Aqueous AcidBase Equilibria Acids and bases Acidbase properties

Aqueous Acid-Base Equilibria • • Acids and bases Acid-base properties of water (Kw) p. H scale Strength of Acids and Bases Weak acid (Ka) Weak base (Kb) Relation between conjugate acid-base ionization constants (Ka and Kb) • Chemical structure and acidity • Acid-base properties of salt solutions
![Arrhenius Acids and Bases Arrhenius acid - a compound that increases [H+] in water Arrhenius Acids and Bases Arrhenius acid - a compound that increases [H+] in water](http://slidetodoc.com/presentation_image_h/14f60e19549c0e6e76232bd8a7751d31/image-2.jpg)
Arrhenius Acids and Bases Arrhenius acid - a compound that increases [H+] in water another example HNO 3(aq) + H 2 O(l) H 3 O+(aq) + NO 3 -(aq) Arrhenius Base : a compound that increases [OH-] in water NH 3(aq) + H 2 O (l) NH 4+(aq) + OH-(aq)
![Arrhenius A. Arrhenius acid - a compound that increases [H+] in water Chemists often Arrhenius A. Arrhenius acid - a compound that increases [H+] in water Chemists often](http://slidetodoc.com/presentation_image_h/14f60e19549c0e6e76232bd8a7751d31/image-3.jpg)
Arrhenius A. Arrhenius acid - a compound that increases [H+] in water Chemists often use the notation H+(aq) for the H 3 O+(aq) ion, and call it the hydrogen ion. Remember, however, that the aqueous hydrogen ion is actually chemically bonded to water, that is, H 3 O+. Even that is a shorthand, as each H 3 O+ ion coordinates with the water molecules around it! Examples of hydrogen ion structures from Martin Chaplin under CC License

