inhibitors Insulin-independent, weight reduction, BPlowering e. GFR dependent, mycotic infection, risk of dehydration

Diabetes Management in Peritoneal Dialysis III • They may be best managed in a multidisciplinary diabetic–renal clinic setting, using the skills of: – diabetologists – nephrologists. – clinical nurse specialists in nephrology and diabetes. – Dietitians. – podiatrists.

Diabetes Management in Peritoneal Dialysis II • Standard drug therapy for hyperglycemia is clearly not possible in patients on dialysis. • Sulphonylureas and insulin are the mainstay of treatment. • Newer therapies for hyperglycaemia have become available, but until recently, renal failure has precluded their use. • Newer gliptins, however, are now licensed for use in ‘severe renal failure’. They have yet to be trialled in dialysis patients. • Diabetic patients on dialysis have special needs, as they have a much greater burden of complications (cardiac, retinal and foot)

Diabetes Management in Peritoneal Dialysis I • The evidence for improving glycemic control in patients on dialysis having an impact on mortality or morbidity is sparse. • Improving glycaemic control in patients on dialysis is very challenging – difficulties with hypoglycemic drugs – monitoring difficulties – dialysis strategies that exacerbate hyperglycemia or hypoglycemia – Therapeutic nihilism or inertia.
- Slides: 55