Approval Estimates UPDATED Calculations and assumptions Blueocean estimates
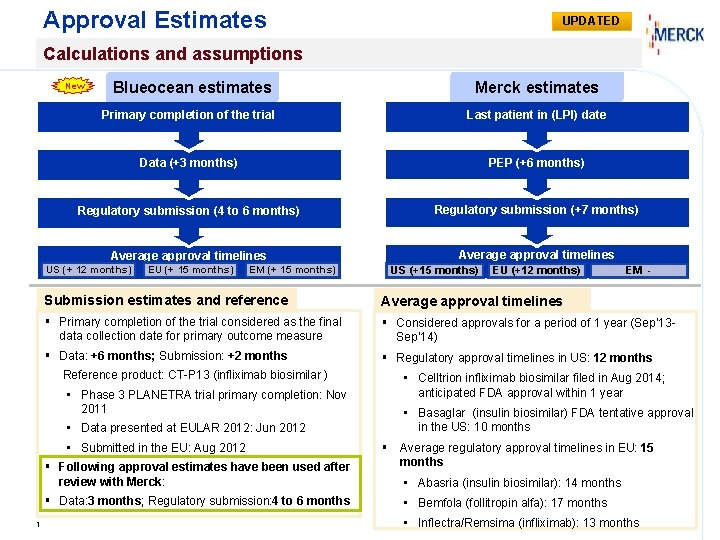
Approval Estimates UPDATED Calculations and assumptions Blueocean estimates Merck estimates Primary completion of the trial Last patient in (LPI) date Data (+3 months) PEP (+6 months) Regulatory submission (4 to 6 months) Regulatory submission (+7 months) Average approval timelines New US (+ 12 months) EU (+ 15 months) EM (+ 15 months) US (+15 months) EM - Submission estimates and reference Average approval timelines § Primary completion of the trial considered as the final data collection date for primary outcome measure § Considered approvals for a period of 1 year (Sep’ 13 Sep’ 14) § Data: +6 months; Submission: +2 months § Regulatory approval timelines in US: 12 months Reference product: CT-P 13 (infliximab biosimilar ) • Celltrion infliximab biosimilar filed in Aug 2014; anticipated FDA approval within 1 year • Phase 3 PLANETRA trial primary completion: Nov 2011 • Basaglar (insulin biosimilar) FDA tentative approval in the US: 10 months • Data presented at EULAR 2012: Jun 2012 • Submitted in the EU: Aug 2012 § Following approval estimates have been used after review with Merck: § Data: 3 months; Regulatory submission: 4 to 6 months 1 EU (+12 months) § Average regulatory approval timelines in EU: 15 months • Abasria (insulin biosimilar): 14 months • Bemfola (follitropin alfa): 17 months • Inflectra/Remsima (infliximab): 13 months
- Slides: 1