Approaches to COPD Assessment and Treatment US16960 1217



























- Slides: 46

Approaches to COPD Assessment and Treatment US-16960 12/17

Considerations in the Management of COPD Including GOLD Report Updates COPD=chronic obstructive pulmonary disease. US-16958 1/18

Welcome to This Promotional Program Sponsored by Astra. Zeneca The purpose of this presentation is for disease state education and is not meant to imply efficacy or safety of any Astra. Zeneca or Competitor products. 3

Objectives • Review key updates in the GOLD 2017 Report with additions from GOLD 2018 • Understand the role of spirometry and recognize the importance of full symptom evaluation in patients with COPD • Acknowledge the need for individualized management approaches including those outlined in the GOLD Report • Appreciate the importance of appropriate device selection for individual patients GOLD=Global Initiative for Chronic Obstructive Lung Disease. 4

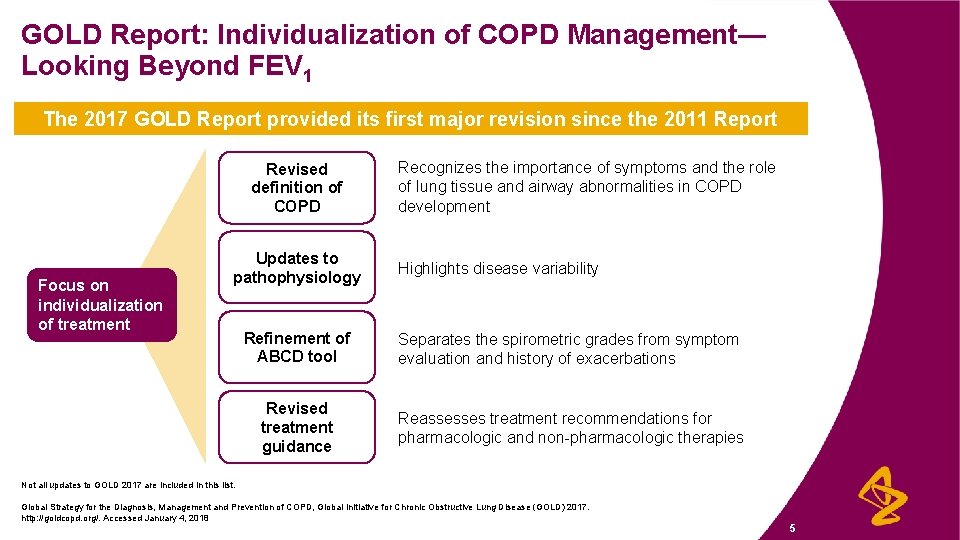
GOLD Report: Individualization of COPD Management— Looking Beyond FEV 1 The 2017 GOLD Report provided its first major revision since the 2011 Report Revised definition of COPD Focus on individualization of treatment Updates to pathophysiology Recognizes the importance of symptoms and the role of lung tissue and airway abnormalities in COPD development Highlights disease variability Refinement of ABCD tool Separates the spirometric grades from symptom evaluation and history of exacerbations Revised treatment guidance Reassesses treatment recommendations for pharmacologic and non-pharmacologic therapies Not all updates to GOLD 2017 are included in this list. Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2017. http: //goldcopd. org/. Accessed January 4, 2018 5

GOLD Report: Updated Definition of COPD 2011 COPD is a common preventable and treatable disease, characterized by persistent airflow limitation that is usually progressive and associated with an enhanced chronic inflammatory response in the airways and the lung to noxious particles or gases 1 2017 and 2018 COPD is a common preventable and treatable disease, characterized by persistent respiratory symptoms and airflow limitation that is due to airway and/or alveolar abnormalities usually caused by significant exposure to noxious particles and gases 2, 3 2017 definition included the impact of respiratory symptoms and the role of lung tissue and airway abnormalities in the development of COPD. The same definition appears in the 2018 update. 2, 3 1. Yusen R. Am J Resp Crit Care Med. 2013; 188: 4 -5. 2. Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2017. http: //goldcopd. org/. Accessed January 4, 2017. 3. Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018. http: //goldcopd. org/. Accessed January 4, 2018. 6

Role of Spirometry in Diagnosis and Monitoring Spirometry Airflow Limitation Post-bronchodilator FEV 1/FVC <0. 70* GOLD Recommends Active Case Finding: ie, performing spirometry in patients with symptoms and/or risk factors, but not screening spirometry • Spirometry confirms the diagnosis of COPD. It is used to assess airflow limitation • Observed disconnect between FEV 1 and symptoms Classification of Severity of Airflow Limitation in COPD (Based on Post-Bronchodilator FEV 1) In patients with FEV 1/FVC <0. 70 GOLD 1: Mild FEV 1 ≥ 80% predicted GOLD 2: Moderate 50% ≤FEV 1 <80% predicted GOLD 3: Severe 30% ≤FEV 1 <50% predicted GOLD 4: Very Severe FEV 1 <30% predicted *Required for the diagnosis of COPD. For FEV 1 between 0. 6 and 0. 8, repeat spirometry should be conducted on a separate occasion to confirm diagnosis. FEV 1=forced expiratory volume in 1 second; FVC=forced vital capacity. Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018. http: //goldcopd. org/. Accessed January 4, 2018. 7

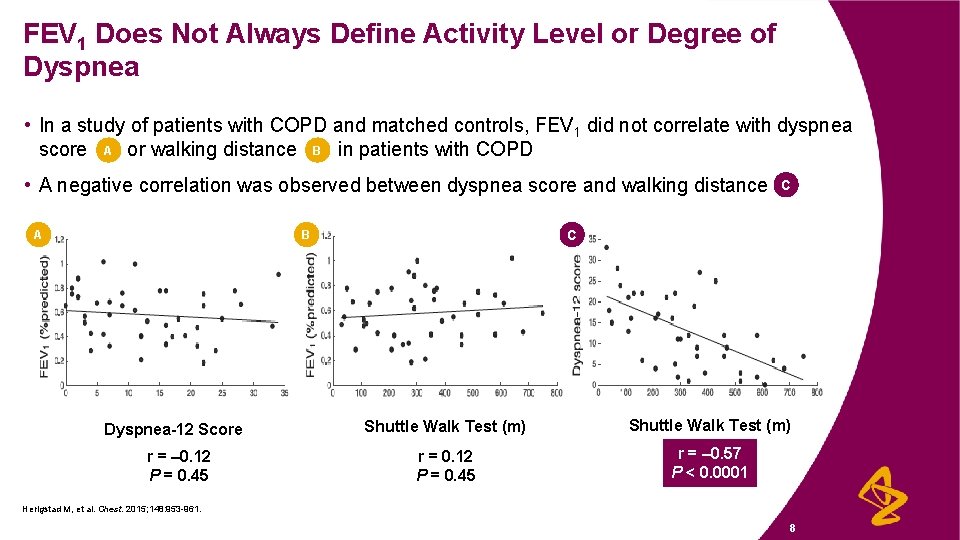
FEV 1 Does Not Always Define Activity Level or Degree of Dyspnea • In a study of patients with COPD and matched controls, FEV 1 did not correlate with dyspnea A or walking distance B in patients with COPD score • A negative correlation was observed between dyspnea score and walking distance A C B Dyspnea-12 Score r = – 0. 12 P = 0. 45 C Shuttle Walk Test (m) r = 0. 12 P = 0. 45 r = – 0. 57 P < 0. 0001 Herigstad M, et al. Chest. 2015; 148: 953 -961. 8

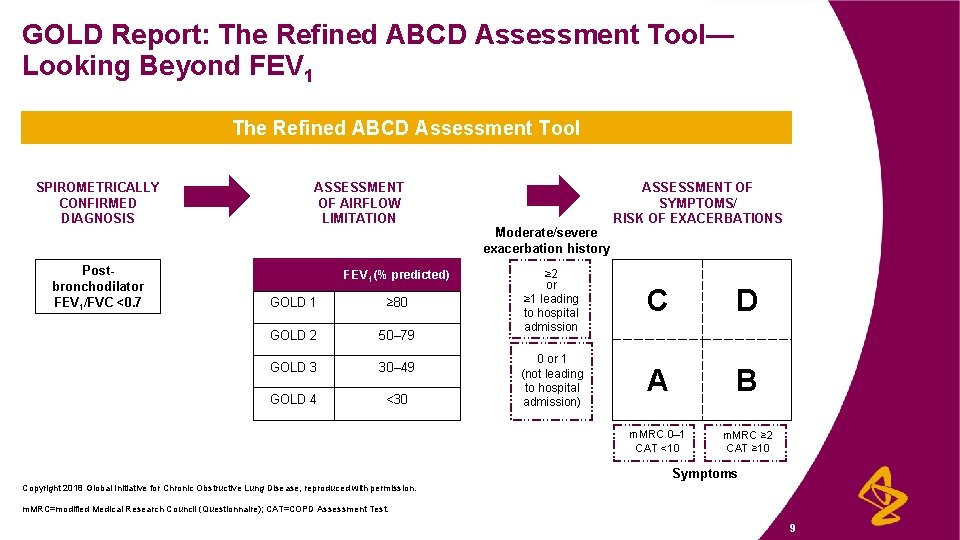
GOLD Report: The Refined ABCD Assessment Tool— Looking Beyond FEV 1 The Refined ABCD Assessment Tool SPIROMETRICALLY CONFIRMED DIAGNOSIS Postbronchodilator FEV 1/FVC <0. 7 ASSESSMENT OF AIRFLOW LIMITATION FEV 1 (% predicted) GOLD 1 ≥ 80 GOLD 2 50– 79 GOLD 3 30– 49 GOLD 4 <30 Moderate/severe exacerbation history ASSESSMENT OF SYMPTOMS/ RISK OF EXACERBATIONS ≥ 2 or ≥ 1 leading to hospital admission C D 0 or 1 (not leading to hospital admission) A B m. MRC 0– 1 CAT <10 m. MRC ≥ 2 CAT ≥ 10 Symptoms Copyright 2018 Global Initiative for Chronic Obstructive Lung Disease, reproduced with permission. m. MRC=modified Medical Research Council (Questionnaire); CAT=COPD Assessment Test. 9