Limitations of the Arrhenius Concept The Arrhenius concept is limited in that it looks at acids and bases in aqueous solutions only. All Arrhenius acids must be protic In addition, it singles out the OH- ion as the source of base character, when other species can play a similar role. Broader definitions of acids and bases are discussed in the next sections. All Arrhenius acids must be protic
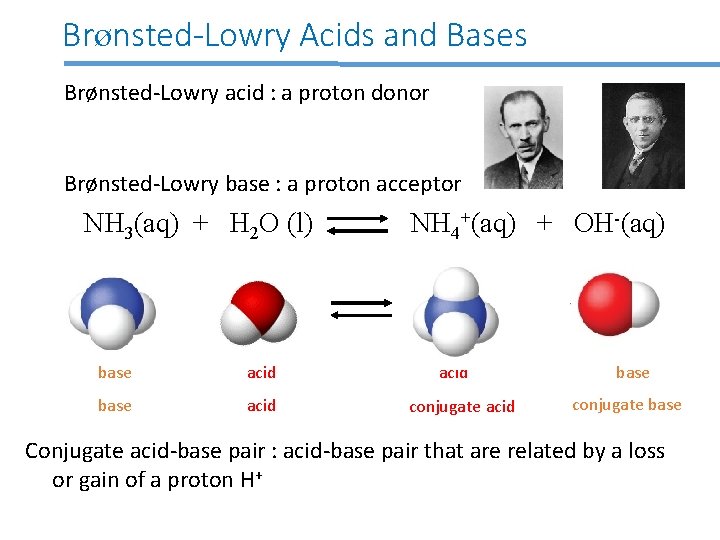
Brønsted-Lowry Acids and Bases Brønsted-Lowry acid : a proton donor Brønsted-Lowry base : a proton acceptor NH 3(aq) + H 2 O (l) base acid NH 4+(aq) + OH-(aq) acid conjugate acid base conjugate base Conjugate acid-base pair : acid-base pair that are related by a loss or gain of a proton H+
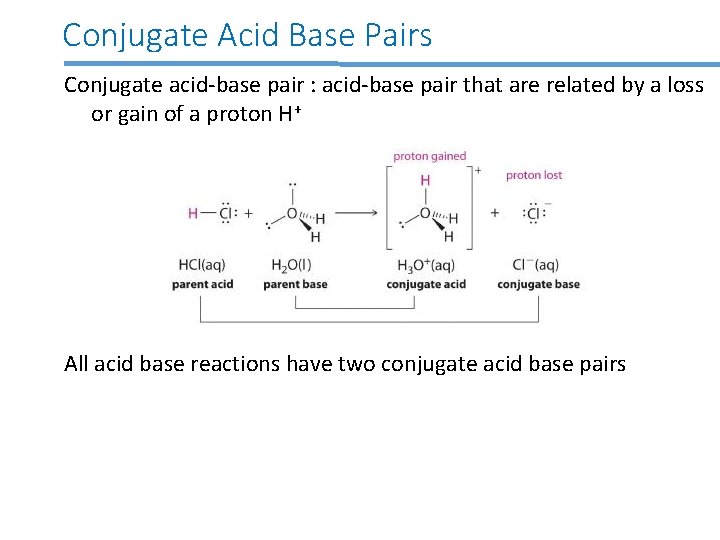
Conjugate Acid Base Pairs Conjugate acid-base pair : acid-base pair that are related by a loss or gain of a proton H+ All acid base reactions have two conjugate acid base pairs
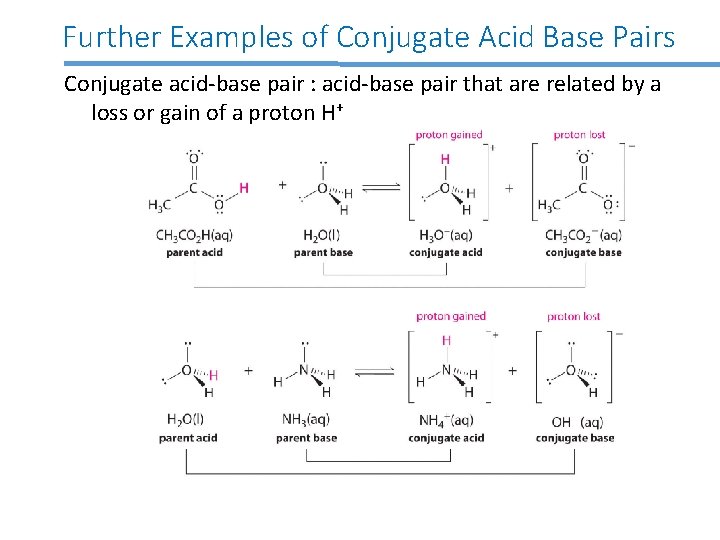
Further Examples of Conjugate Acid Base Pairs Conjugate acid-base pair : acid-base pair that are related by a loss or gain of a proton H+
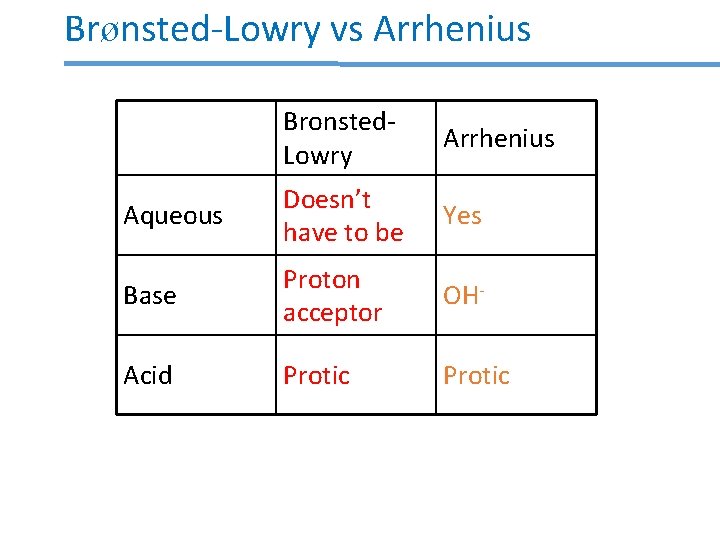
Brønsted-Lowry vs Arrhenius Bronsted. Lowry Arrhenius Aqueous Doesn’t have to be Yes Base Proton acceptor OH- Acid Protic
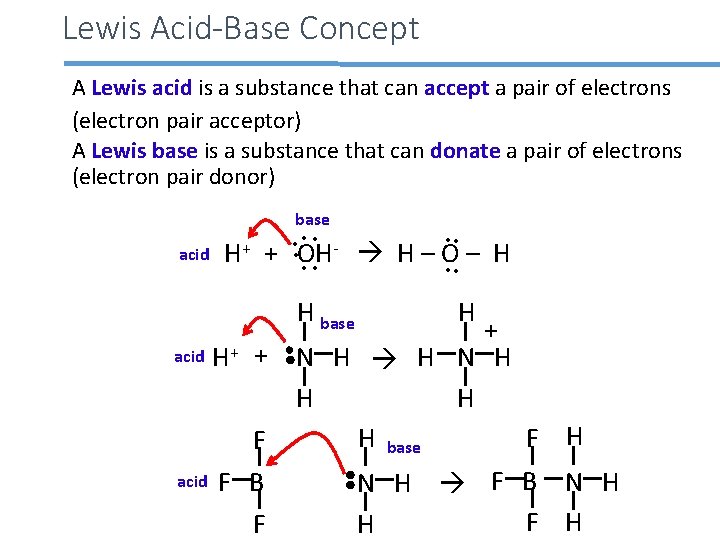
Lewis Acid-Base Concept A Lewis acid is a substance that can accept a pair of electrons (electron pair acceptor) A Lewis base is a substance that can donate a pair of electrons (electron pair donor) base • • + acid H + OH H – O – H • • H base H • • + acid H+ + N H H N H H base F H F acid F B N H F H
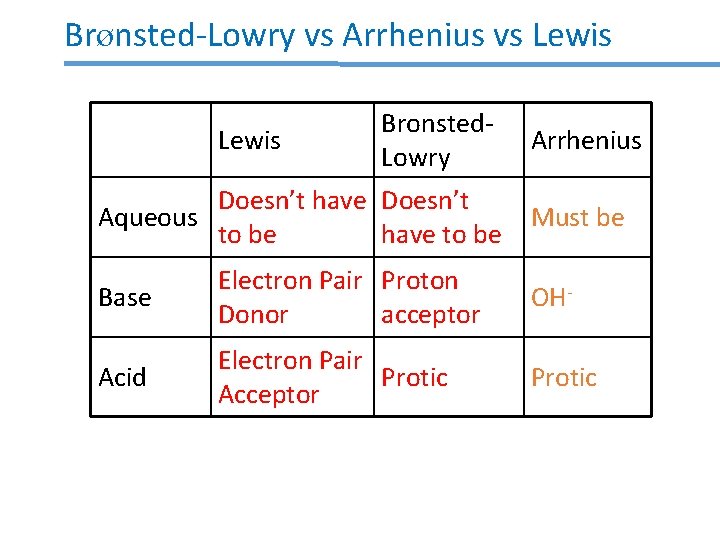
Brønsted-Lowry vs Arrhenius vs Lewis Bronsted. Lowry Doesn’t have Doesn’t Aqueous to be have to be Arrhenius Must be Base Electron Pair Proton Donor acceptor OH- Acid Electron Pair Protic Acceptor Protic

