Aquatic Biogeochemistry Most of Earth is covered by

Aquatic Biogeochemistry Most of Earth is covered by water and this greatly affects climate

Topics • • • Wetlands Lakes – Simple physics – Production & nutrients Streams and Rivers – Physics, Load Estuaries – Conservative vs. nonconservative Oceans – Circulation – Composition – Production & nutrients

Wetlands • Flooded systems (definitions for government • • purposes depend on number of days flooded) Often anaerobic, so use of alternate electron acceptors is important (sulfate, nitrate, metals) Organic matter degrades slowly without O 2, and may persist

Wetlands • • Websters: land or areas (as marshes or swamps) that are covered often intermittently with shallow water or have soil saturated with moisture. EPA: "those areas that are inundated or saturated by surface or groundwater at a frequency and duration sufficient to support, and that under normal circumstances do support, a prevalence of vegetation typically adapted for life in saturated soil conditions. Wetlands generally include swamps, marshes, bogs and similar areas. ”

Redox and Wetlands Uplands Wetlands O 2 H 2 O NO 3 - NH 4+ Mn 2+ Fe 3+ Fe 2+ SO 42 - H 2 S CO 2 CH 4
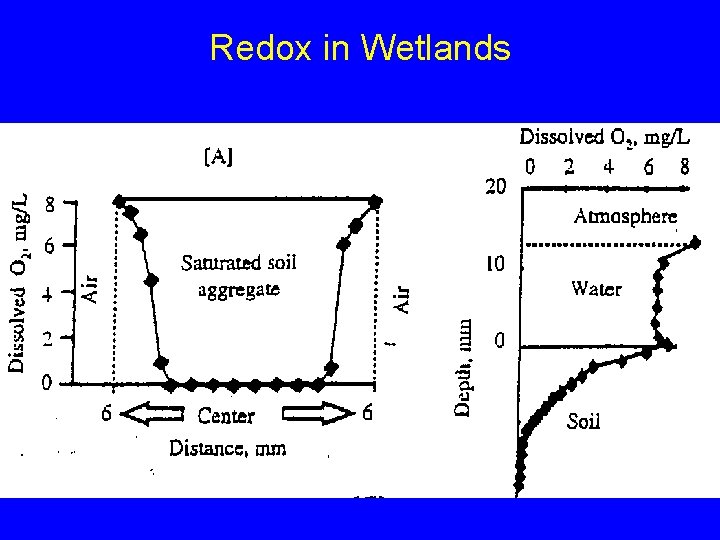
Redox in Wetlands
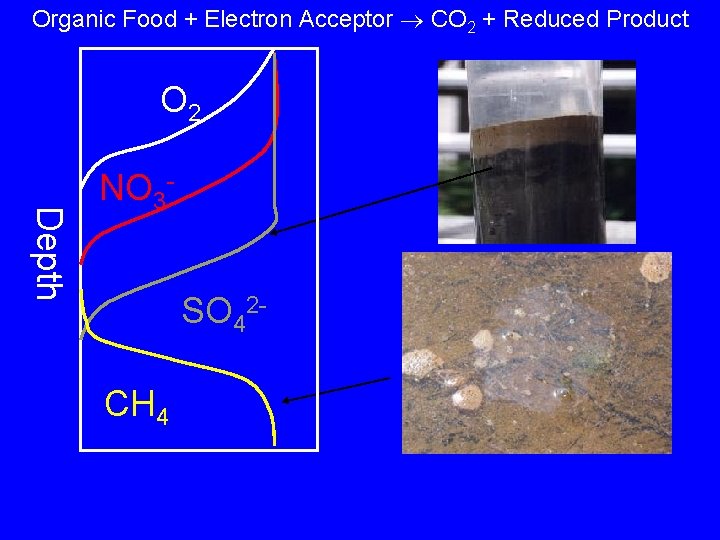
Organic Food + Electron Acceptor CO 2 + Reduced Product O 2 Depth NO 3 SO 42 CH 4
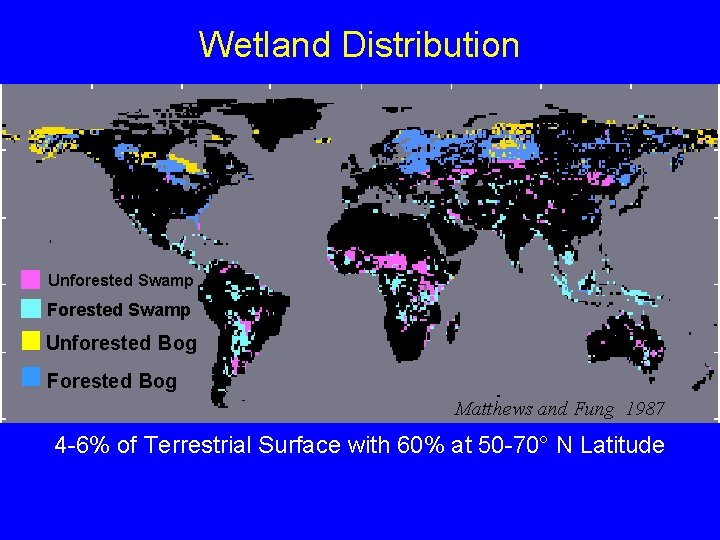
Wetland Distribution Unforested Swamp Forested Swamp Unforested Bog Forested Bog Matthews and Fung 1987 4 -6% of Terrestrial Surface with 60% at 50 -70° N Latitude
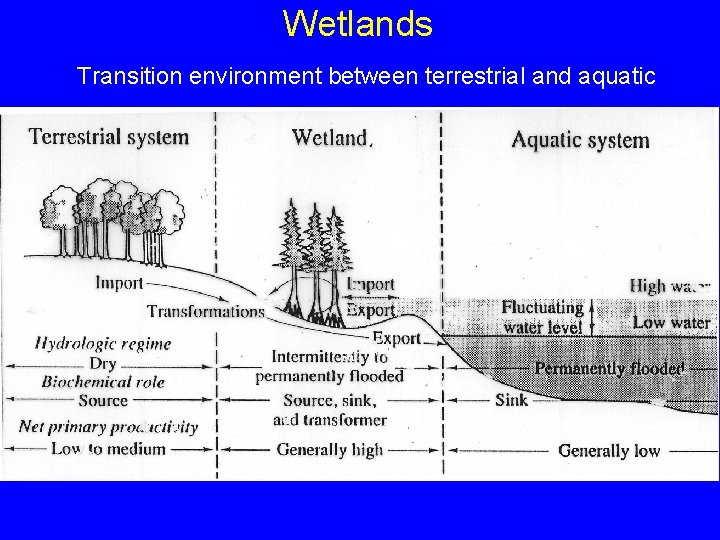
Wetlands Transition environment between terrestrial and aquatic

Wetland Types - Freshwater • Inland – Freshwater marshes • Emergent soft-stemmed – Peatlands • Deep peats (boreal), bogs and fens – Freshwater swamps • Forested (SE USA) – Riparian systems • Along rivers and streams

Wetland Types - Coastal (Marine) • Coastal – Tidal salt marshes • Salt-adapted (Spartina) – Tidal freshwater marshes – Mangroves

