Approved indications and breakdown by region 22 Indications
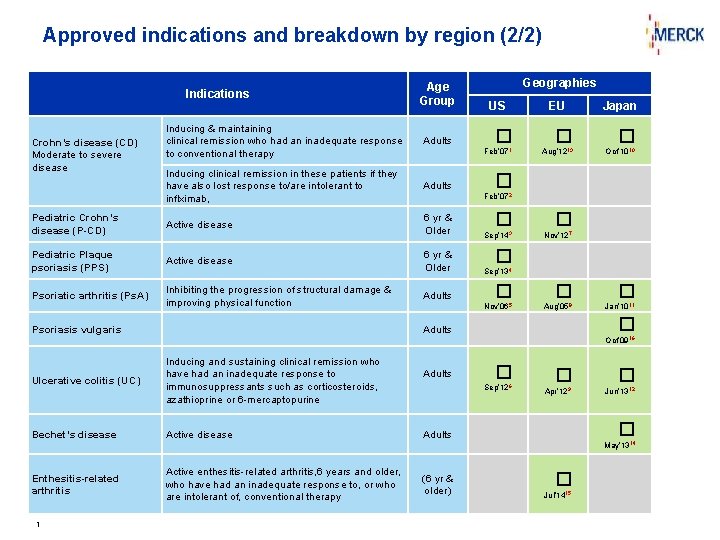
Approved indications and breakdown by region (2/2) Indications Age Group Geographies US EU Inducing & maintaining clinical remission who had an inadequate response to conventional therapy Adults Inducing clinical remission in these patients if they have also lost response to/are intolerant to inflximab, Adults Pediatric Crohn’s disease (P-CD) Active disease 6 yr & Older � � Sep’ 143 Nov’ 127 Pediatric Plaque psoriasis (PPS) Active disease 6 yr & Older Sep’ 134 Psoriatic arthritis (Ps. A) Inhibiting the progression of structural damage & improving physical function Crohn’s disease (CD) Moderate to severe disease Psoriasis vulgaris Japan � � � Feb’ 071 Aug’ 1213 Oct’ 1010 � Feb’ 072 Adults � � Nov’ 065 Aug’ 058 Jan’ 1011 � Adults Oct’ 0916 Ulcerative colitis (UC) Bechet’s disease Inducing and sustaining clinical remission who have had an inadequate response to immunosuppressants such as corticosteroids, azathioprine or 6 -mercaptopurine Active disease Adults � Sep’ 126 � � Apr’ 129 Jun’ 1312 � Adults May’ 1314 Enthesitis-related arthritis 1 Active enthesitis-related arthritis, 6 years and older, who have had an inadequate response to, or who are intolerant of, conventional therapy (6 yr & older) � Jul’ 1415
- Slides: 1