Approach to Dyslipidemia Among Diabetic Patients Farzad Hadaegh


Approach to Dyslipidemia Among Diabetic Patients Farzad Hadaegh, MD Prevention of Metabolic Disorders Research Center Research Institute for Endocrine Sciences Shahid Beheshti University of Medical Sciences Tehran, Oct 2019 12/5/2020 2

Agenda ■ Overview of dyslipidemia among Tehranian diabetic population ■ Definition and pathophysiology of diabetic dyslipidemia ■ Statin therapy in diabetic patients ■ Residual risk and add on statin therapy(ezetimibe , highly purified omega 3 and PCSK 9 inhibotors) in individuals with diabetes mellitus ■ Guideline (ADA/2019, AHA/2019, 2019 ESC/EAS) ■ Take home message 12/5/2020 3

Case Scenario A 58 -year-old obese male patient with type 2 diabetes mellitus (T 2 DM) presents with a history of ACS and previous CABG 2 years earlier. His Hb. A 1 C value has been stable at 7. 2% with metformin and liraglutide 1. 8 mg daily. He is currently normotensive with Lozartan/HCT 50 mg/12. 5 mg with a urine/albumin creatinine ratio at 80 mg/mg and an e. GFR of 48 ml/min. The patient’s current lipid profile with rosuvastatin 40 mg and ezetimibe 10 mgdaily is as follows: (LDL-C), 66 mg/dl; (TGs), 320 mg/dl; (HDL-C), 38 mg/dl; and non– HDL-C, 130 mg/dl. The patient and his primary care physician are concerned about his residual risk of recurrent ASCVD events and his overall prognosis. What is your recommendation for the patient? 12/5/2020 4

Aims/Hypothesis: To investigate secular trends in cardiovascular disease (CVD) risk factors during a decade of follow-up in a Middle Eastern cohort, and to compare observed trends between diabetic and non-diabetic populations. Methods: In a population of 6181 participants (2622 males and 3559 females), diabetes status and CVD risk factors were evaluated in 4 study phases from 1999– 2011. 1045 subjects had type 2 diabetes mellitus at baseline and 5136 participants were diabetes-free. 12/5/2020 5
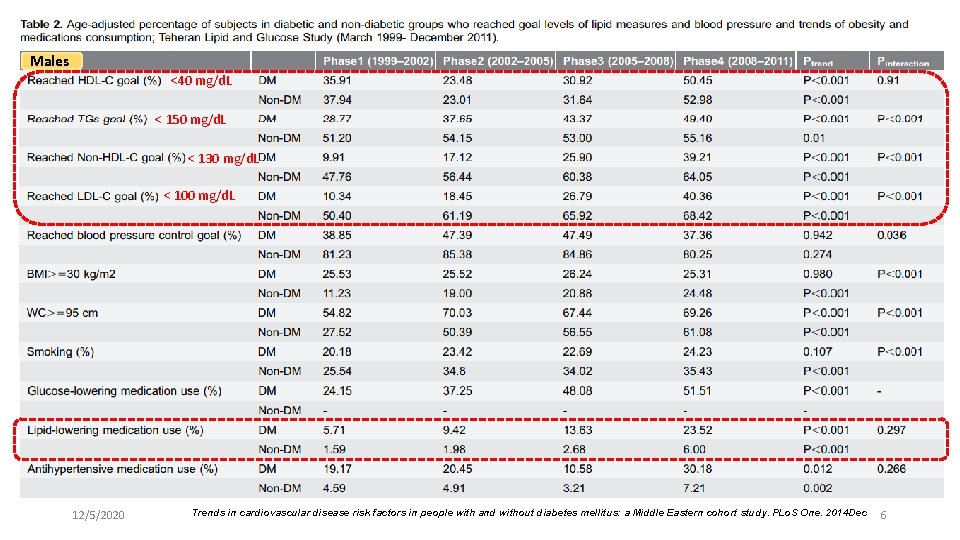
Males <40 mg/d. L < 150 mg/d. L < 130 mg/d. L < 100 mg/d. L 12/5/2020 Trends in cardiovascular disease risk factors in people with and without diabetes mellitus: a Middle Eastern cohort study. PLo. S One. 2014 Dec 6
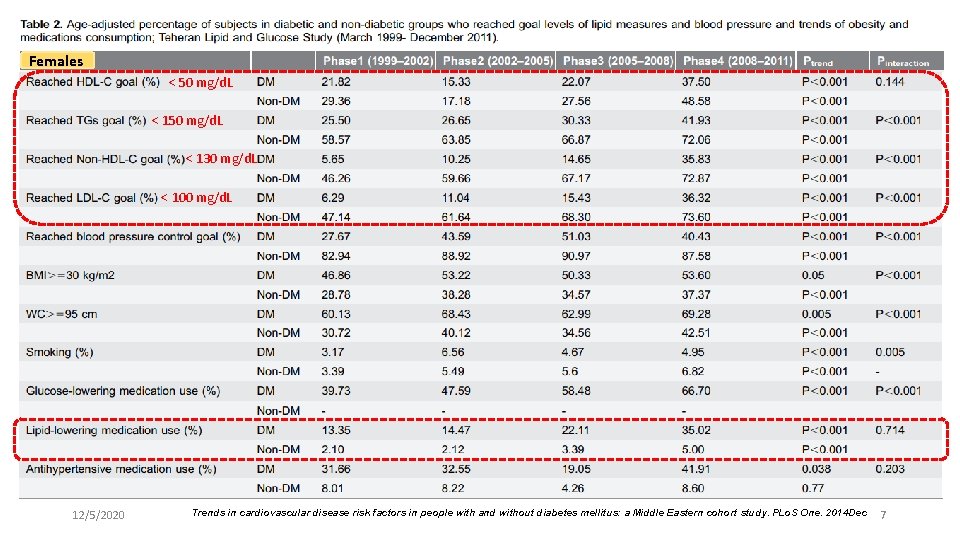
Females < 50 mg/d. L < 130 mg/d. L < 100 mg/d. L 12/5/2020 Trends in cardiovascular disease risk factors in people with and without diabetes mellitus: a Middle Eastern cohort study. PLo. S One. 2014 Dec 7

Results l During the follow-up period diabetic females significantly gained better control of their blood pressure, serum LDL-C and general and central obesity measures compared to non-diabetic counterparts, although 60% of them had high BP and 64% had high serum LDL-C levels till the end of the study. l Diabetic males however, experienced significantly better control on their serum LDL-C and general and central obesity measures compared to their non-diabetic controls; but 24% of them were still smoker, 63% had high BP and 60% had high serum LDL-C levels at the end of the follow-up (all Ps interaction , 0. 05). l Use of lipid-lowering and antihypertensive medications increased consistently in both diabetic and nondiabetic populations. 12/5/2020 Trends in cardiovascular disease risk factors in people with and without diabetes mellitus: a Middle Eastern cohort study. PLo. S One. 2014 Dec 8

Diabetic Dyslipidemia l In patients with type 2 diabetes mellitus, triglycerides are often elevated, HDL-C is often decreased, and LDL-C may be elevated, borderline, or normal. l LDL particles are small and dense. Thus, the LDL-C concentration may be misleading because there will be more LDL particles for any cholesterol concentration l The prevalence of DD reported in various studies varied between 35 and 56% and is strongly associated with increased cardiovascular risk Diabtic Diabetic. Dyslipidemia 12/5/2020 Atherosclerosis 2015. 239: 483 -95 9
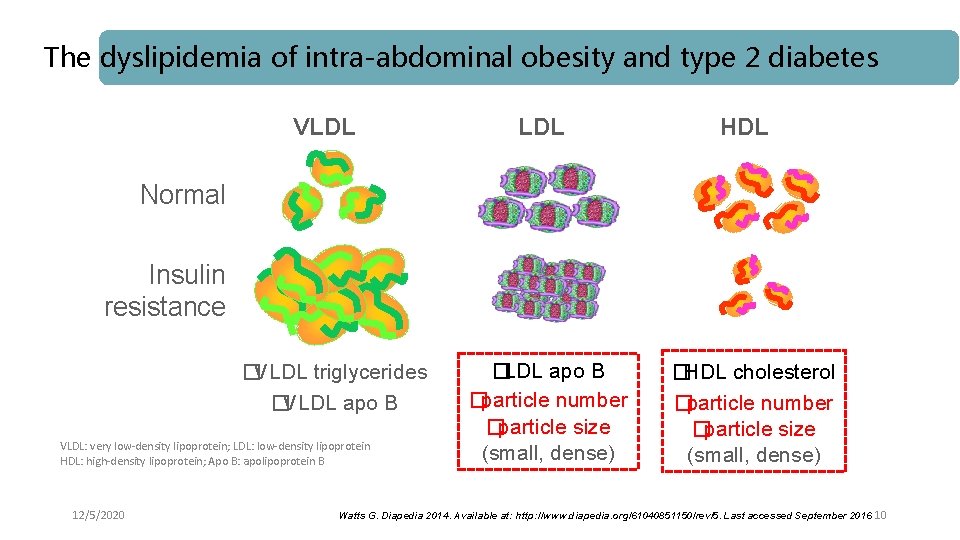
The dyslipidemia of intra-abdominal obesity and type 2 diabetes VLDL HDL Normal Insulin resistance �VLDL triglycerides �VLDL apo B VLDL: very low-density lipoprotein; LDL: low-density lipoprotein HDL: high-density lipoprotein; Apo B: apolipoprotein B 12/5/2020 �LDL apo B �particle number �particle size (small, dense) �HDL cholesterol �particle number �particle size (small, dense) Watts G. Diapedia 2014. Available at: http: //www. diapedia. org/61040851150/rev/5. Last accessed September 2016 10

Two Ways To Measure LDL Quantity Ø LDL cholesterol (LDL-C) is the traditional measure of LDL, chosen for historical, not analytic or clinical reasons. Ø Alternatively, LDL can be measured by particle number (LDL-P), or estimated by apolipoprotein B. Ø Due to differences in the amount of cholesterol contained in LDL, alternate LDL measures (LDL-C vs. LDL-P) frequently disagree (discordance). 1 -7 Triglycerides LDL Particle LDL-P LDL-C LDL Particle LDL-P Cholesterol LDL-C Cholesterol 12/5/2020 1. Otvos JD, et al. Am J Cardiol. 2002; 90(8 A): 22 i-29 i. 2. Cromwell WC, Otvos JD. Am J Cardiol. 2006; 98(12): 1599 -1602. 3. Cromwell WC, et al. . J Clin Lipidol. 2007; 1(6): 583 -592. 4. Otvos JD, et al. J Clin Lipidol. 2011; 5(2): 105 -113. 5. Sniderman AD, et al. Am J Cardiol. 2003; 91(10): 1173 -1177. 6. Sniderman AD, et al. Am J Cardiol. 2001; 87(6): 792 -793, A 798. 7. Sniderman AD. J Clin Lipidol. 2008; 2(1): 36 -42. 11

Alternate LDL Measures and Cardiovascular Disease Cardiovascular risk tracks with LDL particle number § When alternate LDL measures (LDL-C vs LDL particle number) agree (concordance) each measure is equally associated with CVD risk. § When alternate measures are discordant (e. g. , diabetes, metabolic syndrome, statin therapy), risk tracks with LDL-P, not LDL-C. § The majority of atherogenic lipoproteins in individuals with IR, metabolic syndrome or T 2 D are smaller, cholesterol-depleted LDL particles. These compositional changes in LDL particles may lead to a disagreement between measures of LDL-C and LDL-P resulting in “discordance” 12/5/2020 1. Otvos JD, et al. Am J Cardiol. 2002; 90(8 A): 22 i-29 i. 2. Cromwell WC, Otvos JD. Am J Cardiol. 2006; 98(12): 1599 -1602. 3. Cromwell WC, et al. . J Clin Lipidol. 2007; 1(6): 583 -592. 4. Otvos JD, et al. J Clin Lipidol. 2011; 5(2): 105 -113. 5. Sniderman AD, et al. Am J Cardiol. 2003; 91(10): 1173 -1177. 6. Sniderman AD, et al. Am J Cardiol. 2001; 87(6): 792 -793, A 798. 7. Sniderman AD. J Clin Lipidol. 2008; 2(1): 36 -42. 12
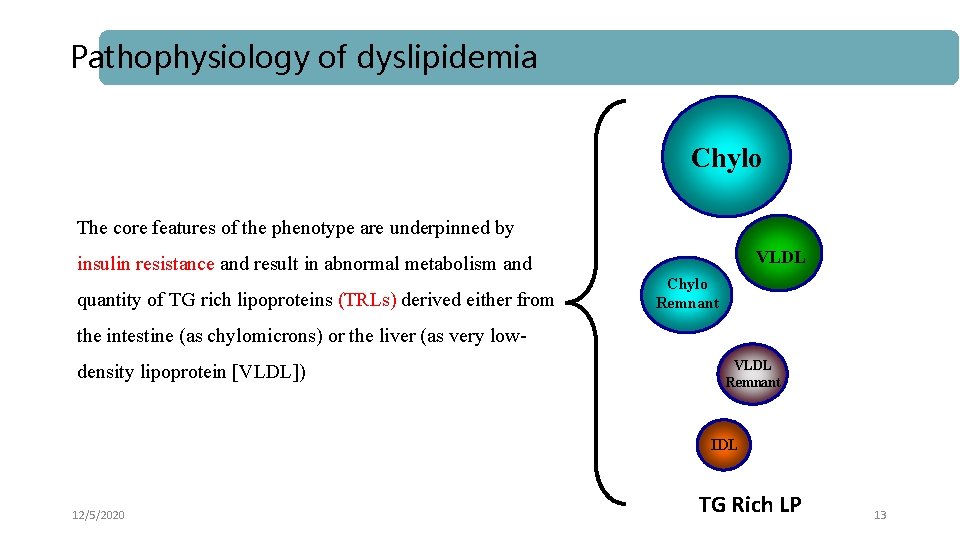
Pathophysiology of dyslipidemia Chylo The core features of the phenotype are underpinned by VLDL insulin resistance and result in abnormal metabolism and quantity of TG rich lipoproteins (TRLs) derived either from Chylo Remnant the intestine (as chylomicrons) or the liver (as very lowdensity lipoprotein [VLDL]) VLDL Remnant IDL 12/5/2020 TG Rich LP 13
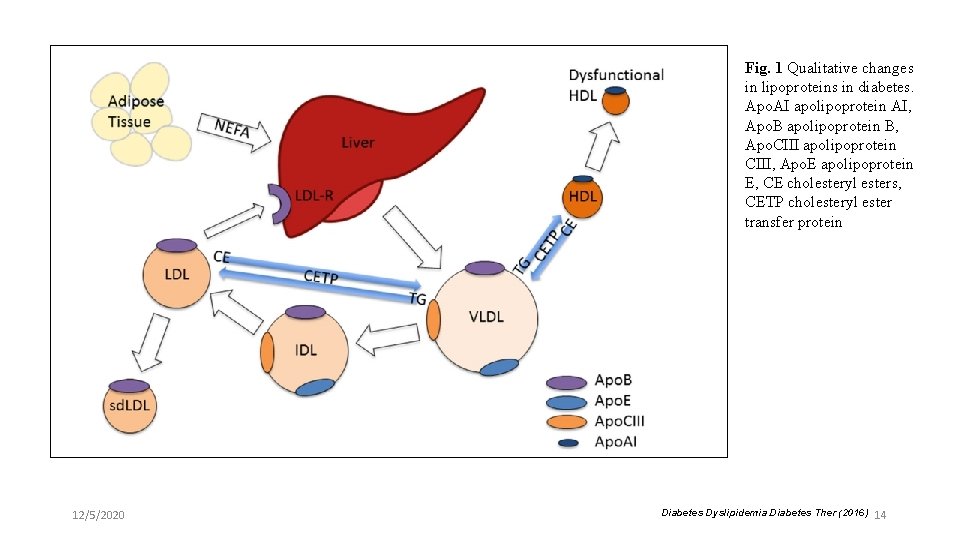
Fig. 1 Qualitative changes in lipoproteins in diabetes. Apo. AI apolipoprotein AI, Apo. B apolipoprotein B, Apo. CIII apolipoprotein CIII, Apo. E apolipoprotein E, CE cholesteryl esters, CETP cholesteryl ester transfer protein 12/5/2020 Diabetes Dyslipidemia. Diabetes Ther (2016) 14
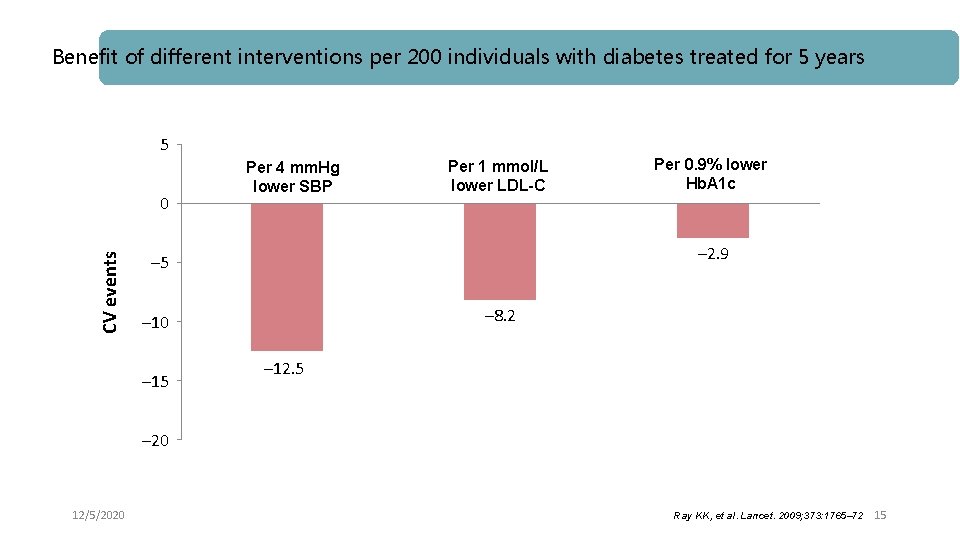
Benefit of different interventions per 200 individuals with diabetes treated for 5 years 5 CV events 0 Per 4 mm. Hg lower SBP Per 1 mmol/L lower LDL-C – 2. 9 – 5 – 8. 2 – 10 – 15 Per 0. 9% lower Hb. A 1 c – 12. 5 – 20 12/5/2020 Ray KK, et al. Lancet. 2009; 373: 1765– 72 15