Wetland Importance • • Wildlife habitat – Waterfowl, fish, shellfish, fur animals, Physical – Moderate flooding – Aesthetic and heritage value Biogeochemical – Improve water quality – Sinks for nutrients and metals – Stability of important gases (S, C, N) Recreation – Hunting, fishing, boating – Mosquito watching
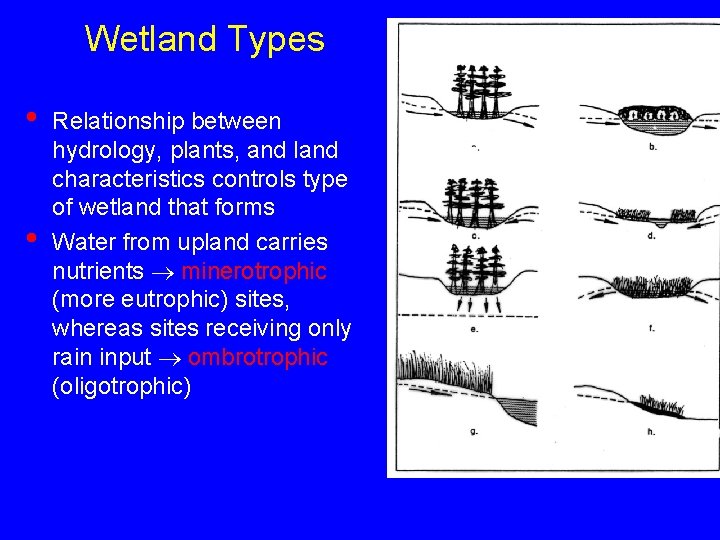
Wetland Types • • Relationship between hydrology, plants, and land characteristics controls type of wetland that forms Water from upland carries nutrients minerotrophic (more eutrophic) sites, whereas sites receiving only rain input ombrotrophic (oligotrophic)
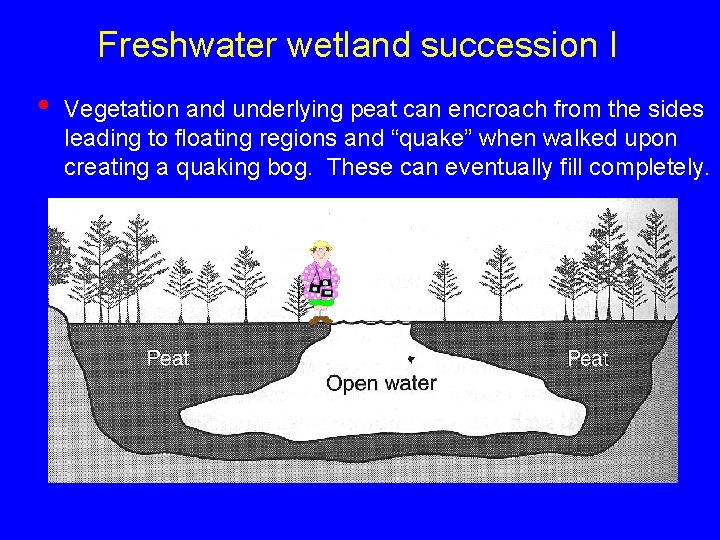
Freshwater wetland succession I • Vegetation and underlying peat can encroach from the sides leading to floating regions and “quake” when walked upon creating a quaking bog. These can eventually fill completely.

Freshwater wetland succession I • Vegetation and underlying peat can encroach from the sides leading to floating regions and “quake” when walked upon creating a quaking bog. These can eventually fill completely.

Freshwater wetland succession I • • Anoxic, low p. H peats degrade very slowly and allow organic matter to persist and to be preserved in some cases Lucky fellow at right was found strangled (a noose around his neck) in a bog in Tolland DK, and was completely intact. He was radiocarbon dated at 2000 years old! Tolland Man
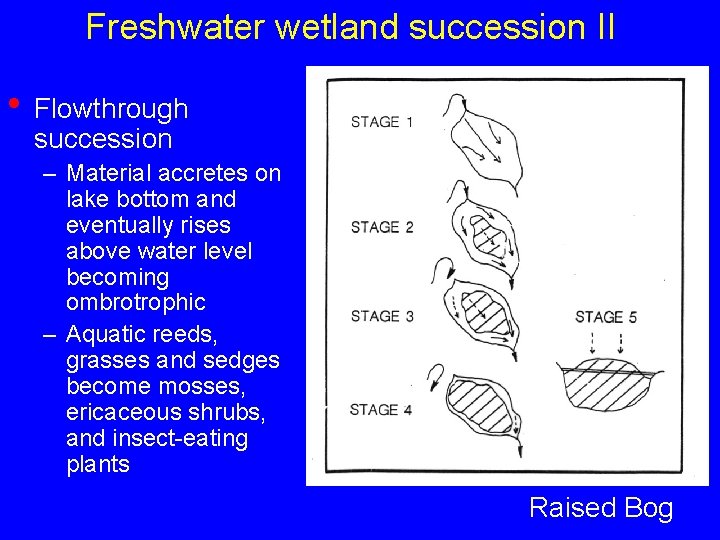
Freshwater wetland succession II • Flowthrough succession – Material accretes on lake bottom and eventually rises above water level becoming ombrotrophic – Aquatic reeds, grasses and sedges become mosses, ericaceous shrubs, and insect-eating plants Raised Bog

Freshwater wetland plant succession • Although succession is complex due to hydrologic, climate, and terrain differences, you often see the following order: – – – Submersed vegetation Floating vegetation Reed swamp Aquatic grasses and sedges Mosses (sphagnum) p. H decreases from ~7 to as low as 3. 5
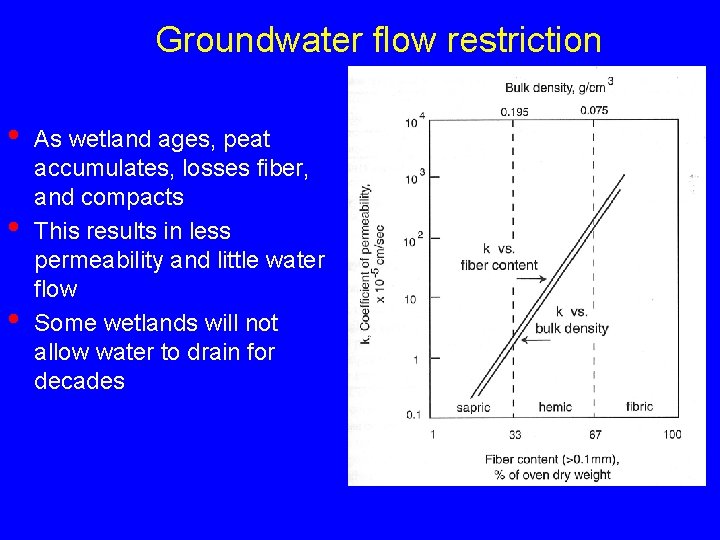
Groundwater flow restriction • • • As wetland ages, peat accumulates, losses fiber, and compacts This results in less permeability and little water flow Some wetlands will not allow water to drain for decades

Freshwater wetland productivity • Production can be very high • Affected greatly by hydraulics – wetlands with high water flow receive sufficient nutrients and are very productive – Wetlands that dry periodically recycle nutrients and destroy reduced compounds that are toxic (e. g. , sulfides) • In general, marshes are most productive and bogs least – Marshes have high hydraulic conductivity that brings in minerals and removes toxins – Bogs have low p. H, very poor hydraulic conductivity, and very low mineral content
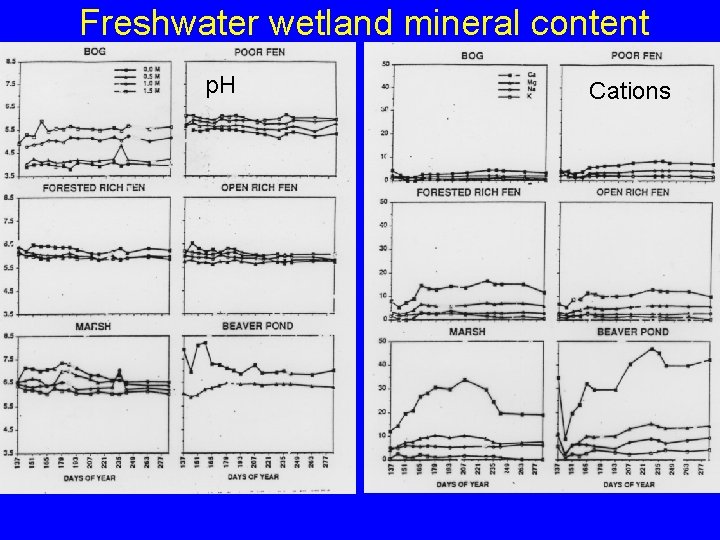
Freshwater wetland mineral content p. H Cations

Sphagnum mosses • • Mosses are the ‘climax’ species Aid in decreasing the p. H by replacing consumed cations with H+ ions leading to acidification Produce anti-degradative compounds Adapted to hold on to water so system can stay moist even when water table is decreasing

Marine Wetlands (salt marshes) • • • Salt marshes distributed in middle and high latitudes worldwide where sediment accumulation is equal to or greater than land subsidence or sea level rise. Protect land from the sea Support fish spawning and feeding Highly productive Outwell organic matter and nutrients

• Marine Wetlands (salt marshes) Widely distributed, but vegetation composition varies as environment varies
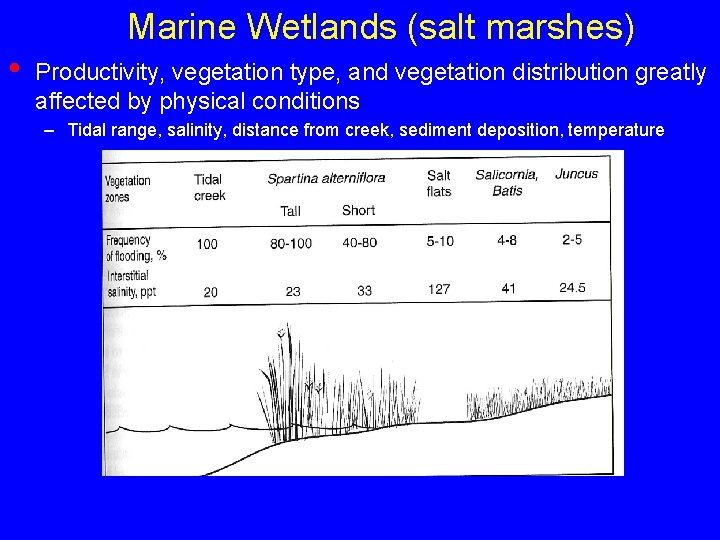
Marine Wetlands (salt marshes) • Productivity, vegetation type, and vegetation distribution greatly affected by physical conditions – Tidal range, salinity, distance from creek, sediment deposition, temperature

