Aqueous Alteration on Mars Importance Some background Alteration

Aqueous Alteration on Mars • Importance • Some background • Alteration products in Martian meteorites – Martian origin for products – Nature of products • Experimental data • Ages of weathering products • Evidence from Viking, Pathfinder, and MER analyses Jeff Taylor Aqueous Alteration on Mars 1

Importance of Understanding Aqueous Alteration on Mars • Understand geologic environments at surface, including habitability • Estimate duration of surfacewater events • Estimate conditions of groundwater-cryosphere system • Date times of aqueous alterations • Understand history of climate on Mars Jeff Taylor Aqueous Alteration on Mars 2

Aqueous Alteration • Definition: Change in chemical and/or mineralogical composition of rock in response to interactions with H 2 Obearing ices, liquids, and vapors at ambient or hydrothermal (above ambient) temperatures. • Chemical alteration takes place by hydrolysis, hydration, oxidation, carbonation, ion exchange (between crystals and solutions) • Alteration without removal of elements is isochemical (closed system). Opposite is open system alteration Jeff Taylor Aqueous Alteration on Mars 3

Variables • Initial rock mineralogy – Anhydrous igneous minerals not stable at surface with water • Temperature • Water chemistry (e. g. , no SO 4 -2, no sulfate forms) • p. H (H+ activity) • Pressure Jeff Taylor Aqueous Alteration on Mars 4

Settings • • Weathering—Surfaces of Earth and Mars Hydrothermal alteration (> ~25 C, ≥ 1 bar) Late-stage magmatic fluids Fumeroles (magmatic fluids and rain or groundwater) • Burial • Subduction • Internal heating of rock/ice mixtures Jeff Taylor Aqueous Alteration on Mars 5

p. H, in case you forgot H 2 O H+ + OHKwater = m. H+ + m. OH- = 10 -14 (dissociation constant of water) m = moles of solute/kg of water (Always some of both H+ and OH-) Can give acidity by conc of H+ alone: p. H = -log(m. H+), hence log of the activity of H+ Acidic, < 7 m. H+ Neutral, 7 m. H+ Alkaline (Basic), >7 Jeff Taylor Aqueous Alteration on Mars 6
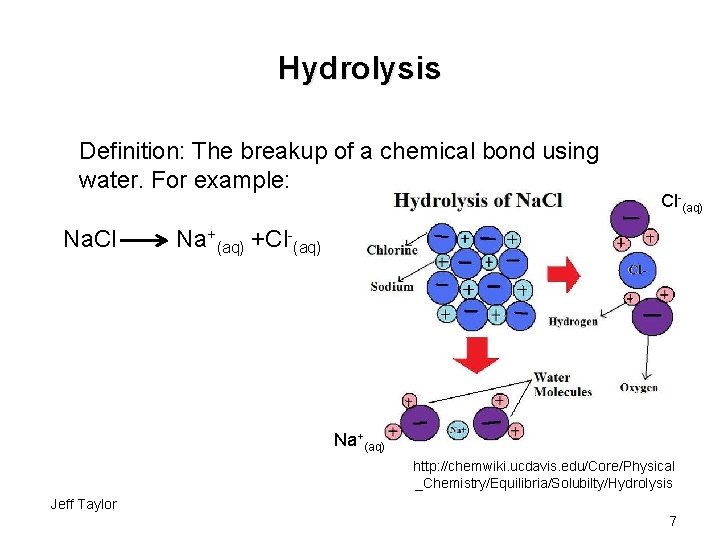
Hydrolysis Definition: The breakup of a chemical bond using water. For example: Na. Cl Cl-(aq) Na+(aq) +Cl-(aq) Na+(aq) http: //chemwiki. ucdavis. edu/Core/Physical _Chemistry/Equilibria/Solubilty/Hydrolysis Jeff Taylor 7

Hydrolysis of Silicates Mg 2 Si. O 4 + 4 H 2 O Mg 2 Si. O 4 + 4 H+ 2 Mg 2+ + 4 OH- +H 4 Si. O 4 2 Mg 2+ +H 4 Si. O 4 Fe 2 Si. O 4 + 4 H+ 2 Fe 2+ + H 4 Si. O 4 The Fe 2+, Mg 2+ and other ions may not stay in solution, depending on water chemistry: (Fe, Mg)2 Si. O 4 +4 H+ = Fe 2+ + Mg 2+ +H 4 Si. O 4 Fe 3 O 4, Fe 2 O 3 Fe. O(OH) Fe. SO 4 Jeff Taylor Mg. SO 4 Mg. Cl 2 Mg 3 Si 2 O 5(OH)4 Si. O 2 (amorphous) Silicious clays Aqueous Alteration on Mars 8