Efficacy of statins in diabetic patients 12/5/2020 16
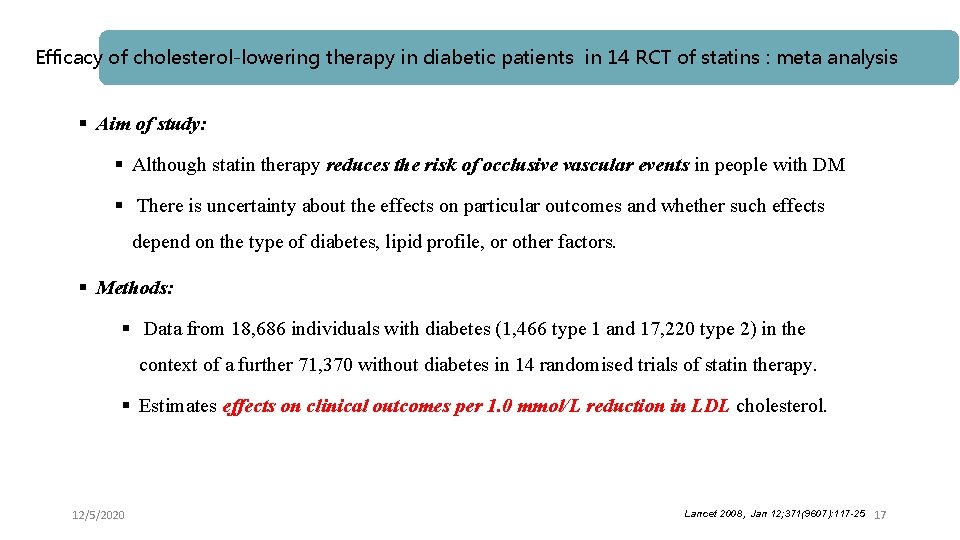
Efficacy of cholesterol-lowering therapy in diabetic patients in 14 RCT of statins : meta analysis § Aim of study: § Although statin therapy reduces the risk of occlusive vascular events in people with DM § There is uncertainty about the effects on particular outcomes and whether such effects depend on the type of diabetes, lipid profile, or other factors. § Methods: § Data from 18, 686 individuals with diabetes (1, 466 type 1 and 17, 220 type 2) in the context of a further 71, 370 without diabetes in 14 randomised trials of statin therapy. § Estimates effects on clinical outcomes per 1. 0 mmol/L reduction in LDL cholesterol. 12/5/2020 Lancet 2008, Jan 12; 371(9607): 117 -25 17
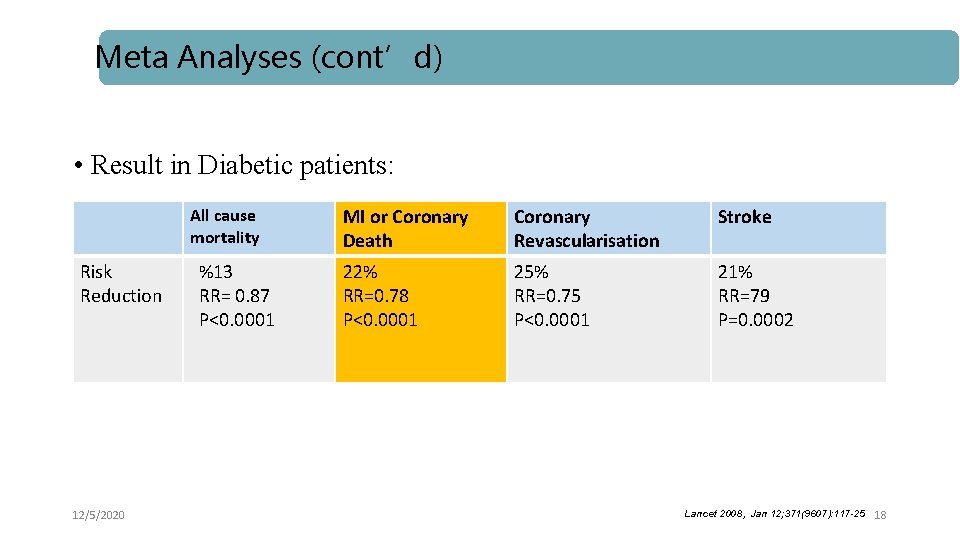
Meta Analyses (cont’d) • Result in Diabetic patients: Risk Reduction 12/5/2020 All cause mortality MI or Coronary Death Coronary Revascularisation Stroke %13 RR= 0. 87 P<0. 0001 22% RR=0. 78 P<0. 0001 25% RR=0. 75 P<0. 0001 21% RR=79 P=0. 0002 Lancet 2008, Jan 12; 371(9607): 117 -25 18

Results l After 5 years, 42 (95% CI 30– 55) fewer people with diabetes had major vascular events per 1000 allocated statin therapy. 12/5/2020 19
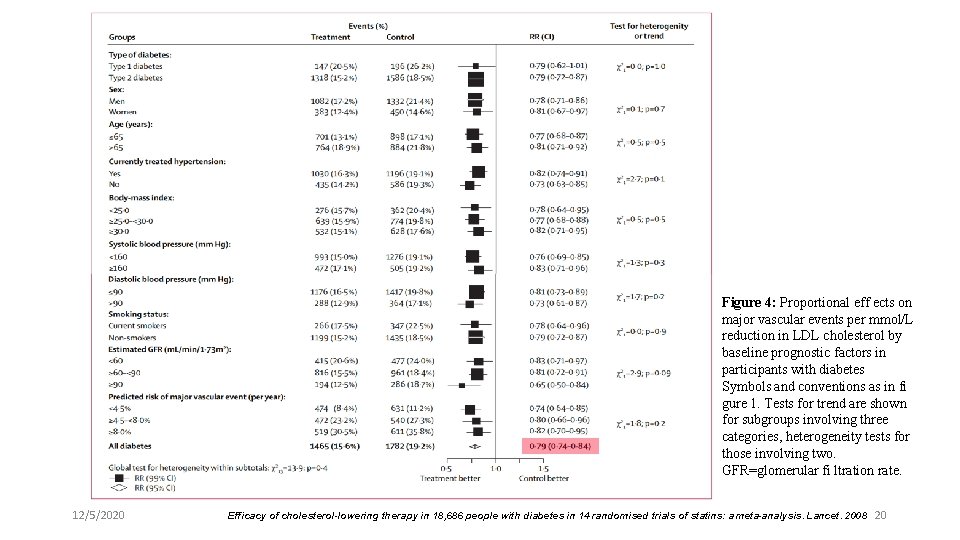
Figure 4: Proportional eff ects on major vascular events per mmol/L reduction in LDL cholesterol by baseline prognostic factors in participants with diabetes Symbols and conventions as in fi gure 1. Tests for trend are shown for subgroups involving three categories, heterogeneity tests for those involving two. GFR=glomerular fi ltration rate. 12/5/2020 Efficacy of cholesterol-lowering therapy in 18, 686 people with diabetes in 14 randomised trials of statins: a meta-analysis. Lancet. 2008 20
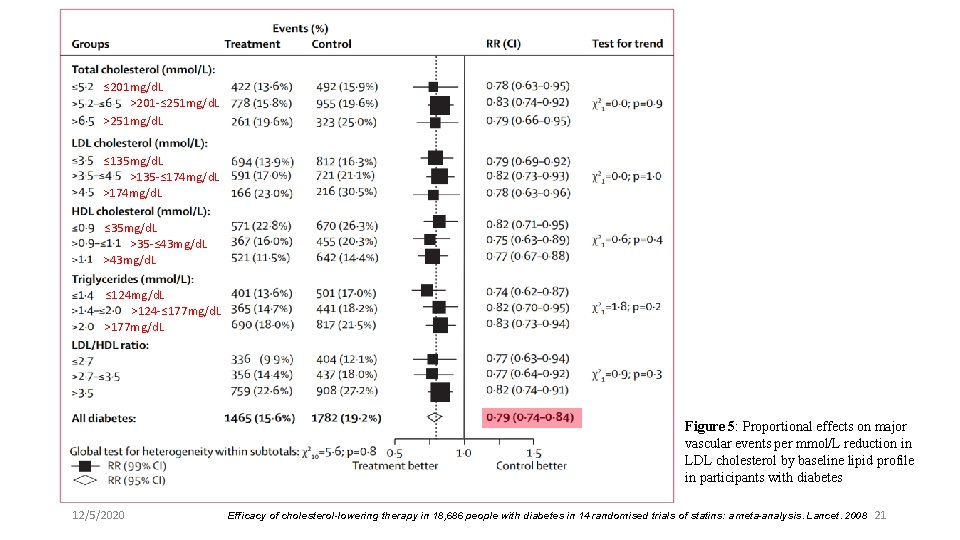
≤ 201 mg/d. L >201 -≤ 251 mg/d. L >251 mg/d. L ≤ 135 mg/d. L >135 -≤ 174 mg/d. L >174 mg/d. L ≤ 35 mg/d. L >35 -≤ 43 mg/d. L >43 mg/d. L ≤ 124 mg/d. L >124 -≤ 177 mg/d. L >177 mg/d. L Figure 5: Proportional effects on major vascular events per mmol/L reduction in LDL cholesterol by baseline lipid profile in participants with diabetes 12/5/2020 Efficacy of cholesterol-lowering therapy in 18, 686 people with diabetes in 14 randomised trials of statins: a meta-analysis. Lancet. 2008 21

Key message l Statin therapy safely reduces the 5 -year incidence of major coronary events, coronary revascularization, and stroke by about a fifth per mmol/L reduction in LDL cholesterol, largely irrespective of initial lipid profile or other baseline characteristics l Conclusion: Statin therapy should be considered for all diabetic individuals who are at sufficiently high risk of vascular events 12/5/2020 22
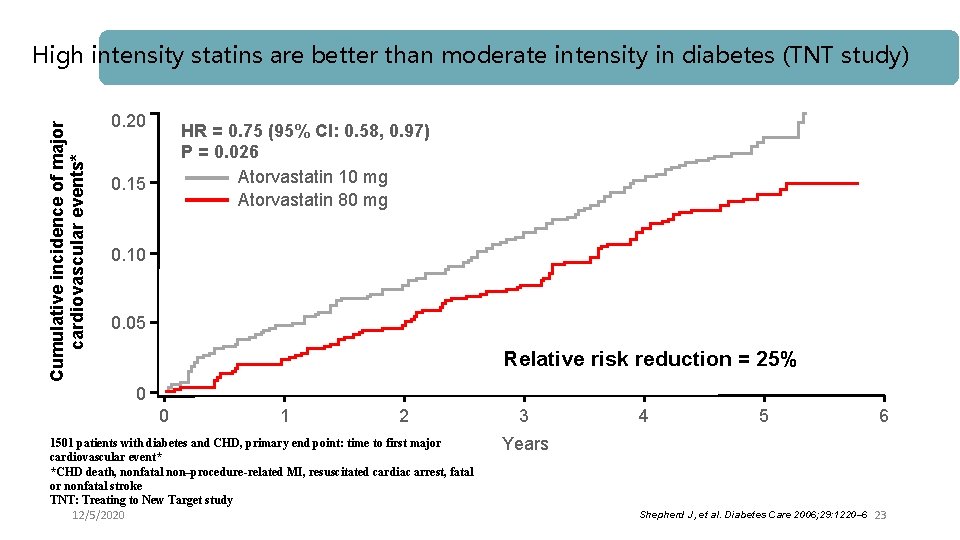
Cumulative incidence of major cardiovascular events* High intensity statins are better than moderate intensity in diabetes (TNT study) 0. 20 HR = 0. 75 (95% CI: 0. 58, 0. 97) P = 0. 026 Atorvastatin 10 mg Atorvastatin 80 mg 0. 15 0. 10 0. 05 Relative risk reduction = 25% 0 0 1 2 1501 patients with diabetes and CHD, primary end point: time to first major cardiovascular event* *CHD death, nonfatal non–procedure-related MI, resuscitated cardiac arrest, fatal or nonfatal stroke TNT: Treating to New Target study 12/5/2020 3 4 5 6 Years Shepherd J, et al. Diabetes Care 2006; 29: 1220– 6 23