Full Symptom Evaluation in COPD Is Critical Symptom Evaluation Is Critical for Diagnosis and Management • There is only a weak correlation between FEV 1, symptoms, and impairment in a patient’s health status • Full symptom assessment is critical Common Symptoms of COPD • • • Dyspnea Cough Sputum production Wheezing Chest tightness Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018. http: //goldcopd. org/. Accessed January 4, 2018. 10

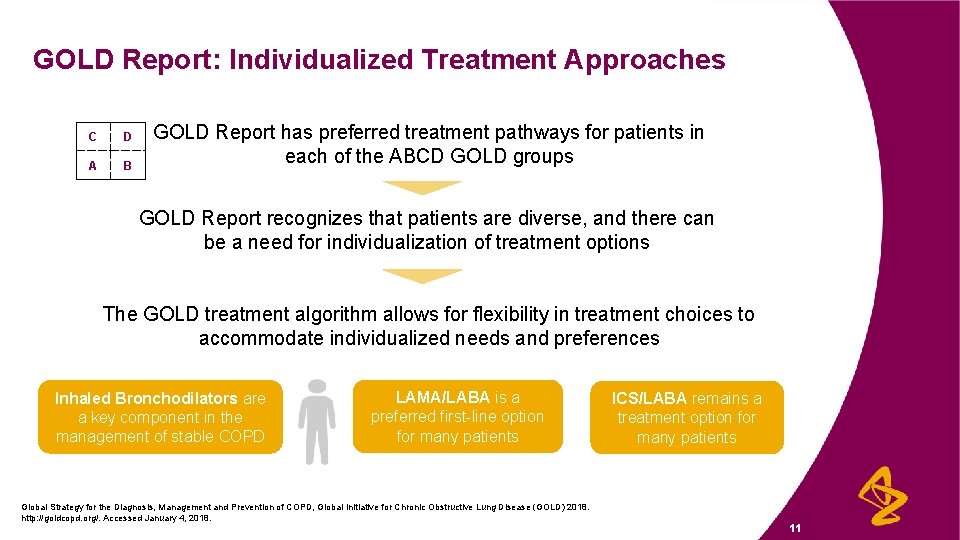
GOLD Report: Individualized Treatment Approaches C D A B GOLD Report has preferred treatment pathways for patients in each of the ABCD GOLD groups GOLD Report recognizes that patients are diverse, and there can be a need for individualization of treatment options The GOLD treatment algorithm allows for flexibility in treatment choices to accommodate individualized needs and preferences Inhaled Bronchodilators are a key component in the management of stable COPD LAMA/LABA is a preferred first-line option for many patients Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018. http: //goldcopd. org/. Accessed January 4, 2018. ICS/LABA remains a treatment option for many patients 11

The GOLD Report Highlights the Importance of Device Selection for Individualization of the Management of COPD • More than two-thirds of patients make at least 1 device error • There may be a relationship between poor inhaler use and symptom control Right patient Right device Individualization • Randomized, controlled trials have not identified superiority of 1 device/formulation • Physicians should gain a working knowledge of inhalation devices • The choice of inhaler device should be matched to the patient • Patient training is essential • Inhaler technique should be assessed regularly to help ensure correct device use Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018. http: //goldcopd. org/. Accessed January 4, 2018. 12

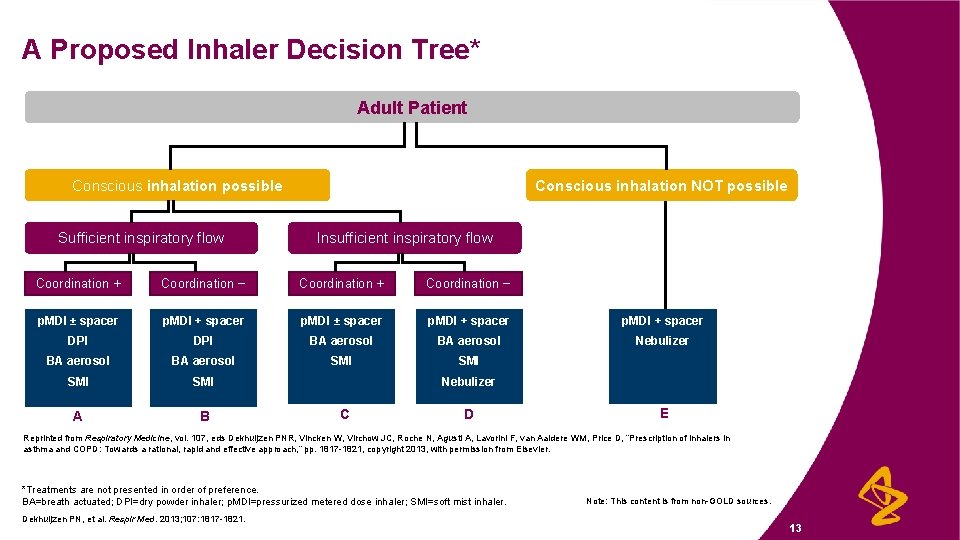
A Proposed Inhaler Decision Tree* Adult Patient Conscious inhalation possible Sufficient inspiratory flow Conscious inhalation NOT possible Insufficient inspiratory flow Coordination + Coordination − p. MDI ± spacer p. MDI + spacer DPI BA aerosol Nebulizer BA aerosol SMI SMI A B Nebulizer C D E Reprinted from Respiratory Medicine, vol. 107, eds Dekhuijzen PNR, Vincken W, Virchow JC, Roche N, Agusti A, Lavorini F, van Aaldere WM, Price D, “Prescription of inhalers in asthma and COPD: Towards a rational, rapid and effective approach, ” pp. 1817 -1821, copyright 2013, with permission from Elsevier. *Treatments are not presented in order of preference. BA=breath actuated; DPI=dry powder inhaler; p. MDI=pressurized metered dose inhaler; SMI=soft mist inhaler. Dekhuijzen PN, et al. Respir Med. 2013; 107: 1817 -1821. Note: This content is from non-GOLD sources. 13

Summary • The GOLD Report acknowledges the need for individualization of assessment and management approaches in COPD • Symptom severity and exacerbation history are recommended to guide treatment approach • Full evaluation of symptoms is important in patients with COPD • Device choice can be a critical part of management in patients with COPD 14

Comments to Astra. Zeneca is committed to conducting business with the highest standards of integrity and professionalism. If you have comments that could improve the delivery of our promotional educational programs, please contact Astra. Zeneca at 1 -800 -236 -9933. © 2018 Astra. Zeneca. All rights reserved. US-16958 1/18 15

Managing Patients With COPD: Individualizing Treatment COPD=chronic obstructive pulmonary disease. US-16961 1/18

Welcome to this promotional program sponsored by Astra. Zeneca Please note that this entire presentation is a promotional presentation sponsored directly by Astra. Zeneca. There are no continuing medical education (CME) credits associated with the activity.

Options for the Treatment of Patients With COPD Predominant Presentation: Bronchoconstriction and inflammation Hypothetical patients 18

BEVESPI AEROSPHERE® (glycopyrrolate 9 mcg/ formoterol fumarate 4. 8 mcg) Inhalation Aerosol Please see the full Prescribing Information, including Boxed WARNING and Medication Guide , available at this presentation. 19

BEVESPI AEROSPHERE for the Maintenance Treatment of COPD Indication • BEVESPI AEROSPHERE is indicated for the long-term maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema • BEVESPI AEROSPHERE is not indicated for the relief of acute bronchospasm, or for the treatment of asthma Boxed WARNING: • Long‐acting beta 2‐adrenergic agonists (LABAs), such as formoterol fumarate, one of the active ingredients in BEVESPI AEROSPHERE, increase the risk of asthma‐related death. A placebo-controlled trial with another LABA (salmeterol) showed an increase in asthma‐related deaths in subjects receiving salmeterol. This finding with salmeterol is considered a class effect of all LABAs, including formoterol fumarate • The safety and efficacy of BEVESPI AEROSPHERE in patients with asthma have not been established. BEVESPI AEROSPHERE is not indicated for the treatment of asthma Please see the full Prescribing Information, including Boxed WARNING and Medication Guide, for BEVESPI AEROSPHERE, available at this presentation. 20

BEVESPI AEROSPHERE May Be Considered For a Patient With COPD With the Following Characteristics Predominant presentation is bronchoconstriction • Short of breath; may be due to hyperinflation • Increasing rescue use • No ICS required • Historically may have been treated with a LAMA Hypothetical patient ICS=inhaled corticosteroids; LAMA=inhaled long-acting muscarinic antagonist. 21