Jeff Taylor Aqueous Alteration on Mars 9

• Giant’s Causeway • 30 -meter thick soil layer • 1 -2 My time gap between lower and upper flows Jeff Taylor Aqueous Alteration on Mars 10

Giant’s Causeway Corestone Jeff Taylor Aqueous Alteration on Mars 11
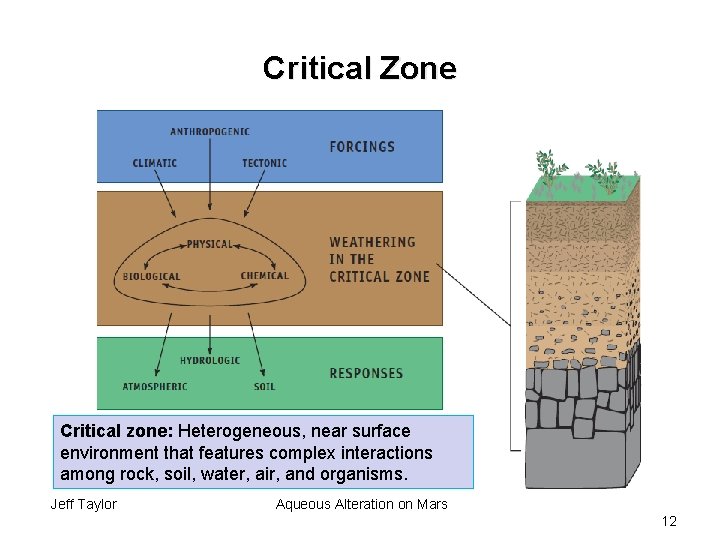
Critical Zone Critical zone: Heterogeneous, near surface environment that features complex interactions among rock, soil, water, air, and organisms. Jeff Taylor Aqueous Alteration on Mars 12
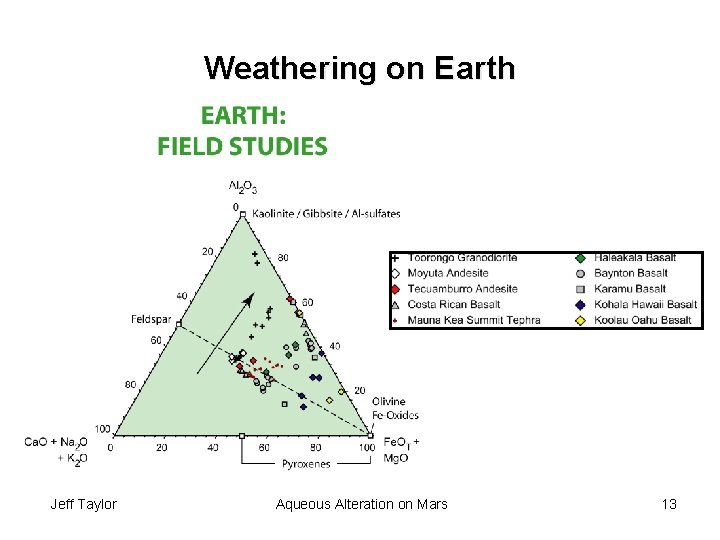
Weathering on Earth Jeff Taylor Aqueous Alteration on Mars 13
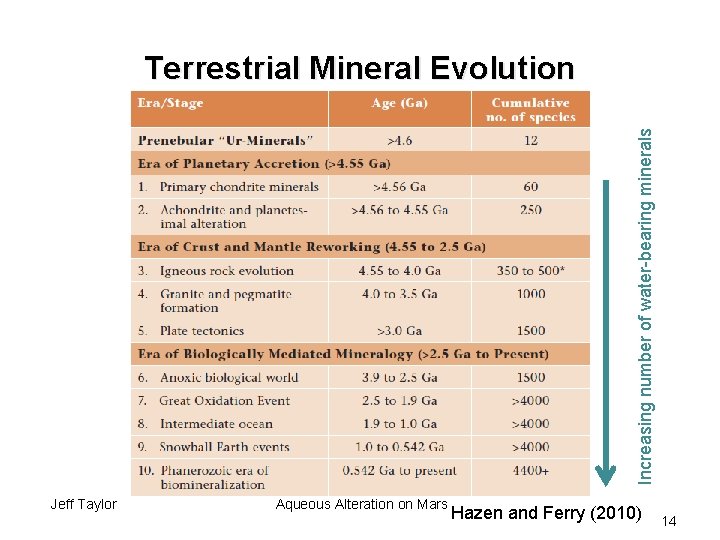