Diabetes with statin use A meta-analysis of 13 randomized statin trials with 91, 140 participants showed an odds ratio of 1. 09 for a new diagnosis of diabetes, so that (on average) treatment of 255 patients with statins for 4 years resulted in 1 additional case of diabetes while simultaneously preventing 5. 4 vascular events among those 255 patients. 12/5/2020 Sattar N, Preiss D, Murray HM, et al. Statins and risk of incident diabetes: a collaborative meta-analysis of randomised statin trials. Lancet 2010; 375: 735– 742 24

Residual Cardiovascular Risk Remaining After Statin Treatment 12/5/2020 25
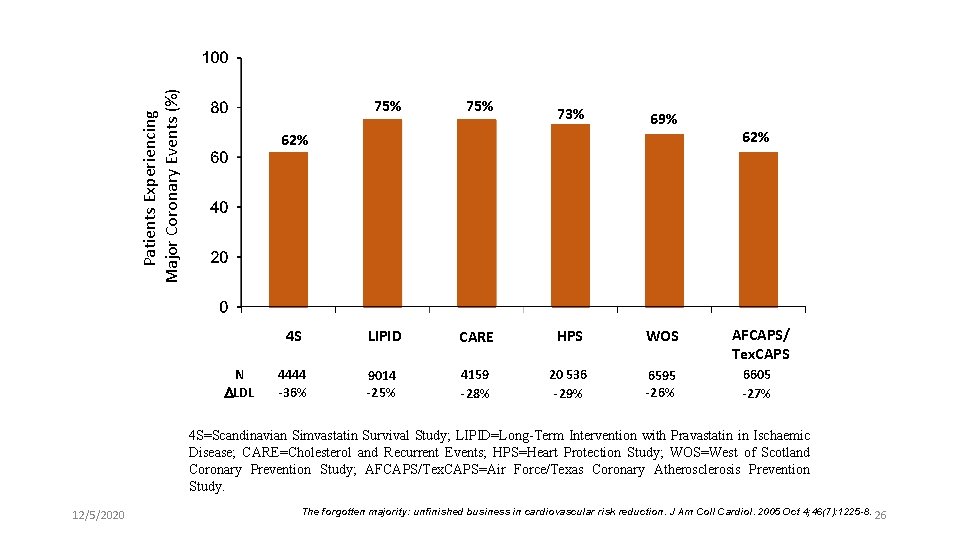
Patients Experiencing Major Coronary Events (%) 75% 73% 69% 62% N LDL 62% 4 S LIPID CARE HPS WOS AFCAPS/ Tex. CAPS 4444 -36% 9014 -25% 4159 -28% 20 536 -29% 6595 -26% 6605 -27% 4 S=Scandinavian Simvastatin Survival Study; LIPID=Long-Term Intervention with Pravastatin in Ischaemic Disease; CARE=Cholesterol and Recurrent Events; HPS=Heart Protection Study; WOS=West of Scotland Coronary Prevention Study; AFCAPS/Tex. CAPS=Air Force/Texas Coronary Atherosclerosis Prevention Study. 12/5/2020 The forgotten majority: unfinished business in cardiovascular risk reduction. J Am Coll Cardiol. 2005 Oct 4; 46(7): 1225 -8. 26
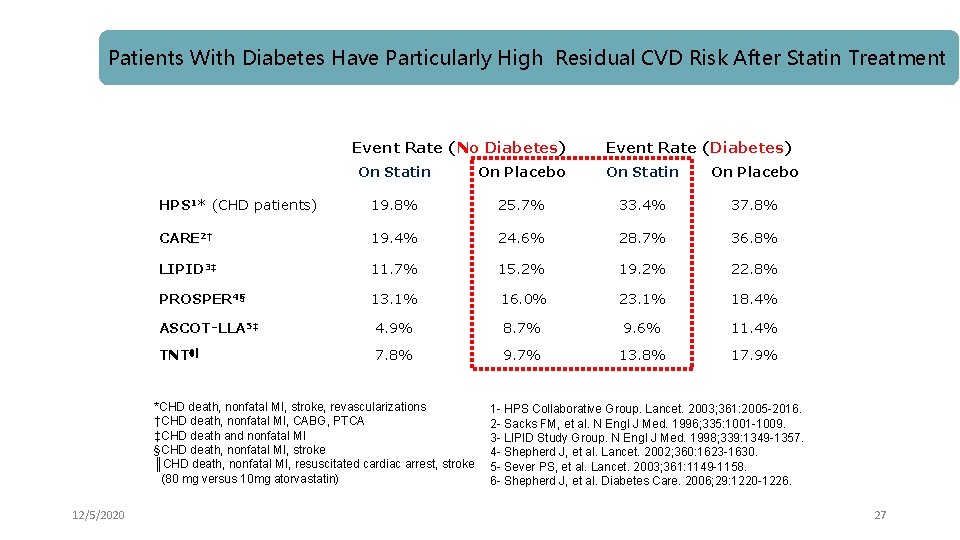
Patients With Diabetes Have Particularly High Residual CVD Risk After Statin Treatment Event Rate (No Diabetes) On Statin On Placebo HPS 1* (CHD patients) 19. 8% 25. 7% 33. 4% 37. 8% CARE 2† 19. 4% 24. 6% 28. 7% 36. 8% LIPID 3‡ 11. 7% 15. 2% 19. 2% 22. 8% PROSPER 4§ 13. 1% 16. 0% 23. 1% 18. 4% ASCOT-LLA 5‡ 4. 9% 8. 7% 9. 6% 11. 4% TNT 6║ 7. 8% 9. 7% 13. 8% 17. 9% *CHD death, nonfatal MI, stroke, revascularizations †CHD death, nonfatal MI, CABG, PTCA ‡CHD death and nonfatal MI §CHD death, nonfatal MI, stroke ║CHD death, nonfatal MI, resuscitated cardiac arrest, stroke (80 mg versus 10 mg atorvastatin) 12/5/2020 Event Rate (Diabetes) 1 - HPS Collaborative Group. Lancet. 2003; 361: 2005 -2016. 2 - Sacks FM, et al. N Engl J Med. 1996; 335: 1001 -1009. 3 - LIPID Study Group. N Engl J Med. 1998; 339: 1349 -1357. 4 - Shepherd J, et al. Lancet. 2002; 360: 1623 -1630. 5 - Sever PS, et al. Lancet. 2003; 361: 1149 -1158. 6 - Shepherd J, et al. Diabetes Care. 2006; 29: 1220 -1226. 27

Residual risk ? Blood Pressure control Role of other risk factors Beyond LDL-C, non HDL-C a better predictor Elevated Triglyceride level Role of TG rich lipoproteins 12/5/2020 28

Role of non-HDL-C beyond LDL-C • To determine if non-HDL cholesterol is a more useful predictor of coronary heart disease (CHD) risk than LDL cholesterol and if VLDL cholesterol is an independent predictor of CHD risk. • All subjects aged >30 years and free of CHD at baseline, followed for incident CHD (618 men, 372 women). 12/5/2020 29
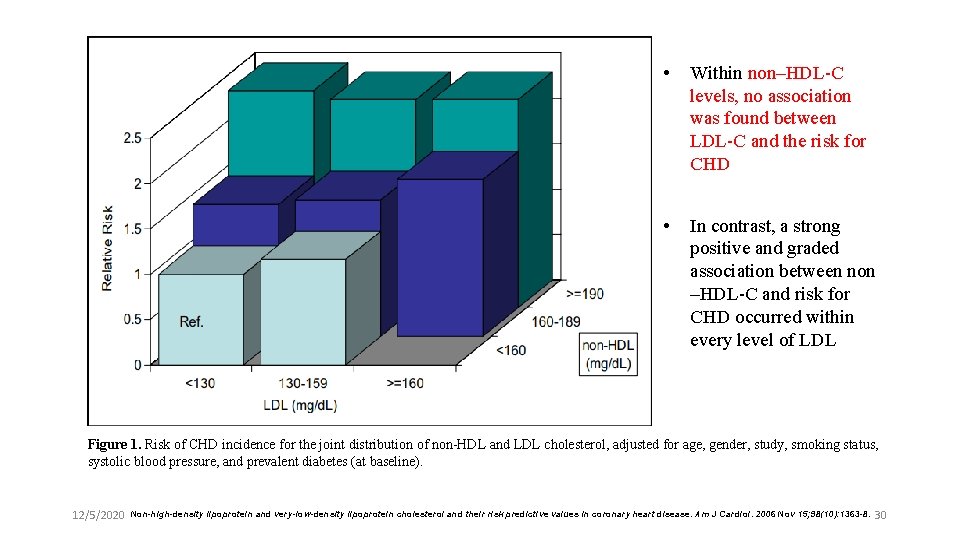
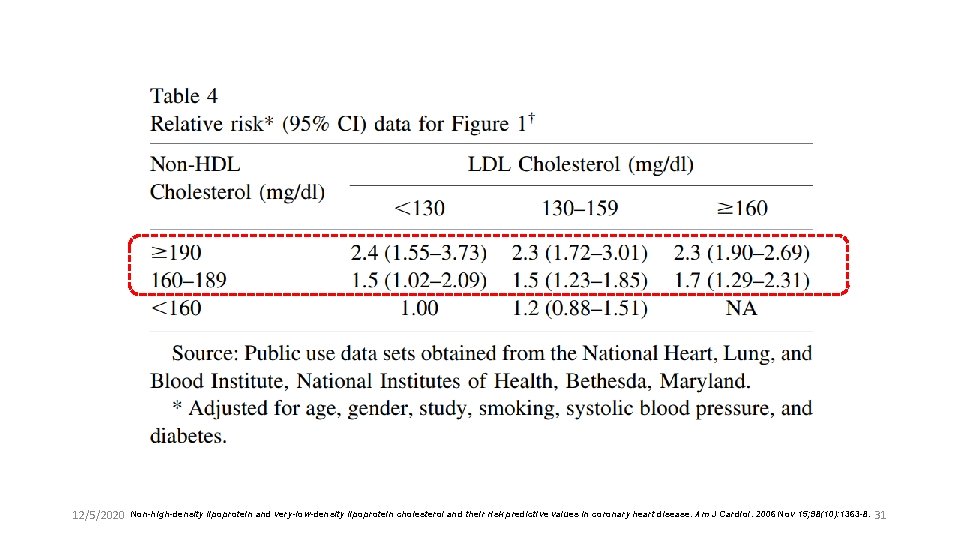
• Within non–HDL-C levels, no association was found between LDL-C and the risk for CHD • In contrast, a strong positive and graded association between non –HDL-C and risk for CHD occurred within every level of LDL Figure 1. Risk of CHD incidence for the joint distribution of non-HDL and LDL cholesterol, adjusted for age, gender, study, smoking status, systolic blood pressure, and prevalent diabetes (at baseline). 12/5/2020 Non-high-density lipoprotein and very-low-density lipoprotein cholesterol and their risk predictive values in coronary heart disease. Am J Cardiol. 2006 Nov 15; 98(10): 1363 -8. 30
12/5/2020 Non-high-density lipoprotein and very-low-density lipoprotein cholesterol and their risk predictive values in coronary heart disease. Am J Cardiol. 2006 Nov 15; 98(10): 1363 -8. 31

Key messages l The results from this study support the conclusions that VLDL cholesterol is an independent predictor of CHD risk and that non-HDL-C overall appears to be a better predictor of CHD risk than LDL -C. l These conclusions appear to be valid for the entire population and when TG levels are either ≥ 200 or< 200 mg/dl. 12/5/2020 Non-high-density lipoprotein and very-low-density lipoprotein cholesterol and their risk predictive values in coronary heart disease. Am J Cardiol. 2006 Nov 15; 98(10): 1363 -8. 32