BEVESPI AEROSPHERE Improved lung function including predose FEV 1 and peak FEV 1 at 24 weeks 1, 2 MAXIMIZE BRONCHODILATION* In a separate study vs placebo, improvement in peak inspiratory capacity at Day 293† INTELLIGENT FORMULATION 1‡ Intelligent formulation for a p. MDI using patented AEROSPHERE™ Delivery Technology 1 In a separate functional respiratory imaging study, an increase in specific-airway volume vs placebo was demonstrated 4§ Adverse reactions with BEVESPI AEROSPHERE with a ≥ 2% incidence and more common than placebo were urinary tract infections and cough 1 BEVESPI AEROSPHERE is NOT a rescue medication and does NOT replace fast-acting inhalers to treat acute symptoms 1 *Improvements in lung function relative to its individual components and placebo in two 24 -week pivotal trials (n=3699). †Peak inspiratory capacity after the evening dose on Day 29. ‡BEVESPI AEROSPHERE is a p. MDI containing the LAMA glycopyrrolate and LABA formoterol fumarate along with phospholipid porous particles that form the co-suspension with the micronized drug crystals. §Geometric least squares mean specific image-based airway volume at Day 15 (n=19). FEV 1=forced expiratory volume in 1 second; p. MDI=pressurized metered dose inhaler. 1. BEVESPI AEROSPHERE [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; 2017. 2. Martinez FJ, et al. Chest. 2017; 151: 340 -357. 3. Reisner C, et al. Resp Res. 2017; 18: 157. 4. Data on File, REF-14480. Astra. Zeneca Pharmaceuticals LP. 22

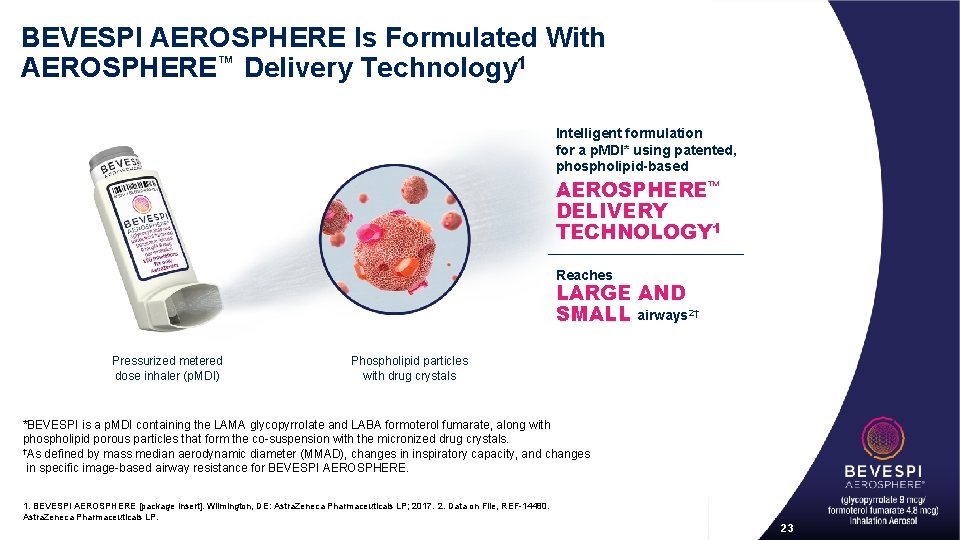
BEVESPI AEROSPHERE Is Formulated With AEROSPHERE™ Delivery Technology 1 Intelligent formulation for a p. MDI* using patented, phospholipid-based AEROSPHERE™ DELIVERY TECHNOLOGY 1 Reaches LARGE AND SMALL airways 2† Pressurized metered dose inhaler (p. MDI) Phospholipid particles with drug crystals *BEVESPI is a p. MDI containing the LAMA glycopyrrolate and LABA formoterol fumarate, along with phospholipid porous particles that form the co-suspension with the micronized drug crystals. †As defined by mass median aerodynamic diameter (MMAD), changes in inspiratory capacity, and changes in specific image-based airway resistance for BEVESPI AEROSPHERE. 1. BEVESPI AEROSPHERE [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; 2017. 2. Data on File, REF-14480. Astra. Zeneca Pharmaceuticals LP. 23

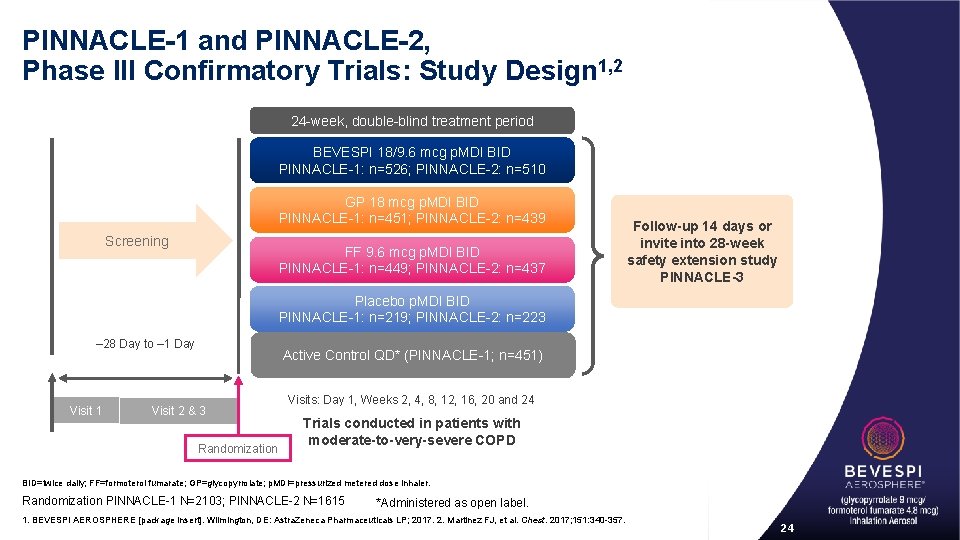
PINNACLE-1 and PINNACLE-2, Phase III Confirmatory Trials: Study Design 1, 2 24 -week, double-blind treatment period BEVESPI 18/9. 6 mcg p. MDI BID PINNACLE-1: n=526; PINNACLE-2: n=510 GP 18 mcg p. MDI BID PINNACLE-1: n=451; PINNACLE-2: n=439 Screening FF 9. 6 mcg p. MDI BID PINNACLE-1: n=449; PINNACLE-2: n=437 Follow-up 14 days or invite into 28 -week safety extension study PINNACLE-3 Placebo p. MDI BID PINNACLE-1: n=219; PINNACLE-2: n=223 – 28 Day to – 1 Day Visit 1 Active Control QD* (PINNACLE-1; n=451) Visit 2 & 3 Randomization Visits: Day 1, Weeks 2, 4, 8, 12, 16, 20 and 24 Trials conducted in patients with moderate-to-very-severe COPD BID=twice daily; FF=formoterol fumarate; GP=glycopyrrolate; p. MDI=pressurized metered dose inhaler. Randomization PINNACLE-1 N=2103; PINNACLE-2 N=1615 *Administered as open label. 1. BEVESPI AEROSPHERE [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; 2017. 2. Martinez FJ, et al. Chest. 2017; 151: 340 -357. 24

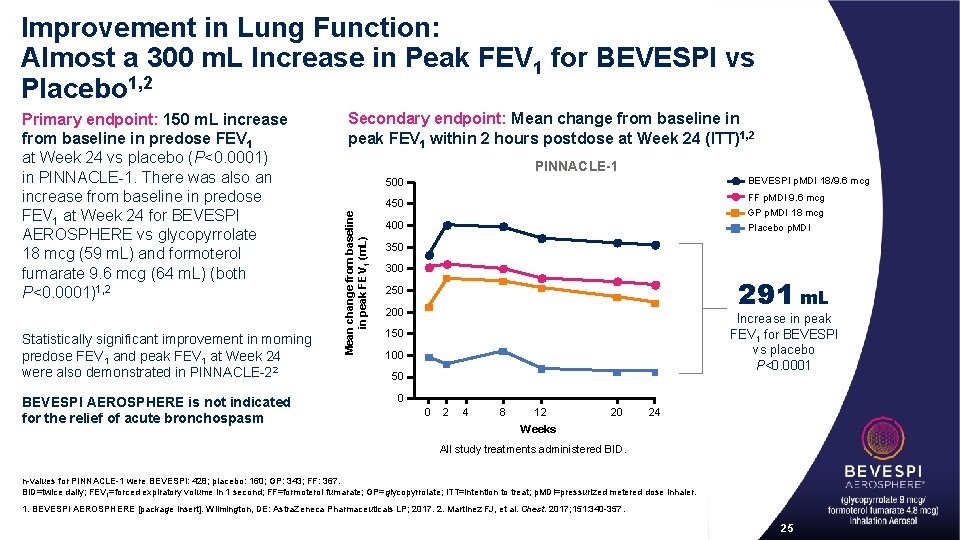
Improvement in Lung Function: Almost a 300 m. L Increase in Peak FEV 1 for BEVESPI vs Placebo 1, 2 Statistically significant improvement in morning predose FEV 1 and peak FEV 1 at Week 24 were also demonstrated in PINNACLE-22 BEVESPI AEROSPHERE is not indicated for the relief of acute bronchospasm Secondary endpoint: Mean change from baseline in peak FEV 1 within 2 hours postdose at Week 24 (ITT)1, 2 PINNACLE-1 BEVESPI p. MDI 18/9. 6 mcg 500 FF p. MDI 9. 6 mcg 450 Mean change from baseline in peak FEV 1 (m. L) Primary endpoint: 150 m. L increase from baseline in predose FEV 1 at Week 24 vs placebo (P<0. 0001) in PINNACLE-1. There was also an increase from baseline in predose FEV 1 at Week 24 for BEVESPI AEROSPHERE vs glycopyrrolate 18 mcg (59 m. L) and formoterol fumarate 9. 6 mcg (64 m. L) (both P<0. 0001)1, 2 GP p. MDI 18 mcg Placebo p. MDI 400 350 300 291 m. L 250 200 Increase in peak FEV 1 for BEVESPI vs placebo P<0. 0001 150 100 50 0 0 2 4 8 12 20 24 Weeks All study treatments administered BID. n-values for PINNACLE-1 were BEVESPI: 428; placebo: 160; GP: 343; FF: 367. BID=twice daily; FEV 1=forced expiratory volume in 1 second; FF=formoterol fumarate; GP=glycopyrrolate; ITT=intention to treat; p. MDI=pressurized metered dose inhaler. 1. BEVESPI AEROSPHERE [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; 2017. 2. Martinez FJ, et al. Chest. 2017; 151: 340 -357. 25