Lakes • Lake stratification • Primary production and nutrients
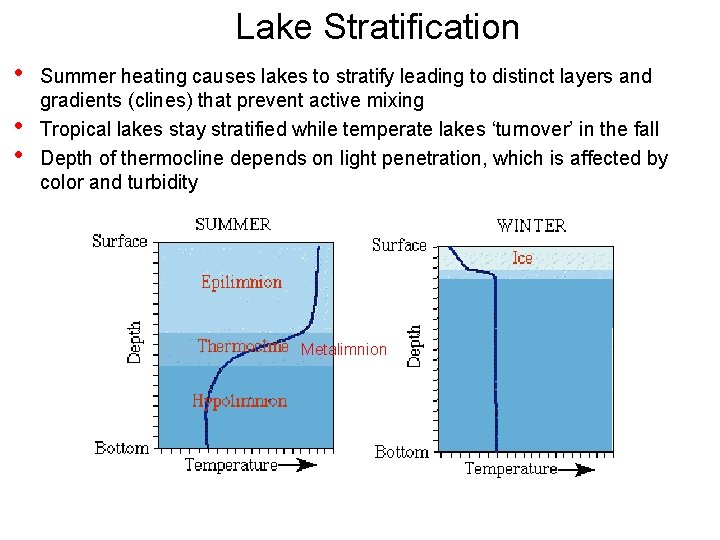
Lake Stratification • • • Summer heating causes lakes to stratify leading to distinct layers and gradients (clines) that prevent active mixing Tropical lakes stay stratified while temperate lakes ‘turnover’ in the fall Depth of thermocline depends on light penetration, which is affected by color and turbidity Metalimnion
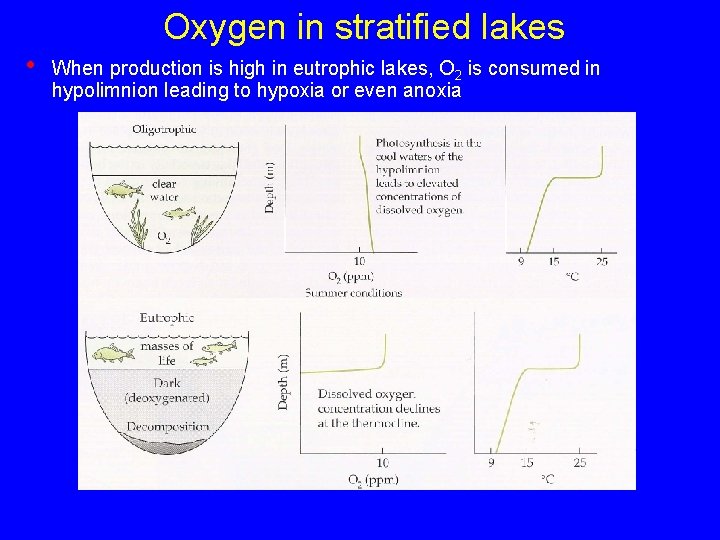
Oxygen in stratified lakes • When production is high in eutrophic lakes, O 2 is consumed in hypolimnion leading to hypoxia or even anoxia
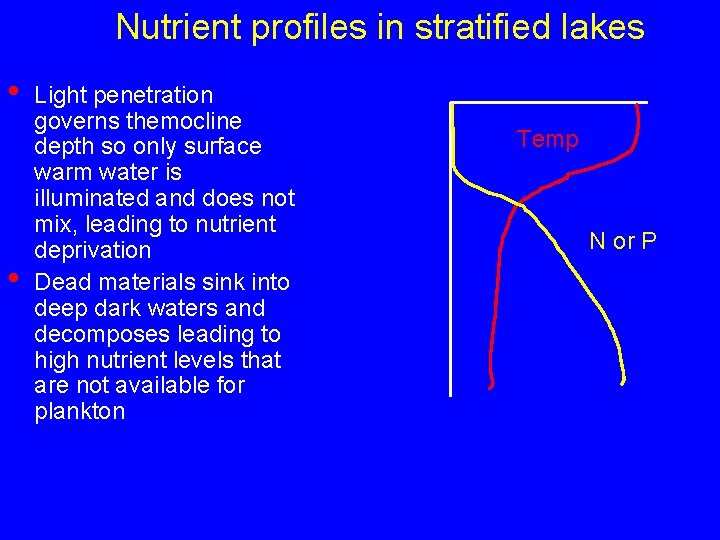
Nutrient profiles in stratified lakes • • Light penetration governs themocline depth so only surface warm water is illuminated and does not mix, leading to nutrient deprivation Dead materials sink into deep dark waters and decomposes leading to high nutrient levels that are not available for plankton Temp N or P
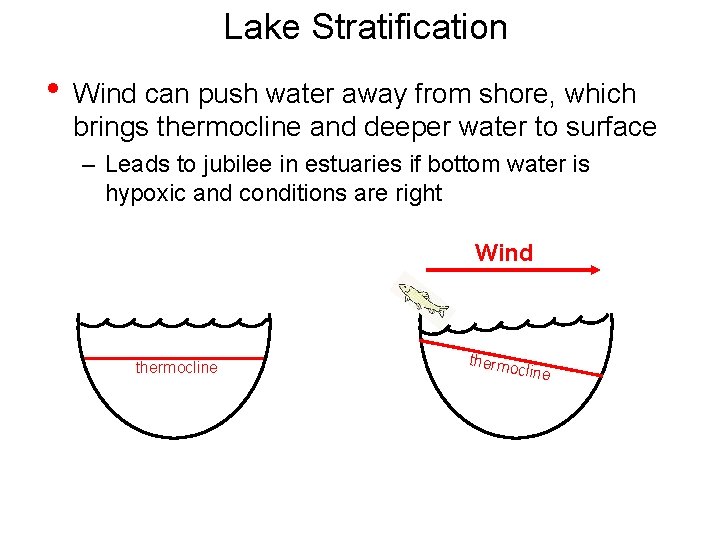
Lake Stratification • Wind can push water away from shore, which brings thermocline and deeper water to surface – Leads to jubilee in estuaries if bottom water is hypoxic and conditions are right Wind thermocline thermo cline

Primary production in lakes • Plankton vs. macrophytes – Small lakes have majority of production as macrophytes – Larger lakes have more open water and phytoplankton dominate – Macrophytes and terrestrial organics have longer residence time than plankton that turn over rapidly – Plankton have relatively fixed C: N: P ratios whereas macrophytes can vary greatly. Therefore, ratio of nutrient inputs (C: N: P) affect production especially in planktondominated lakes – Production within lake is autochthonous while production from the outside is allochthonous
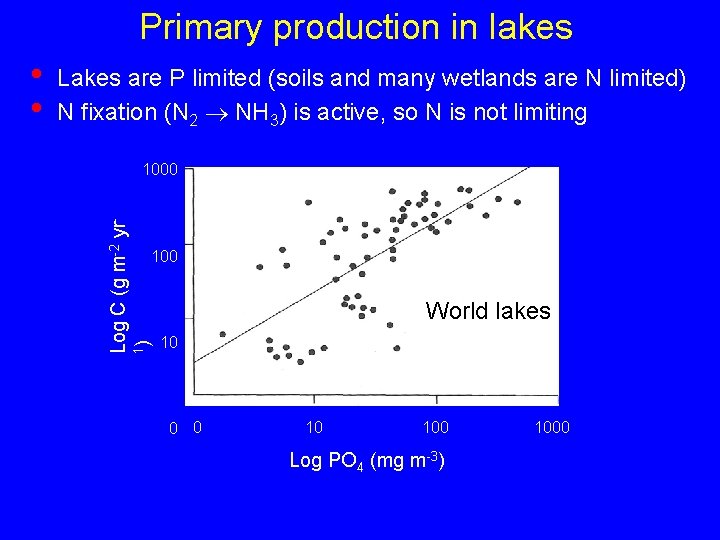
Primary production in lakes Lakes are P limited (soils and many wetlands are N limited) N fixation (N 2 NH 3) is active, so N is not limiting 1000 Log C (g m-2 yr 1) • • 100 World lakes 10 0 0 10 100 Log PO 4 (mg m-3) 1000