Increasing number of water-bearing minerals Terrestrial Mineral Evolution Jeff Taylor Aqueous Alteration on Mars Hazen and Ferry (2010) 14
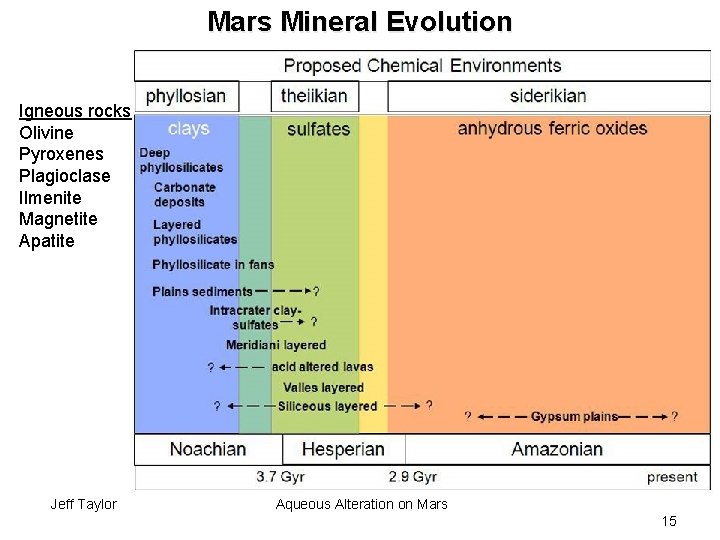
Mars Mineral Evolution Igneous rocks Olivine Pyroxenes Plagioclase Ilmenite Magnetite Apatite Jeff Taylor Aqueous Alteration on Mars 15
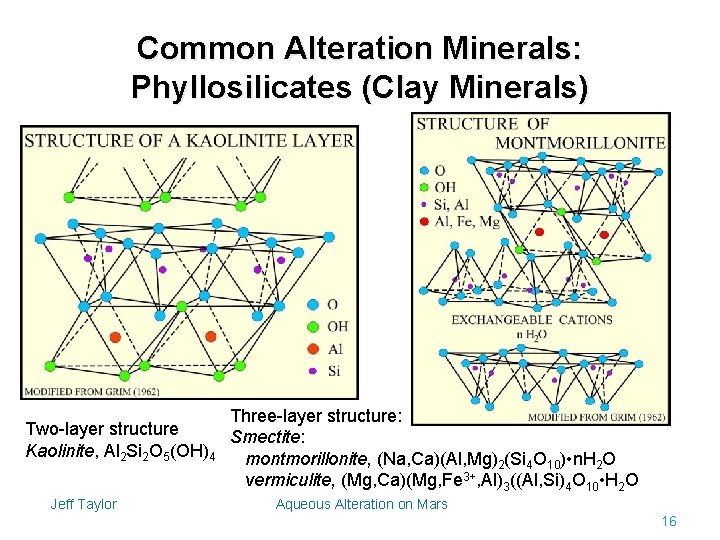
Common Alteration Minerals: Phyllosilicates (Clay Minerals) Three-layer structure: Two-layer structure Smectite: Kaolinite, Al 2 Si 2 O 5(OH)4 montmorillonite, (Na, Ca)(Al, Mg)2(Si 4 O 10) • n. H 2 O vermiculite, (Mg, Ca)(Mg, Fe 3+, Al)3((Al, Si)4 O 10 • H 2 O Jeff Taylor Aqueous Alteration on Mars 16