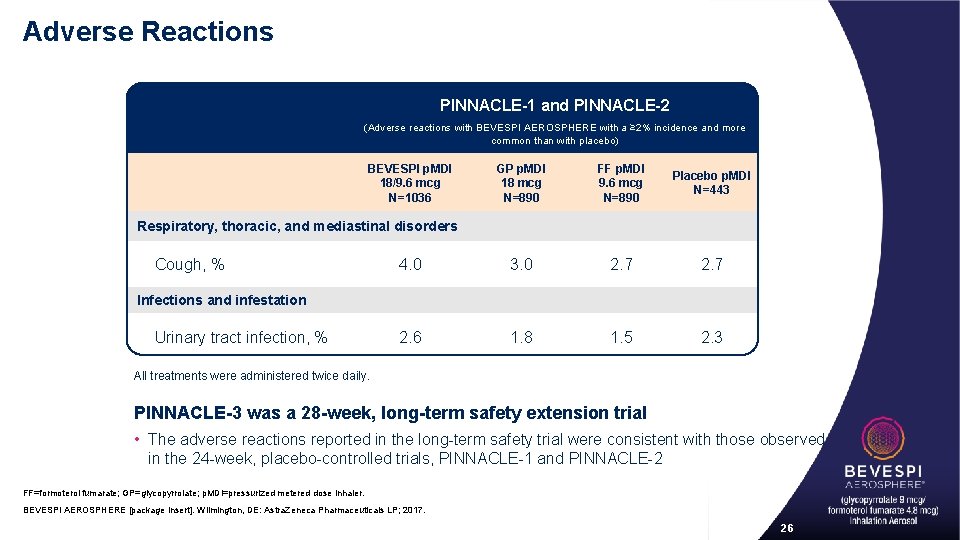
Adverse Reactions PINNACLE-1 and PINNACLE-2 (Adverse reactions with BEVESPI AEROSPHERE with a ≥ 2% incidence and more common than with placebo) BEVESPI p. MDI 18/9. 6 mcg N=1036 GP p. MDI 18 mcg N=890 FF p. MDI 9. 6 mcg N=890 Placebo p. MDI N=443 4. 0 3. 0 2. 7 2. 6 1. 8 1. 5 2. 3 Respiratory, thoracic, and mediastinal disorders Cough, % Infections and infestation Urinary tract infection, % All treatments were administered twice daily. PINNACLE-3 was a 28 -week, long-term safety extension trial • The adverse reactions reported in the long-term safety trial were consistent with those observed in the 24 -week, placebo-controlled trials, PINNACLE-1 and PINNACLE-2 FF=formoterol fumarate; GP=glycopyrrolate; p. MDI=pressurized metered dose inhaler. BEVESPI AEROSPHERE [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; 2017. 26

Inspiratory Capacity (IC) and FEV 1 Can Serve Different Purposes in the Assessment of COPD FEV 1 IC • FEV 1 measures the volume of air that can be forcibly breathed out in the first second 1 • IC is the maximum amount of air that can be taken in after a normal breath 3 • FEV 1 does not always correlate with activity level or degree of dyspnea 2 • In COPD, residual air trapped in the lungs after expiration reduces patients’ IC 3, 4 1. Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018. http: //goldcopd. org/. Accessed January 4, 2018. 2. Herigstad M et al. Chest. 2015; 148(4): 953 -961. 3. Seeley R. Respiratory System. In: Seeley R, Stephens T, Tate P, eds. Anatomy & Physiology. 6 th ed. New York, NY: Mc. Graw-Hill; 2003: 813 -859. 4. O'Donnell DE et al. COPD Res Pract. 2015: 1: 4. 27

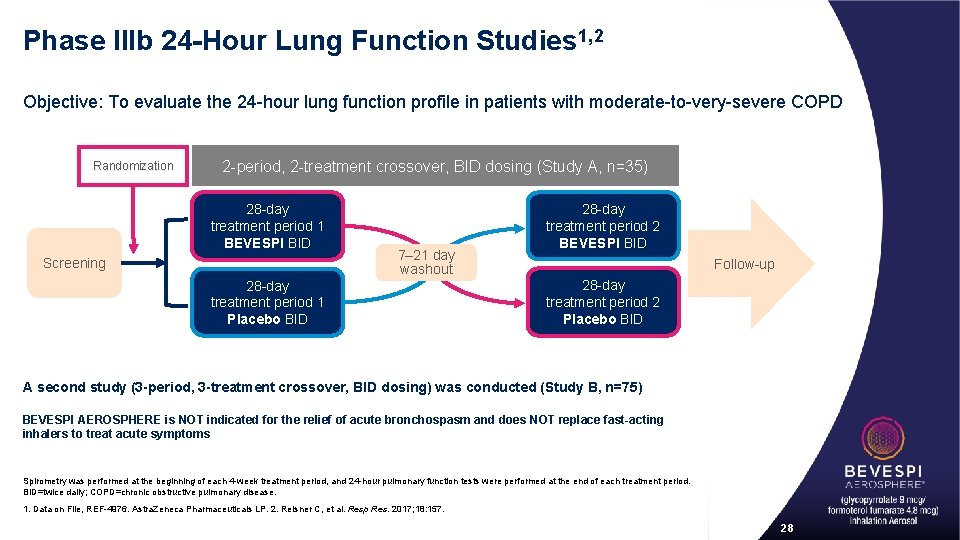
Phase IIIb 24 -Hour Lung Function Studies 1, 2 Objective: To evaluate the 24 -hour lung function profile in patients with moderate-to-very-severe COPD Randomization 2 -period, 2 -treatment crossover, BID dosing (Study A, n=35) 28 -day treatment period 1 BEVESPI BID Screening 7– 21 day washout 28 -day treatment period 1 Placebo BID 28 -day treatment period 2 BEVESPI BID Follow-up 28 -day treatment period 2 Placebo BID A second study (3 -period, 3 -treatment crossover, BID dosing) was conducted (Study B, n=75) BEVESPI AEROSPHERE is NOT indicated for the relief of acute bronchospasm and does NOT replace fast-acting inhalers to treat acute symptoms Spirometry was performed at the beginning of each 4 -week treatment period, and 24 -hour pulmonary function tests were performed at the end of each treatment period. BID=twice daily; COPD=chronic obstructive pulmonary disease. 1. Data on File, REF-4976. Astra. Zeneca Pharmaceuticals LP. 2. Reisner C, et al. Resp Res. 2017; 18: 157. 28

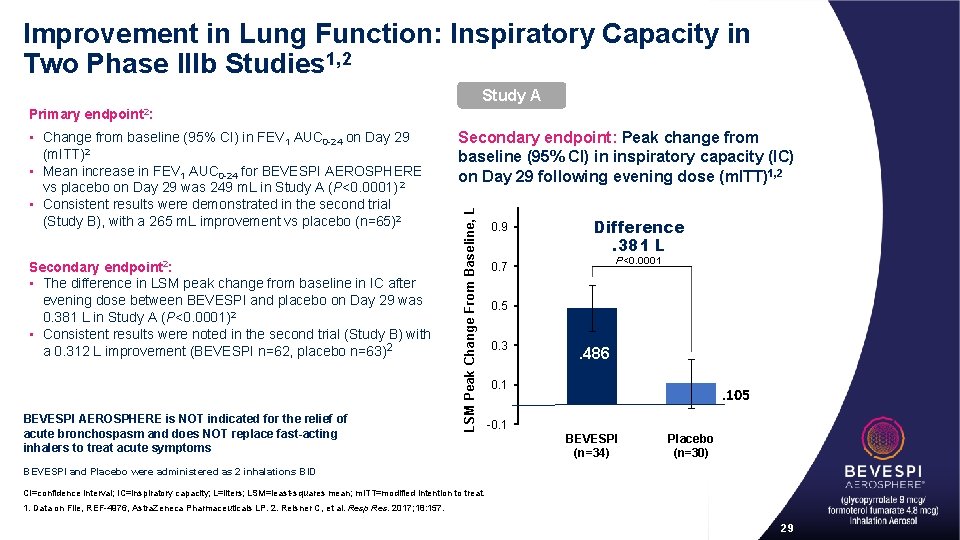
Improvement in Lung Function: Inspiratory Capacity in Two Phase IIIb Studies 1, 2 Study A • Change from baseline (95% CI) in FEV 1 AUC 0 -24 on Day 29 (m. ITT)2 • Mean increase in FEV 1 AUC 0 -24 for BEVESPI AEROSPHERE vs placebo on Day 29 was 249 m. L in Study A (P<0. 0001) 2 • Consistent results were demonstrated in the second trial (Study B), with a 265 m. L improvement vs placebo (n=65)2 Secondary endpoint 2: • The difference in LSM peak change from baseline in IC after evening dose between BEVESPI and placebo on Day 29 was 0. 381 L in Study A (P<0. 0001)2 • Consistent results were noted in the second trial (Study B) with a 0. 312 L improvement (BEVESPI n=62, placebo n=63)2 BEVESPI AEROSPHERE is NOT indicated for the relief of acute bronchospasm and does NOT replace fast-acting inhalers to treat acute symptoms Secondary endpoint: Peak change from baseline (95% CI) in inspiratory capacity (IC) on Day 29 following evening dose (m. ITT)1, 2 LSM Peak Change From Baseline, L Primary endpoint 2: 0. 9 Difference. 381 L P<0. 0001 0. 7 0. 5 0. 3 . 486 0. 1 . 105 -0. 1 BEVESPI (n=34) Placebo (n=30) BEVESPI and Placebo were administered as 2 inhalations BID CI=confidence interval; IC=inspiratory capacity; L=liters; LSM=least-squares mean; m. ITT=modified intention to treat. 1. Data on File, REF-4976, Astra. Zeneca Pharmaceuticals LP. 2. Reisner C, et al. Resp Res. 2017; 18: 157. 29