Primary production in lakes • Low productivity (oligotrophic) lakes have large volume relative the area that drains nutrients. Therefore, more nutrients come from precipitation than in high productivity lakes (eutrophic) that usually have smaller volumes and are influenced by runoff Sources of N and P as % of total input Precipitation Runoff N P Oligotrophic lakes 56 50 44 50 Eutrophic lakes 12 7 88 93

Rivers and streams • Stream flow affected by – Infiltration rates in soils, which are affected by • Soil porosity (clays are low while sand is high) • Vegetation (little surface runoff in forests) – lowers rain impact allowing seepage into soil – Produces conduits to depth – Removes water via transpiration – Groundwater flow • Hydraulic conductivity • Hydraulic gradient – Downward flow that occurs when soils exceed waterholding capacity (amount of water held against gravity)
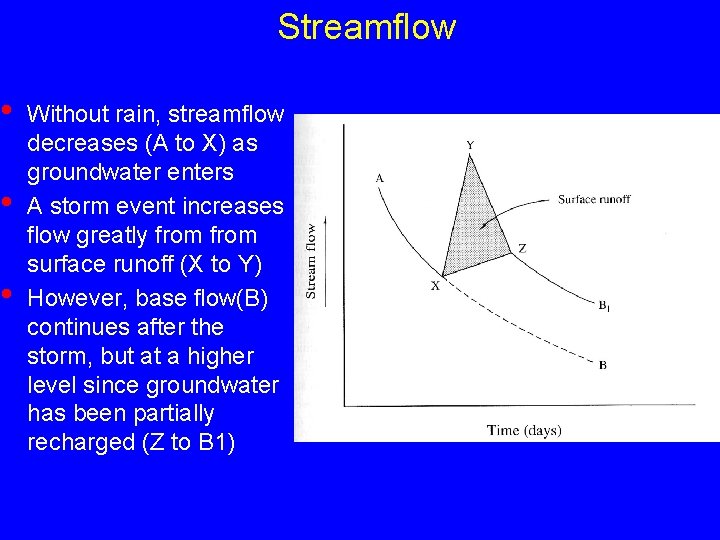
Streamflow • • • Without rain, streamflow decreases (A to X) as groundwater enters A storm event increases flow greatly from surface runoff (X to Y) However, base flow(B) continues after the storm, but at a higher level since groundwater has been partially recharged (Z to B 1)

Stream load • Material transported by streams in either dissolved or particulate form – Dissolved from rainfall and soil solutions (from weathering) (dissolved solids) – Particulate from weathering and erosion • Includes suspended load and bed load (moves along bottom) • In a system with no significant chemistry or biology, ions are simply diluted as flow increases – Works somewhat for unreactive species like major ions (Mg, Na, Si, Cl) – Constituents consumed by biota affected by growing season, etc • Concentrations higher on rising limb of flow compared to declining limb (hysteresis)
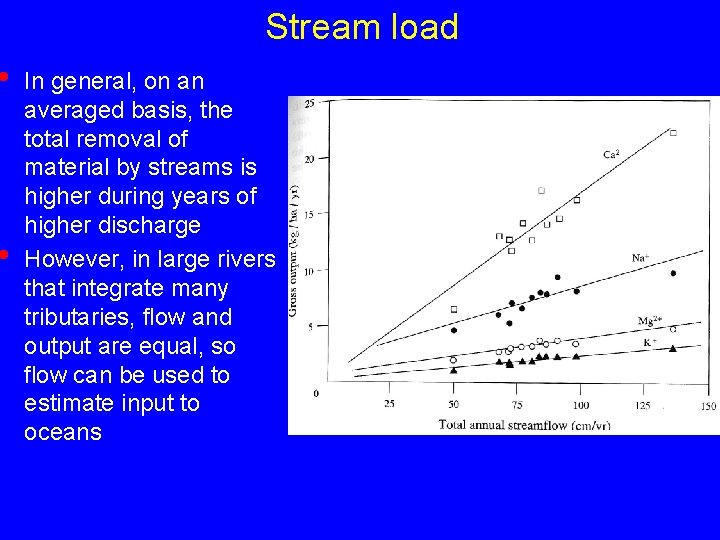
• • Stream load In general, on an averaged basis, the total removal of material by streams is higher during years of higher discharge However, in large rivers that integrate many tributaries, flow and output are equal, so flow can be used to estimate input to oceans
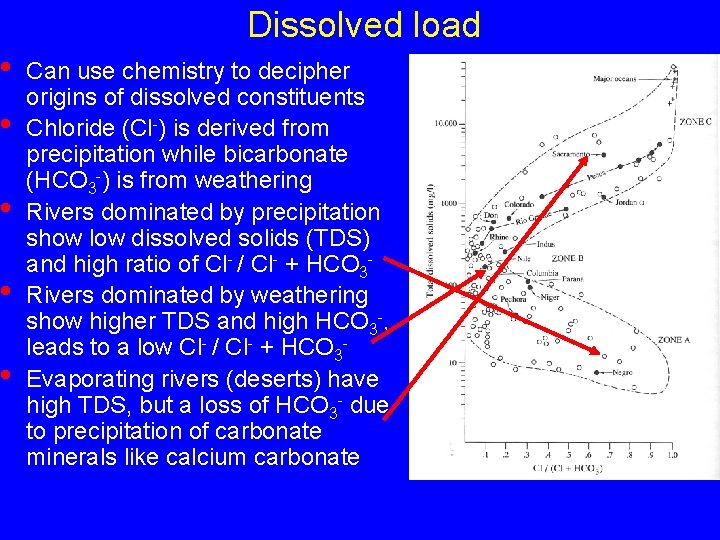
• • • Dissolved load Can use chemistry to decipher origins of dissolved constituents Chloride (Cl-) is derived from precipitation while bicarbonate (HCO 3 -) is from weathering Rivers dominated by precipitation show low dissolved solids (TDS) and high ratio of Cl- / Cl- + HCO 3 Rivers dominated by weathering show higher TDS and high HCO 3 -, leads to a low Cl- / Cl- + HCO 3 Evaporating rivers (deserts) have high TDS, but a loss of HCO 3 - due to precipitation of carbonate minerals like calcium carbonate

Particulate load • Increases exponentially with water flow • Most during episodic events • Affected by vegetation, • elevation, relief, degree of runoff Sediments carry P, pollutant metals and organics, and bury habitat
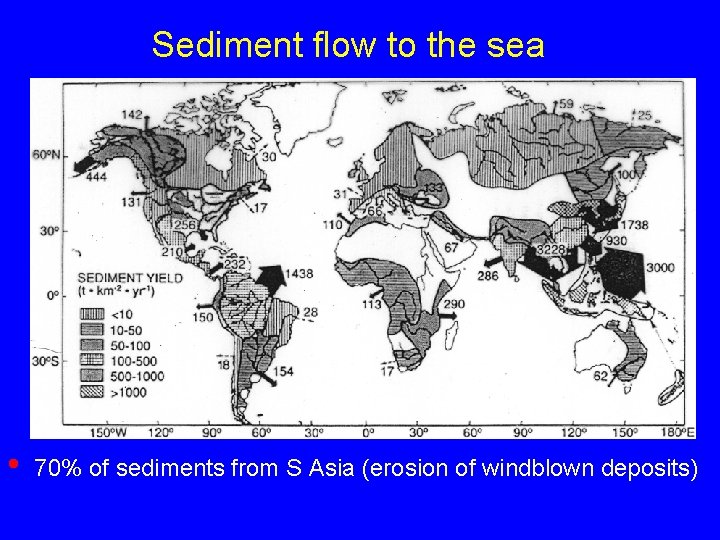
Sediment flow to the sea • 70% of sediments from S Asia (erosion of windblown deposits)