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 33

Role of TG beyond LDL-C • • • 12/5/2020 The PROVE IT-TIMI 22 trial demonstrated that low-density lipoprotein cholesterol (LDL-C)< 70 mg/dl was associated with greater CHD event reduction than LDL-C <100 mg/dl after ACS. However, the impact of low on-treatment TG on CHD risk beyond LDL-C <70 mg/dl has not been explored. Purpose: to assess the impact of on treatment TG on CHD risk after an ACS Sample size: 4, 162 patients hospitalized for ACS Intervention: Atorvastatin 80 mg VS pravastatin 40 mg daily Length of F/U: 2 years 34
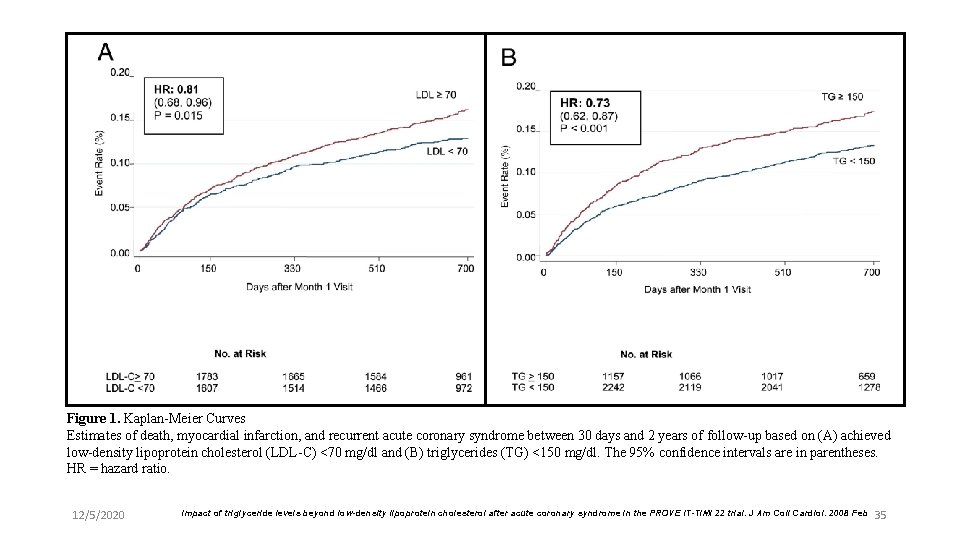
Figure 1. Kaplan-Meier Curves Estimates of death, myocardial infarction, and recurrent acute coronary syndrome between 30 days and 2 years of follow-up based on (A) achieved low-density lipoprotein cholesterol (LDL-C) <70 mg/dl and (B) triglycerides (TG) <150 mg/dl. The 95% confidence intervals are in parentheses. HR = hazard ratio. 12/5/2020 Impact of triglyceride levels beyond low-density lipoprotein cholesterol after acute coronary syndrome in the PROVE IT-TIMI 22 trial. J Am Coll Cardiol. 2008 Feb 35
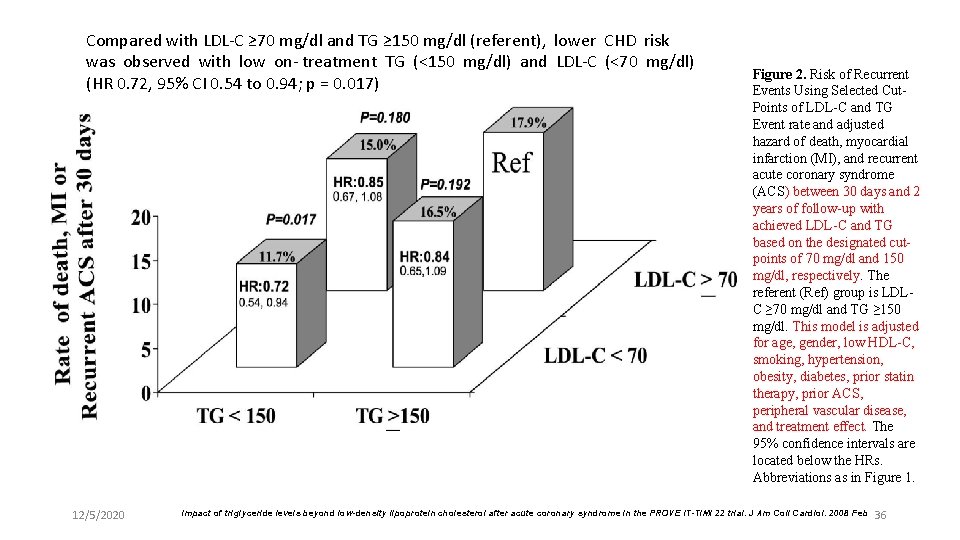
Compared with LDL-C ≥ 70 mg/dl and TG ≥ 150 mg/dl (referent), lower CHD risk was observed with low on- treatment TG (<150 mg/dl) and LDL-C (<70 mg/dl) (HR 0. 72, 95% CI 0. 54 to 0. 94; p = 0. 017) 12/5/2020 Figure 2. Risk of Recurrent Events Using Selected Cut. Points of LDL-C and TG Event rate and adjusted hazard of death, myocardial infarction (MI), and recurrent acute coronary syndrome (ACS) between 30 days and 2 years of follow-up with achieved LDL-C and TG based on the designated cutpoints of 70 mg/dl and 150 mg/dl, respectively. The referent (Ref) group is LDLC ≥ 70 mg/dl and TG ≥ 150 mg/dl. This model is adjusted for age, gender, low HDL-C, smoking, hypertension, obesity, diabetes, prior statin therapy, prior ACS, peripheral vascular disease, and treatment effect. The 95% confidence intervals are located below the HRs. Abbreviations as in Figure 1. Impact of triglyceride levels beyond low-density lipoprotein cholesterol after acute coronary syndrome in the PROVE IT-TIMI 22 trial. J Am Coll Cardiol. 2008 Feb 36

Key messages l In this analysis , the most noteworthy finding was the reduced risk of CHD with low on-treatment TG (<150 mg/dl) that was independent of the level of LDL-C. l For each 10 -mg/dl decline in on-treatment TG, we observed a 1. 4% lower risk of the composite end point (p <0. 001) after adjustment for LDL-C, non. HDL-C and other covariates. l Moreover, the combination of low LDL-C (<70 mg/dl) and low TG (<150 mg/dl) was associated with the lowest event rates compared with higher LDL-C, higher TG, or both. 12/5/2020 Impact of triglyceride levels beyond low-density lipoprotein cholesterol after acute coronary syndrome in the PROVE IT-TIMI 22 trial. J Am Coll Cardiol. 2008 Feb 37
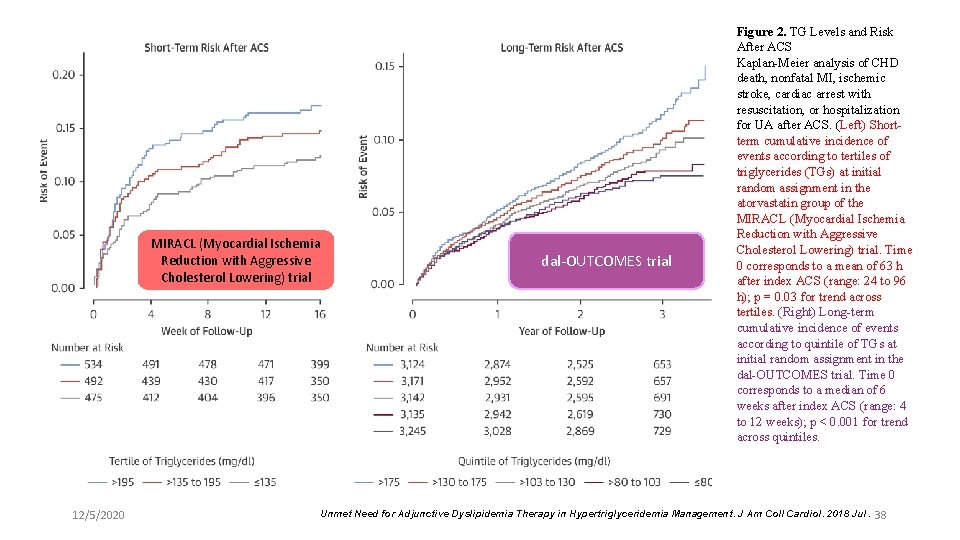
MIRACL (Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering) trial 12/5/2020 dal-OUTCOMES trial Figure 2. TG Levels and Risk After ACS Kaplan-Meier analysis of CHD death, nonfatal MI, ischemic stroke, cardiac arrest with resuscitation, or hospitalization for UA after ACS. (Left) Shortterm cumulative incidence of events according to tertiles of triglycerides (TGs) at initial random assignment in the atorvastatin group of the MIRACL (Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering) trial. Time 0 corresponds to a mean of 63 h after index ACS (range: 24 to 96 h); p = 0. 03 for trend across tertiles. (Right) Long-term cumulative incidence of events according to quintile of TGs at initial random assignment in the dal-OUTCOMES trial. Time 0 corresponds to a median of 6 weeks after index ACS (range: 4 to 12 weeks); p < 0. 001 for trend across quintiles. Unmet Need for Adjunctive Dyslipidemia Therapy in Hypertriglyceridemia Management. J Am Coll Cardiol. 2018 Jul. 38

Combination therapy of statins with Fibrate 12/5/2020 39

• Randomized, placebo-controlled, double-blind clinical trial conducted in 77 clinical sites in the U. S. and Canada • Sample size: 5518 patients with type 2 diabetes • Mean follow-up : 4. 7 years. 12/5/2020 40
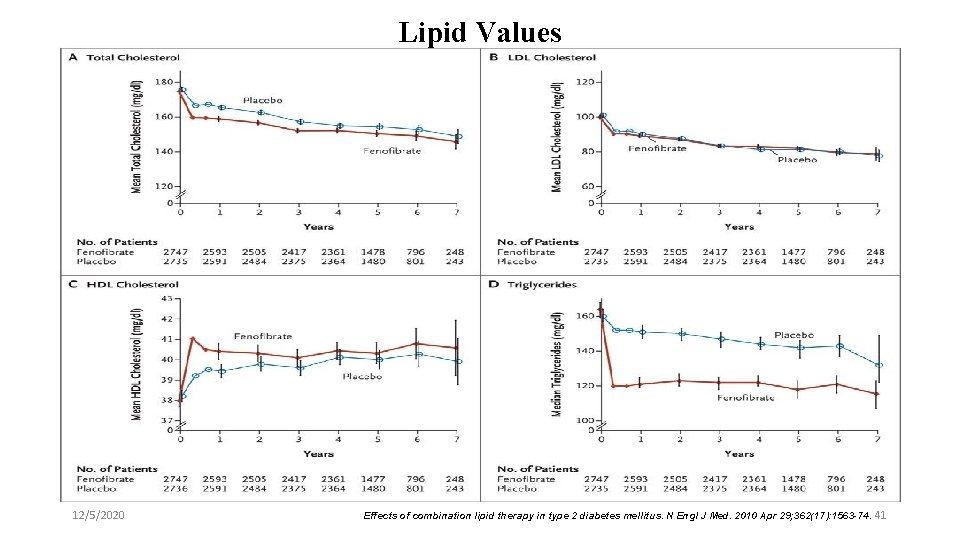
Lipid Values 12/5/2020 Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010 Apr 29; 362(17): 1563 -74. 41
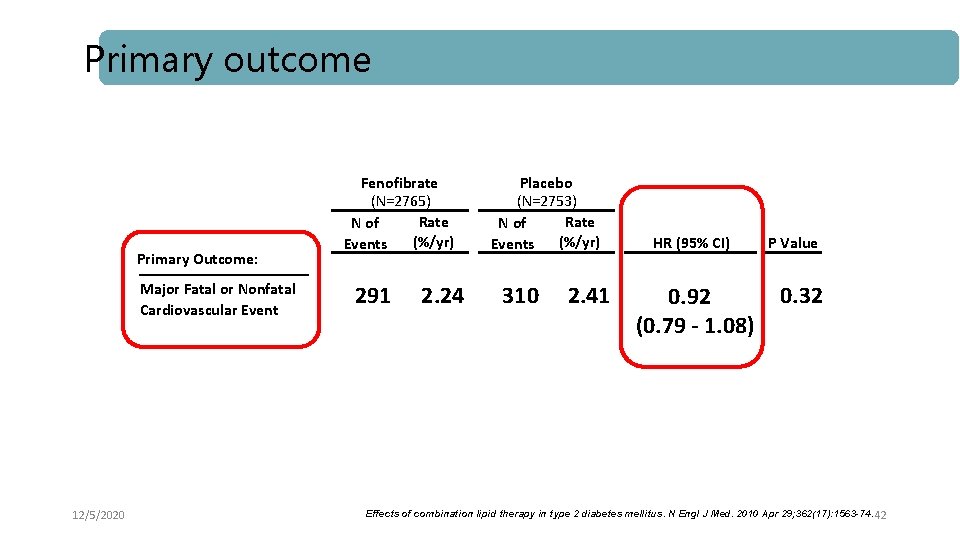
Primary outcome Primary Outcome: Major Fatal or Nonfatal Cardiovascular Event 12/5/2020 Fenofibrate (N=2765) Rate N of (%/yr) Events 291 2. 24 Placebo (N=2753) Rate N of (%/yr) Events 310 2. 41 HR (95% CI) 0. 92 (0. 79 - 1. 08) P Value 0. 32 Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010 Apr 29; 362(17): 1563 -74. 42
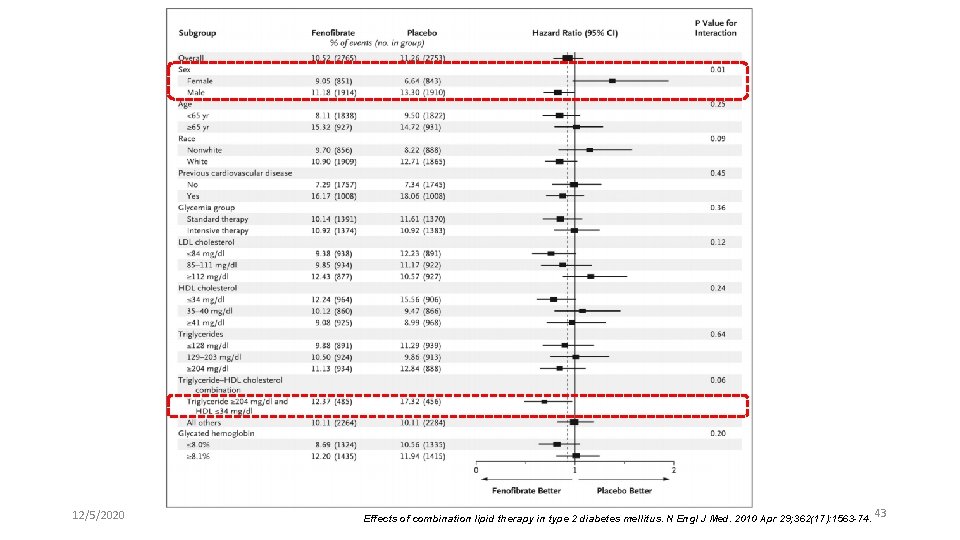
12/5/2020 Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010 Apr 29; 362(17): 1563 -74. 43