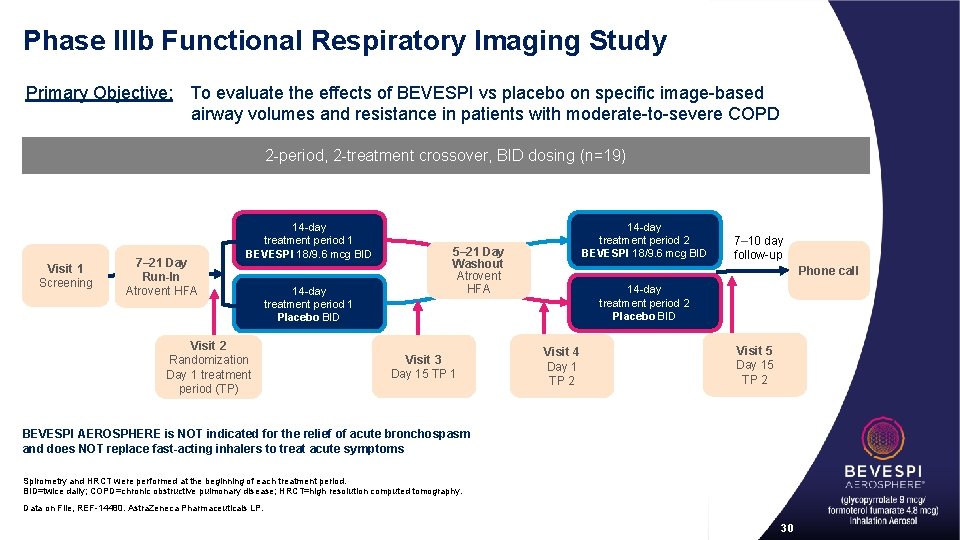
Phase IIIb Functional Respiratory Imaging Study Primary Objective: To evaluate the effects of BEVESPI vs placebo on specific image-based airway volumes and resistance in patients with moderate-to-severe COPD 2 -period, 2 -treatment crossover, BID dosing (n=19) Visit 1 Screening 7– 21 Day Run-In Atrovent HFA 14 -day treatment period 1 BEVESPI 18/9. 6 mcg BID Visit 2 Randomization Day 1 treatment period (TP) 14 -day treatment period 1 Placebo BID 14 -day treatment period 2 BEVESPI 18/9. 6 mcg BID 5– 21 Day Washout Atrovent HFA Visit 3 Day 15 TP 1 7– 10 day follow-up Phone call 14 -day treatment period 2 Placebo BID Visit 4 Day 1 TP 2 Visit 5 Day 15 TP 2 BEVESPI AEROSPHERE is NOT indicated for the relief of acute bronchospasm and does NOT replace fast-acting inhalers to treat acute symptoms Spirometry and HRCT were performed at the beginning of each treatment period. BID=twice daily; COPD=chronic obstructive pulmonary disease; HRCT=high resolution computed tomography. Data on File, REF-14480. Astra. Zeneca Pharmaceuticals LP. 30

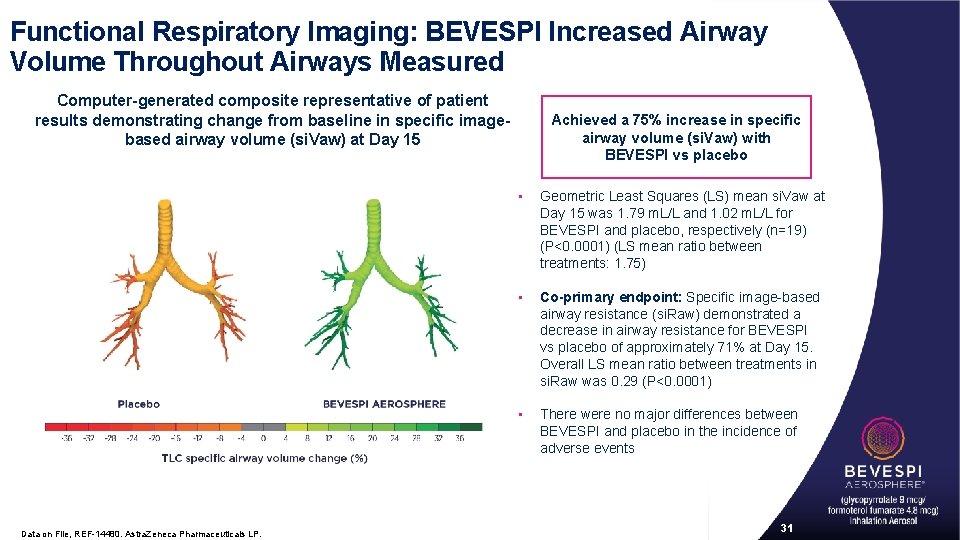
Functional Respiratory Imaging: BEVESPI Increased Airway Volume Throughout Airways Measured Computer-generated composite representative of patient results demonstrating change from baseline in specific imagebased airway volume (si. Vaw) at Day 15 Data on File, REF-14480. Astra. Zeneca Pharmaceuticals LP. Achieved a 75% increase in specific airway volume (si. Vaw) with BEVESPI vs placebo • Geometric Least Squares (LS) mean si. Vaw at Day 15 was 1. 79 m. L/L and 1. 02 m. L/L for BEVESPI and placebo, respectively (n=19) (P<0. 0001) (LS mean ratio between treatments: 1. 75) • Co-primary endpoint: Specific image-based airway resistance (si. Raw) demonstrated a decrease in airway resistance for BEVESPI vs placebo of approximately 71% at Day 15. Overall LS mean ratio between treatments in si. Raw was 0. 29 (P<0. 0001) • There were no major differences between BEVESPI and placebo in the incidence of adverse events 31

Important Safety Information, Including Boxed WARNING* WARNING: Long‐acting beta 2‐adrenergic agonists (LABAs), such as formoterol fumarate, one of the active ingredients in BEVESPI AEROSPHERE, increase the risk of asthma‐related death. A placebo-controlled trial with another LABA (salmeterol) showed an increase in asthma‐related deaths in subjects receiving salmeterol. This finding with salmeterol is considered a class effect of all LABAs, including formoterol fumarate. The safety and efficacy of BEVESPI AEROSPHERE in patients with asthma have not been established. BEVESPI AEROSPHERE is not indicated for the treatment of asthma. CONTRAINDICATIONS: All LABAs are contraindicated in patients with asthma without use of a long‐term asthma control medication. BEVESPI is contraindicated in patients with hypersensitivity to glycopyrrolate, formoterol fumarate, or to any component of the product. WARNINGS AND PRECAUTIONS • BEVESPI should not be initiated in patients with acutely deteriorating chronic obstructive pulmonary disease (COPD), which may be a lifethreatening condition • BEVESPI should not be used for the relief of acute symptoms (ie, as rescue therapy for the treatment of acute episodes of bronchospasm). Acute symptoms should be treated with an inhaled short-acting beta 2‐agonist • BEVESPI should not be used more often or at higher doses than recommended, or with other LABAs, as an overdose may result • If paradoxical bronchospasm occurs, discontinue BEVESPI immediately and institute alternative therapy • If immediate hypersensitivity reactions occur, in particular, angioedema, urticaria, or skin rash, discontinue BEVESPI at once and consider alternative treatment • BEVESPI can produce a clinically significant cardiovascular effect in some patients, as measured by increases in pulse rate, blood pressure, or symptoms. If such effects occur, BEVESPI may need to be discontinued • Use with caution in patients with convulsive disorders, thyrotoxicosis, diabetes mellitus, ketoacidosis, and in patients who are unusually responsive to sympathomimetic amines • Be alert to hypokalemia and hyperglycemia • Worsening of narrow-angle glaucoma or urinary retention may occur. Use with caution in patients with narrow-angle glaucoma, prostatic hyperplasia, or bladder-neck obstruction, and instruct patients to contact a physician immediately if symptoms occur *Please see the full Prescribing Information for BEVESPI AEROSPHERE, including Boxed WARNING and Medication Guide, available at this presentation. BEVESPI AEROSPHERE [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; 2017. 32