Estuaries • Transition between land seas and fresh and • seawater Dynamic mixing zone since – p. H of rivers is slightly acid while seawater is slightly basic – Some materials precipitate while others coagulate – Degree of tidal inundation affects whether estuary is water column or sediment dominated – Rivers tends to be P limited while coastal oceans can be N limited (or Si), so nutrient mixing can occur – Highly productive
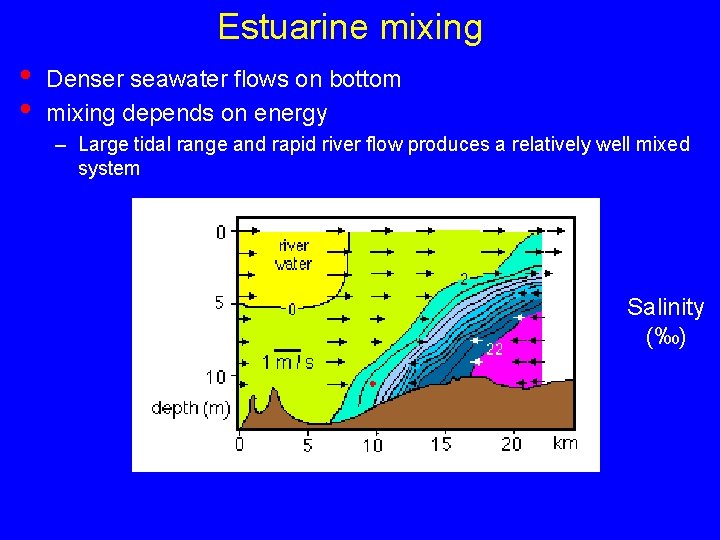
Estuarine mixing • • Denser seawater flows on bottom mixing depends on energy – Large tidal range and rapid river flow produces a relatively well mixed system Salinity (‰)
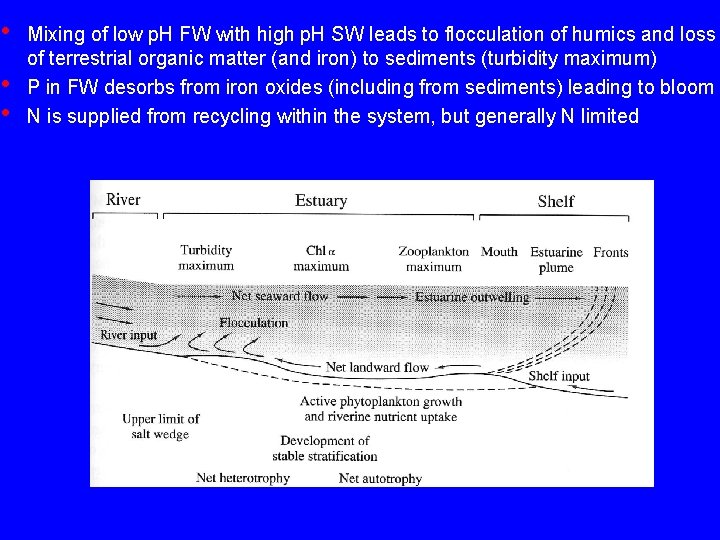
• • • Mixing of low p. H FW with high p. H SW leads to flocculation of humics and loss of terrestrial organic matter (and iron) to sediments (turbidity maximum) P in FW desorbs from iron oxides (including from sediments) leading to bloom N is supplied from recycling within the system, but generally N limited

Estuarine mixing • • Conservative elements are unreactive with little to no biological uptake or production (e. g. , Cl, Na, Mg, K), and do not vary in seawater relative to salinity (or each other) Non-conservative elements display significant reactivity, are utilized by life, and display variation Estuarine mixing diagrams Conservative Element Mixing Sink Salinity Source

Oceans • Plate tectonics • Circulation – Surface and deep • Chemical composition • Primary production and • • • O 2 Carbonate biogeochemistry Nutrient cycling Sediments
Earth Topography
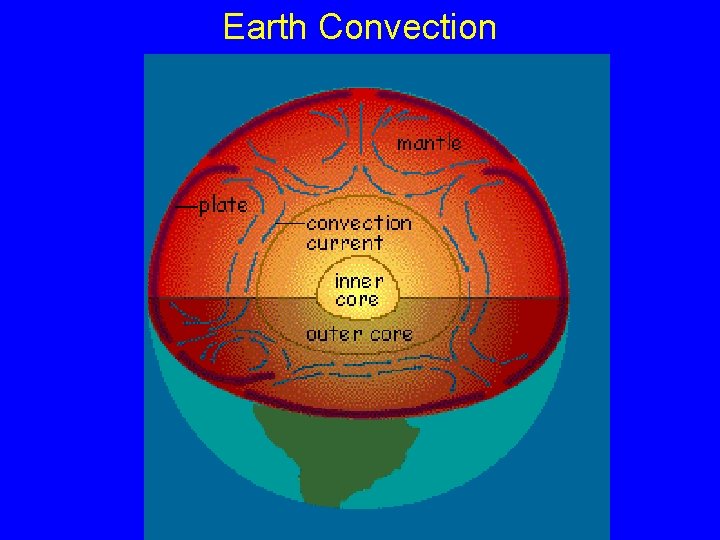
Earth Convection
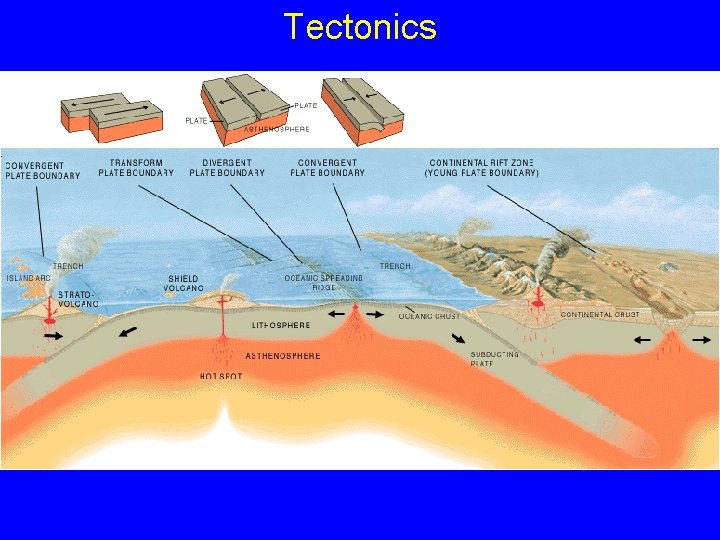
Tectonics
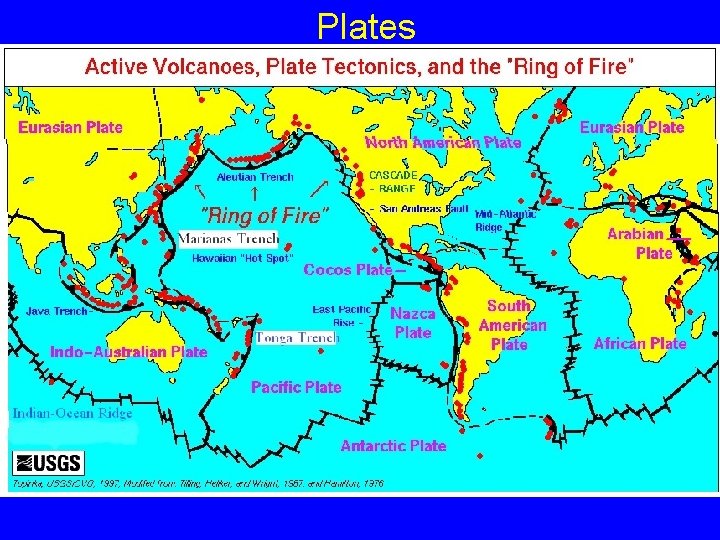
Plates
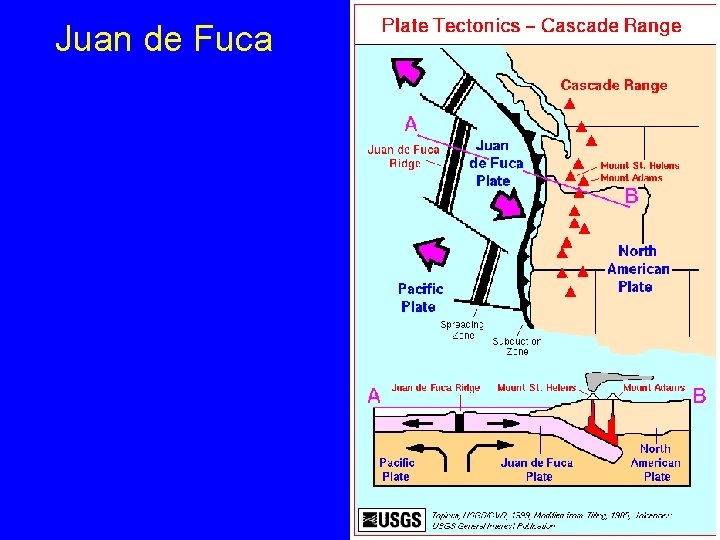
Juan de Fuca
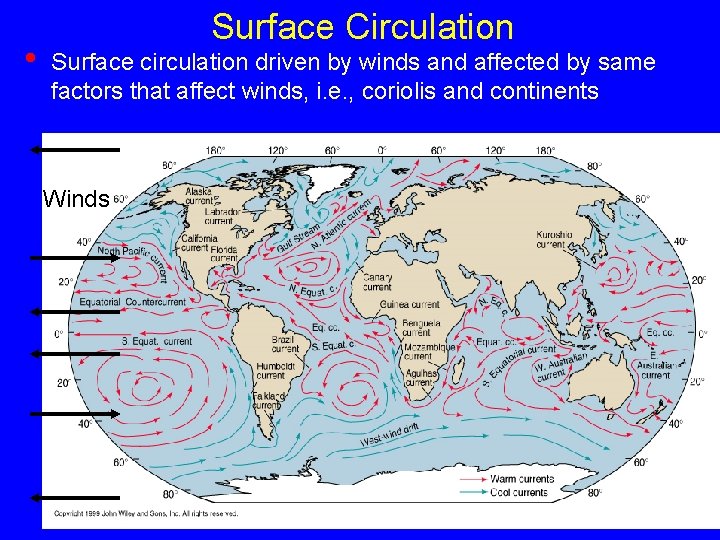
• Surface Circulation Surface circulation driven by winds and affected by same factors that affect winds, i. e. , coriolis and continents Winds
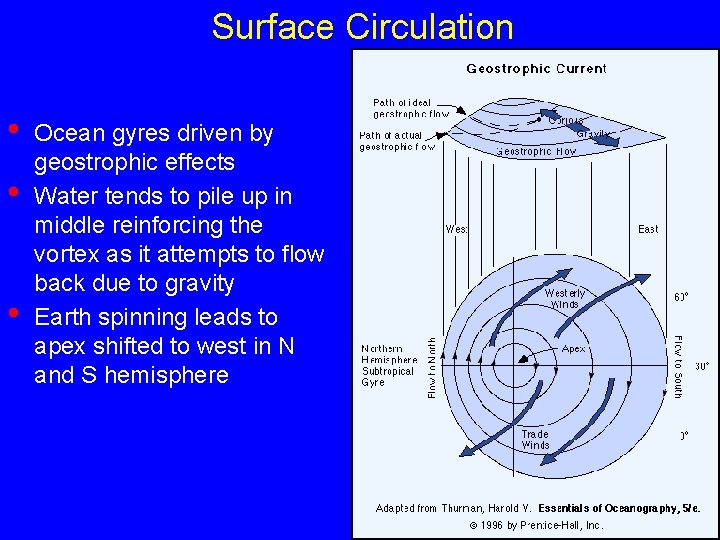
Surface Circulation • • • Ocean gyres driven by geostrophic effects Water tends to pile up in middle reinforcing the vortex as it attempts to flow back due to gravity Earth spinning leads to apex shifted to west in N and S hemisphere
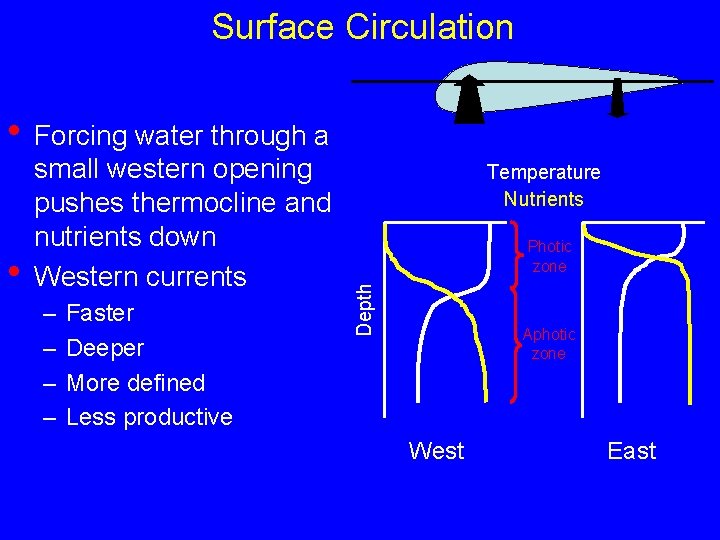
Surface Circulation • Forcing water through a – – Faster Deeper More defined Less productive Temperature Nutrients Photic zone Depth • small western opening pushes thermocline and nutrients down Western currents Aphotic zone West East