Key messages l ACCORD Lipid trial does not support use of the combination of fenofibrate and simvastatin, compared to simvastatin alone, to reduce CVD events in the majority of patients with T 2 DM who are at high risk for CVD. 12/5/2020 Effects of combination lipid therapy in type 2 diabetes mellitus. N Engl J Med. 2010 Apr 29; 362(17): 1563 -74. 44
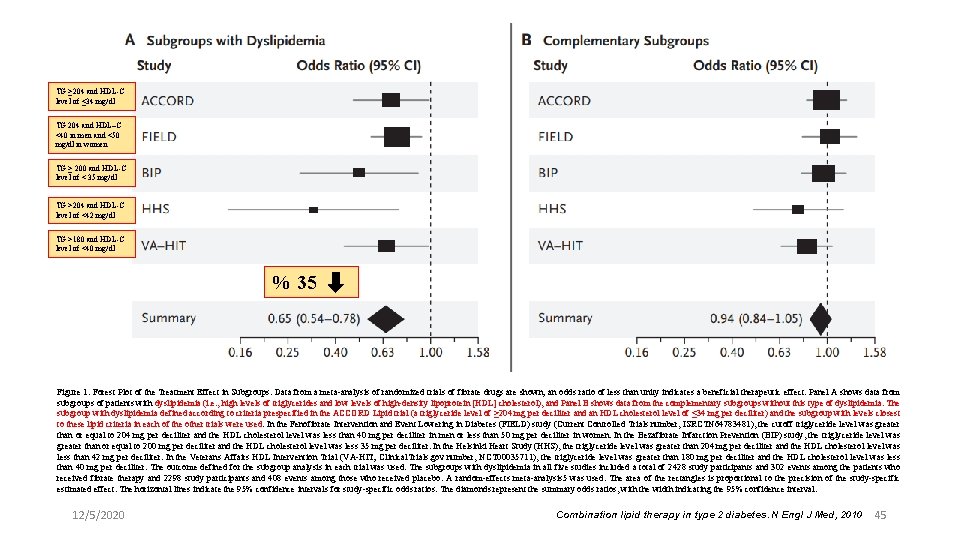
TG ≥ 204 and HDL-C level of ≤ 34 mg/dl TG 204 and HDL–C <40 in men and <50 mg/dl in women TG ≥ 200 and HDL-C level of < 35 mg/dl TG >204 and HDL-C level of <42 mg/dl TG >180 and HDL-C level of <40 mg/dl % 35 Figure 1. Forest Plot of the Treatment Effect in Subgroups. Data from a meta-analysis of randomized trials of fibrate drugs are shown; an odds ratio of less than unity indicates a beneficial therapeutic effect. Panel A shows data from subgroups of patients with dyslipidemia (i. e. , high levels of triglycerides and low levels of high-density lipoprotein [HDL] cholesterol), and Panel B shows data from the complementary subgroups without this type of dyslipidemia. The subgroup with dyslipidemia defined according to criteria prespecified in the ACCORD Lipid trial (a triglyceride level of ≥ 204 mg per deciliter and an HDL cholesterol level of ≤ 34 mg per deciliter) and the subgroup with levels closest to these lipid criteria in each of the other trials were used. In the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study (Current Controlled Trials number, ISRCTN 64783481), the cutoff triglyceride level was greater than or equal to 204 mg per deciliter and the HDL cholesterol level was less than 40 mg per deciliter in men or less than 50 mg per deciliter in women. In the Bezafibrate Infarction Prevention (BIP) study, the triglyceride level was greater than or equal to 200 mg per deciliter and the HDL cholesterol level was less 35 mg per deciliter. In the Helsinki Heart Study (HHS), the triglyceride level was greater than 204 mg per deciliter and the HDL cholesterol level was less than 42 mg per deciliter. In the Veterans Affairs HDL Intervention Trial (VA-HIT; Clinical. Trials. gov number, NCT 00035711), the triglyceride level was greater than 180 mg per deciliter and the HDL cholesterol level was less than 40 mg per deciliter. The outcome defined for the subgroup analysis in each trial was used. The subgroups with dyslipidemia in all five studies included a total of 2428 study participants and 302 events among the patients who received fibrate therapy and 2298 study participants and 408 events among those who received placebo. A random-effects meta-analysis 5 was used. The area of the rectangles is proportional to the precision of the study-specific estimated effect. The horizontal lines indicate the 95% confidence intervals for study-specific odds ratios. The diamonds represent the summary odds ratios, with the width indicating the 95% confidence interval. 12/5/2020 Combination lipid therapy in type 2 diabetes. N Engl J Med, 2010 45

ADA 2019, Combination therapy of Fibrate with Statin 10. 28 Combination therapy (statin/ fibrate) has not been shown to improve atherosclerotic cardiovascular disease outcomes and is generally not recommended. A 10. 29 Combination therapy (statin/ niacin) has not been shown to provide additional cardiovascular benefit above statin therapy alone, may increase the risk of stroke with additional side effects, and is generally not recommended. A 12/5/2020 46

12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 47

12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 48

Combination therapy of statins with Ezethimibe 12/5/2020 49
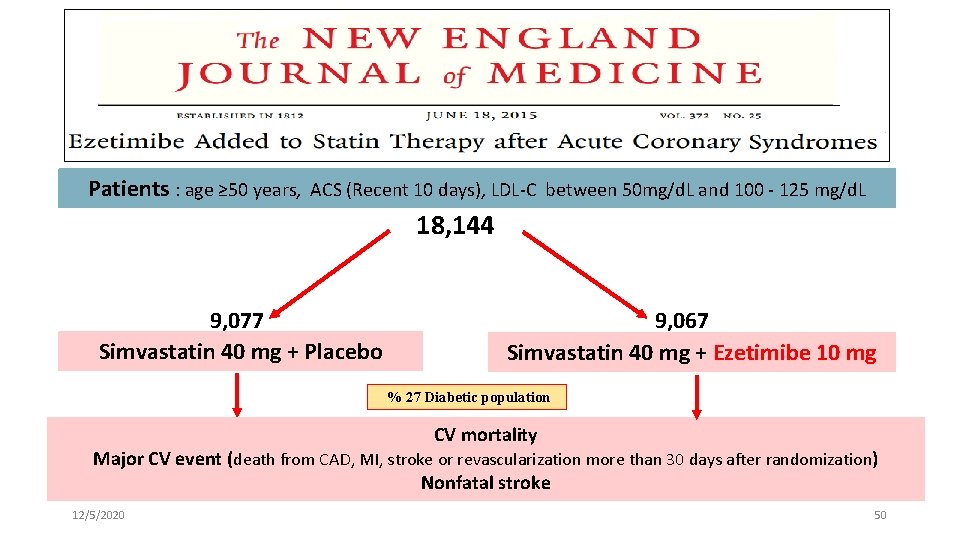
Patients : age ≥ 50 years, ACS (Recent 10 days), LDL-C between 50 mg/d. L and 100 - 125 mg/d. L 18, 144 9, 077 Simvastatin 40 mg + Placebo 9, 067 Simvastatin 40 mg + Ezetimibe 10 mg % 27 Diabetic population CV mortality Major CV event (death from CAD, MI, stroke or revascularization more than 30 days after randomization) Nonfatal stroke 12/5/2020 50

Result l Primary Outcome - CV mortality, major CV event, or nonfatal stroke 34. 7% vs. 32. 7% (HR 0. 94; 95% CI 0. 89 -0. 99; P=0. 016) l Secondary Outcomes - All-cause mortality, major CV event , or nonfatal stroke 40. 3% vs. 38. 7% (HR 0. 95; 95% CI 0. 90 -1. 00; P=0. 03) - CV mortality, nonfatal MI, urgent coronary revascularization ≥ 30 days 18. 9% vs. 17. 5% (HR 0. 91; 95% CI 0. 85 -0. 98; P=0. 02) - CV mortality, nonfatal MI, unstable angina hospitalization, any revascularization ≥ 30 days, nonfatal stroke 36. 2% vs. 34. 5% (HR 0. 95; 95% CI 0. 90 -1. 00; P=0. 04) 12/5/2020 Non-high-density lipoprotein and very-low-density lipoprotein cholesterol and their risk predictive values in coronary heart disease. Am J Cardiol. 2006 51
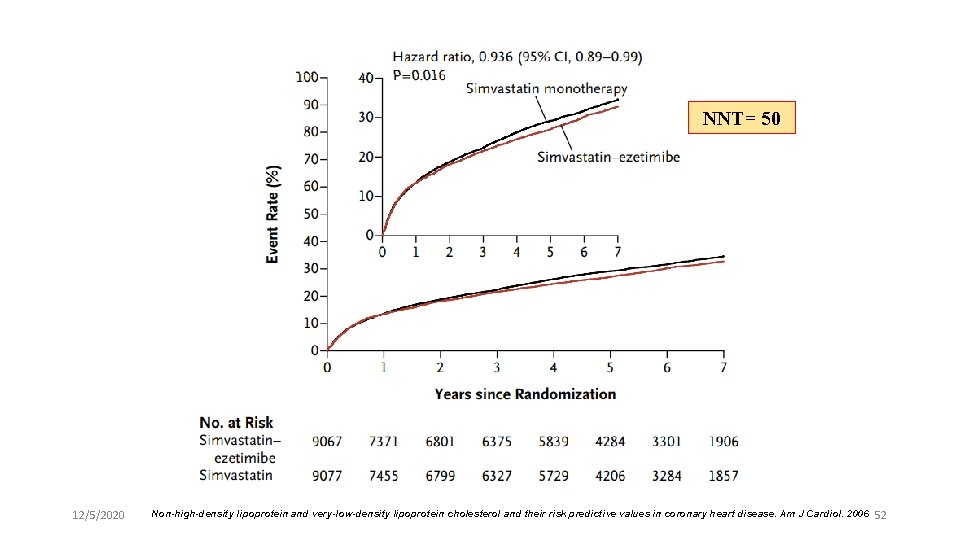
Result NNT= 50 12/5/2020 Non-high-density lipoprotein and very-low-density lipoprotein cholesterol and their risk predictive values in coronary heart disease. Am J Cardiol. 2006 52

Key messages l IMPROVE-IT is the first trial to show that adding a non-statin lipid-modifying agent in concert with a statin improves CV outcomes in addition to enhancing lipid lowering effects 12/5/2020 Non-high-density lipoprotein and very-low-density lipoprotein cholesterol and their risk predictive values in coronary heart disease. Am J Cardiol. 2006 53

Combination therapy of statins with OM 3 FA 12/5/2020 54

v In view of the disappointing results with fibrates and niacin when coadministered in statin-treated patients, there is considerable interest in OM 3 FA (eicosapentaenoic acid [EPA] and docosahexaenoic acid [DHA], or EPA alone) when added to high-potency statin therapy. v These agents have been associated with significant reductions in both TG and non–HDL-C levels during short-term Phase III studies. There is evidence for a modest increase in LDL-C with DHA but not with EPA 12/5/2020 55
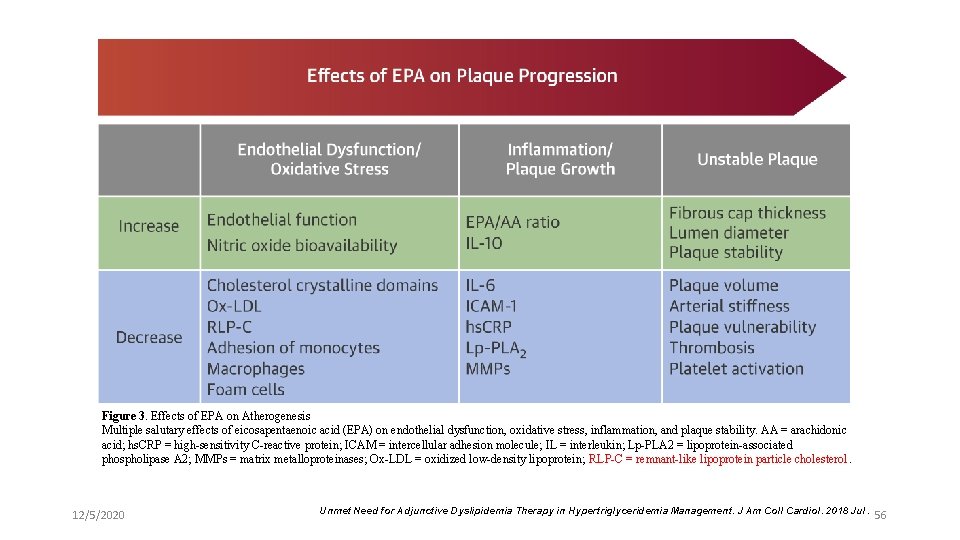
Figure 3. Effects of EPA on Atherogenesis Multiple salutary effects of eicosapentaenoic acid (EPA) on endothelial dysfunction, oxidative stress, inflammation, and plaque stability. AA = arachidonic acid; hs. CRP = high-sensitivity C-reactive protein; ICAM = intercellular adhesion molecule; IL = interleukin; Lp-PLA 2 = lipoprotein-associated phospholipase A 2; MMPs = matrix metalloproteinases; Ox-LDL = oxidized low-density lipoprotein; RLP-C = remnant-like lipoprotein particle cholesterol. 12/5/2020 Unmet Need for Adjunctive Dyslipidemia Therapy in Hypertriglyceridemia Management. J Am Coll Cardiol. 2018 Jul. 56
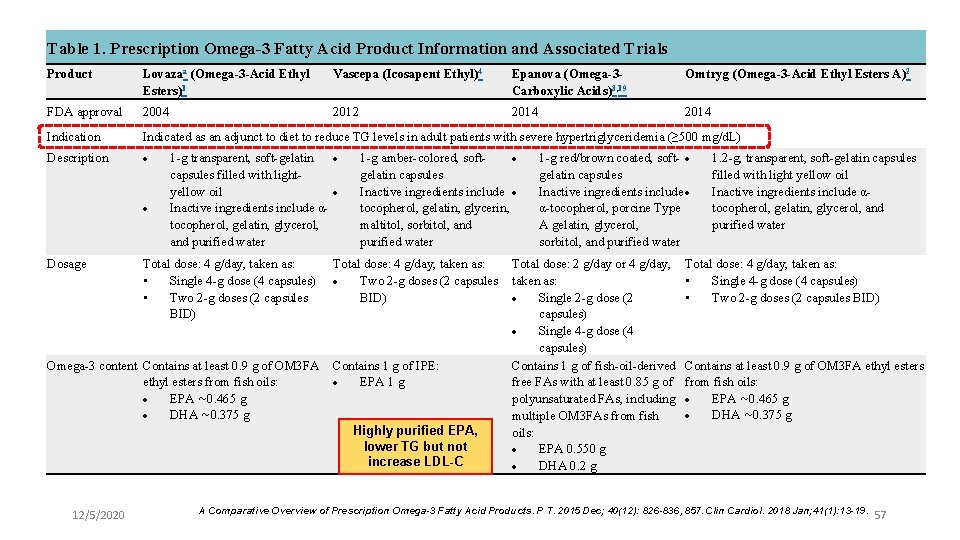
Table 1. Prescription Omega-3 Fatty Acid Product Information and Associated Trials Product Lovazaa (Omega-3 -Acid Ethyl Esters)1 Vascepa (Icosapent Ethyl)4 Epanova (Omega-3 Carboxylic Acids)3, 19 Omtryg (Omega-3 -Acid Ethyl Esters A)2 FDA approval 2004 2012 2014 Indication Indicated as an adjunct to diet to reduce TG levels in adult patients with severe hypertriglyceridemia (≥ 500 mg/d. L) Description Dosage 1 -g transparent, soft-gelatin capsules filled with lightyellow oil Inactive ingredients include αtocopherol, gelatin, glycerol, and purified water 1 -g amber-colored, soft gelatin capsules Inactive ingredients include tocopherol, gelatin, glycerin, maltitol, sorbitol, and purified water 1 -g red/brown coated, soft- gelatin capsules Inactive ingredients include α-tocopherol, porcine Type A gelatin, glycerol, sorbitol, and purified water 1. 2 -g, transparent, soft-gelatin capsules filled with light yellow oil Inactive ingredients include αtocopherol, gelatin, glycerol, and purified water Total dose: 4 g/day, taken as: Total dose: 2 g/day or 4 g/day, Total dose: 4 g/day, taken as: Two 2 -g doses (2 capsules taken as: • Single 4 -g dose (4 capsules) BID) Single 2 -g dose (2 • Two 2 -g doses (2 capsules BID) capsules) Single 4 -g dose (4 capsules) Omega-3 content Contains at least 0. 9 g of OM 3 FA Contains 1 g of IPE: Contains 1 g of fish-oil-derived Contains at least 0. 9 g of OM 3 FA ethyl esters from fish oils: EPA 1 g free FAs with at least 0. 85 g of from fish oils: EPA ∼ 0. 465 g polyunsaturated FAs, including DHA ∼ 0. 375 g multiple OM 3 FAs from fish Highly purified EPA, oils: lower TG but not EPA 0. 550 g increase LDL-C DHA 0. 2 g 12/5/2020 Total dose: 4 g/day, taken as: • Single 4 -g dose (4 capsules) • Two 2 -g doses (2 capsules BID) A Comparative Overview of Prescription Omega-3 Fatty Acid Products. P T. 2015 Dec; 40(12): 826 -836, 857. Clin Cardiol. 2018 Jan; 41(1): 13 -19. 57