Important Safety Information, Including Boxed WARNING* (Continued) ADVERSE REACTIONS: The most common adverse reactions with INDICATION: BEVESPI AEROSPHERE is a combination of BEVESPI (≥ 2% and more common than placebo) were: cough, 4. 0% (2. 7%), and urinary tract infection, 2. 6% (2. 3%). glycopyrrolate, an anticholinergic, and formoterol fumarate, a long-acting beta 2 -adrenergic agonist (LABA), indicated for the long-term, maintenance treatment of airflow obstruction in patients with COPD, including chronic bronchitis and/or emphysema. DRUG INTERACTIONS • Use caution if administering additional adrenergic drugs because the sympathetic effects of formoterol may be potentiated • Concomitant treatment with xanthine derivatives, steroids, or diuretics may potentiate any hypokalemic effect of formoterol • Use with caution in patients taking non–potassium‐sparing diuretics, as the ECG changes and/or hypokalemia may worsen with concomitant beta 2‐agonists • The action of adrenergic agonists on the cardiovascular system may be potentiated by monoamine oxidase inhibitors, tricyclic antidepressants, or other drugs known to prolong the QTc interval. Therefore, BEVESPI should be used with extreme caution in patients being treated with these agents • Use beta‐blockers with caution as they not only block therapeutic effects of beta‐agonists, but may produce severe bronchospasm in patients with COPD • Avoid co‐administration of BEVESPI with other anticholinergic‐ containing drugs as this may lead to an increase in anticholinergic adverse effects LIMITATION OF USE: Not indicated for the relief of acute bronchospasm or for the treatment of asthma. *Please see the full Prescribing Information for BEVESPI AEROSPHERE, including Boxed WARNING and Medication Guide, available at this presentation. BEVESPI AEROSPHERE [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; 2017. 33

SYMBICORT® (budesonide/formoterol fumarate dihydrate) 34

Indication, Dosage, and Important Safety Information for Patients With COPD 160/4. 5 µg, 2 inhalations twice daily* § SYMBICORT 160/4. 5 is indicated for the maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD) including chronic bronchitis and/or emphysema. SYMBICORT 160/4. 5 is also indicated to reduce exacerbations of COPD. SYMBICORT 160/4. 5 is the only strength indicated for the treatment of COPD § SYMBICORT is NOT indicated for relief of acute bronchospasm § Use of long-acting beta 2 -adrenergic agonists (LABA) as monotherapy (without inhaled corticosteroids [ICS]) for asthma is associated with an increased risk of asthma-related death. These findings are considered a class effect of LABA. When LABA are used in fixed dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared to ICS alone *Administered in the morning and in the evening. Please see full Prescribing Information, including Patient Information, available at this presentation. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; December 2017. 35

SYMBICORT May Be Considered for a Patient With COPD With the Following Characteristics Predominant presentation is bronchoconstriction and inflammation A patient with COPD who § Needs an ICS § May have asthma-like features and a history of exacerbations 1 -3 Hypothetical patient 1. Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2018. http: //goldcopd. org. Accessed January 4, 2018. 2. Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention. www. ginasthma. org. Updated 2017. Accessed January 18, 2018. 3. Data on File, REF-5531, Astra. Zeneca Pharmaceuticals LP. 36

SYMBICORT 160/4. 5 Inhalation Aerosol Majority of patients’ FEV 1* improvement occurred at 5 minutes in SPEED COPD† 1 -3 CONTR Reduced COPD exacerbations vs formoterol alone 3, 4 OL ‡ 5 Reduced COPD symptom scores vs formoterol alone SYMBICORT is NOT a rescue medication and does NOT replace fast-acting inhalers to treat acute symptoms. The most common adverse reactions ≥ 3% reported in COPD lung function clinical trials included nasopharyngitis, oral candidiasis, bronchitis, sinusitis, and upper respiratory tract infection. The safety findings from the two exacerbation clinical trials were consistent with the lung function studies. *1 -hour postdose FEV 1. †Serial spirometry subset of SUN study patients taking SYMBICORT 160/4. 5. ‡In the SUN study, COPD symptom scores were assessed with The Breathlessness, Cough, and Sputum Scale (BCSS). BCSS was measured in diary cards every day before the evening dose. BCSS measurement was a secondary endpoint, with the primary comparison being SYMBICORT vs placebo. 5 1. Data on File, 1084400, Astra. Zeneca Pharmaceuticals LP. 2. Rennard SI, et al. Drugs. 2009; 69(5): 549 -565. 3. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; December 2017. 4. Data on File, REF-16658, Astra. Zeneca Pharmaceuticals LP. 5. Data on File, 1814606, Astra. Zeneca Pharmaceuticals LP. 37

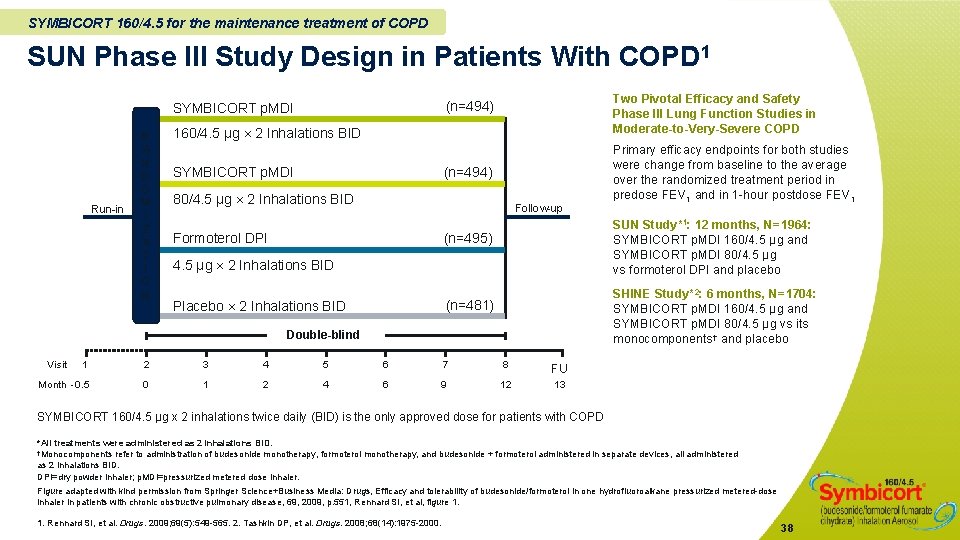
SYMBICORT 160/4. 5 for the maintenance treatment of COPD SUN Phase III Study Design in Patients With COPD 1 Run-in R A N D O M I Z A T I O N Two Pivotal Efficacy and Safety Phase III Lung Function Studies in Moderate-to-Very-Severe COPD (n=494) SYMBICORT p. MDI 160/4. 5 µg 2 Inhalations BID SYMBICORT p. MDI (n=494) 80/4. 5 µg 2 Inhalations BID Follow-up Formoterol DPI SUN Study*1: 12 months, N=1964: SYMBICORT p. MDI 160/4. 5 µg and SYMBICORT p. MDI 80/4. 5 µg vs formoterol DPI and placebo (n=495) 4. 5 µg 2 Inhalations BID SHINE Study*2: 6 months, N=1704: SYMBICORT p. MDI 160/4. 5 µg and SYMBICORT p. MDI 80/4. 5 µg vs its monocomponents † and placebo (n=481) Placebo 2 Inhalations BID Double-blind Visit Primary efficacy endpoints for both studies were change from baseline to the average over the randomized treatment period in predose FEV 1 and in 1 -hour postdose FEV 1 1 2 3 4 5 6 7 8 FU Month -0. 5 0 1 2 4 6 9 12 13 SYMBICORT 160/4. 5 µg x 2 inhalations twice daily (BID) is the only approved dose for patients with COPD *All treatments were administered as 2 inhalations BID. †Monocomponents refer to administration of budesonide monotherapy, formoterol monotherapy, and budesonide + formoterol administered in separate devices, all administered as 2 inhalations BID. DPI=dry powder inhaler; p. MDI=pressurized metered dose inhaler. Figure adapted with kind permission from Springer Science+Business Media: Drugs, Efficacy and tolerability of budesonide/formoterol in one hydrofluoroalkane pressurized metered-dose inhaler in patients with chronic obstructive pulmonary disease, 69, 2009, p. 551, Rennard SI, et al, figure 1. 1. Rennard SI, et al. Drugs. 2009; 69(5): 549 -565. 2. Tashkin DP, et al. Drugs. 2008; 68(14): 1975 -2000. 38