Currents in western sides of oceans • • Western currents (like the Gulf Stream) display sharp demarcation from coastal and mid-gyre waters Tend to meander and break into rings, which can persist for several months
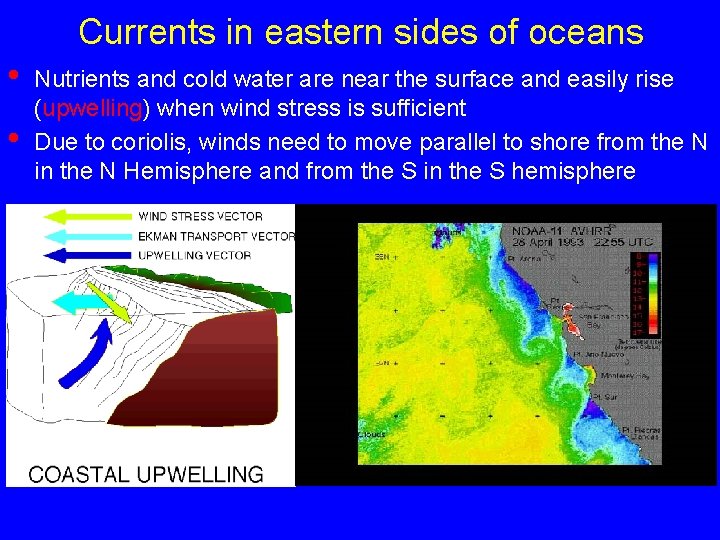
Currents in eastern sides of oceans • • Nutrients and cold water are near the surface and easily rise (upwelling) when wind stress is sufficient Due to coriolis, winds need to move parallel to shore from the N in the N Hemisphere and from the S in the S hemisphere
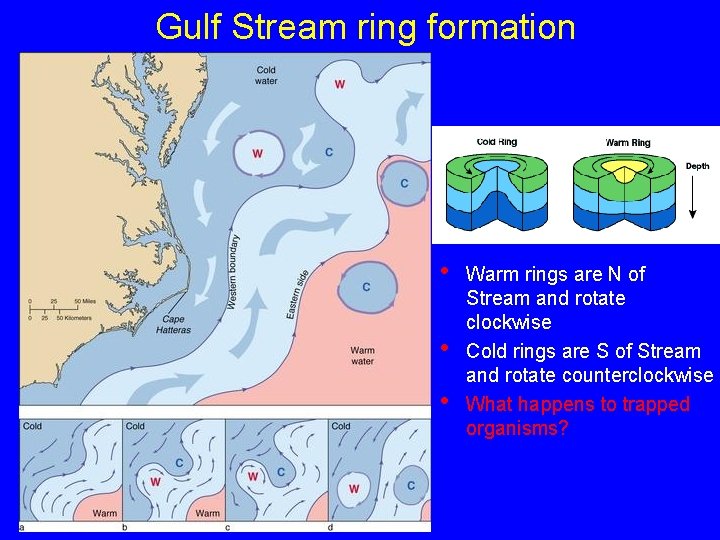
Gulf Stream ring formation • • • Warm rings are N of Stream and rotate clockwise Cold rings are S of Stream and rotate counterclockwise What happens to trapped organisms?
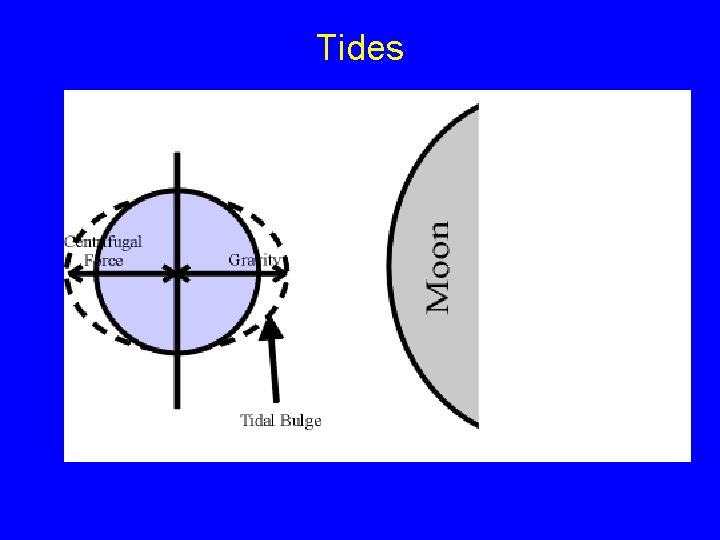
Tides
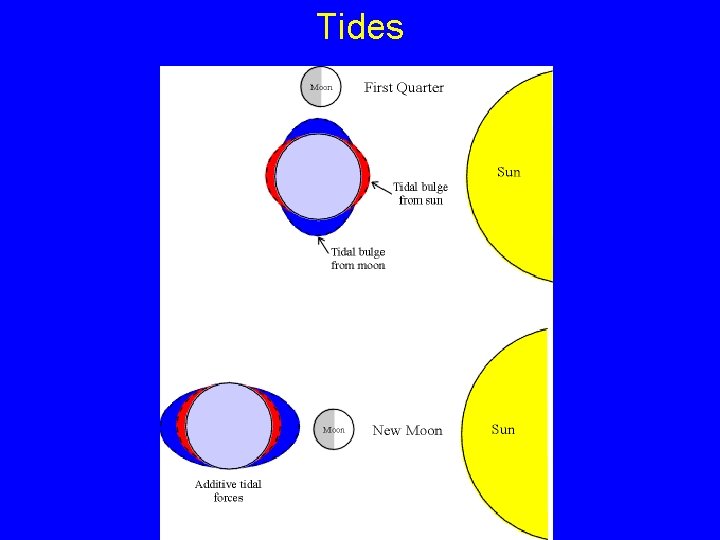
Tides
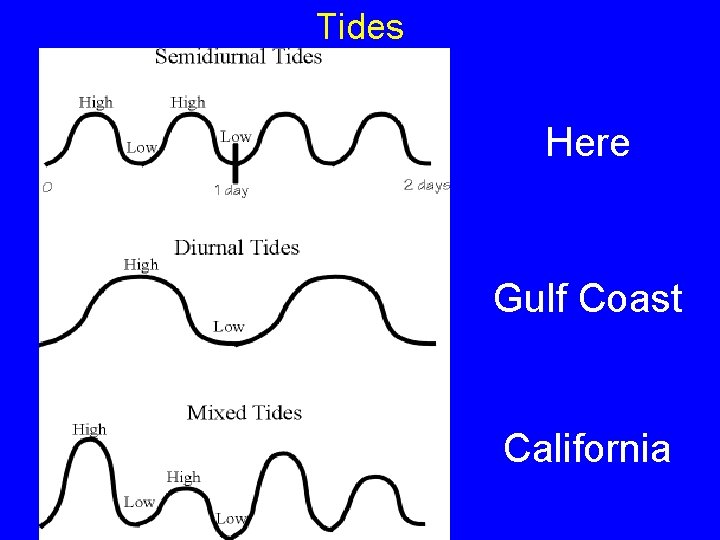
Tides Here Gulf Coast California
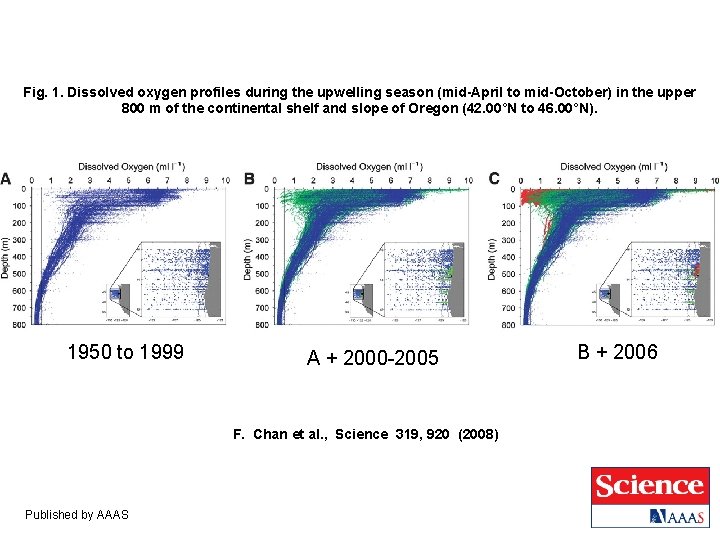
Fig. 1. Dissolved oxygen profiles during the upwelling season (mid-April to mid-October) in the upper 800 m of the continental shelf and slope of Oregon (42. 00°N to 46. 00°N). 1950 to 1999 A + 2000 -2005 F. Chan et al. , Science 319, 920 (2008) Published by AAAS B + 2006
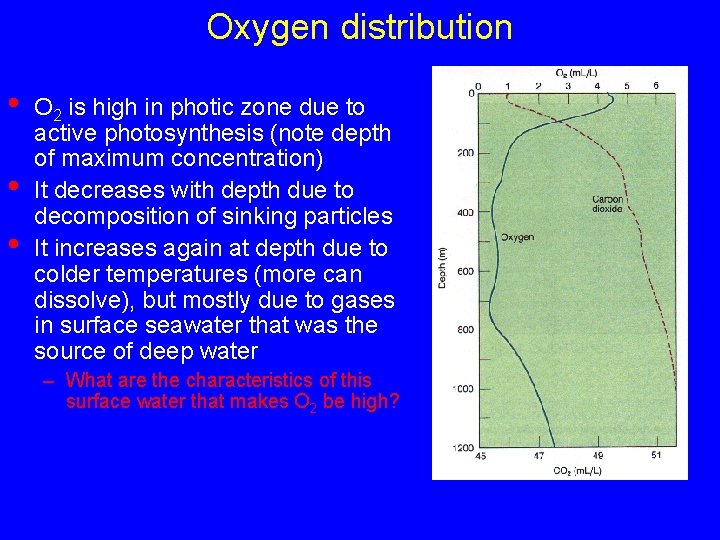
Oxygen distribution • • • O 2 is high in photic zone due to active photosynthesis (note depth of maximum concentration) It decreases with depth due to decomposition of sinking particles It increases again at depth due to colder temperatures (more can dissolve), but mostly due to gases in surface seawater that was the source of deep water – What are the characteristics of this surface water that makes O 2 be high?