BACKGROUND Patients with elevated triglyceride levels are at increased risk for ischemic events. Icosapent ethyl, a highly purified eicosapentaenoic acid ethyl ester, lowers triglyceride levels, but data are needed to determine its effects on ischemic events. METHODS We performed a multicenter, randomized, double-blind, placebo-controlled trial involving patients with established CVD or with diabetes and other risk factors, who had been receiving statin therapy and who had a fasting triglyceride level of 135 to 499 mg per deciliter and a LDL-C level of 41 to 100 mg per deciliter. The patients were randomly assigned to receive 2 g of icosapent ethyl twice daily (total daily dose, 4 g) or placebo. The primary end point was a composite of CV-death, nonfatal MI, nonfatal stroke, coronary revascularization, or unstable angina. The key secondary end point was a composite of CV- death, nonfatal MI, or nonfatal stroke. RESULTS A total of 8179 patients were enrolled (70. 7% for secondary prevention of CVD ) and were followed for a median of 4. 9 years. A primary end-point event occurred in 17. 2% of the patients in the icosapent ethyl group, as compared with 22. 0% of the patients in the placebo group 12/5/2020 58
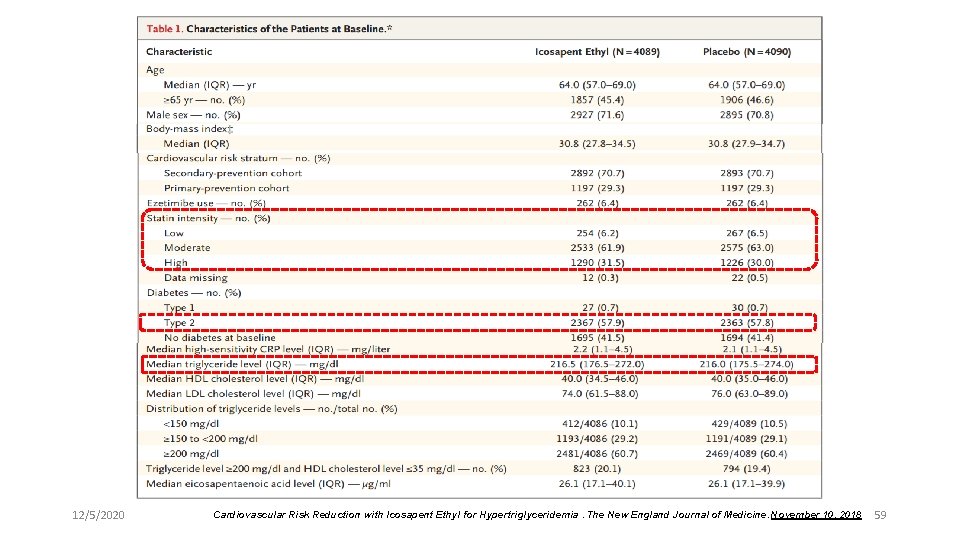
12/5/2020 Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. The New England Journal of Medicine. November 10, 2018 59
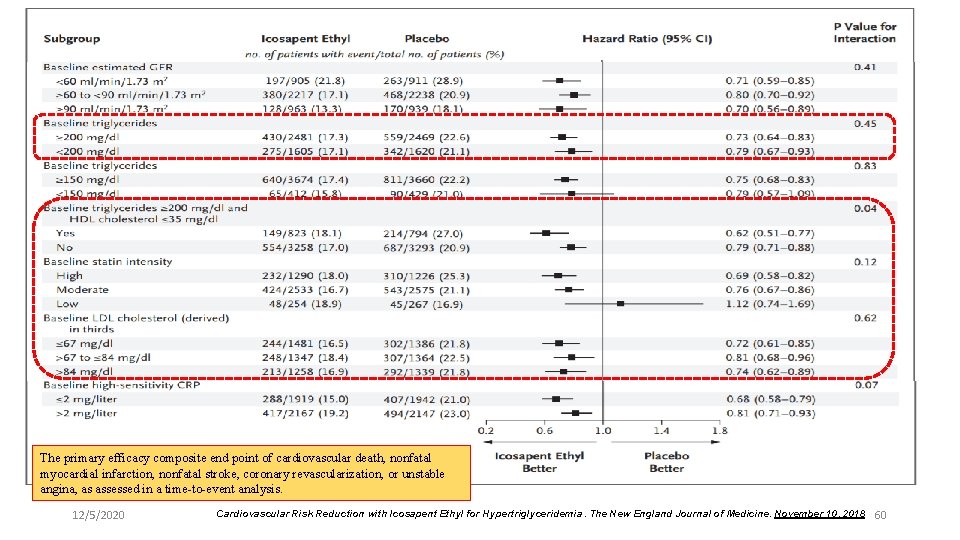
The primary efficacy composite end point of cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, coronary revascularization, or unstable angina, as assessed in a time-to-event analysis. 12/5/2020 Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. The New England Journal of Medicine. November 10, 2018 60
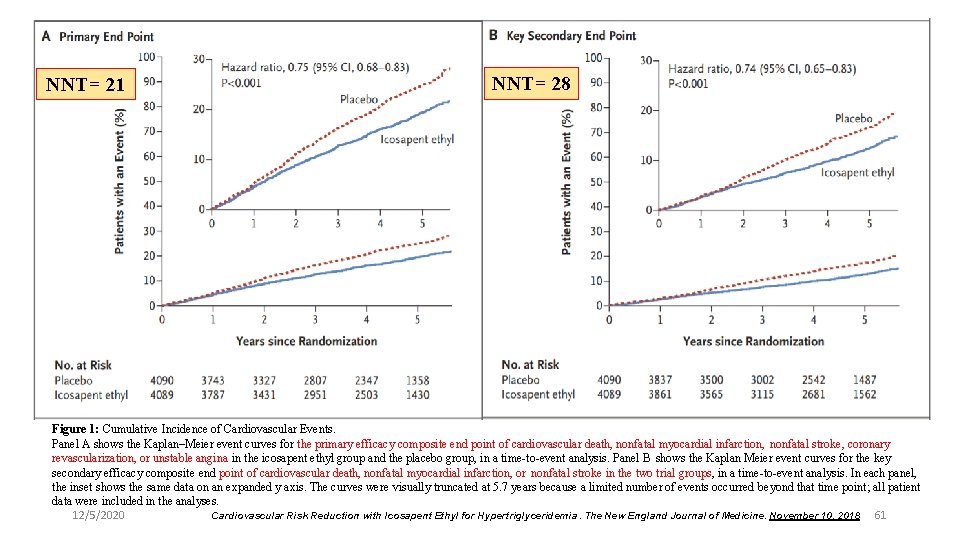
NNT= 21 NNT= 28 Figure 1: Cumulative Incidence of Cardiovascular Events. Panel A shows the Kaplan–Meier event curves for the primary efficacy composite end point of cardiovascular death, nonfatal myocardial infarction, nonfatal stroke, coronary revascularization, or unstable angina in the icosapent ethyl group and the placebo group, in a time-to-event analysis. Panel B shows the Kaplan Meier event curves for the key secondary efficacy composite end point of cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke in the two trial groups, in a time-to-event analysis. In each panel, the inset shows the same data on an expanded y axis. The curves were visually truncated at 5. 7 years because a limited number of events occurred beyond that time point; all patient data were included in the analyses. 12/5/2020 Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. The New England Journal of Medicine. November 10, 2018 61
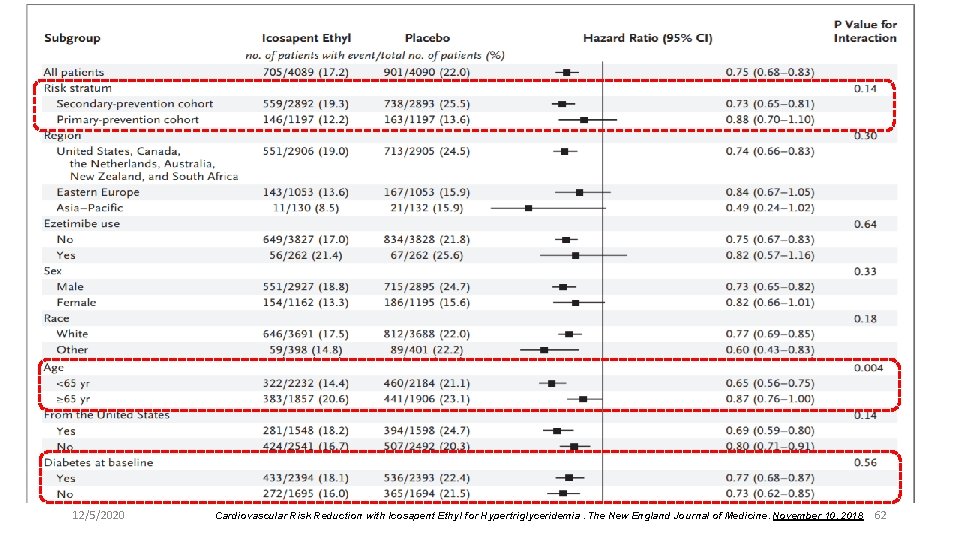
12/5/2020 Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. The New England Journal of Medicine. November 10, 2018 62
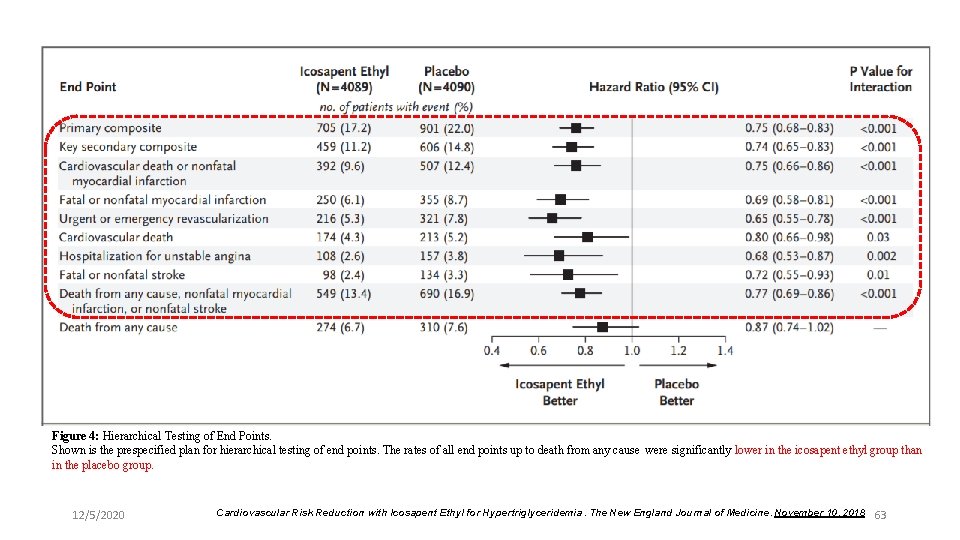
Figure 4: Hierarchical Testing of End Points. Shown is the prespecified plan for hierarchical testing of end points. The rates of all end points up to death from any cause were significantly lower in the icosapent ethyl group than in the placebo group. 12/5/2020 Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. The New England Journal of Medicine. November 10, 2018 63

Conclusions Among patients with elevated triglyceride levels despite the use of statins, the risk of ischemic events, including CV-death, was significantly lower among those who received 2 g of icosapent ethyl twice daily than among those who received placebo. (Funded by Amarin Pharma; REDUCE-IT Clinical. Trials. gov number, NCT 01492361) 12/5/2020 Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. The New England Journal of Medicine. November 10, 2018 64