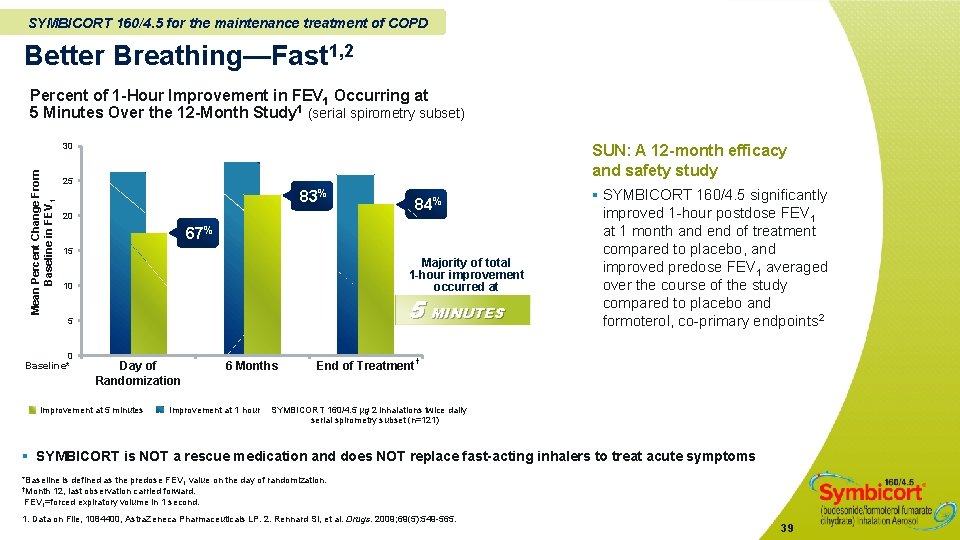
SYMBICORT 160/4. 5 for the maintenance treatment of COPD Better Breathing—Fast 1, 2 Percent of 1 -Hour Improvement in FEV 1 Occurring at 5 Minutes Over the 12 -Month Study 1 (serial spirometry subset) Mean Percent Change From Baseline in FEV 1 30 SUN: A 12 -month efficacy and safety study 25 83% 20 84% 67% 15 Majority of total 1 -hour improvement occurred at 10 5 MINUTES 5 0 Baseline* Day of Randomization 6 Months End of Treatment § SYMBICORT 160/4. 5 significantly improved 1 -hour postdose FEV 1 at 1 month and end of treatment compared to placebo, and improved predose FEV 1 averaged over the course of the study compared to placebo and formoterol, co-primary endpoints 2 † Improvement at 5 minutes Improvement at 1 hour SYMBICORT 160/4. 5 µg 2 inhalations twice daily serial spirometry subset (n=121) § SYMBICORT is NOT a rescue medication and does NOT replace fast-acting inhalers to treat acute symptoms *Baseline is defined as the predose FEV 1 value on the day of randomization. †Month 12, last observation carried forward. FEV 1=forced expiratory volume in 1 second. 1. Data on File, 1084400, Astra. Zeneca Pharmaceuticals LP. 2. Rennard SI, et al. Drugs. 2009; 69(5): 549 -565. 39

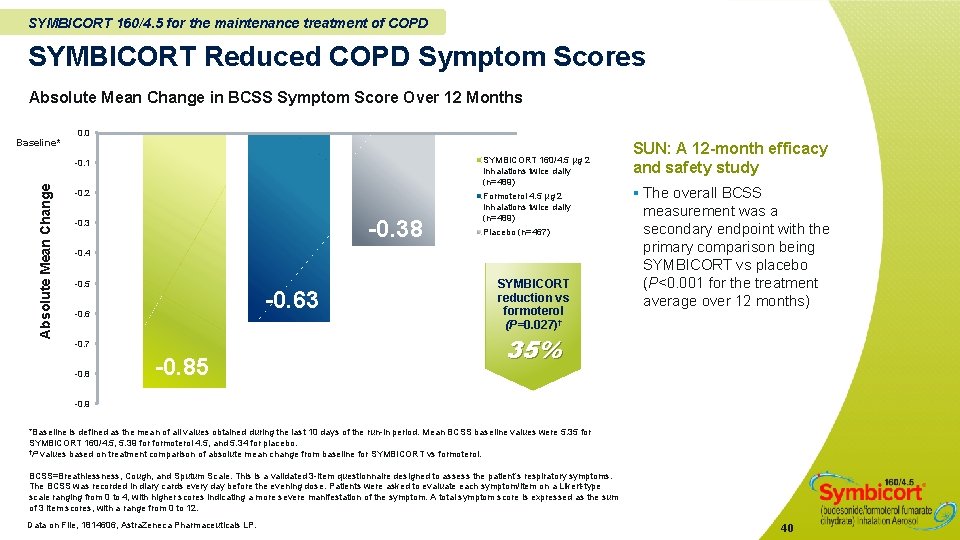
SYMBICORT 160/4. 5 for the maintenance treatment of COPD SYMBICORT Reduced COPD Symptom Scores Absolute Mean Change in BCSS Symptom Score Over 12 Months Absolute Mean Change Baseline* 0. 0 -0. 1 SYMBICORT 160/4. 5 µg 2 inhalations twice daily (n=489) -0. 2 Formoterol 4. 5 µg 2 inhalations twice daily (n=489) -0. 38 -0. 3 Placebo (n=467) -0. 4 -0. 5 -0. 63 -0. 6 -0. 7 -0. 85 SYMBICORT reduction vs formoterol (P=0. 027)† SUN: A 12 -month efficacy and safety study § The overall BCSS measurement was a secondary endpoint with the primary comparison being SYMBICORT vs placebo (P<0. 001 for the treatment average over 12 months) 35% -0. 9 *Baseline is defined as the mean of all values obtained during the last 10 days of the run-in period. Mean BCSS baseline values were 5. 35 for SYMBICORT 160/4. 5, 5. 39 formoterol 4. 5, and 5. 34 for placebo. †P values based on treatment comparison of absolute mean change from baseline for SYMBICORT vs formoterol. BCSS=Breathlessness, Cough, and Sputum Scale. This is a validated 3 -item questionnaire designed to assess the patient’s respiratory symptoms. The BCSS was recorded in diary cards every day before the evening dose. Patients were asked to evaluate each symptom/item on a Likert-type scale ranging from 0 to 4, with higher scores indicating a more severe manifestation of the symptom. A total symptom score is expressed as the sum of 3 item scores, with a range from 0 to 12. Data on File, 1814606, Astra. Zeneca Pharmaceuticals LP. 40

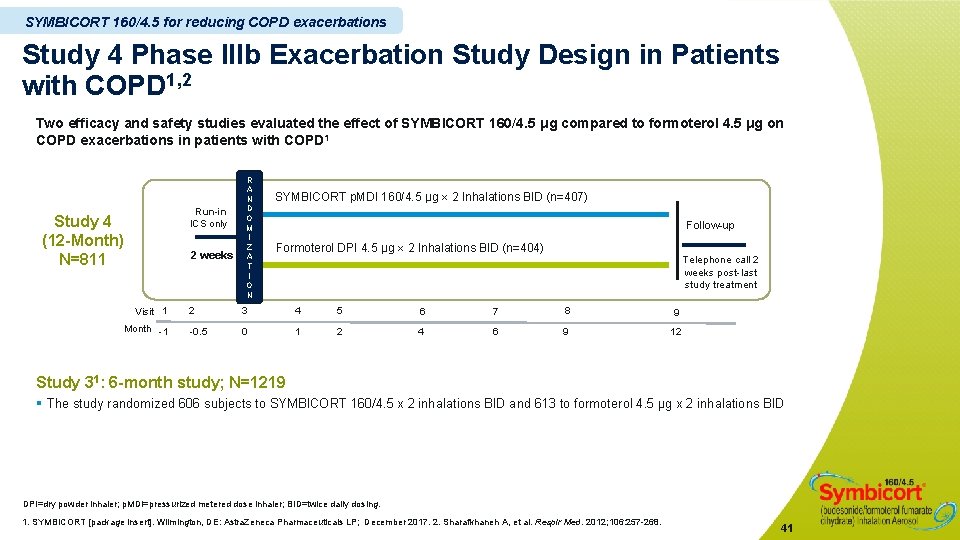
SYMBICORT 160/4. 5 for reducing COPD exacerbations Study 4 Phase IIIb Exacerbation Study Design in Patients with COPD 1, 2 Two efficacy and safety studies evaluated the effect of SYMBICORT 160/4. 5 μg compared to formoterol 4. 5 μg on COPD exacerbations in patients with COPD 1 Run-in ICS only Study 4 (12 -Month) N=811 2 weeks Visit 1 Month -1 R A N D O M I Z A T I O N SYMBICORT p. MDI 160/4. 5 µg 2 Inhalations BID (n=407) Follow-up Formoterol DPI 4. 5 µg 2 Inhalations BID (n=404) Telephone call 2 weeks post-last study treatment 2 3 4 5 6 7 8 9 -0. 5 0 1 2 4 6 9 12 Study 31: 6 -month study; N=1219 § The study randomized 606 subjects to SYMBICORT 160/4. 5 x 2 inhalations BID and 613 to formoterol 4. 5 µg x 2 inhalations BID DPI=dry powder inhaler; p. MDI=pressurized metered dose inhaler; BID=twice daily dosing. 1. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; December 2017. 2. Sharafkhaneh A, et al. Respir Med. 2012; 106: 257 -268. 41