El Niño and La Niña • • • In normal years (middle), trades are strong and pile water up in Indonesia while upwelling is strong in Peru In el niño years ( bottom), trades relax and upwelling ceases as thermocline deepens La niña is the cold part of the cycle, but the magnitude can be small or large La niña (Dec 1998) Normal (Dec 1993) El niño (Dec 1997)

Deep ocean circulation • Winds have no effect at depth so deep • • circulation is driven by density differences due to temperature and salinity (thermohaline circulation) Deep water refers to everything below the surface layer (<100 -200 meters) There are intermediate waters too, but the deep water largely control atmospheric processes on the long term

Deep ocean circulation • • Almost all deep water originates in the Atlantic Ice formation in the N leads to cold salty water that sinks (NADW) Colder, fresher water sinks in the S (ABW), but stays in Atlantic water moves into Indian and then Pacific N. Atlantic Deep Water (NADW) Antarctic Bottom Water (ABW)
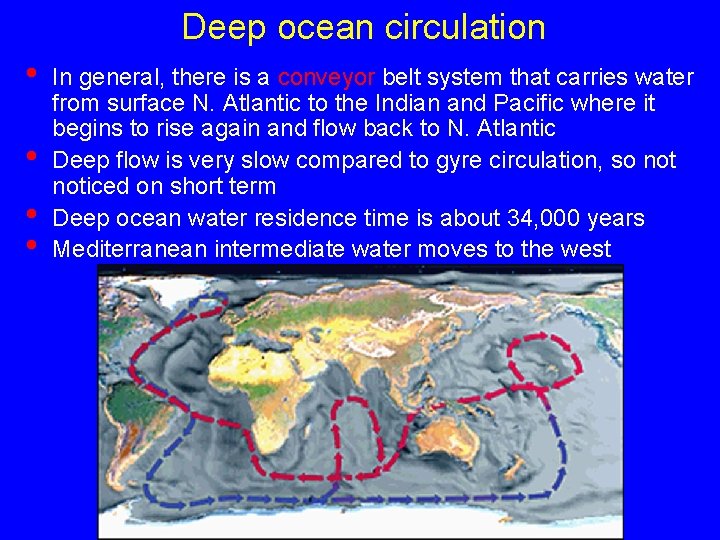
Deep ocean circulation • • In general, there is a conveyor belt system that carries water from surface N. Atlantic to the Indian and Pacific where it begins to rise again and flow back to N. Atlantic Deep flow is very slow compared to gyre circulation, so noticed on short term Deep ocean water residence time is about 34, 000 years Mediterranean intermediate water moves to the west
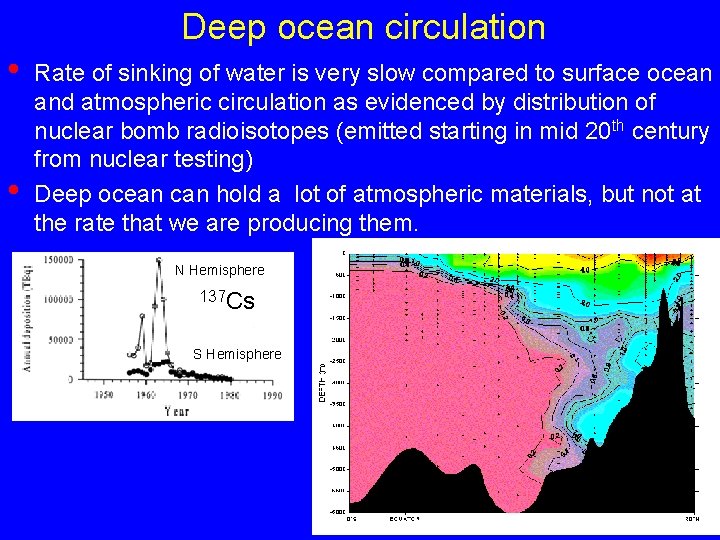
Deep ocean circulation • • Rate of sinking of water is very slow compared to surface ocean and atmospheric circulation as evidenced by distribution of nuclear bomb radioisotopes (emitted starting in mid 20 th century from nuclear testing) Deep ocean can hold a lot of atmospheric materials, but not at the rate that we are producing them. N Hemisphere 137 Cs S Hemisphere