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 65
Combination therapy of statins with proprotein convertase subtilisin–kexin type 9 (PCSK 9) inhibitors 12/5/2020 66
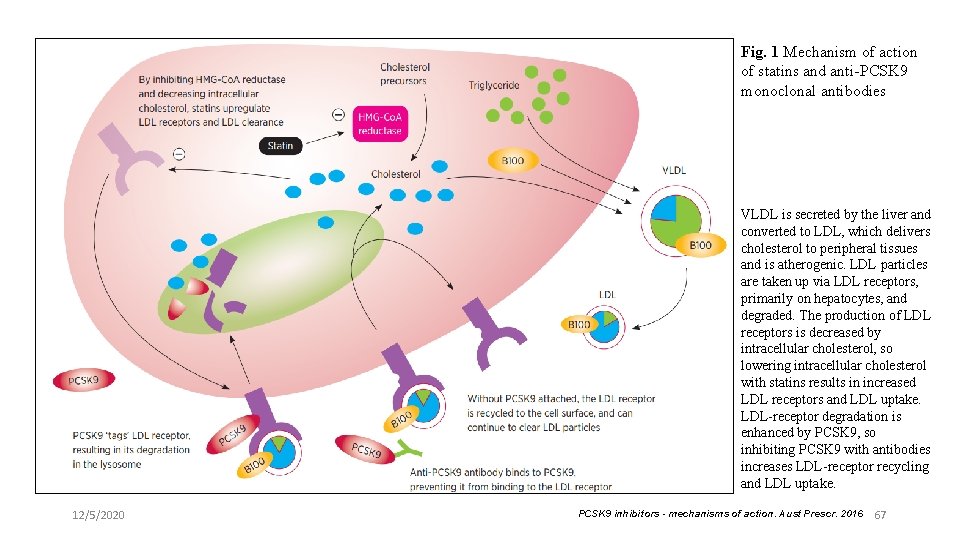
Fig. 1 Mechanism of action of statins and anti-PCSK 9 monoclonal antibodies VLDL is secreted by the liver and converted to LDL, which delivers cholesterol to peripheral tissues and is atherogenic. LDL particles are taken up via LDL receptors, primarily on hepatocytes, and degraded. The production of LDL receptors is decreased by intracellular cholesterol, so lowering intracellular cholesterol with statins results in increased LDL receptors and LDL uptake. LDL-receptor degradation is enhanced by PCSK 9, so inhibiting PCSK 9 with antibodies increases LDL-receptor recycling and LDL uptake. 12/5/2020 PCSK 9 inhibitors - mechanisms of action. Aust Prescr. 2016 67
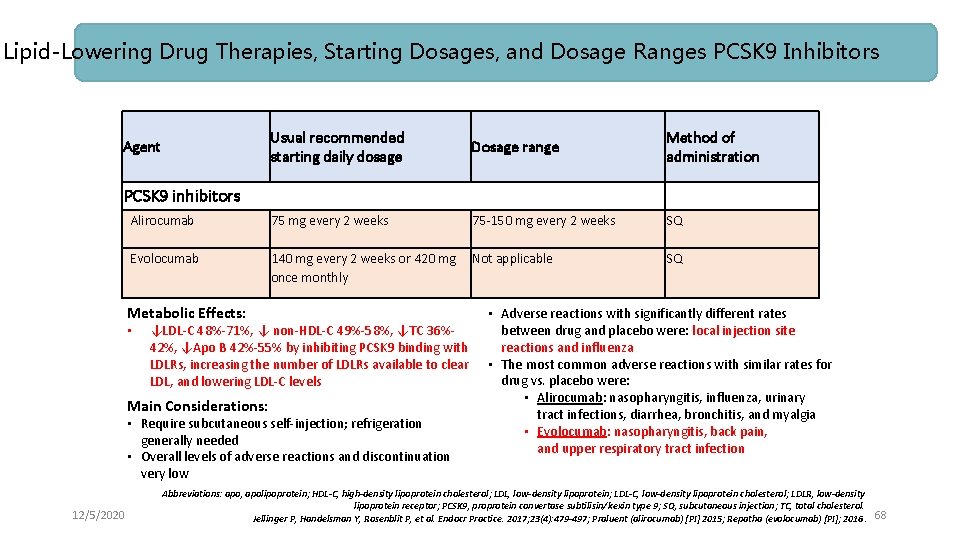
Lipid-Lowering Drug Therapies, Starting Dosages, and Dosage Ranges PCSK 9 Inhibitors Usual recommended starting daily dosage Agent Dosage range Method of administration PCSK 9 inhibitors Alirocumab 75 mg every 2 weeks 75 -150 mg every 2 weeks Evolocumab 140 mg every 2 weeks or 420 mg Not applicable once monthly Metabolic Effects: • ↓LDL-C 48%-71%, ↓ non-HDL-C 49%-58%, ↓TC 36%42%, ↓Apo B 42%-55% by inhibiting PCSK 9 binding with LDLRs, increasing the number of LDLRs available to clear LDL, and lowering LDL-C levels Main Considerations: • Require subcutaneous self-injection; refrigeration generally needed • Overall levels of adverse reactions and discontinuation very low 12/5/2020 SQ SQ • Adverse reactions with significantly different rates between drug and placebo were: local injection site reactions and influenza • The most common adverse reactions with similar rates for drug vs. placebo were: • Alirocumab: nasopharyngitis, influenza, urinary tract infections, diarrhea, bronchitis, and myalgia • Evolocumab: nasopharyngitis, back pain, and upper respiratory tract infection Abbreviations: apo, apolipoprotein; HDL-C, high-density lipoprotein cholesterol; LDL, low-density lipoprotein; LDL-C, low-density lipoprotein cholesterol; LDLR, low-density lipoprotein receptor; PCSK 9, proprotein convertase subtilisin/kexin type 9; SQ, subcutaneous injection; TC, total cholesterol. Jellinger P, Handelsman Y, Rosenblit P, et al. Endocr Practice. 2017; 23(4): 479 -497; Praluent (alirocumab) [PI] 2015; Repatha (evolocumab) [PI]; 2016. 68

FOURIER Clinical Question Among patients with clinical atherosclerotic disease and LDL >70 despite high- or moderate-intensity statin therapy, does the addition of PCSK 9 inhibitor evolocumab reduce major cardiovascular events compared to placebo? Design N= 27, 564 : Evolocumab (n=13, 784), Placebo (n=13, 780) Enrollment: February 2013 - June 2015 Median follow-up: 26 months Analysis: Intention-to-treat Primary outcome: Major CV events (CV death, MI, stroke, hospitalization for unstable angina, or coronary revascularization) 12/5/2020 69
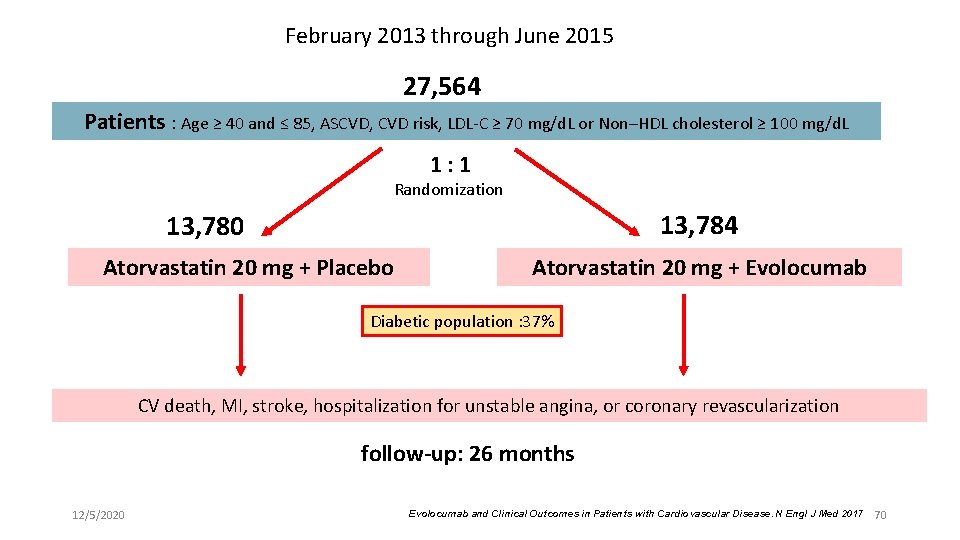
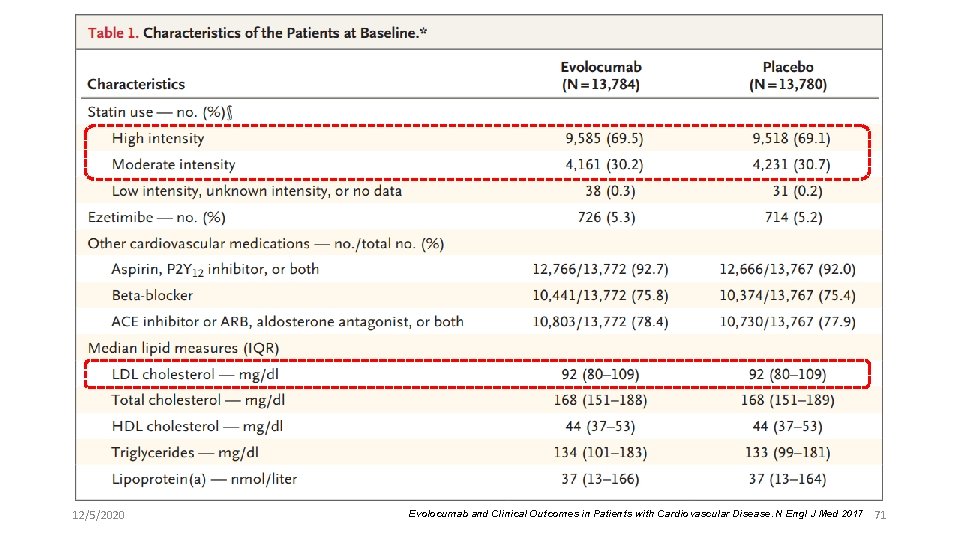
February 2013 through June 2015 27, 564 Patients : Age ≥ 40 and ≤ 85, ASCVD, CVD risk, LDL-C ≥ 70 mg/d. L or Non–HDL cholesterol ≥ 100 mg/d. L 1: 1 Randomization 13, 784 13, 780 Atorvastatin 20 mg + Placebo Atorvastatin 20 mg + Evolocumab Diabetic population : 37% CV death, MI, stroke, hospitalization for unstable angina, or coronary revascularization follow-up: 26 months 12/5/2020 Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med 2017 70
12/5/2020 Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med 2017 71
![At 48 weeks % LDL Reduction = 59 % (95% [CI], 58 to 60; At 48 weeks % LDL Reduction = 59 % (95% [CI], 58 to 60;](http://slidetodoc.com/presentation_image_h/ed5441da5c403f6ec42a497b2457621d/image-72.jpg)
At 48 weeks % LDL Reduction = 59 % (95% [CI], 58 to 60; P<0. 001) Mean absolute reduction = 56 mg/d. L (95% CI, 55 to 57) 12/5/2020 Figure 1. Low-Density Lipoprotein (LDL) Cholesterol Levels over Time. Shown are median values in the two study groups; I bars indicate 95% confidence intervals. Below the graph, the absolute and percentage reductions in LDL cholesterol level in the evolocumab group are compared with those in the placebo group and are presented as least-squares means or means (details are provided in the Methods section in the Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med 2017 72

Results NNT= 68 Primary Outcome Cardiovascular death, MI, stroke, hospitalization for unstable angina, coronary revascularization 1344 (9. 8%) vs. 1563 (11. 3%) [HR 0. 85, 95% CI 0. 79 -0. 92, p<0. 001] Secondary Outcomes Cardiovascular death, MI, stroke : 816 (5. 9%) vs. 1013 (7. 4%) [HR 0. 80, 95% CI 0. 73 -0. 88, p<0. 001] Cardiovascular death : 251 (1. 8%) vs. 240 (1. 7%) [HR 1. 05, 95% CI 0. 88 -1. 25, p=0. 62] All-cause death : 444 (3. 2%) vs. 426 (3. 1%) [HR 1. 04, 95% CI 0. 91 -1. 19, p=0. 54] Myocardial infarction : 468 (3. 4%) vs. 639 (4. 6%) [HR 0. 73, 95% CI 0. 65 -0. 82, p<0. 001] Stroke : 207 (1. 5%) vs. 262 (1. 9%) [HR 0. 79, 95% CI 0. 66 -0. 95, p=0. 01] Coronary revascularization : 759 (5. 5%) vs. 965 (7. 0%) [HR 0. 78, 95% CI 0. 71 -0. 86, p<0. 001] 12/5/2020 Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med 2017 73

Key messages l Inhibition of PCSK 9 with evolocumab on a background of statin therapy lowered LDL cholesterol levels to a median of 30 mg /d. L and reduced the risk of cardiovascular events l These findings show that patients with atherosclerotic cardiovascular disease benefit from the lowering of LDL cholesterol levels below current targets 12/5/2020 Evolocumab and Clinical Outcomes in Patients with Cardiovascular Disease. N Engl J Med 2017 74

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 75
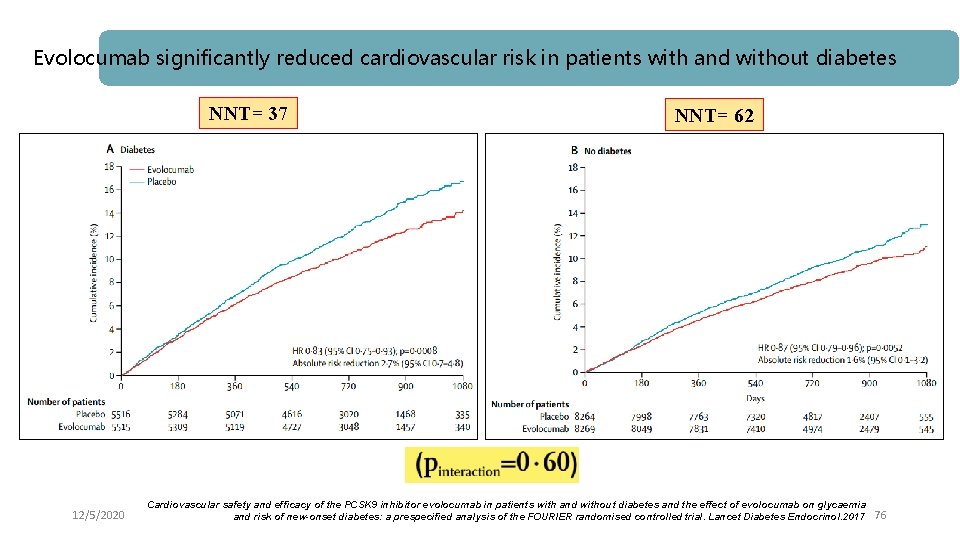
Evolocumab significantly reduced cardiovascular risk in patients with and without diabetes NNT= 37 12/5/2020 NNT= 62 Cardiovascular safety and efficacy of the PCSK 9 inhibitor evolocumab in patients with and without diabetes and the effect of evolocumab on glycaemia and risk of new-onset diabetes: a prespecified analysis of the FOURIER randomised controlled trial. Lancet Diabetes Endocrinol. 2017 76

BACKGROUND Patients who have had an acute coronary syndrome are at high risk for recurrent ischemic cardiovascular events. We sought to determine whether alirocumab, a human monoclonal antibody to proprotein convertase subtilisin–kexin type 9 (PCSK 9), would improve cardiovascular outcomes after an acute coronary syndrome in patients receiving high-intensity statin therapy. METHODS 18, 924 patients who had an acute coronary syndrome 1 to 12 months earlier, LDL cholesterol level of at least 70 mg per deciliter, a non−HDL-C of at least 100 mg per deciliter, or an apolipoprotein B level of at least 80 mg per deciliter, and were receiving statin therapy at a high-intensity dose or at the maximum tolerated dose. Patients were randomly assigned to receive alirocumab subcutaneously at a dose of 75 mg (9462 patients) or matching placebo (9462 patients) every 2 weeks. The dose of alirocumab was adjusted under blinded conditions to target an LDL cholesterol level of 25 to 50 mg per deciliter 12/5/2020 77
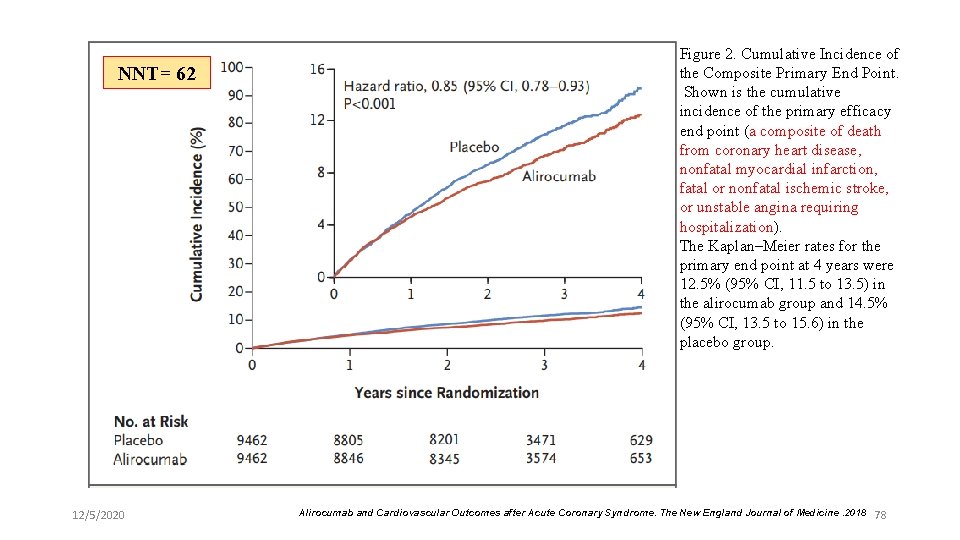
NNT= 62 12/5/2020 Figure 2. Cumulative Incidence of the Composite Primary End Point. Shown is the cumulative incidence of the primary efficacy end point (a composite of death from coronary heart disease, nonfatal myocardial infarction, fatal or nonfatal ischemic stroke, or unstable angina requiring hospitalization). The Kaplan–Meier rates for the primary end point at 4 years were 12. 5% (95% CI, 11. 5 to 13. 5) in the alirocumab group and 14. 5% (95% CI, 13. 5 to 15. 6) in the placebo group. Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. The New England Journal of Medicine. 2018 78