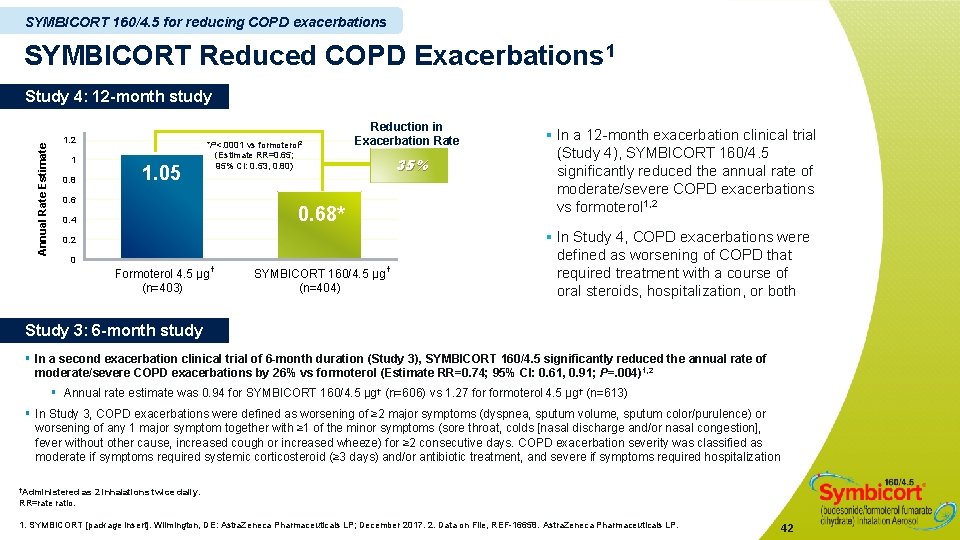
SYMBICORT 160/4. 5 for reducing COPD exacerbations SYMBICORT Reduced COPD Exacerbations 1 Annual Rate Estimate Study 4: 12 -month study 1. 2 1 0. 8 1. 05 *P<. 0001 vs formoterol 2 (Estimate RR=0. 65; 95% CI: 0. 53, 0. 80) 0. 6 Reduction in Exacerbation Rate 35% 0. 68* 0. 4 0. 2 0 Formoterol 4. 5 µg (n=403) † SYMBICORT 160/4. 5 µg (n=404) † § In a 12 -month exacerbation clinical trial (Study 4), SYMBICORT 160/4. 5 significantly reduced the annual rate of moderate/severe COPD exacerbations vs formoterol 1, 2 § In Study 4, COPD exacerbations were defined as worsening of COPD that required treatment with a course of oral steroids, hospitalization, or both Study 3: 6 -month study § In a second exacerbation clinical trial of 6 -month duration (Study 3), SYMBICORT 160/4. 5 significantly reduced the annual rate of moderate/severe COPD exacerbations by 26% vs formoterol (Estimate RR=0. 74; 95% CI: 0. 61, 0. 91; P=. 004)1, 2 § Annual rate estimate was 0. 94 for SYMBICORT 160/4. 5 µg† (n=606) vs 1. 27 formoterol 4. 5 µg† (n=613) § In Study 3, COPD exacerbations were defined as worsening of ≥ 2 major symptoms (dyspnea, sputum volume, sputum color/purulence) or worsening of any 1 major symptom together with ≥ 1 of the minor symptoms (sore throat, colds [nasal discharge and/or nasal congestion], fever without other cause, increased cough or increased wheeze) for ≥ 2 consecutive days. COPD exacerbation severity was classified as moderate if symptoms required systemic corticosteroid (≥ 3 days) and/or antibiotic treatment, and severe if symptoms required hospitalization †Administered as 2 inhalations twice daily. RR=rate ratio. 1. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; December 2017. 2. Data on File, REF-16658. Astra. Zeneca Pharmaceuticals LP. 42

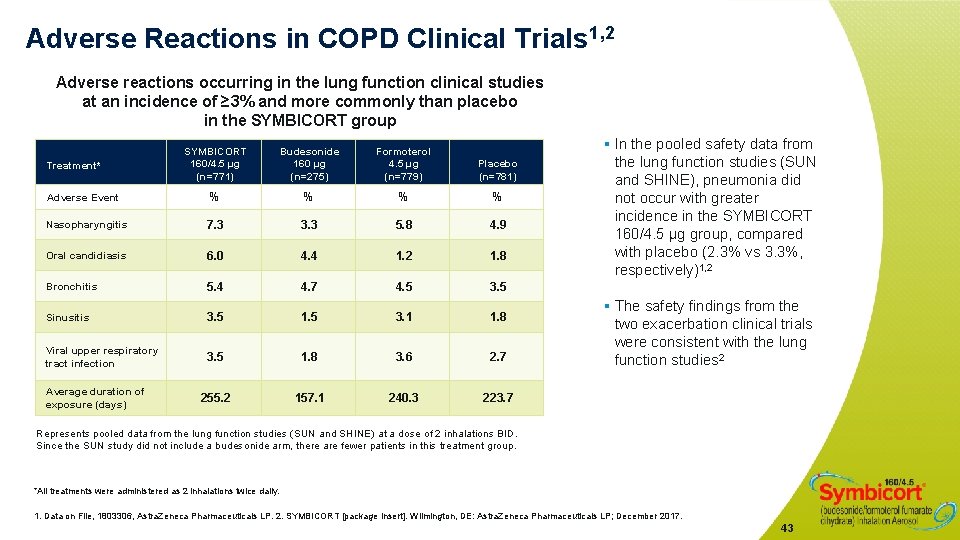
Adverse Reactions in COPD Clinical Trials 1, 2 Adverse reactions occurring in the lung function clinical studies at an incidence of ≥ 3% and more commonly than placebo in the SYMBICORT group SYMBICORT 160/4. 5 µg (n=771) Budesonide 160 µg (n=275) Formoterol 4. 5 µg (n=779) Placebo (n=781) Adverse Event % % Nasopharyngitis 7. 3 3. 3 5. 8 4. 9 Oral candidiasis 6. 0 4. 4 1. 2 1. 8 Bronchitis 5. 4 4. 7 4. 5 3. 5 Sinusitis 3. 5 1. 5 3. 1 1. 8 Viral upper respiratory tract infection 3. 5 1. 8 3. 6 2. 7 255. 2 157. 1 240. 3 223. 7 Treatment* Average duration of exposure (days) § In the pooled safety data from the lung function studies (SUN and SHINE), pneumonia did not occur with greater incidence in the SYMBICORT 160/4. 5 μg group, compared with placebo (2. 3% vs 3. 3%, respectively)1, 2 § The safety findings from the two exacerbation clinical trials were consistent with the lung function studies 2 Represents pooled data from the lung function studies (SUN and SHINE) at a dose of 2 inhalations BID. Since the SUN study did not include a budesonide arm, there are fewer patients in this treatment group. *All treatments were administered as 2 inhalations twice daily. 1. Data on File, 1803306, Astra. Zeneca Pharmaceuticals LP. 2. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; December 2017. 43

Important Safety Information § Use of long-acting beta 2 -adrenergic agonists (LABA) as monotherapy (without inhaled corticosteroids [ICS]) for asthma is associated with an increased risk of asthma-related death. These findings are considered a class effect of LABA. When LABA are used in fixed dose combination with ICS, data from large clinical trials do not show a significant increase in the risk of serious asthma-related events (hospitalizations, intubations, death) compared to ICS alone § It is possible that systemic corticosteroid effects such as hypercorticism and adrenal suppression may occur, particularly at higher doses. Particular care is needed for patients who are transferred from systemically active corticosteroids to ICS. Deaths due to adrenal insufficiency have occurred in asthmatic patients during and after transfer from systemic corticosteroids to less systemically available ICS § SYMBICORT is NOT a rescue medication and does NOT replace fast-acting inhalers to treat acute symptoms § Caution should be exercised when considering administration of SYMBICORT in patients on long-term ketoconazole and other known potent CYP 3 A 4 inhibitors § SYMBICORT should not be initiated in patients during rapidly deteriorating episodes of asthma or COPD § Patients who are receiving SYMBICORT should not use additional formoterol or other LABA for any reason § Localized infections of the mouth and pharynx with Candida albicans has occurred in patients treated with SYMBICORT. Patients should rinse the mouth after inhalation of SYMBICORT § Lower respiratory tract infections, including pneumonia, have been reported following the administration of ICS § Due to possible immunosuppression, potential worsening of infections could occur. A more serious or even fatal course of chickenpox or measles can occur in susceptible patients § As with other inhaled medications, paradoxical bronchospasm may occur with SYMBICORT § Immediate hypersensitivity reactions may occur, as demonstrated by cases of urticaria, angioedema, rash, and bronchospasm § Excessive beta-adrenergic stimulation has been associated with central nervous system and cardiovascular effects. SYMBICORT should be used with caution in patients with cardiovascular disorders, especially coronary insufficiency, cardiac arrhythmias, and hypertension Please see full Prescribing Information, including Patient Information, available at this presentation. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; December 2017. 44

Important Safety Information (Continued) § Long-term use of ICS may result in a decrease in bone mineral density (BMD). Since patients with COPD often have multiple risk factors for reduced BMD, assessment of BMD is recommended prior to initiating SYMBICORT and periodically thereafter § Glaucoma, increased intraocular pressure, and cataracts have been reported following the administration of ICS, including budesonide, a component of SYMBICORT. Close monitoring is warranted in patients with a change in vision or history of increased intraocular pressure, glaucoma, or cataracts § SYMBICORT should be administered with caution to patients being treated with MAO inhibitors or tricyclic antidepressants, or within 2 weeks of discontinuation of such agents § Beta-blockers may not only block the pulmonary effect of betaagonists, such as formoterol, but may produce severe bronchospasm in patients with asthma § ECG changes and/or hypokalemia associated with nonpotassium-sparing diuretics may worsen with concomitant beta-agonists. Use caution with the coadministration of SYMBICORT § In rare cases, patients on ICS may present with systemic eosinophilic conditions INDICATIONS § SYMBICORT should be used with caution in patients with convulsive disorders, thyrotoxicosis, diabetes mellitus, ketoacidosis, and in patients who are unusually responsive to sympathomimetic amines SYMBICORT 160/4. 5 is indicated for the maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema, and to reduce COPD exacerbations. § Beta-adrenergic agonist medications may produce hypokalemia and hyperglycemia in some patients § The most common adverse reactions ≥ 3% reported in COPD clinical trials included nasopharyngitis, oral candidiasis, bronchitis, sinusitis, and upper respiratory tract infection SYMBICORT is NOT indicated for the relief of acute bronchospasm. Please see full Prescribing Information, including Patient Information, available at this presentation. SYMBICORT [package insert]. Wilmington, DE: Astra. Zeneca Pharmaceuticals LP; December 2017. 45

Comments to Astra. Zeneca is committed to conducting business with the highest standards of integrity and professionalism. If you have comments that could improve the delivery of our promotional educational programs, please contact Astra. Zeneca at 1 -800 -236 -9933. © 2018 Astra. Zeneca. All rights reserved. US-16961 1/18