Major ions in seawater • Major ions in order of abundance – Cl-, Na+, SO 42 -, Mg 2+, Ca 2+, K+, HCO 3 -, Br-, Sr – Each is conservative so knowledge of concentration of one (or of salinity in general) can be used to calculate any other – Residence times are long, e. g. , • • • Cl - 120 million years Na - 75 million years SO 4 - 12 million years Mg - 14 million years Ca - 1. 1 million years • These are in steady state. Why is that important?
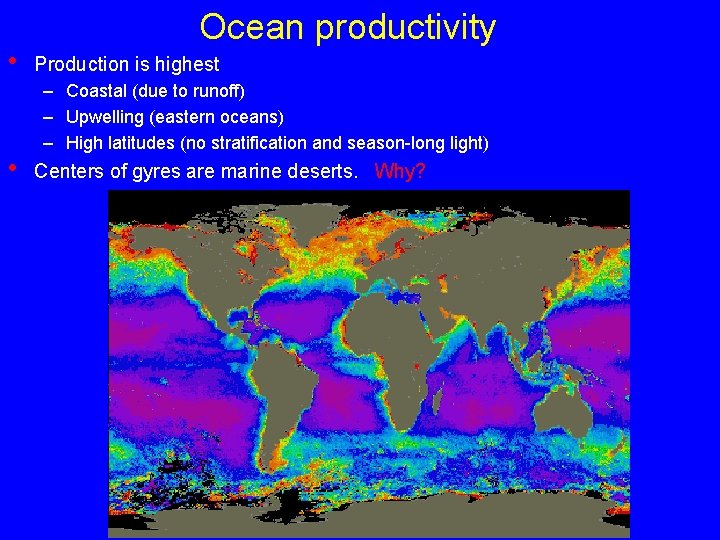
• • Ocean productivity Production is highest – Coastal (due to runoff) – Upwelling (eastern oceans) – High latitudes (no stratification and season-long light) Centers of gyres are marine deserts. Why?

Fate of marine production • • Production equals (slightly less) terrestrial Due to phytoplankton and picoplankton (0. 2 -2. 0 µm) Production is consumed rapidly so primary producer pool is small with short residence time (hours vs. years for terrestrial habitats) 90% is remineralized in surface Sinking particles take an average of 10 days to sink, but small ones take much longer Particles degrade as they sink so <1% of NPP is degraded in sediments No terrestrial organic matter in deep ocean. Why not? What are sediments made of?
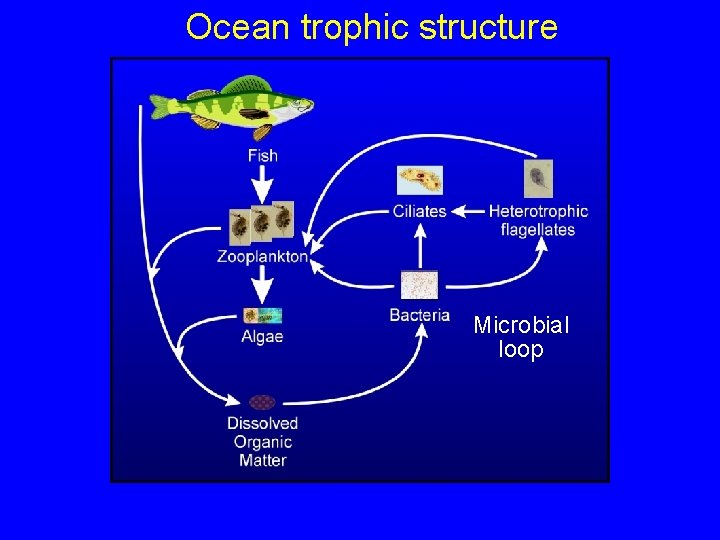
Ocean trophic structure Microbial loop
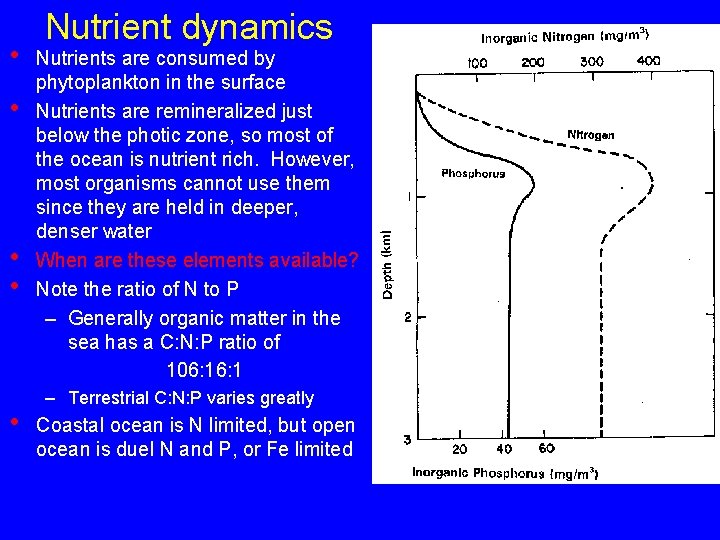
• • • Nutrient dynamics Nutrients are consumed by phytoplankton in the surface Nutrients are remineralized just below the photic zone, so most of the ocean is nutrient rich. However, most organisms cannot use them since they are held in deeper, denser water When are these elements available? Note the ratio of N to P – Generally organic matter in the sea has a C: N: P ratio of 106: 1 – Terrestrial C: N: P varies greatly Coastal ocean is N limited, but open ocean is duel N and P, or Fe limited
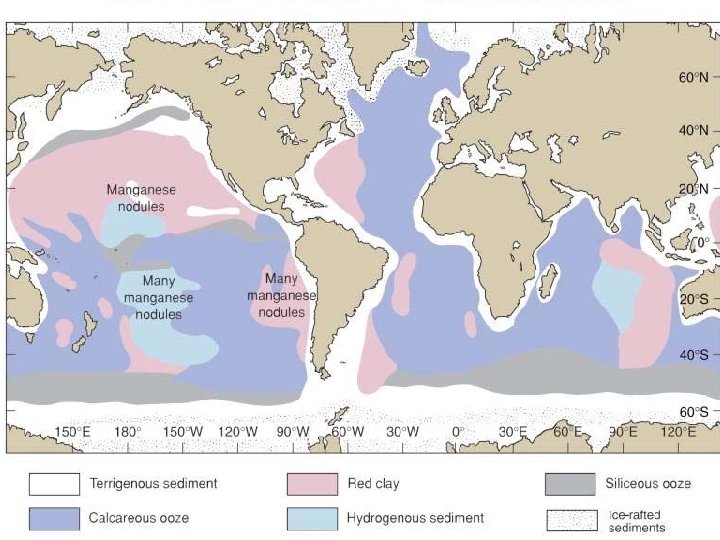
Ocean Sediments

• Ocean Sediments Major sediment types – Siliceous from diatoms (high latitudes and upwelling areas) – Calcium carbonate from coccolithophores and other carbonateprecipitating species • Ca. CO 3 dissolves at high pressure and cold temperatures and when CO 2 concentrations increase • Therefore, not found at depths below ~3, 700 m in Atlantic and ~1, 000 m in Pacific. What causes the difference and why present in mid-ocean? – Red clays (terrestrial silicates) are found everywhere that biogenic sources are absent – Organic deposits in anoxic basins and near shore
- Slides: 74