Common Alteration Minerals: Carbonates • Note miscibility gaps, based on experiments • Compositions in shaded areas not common and if present represent nonequilibrium conditions Jeff Taylor Aqueous Alteration on Mars 17

Common Alteration Minerals: Sulfates and Halides • There are > 100 sulfate minerals • Common in Mars Meteorites and inferred on surface: – – Anhydrite, Ca. SO 4 Gypsum, Ca. SO 4 • 2 H 2 O Epsomite, Mg. SO 4 • 7 H 2 O Kieserite, Mg. SO 4·H 2 O • Halides – Halite, Na. Cl (rock salt) – Sylvite, KCl Jeff Taylor Aqueous Alteration on Mars 18

Common Alteration Minerals: Zeolites • At least 40 different minerals in zeolite group • Framework silicates – Tetrahedra of Si. O 4 and Al. O 4 linked to form channels, cages, rings, loops – Form cage-like openings in structure – Channels and cages permit cations and H 2 O to pass in and out – Result: can be used as molecular sieves in water softeners, or as kitty litter Jeff Taylor • Typically form as secondary minerals in cracks or cavities in mafic igneous rocks • Examples: – – Natrolite, Na 2 Al 2 Si 3 O 10 • 2 H 2 O Chabazite, Ca. Al 2 Si 4 O 12 • 6 H 2 O Heulandite, Ca. Al 2 Si 7 O 18 • 7 H 2 O Stilbite, Ca. Al 2 Si 7 O 18 • 7 H 2 O Aqueous Alteration on Mars 19
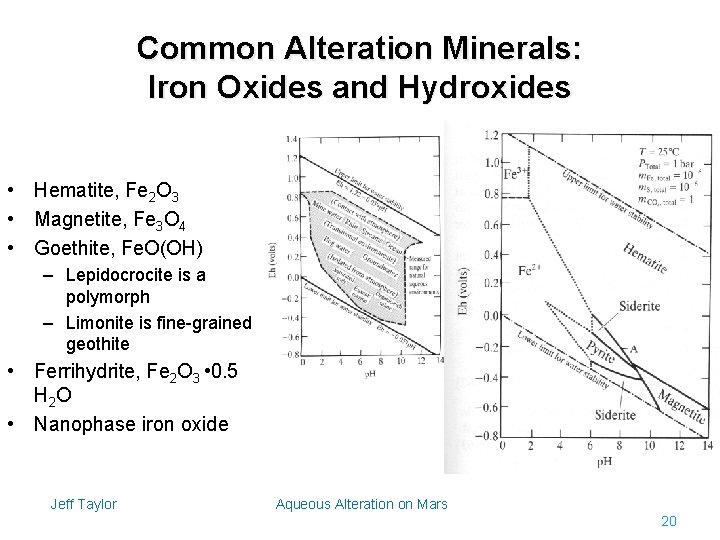
Common Alteration Minerals: Iron Oxides and Hydroxides • Hematite, Fe 2 O 3 • Magnetite, Fe 3 O 4 • Goethite, Fe. O(OH) – Lepidocrocite is a polymorph – Limonite is fine-grained geothite • Ferrihydrite, Fe 2 O 3 • 0. 5 H 2 O • Nanophase iron oxide Jeff Taylor Aqueous Alteration on Mars 20

Common Alteration Minerals: Iddingsite • Rusty red stuff that forms veinlets • Common alteration product • Mixture of Fe-bearing smectite and iron oxides (ferrihydrite and magnetite) • Smectite usually poorly crystalline Jeff Taylor Aqueous Alteration on Mars 21

Aqueous Alteration in SNC Meteorites • Fusion crust cuts across alteration Best evidence for extraterrestrial origin Jeff Taylor Aqueous Alteration on Mars 22

Evidence for Martian Origin • Pre-terrestrial fracturing • D/H in some phases too high to be terrrestrial, but consistent with high D/H in Martian atmosphere • Phases with C, O, and S isotopes inconsistent with terrestrial alteration • Ages far older (> 600 My) than terrestrial residence times • However, except for ALH 84001 and the nakhlites, others have not been as well documented Jeff Taylor Aqueous Alteration on Mars 23

Summary of Alteration Phases in Nakhlites • Lafayette: – Ca-rich siderite, smectite, gypsum • Governador Valadares: – Smectite, siderite, gypsum • Nakhla: – Smectite, siderite, gypsum, anhydrite, halite, epsomite, goethite • These form an evaporation sequence from a brine (Bridges and Grady, 2000): – Lafayette, 25% brine remaining – Governador Valadares, 20% brine remaining – Nakhla, 10% brine remaining Jeff Taylor Aqueous Alteration on Mars 24

Alteration in Nakhlites 50 microns Halite (h), anhydrite (a), and siderite (sd) in Nakhla Jeff Taylor Iddingsite in Lafayette; sd is Siderite, sm is smectite Aqueous Alteration on Mars 25

Alteration in Chassigny • Occurs in veins in olivine; probably deposited from brines • Calcite • Magnesite • Gypsum • Not an alteration product: Amphibole (kaersutite) in melt inclusions – Shows there was water in magma Jeff Taylor Aqueous Alteration on Mars 26

Alteration in Shergottites • Have to worry about effects of shock on alteration phases—can convert low-T products to higher-T products – No systematic study of alterations and shock has been done • Alterations described only from Shergotty and EET 79001: – – Calcite Gypsum Sulfates Phyllosilicates (Shergotty) Jeff Taylor Aqueous Alteration on Mars 27

Alteration in ALH 84001 Carbonates in ALH 84001 form disks with compositions varying continuously among carbonate endmembers (gray field) On the diagram, Lafayette points are crosses, GV are triangles, Nakhla is circles Jeff Taylor Important point: some carbonate on Mars Aqueous Alteration on Mars 28
- Slides: 28