Acid-Base Properties of Water is amphoteric – it can act as either an acid or a base Since water is amphoteric it can autoionize yielding both OH-(aq) and H 3 O+ (aq) ions

Autoionization of water We can write the equilibrium expression for the autoionization of water As For pure water (density 0. 997 g/ml at 25 o. C)

Kw The equilibrium concentrations of OH-(aq) and H 3 O+(aq) at 25 o. C are 1. 003 x 10 -7 M so and numerically For most purposes (e. g. tests unless told otherwise) we will approximate Kw as 1. 0 x 10 -14 and in neutral water [H 3 O+(aq)] = [OH-(aq)] = 1. 0 x 10 -7 M Depending on context we will also use either H 3 O+(aq) or H+(aq)

p. H p. OH p. K Because concentrations of ions can vary over many orders of magnitude we use a logarithmic scale. For example the concentration of H 3 O+(aq) is usually transformed to where p. X indicates minus the base 10 logarithm of X. OH-(aq) concentrations are transformed to A little bit of manipulation shows that p. Kw= -log Kw

p. H Scale • Acids and bases 7 is neutral on the p. H scale. Anything above 7 is basic & anything below 7 is acidic If the p. H of a solution decreases one says it has been acidified or become more acidic. OTOH if the p. H of a solution increases one says it has become more basic.

Acid Equilibrium Ionization Constant Acids that (almost) completely ionize in water are called strong acids and those that only partially ionize are called weak acids. The quantitative amount of ionization is described by the acid ionization equilibrium constant Ka We can also define p. Ka = -log 10 Ka The larger p. Ka the stronger the acid and visa versa

Acid Equilibrium Ionization Constant Similarly for bases we can define Kb and p. Kb= -log 10 Kb There is a simple relationship between Ka, Kb and Kw Ka Kb=Kw And p. Ka + p. Kb =p. Kw = 14. 00

Aqueous Acid-Base Equilibria When an acid ionizes in water it produces positive hydrogen ions and negative conjugate base ions OTOH, if we mixed a soluble salt of the negative (CN-) ion into water the following reaction would occur If we add these two reaction up the net is and Ka. Kb= = [H+][OH-]=Kw
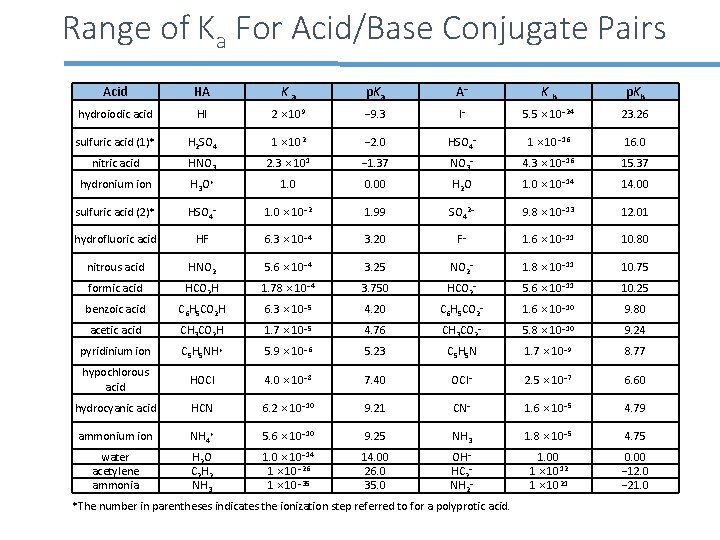
Range of Ka For Acid/Base Conjugate Pairs Acid HA K a p. Ka A− K b p. Kb hydroiodic acid HI 2 × 109 − 9. 3 I− 5. 5 × 10− 24 23. 26 sulfuric acid (1)* H 2 SO 4 1 × 102 − 2. 0 HSO 4− 1 × 10− 16 16. 0 nitric acid HNO 3 2. 3 × 101 − 1. 37 NO 3− 4. 3 × 10− 16 15. 37 hydronium ion H 3 O+ 1. 0 0. 00 H 2 O 1. 0 × 10− 14 14. 00 sulfuric acid (2)* HSO 4− 1. 0 × 10− 2 1. 99 SO 42− 9. 8 × 10− 13 12. 01 hydrofluoric acid HF 6. 3 × 10− 4 3. 20 F− 1. 6 × 10− 11 10. 80 nitrous acid HNO 2 5. 6 × 10− 4 3. 25 NO 2− 1. 8 × 10− 11 10. 75 formic acid HCO 2 H 1. 78 × 10− 4 3. 750 HCO 2− 5. 6 × 10− 11 10. 25 benzoic acid C 6 H 5 CO 2 H 6. 3 × 10− 5 4. 20 C 6 H 5 CO 2− 1. 6 × 10− 10 9. 80 acetic acid CH 3 CO 2 H 1. 7 × 10− 5 4. 76 CH 3 CO 2− 5. 8 × 10− 10 9. 24 pyridinium ion C 5 H 5 NH+ 5. 9 × 10− 6 5. 23 C 5 H 5 N 1. 7 × 10− 9 8. 77 hypochlorous acid HOCl 4. 0 × 10− 8 7. 40 OCl− 2. 5 × 10− 7 6. 60 hydrocyanic acid HCN 6. 2 × 10− 10 9. 21 CN− 1. 6 × 10− 5 4. 79 ammonium ion NH 4+ 5. 6 × 10− 10 9. 25 NH 3 1. 8 × 10− 5 4. 75 water acetylene ammonia H 2 O C 2 H 2 NH 3 1. 0 × 10− 14 1 × 10− 26 1 × 10− 35 14. 00 26. 0 35. 0 OH− HC 2− NH 2− 1. 00 1 × 1012 1 × 1021 0. 00 − 12. 0 − 21. 0 *The number in parentheses indicates the ionization step referred to for a polyprotic acid.

Direction of Acid-Base Reactions We know that if we mix HCl in water the hydrochloric acid will almost completely ionize HCl(aq) +H 2 O(l) H 3 O+(aq) + Cl-(aq) In the language of conjugate acid base pairs the HCl(aq) is a strong acid and the conjugate Cl-(aq) base is a weak base. The table to the left shows the relative strengths of different acid base pairs
- Slides: 20