Aims: Pro‐protein convertase subtilisin/kexin type 9 (PCSK 9) inhibitors bring about a wide reduction in low‐density lipoprotein (LDL) cholesterol, greater than that of other lipid‐lowering agents. The aim of this metanalysis was assessment of the effects of PCSK 9 inhibitors on glucose metabolism, LDL cholesterol, cardiovascular morbidity and mortality in individuals with and without diabetes. Materials and methods: A Medline and Clinicaltrials. gov search for eligible studies published before 1 December 2017 was performed (38 Trials). All randomized trials comparing PCSK‐ 9 inhibitors with placebo or active drugs were included. Primary endpoints included (a) incident diabetes, fasting glucose and Hb. A 1 c, (b) LDL cholesterol at endpoint in patients with diabetes and in the total sample, and (c) major cardiovascular events (MACE) and mortality in individuals with and without diabetes. 12/5/2020 79

The reduction of LDL cholesterol versus placebo in patients with diabetes was 52. 6 [41. 3; 63. 8] mg/dl ; the corresponding figure for all patients (with and without diabetes) was 66. 9[62. 4; 71. 3] mg/dl 12/5/2020 PCSK 9 inhibitor therapy: A systematic review and meta-analysis of metabolic and cardiovascular outcomes in patients with diabetes. Diabetes Obes Metab. 2018 80
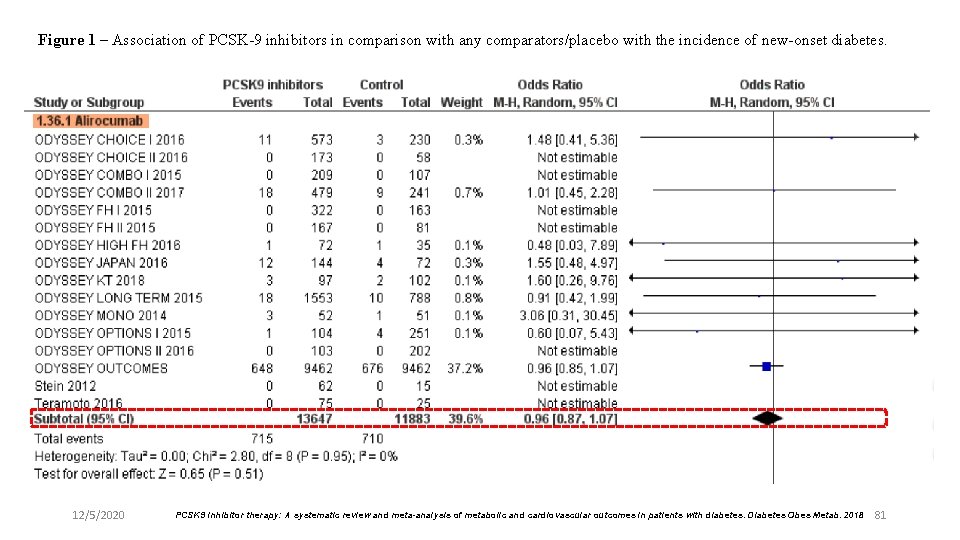
Figure 1 – Association of PCSK-9 inhibitors in comparison with any comparators/placebo with the incidence of new-onset diabetes. 12/5/2020 PCSK 9 inhibitor therapy: A systematic review and meta-analysis of metabolic and cardiovascular outcomes in patients with diabetes. Diabetes Obes Metab. 2018 81
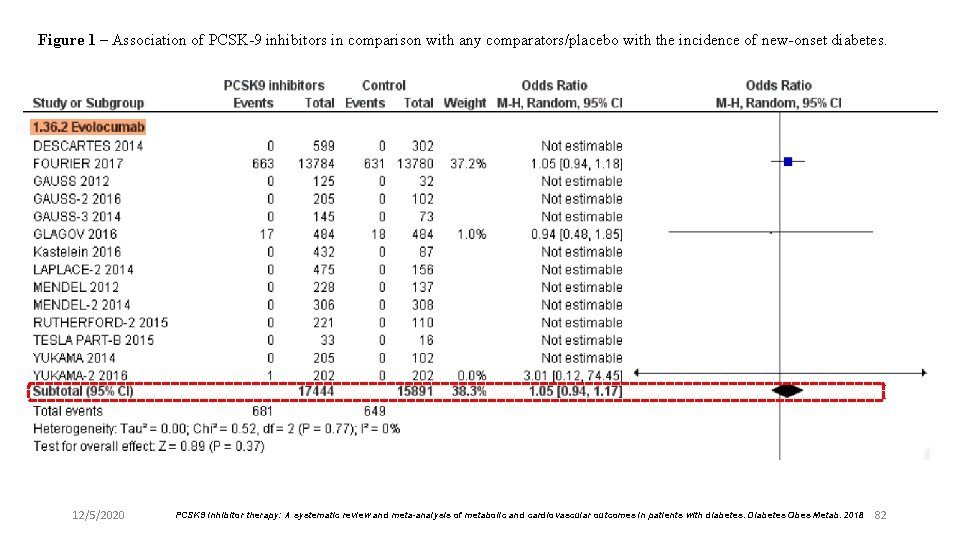
Figure 1 – Association of PCSK-9 inhibitors in comparison with any comparators/placebo with the incidence of new-onset diabetes. 12/5/2020 PCSK 9 inhibitor therapy: A systematic review and meta-analysis of metabolic and cardiovascular outcomes in patients with diabetes. Diabetes Obes Metab. 2018 82
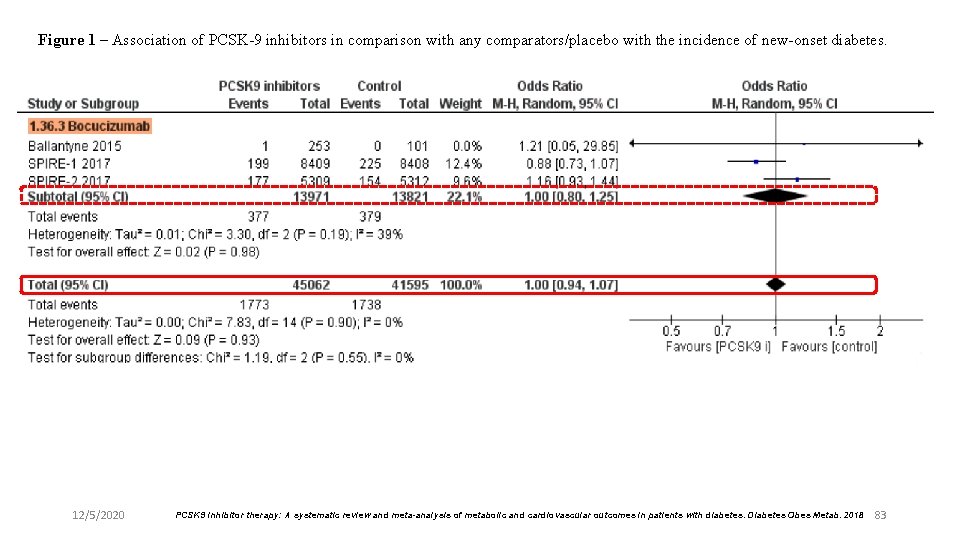
Figure 1 – Association of PCSK-9 inhibitors in comparison with any comparators/placebo with the incidence of new-onset diabetes. 12/5/2020 PCSK 9 inhibitor therapy: A systematic review and meta-analysis of metabolic and cardiovascular outcomes in patients with diabetes. Diabetes Obes Metab. 2018 83
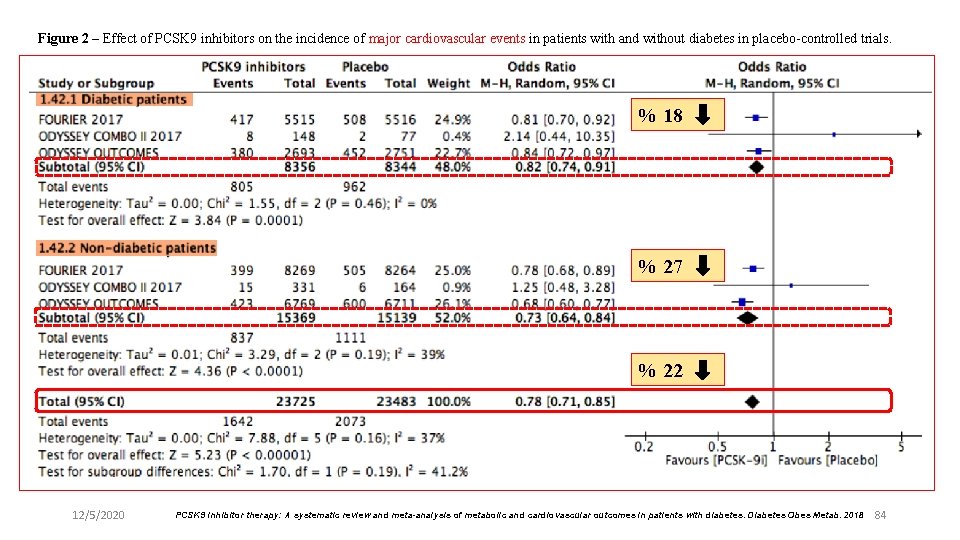
Figure 2 – Effect of PCSK 9 inhibitors on the incidence of major cardiovascular events in patients with and without diabetes in placebo-controlled trials. % 18 % 27 % 22 12/5/2020 PCSK 9 inhibitor therapy: A systematic review and meta-analysis of metabolic and cardiovascular outcomes in patients with diabetes. Diabetes Obes Metab. 2018 84
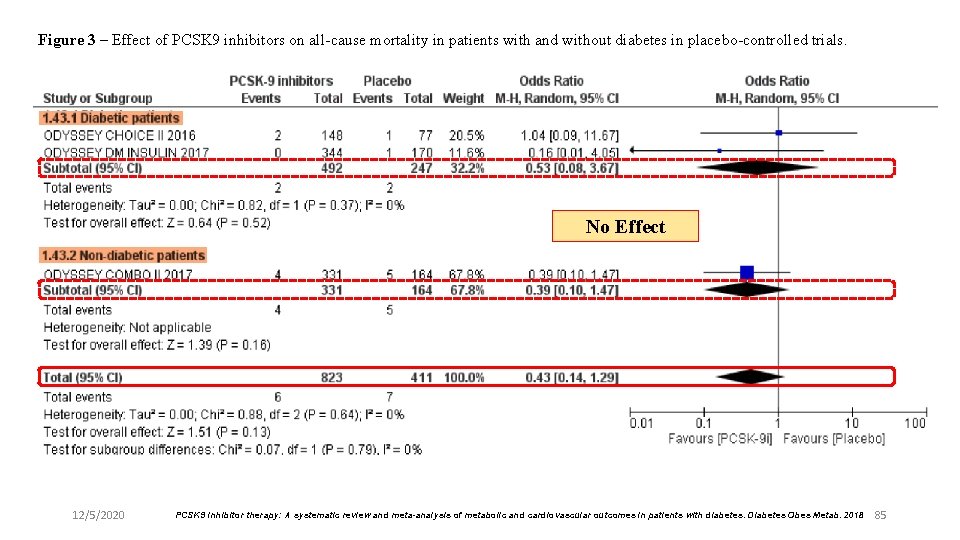
Figure 3 – Effect of PCSK 9 inhibitors on all-cause mortality in patients with and without diabetes in placebo-controlled trials. No Effect 12/5/2020 PCSK 9 inhibitor therapy: A systematic review and meta-analysis of metabolic and cardiovascular outcomes in patients with diabetes. Diabetes Obes Metab. 2018 85

Results l The risk of incident diabetes was not increased by PCSK-9 inhibitors, vs placebo or any comparator. l The reduction in LDL cholesterol vs placebo in patients with diabetes was 52. 6 [41. 3; 63. 8] mg/d. L; the corresponding figure for all patients was 66. 9 [62. 4; 71. 3] mg/d. L. l In studies reporting data on MACE and mortality separately for individuals with and without diabetes, the effect of PCSK-9 did not appear to be affected by diabetes. l Their efficacy on LDL-C and MACE in patients with diabetes does not seem to be very dissimilar from that observed in non-diabetic subjects. 12/5/2020 PCSK 9 inhibitor therapy: A systematic review and meta-analysis of metabolic and cardiovascular outcomes in patients with diabetes. Diabetes Obes Metab. 2018 86

Baseline risk =22% LDL-C =140 mg/dl 140 × % 65=90 mg/dl 90 / 40 =2. 2 × 0. 2=0. 44 22% × 0. 44= % 9. 7 12/5/2020 Figure 3 Expected clinical benefits of low-density lipoprotein cholesterol-lowering therapies. The expected clinical benefits of treatment to lower LDL-C for any person can be estimated; it depends on the intensity of therapy, the baseline LDL-C level, the expected absolute achieved reduction in LDL-C and the baseline estimated risk of atherosclerotic cardiovascular disease. The intensity of therapy should be selected to achieve the recommended proportional reduction in LDL-C based on the person’s estimated risk of atherosclerotic cardiovascular disease. Multiplying the proportional reduction in low-density lipoprotein cholesterol by a person’s baseline low-density lipoprotein cholesterol level estimates the expected absolute reduction in low-density lipoprotein cholesterol that is likely to be achieved with that therapy. Because each 1. 0 mmol/L absolute reduction in low-density lipoprotein cholesterol is associated with a 20% reduction in the risk of cardiovascular events, larger absolute reductions in lowdensity lipoprotein cholesterol lead to larger proportional reductions in risk. Multiplying the proportional reduction in risk expected for the achieved absolute reduction in low-density lipoprotein cholesterol by a person’s estimated baseline risk of atherosclerotic cardiovascular disease determines the expected absolute risk reduction for that person. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 87

ADA & AHA Guidelines 2019 ESC/EAS 2019 12/5/2020 88

Lipid Management Lifestyle Intervention Recommandation 10. 15 Lifestyle modification focusing on weight loss (if indicated); application of a Mediterranean diet or Dietary Approaches to Stop Hypertension (DASH) dietary pattern; reduction of saturated fat and trans fat; increase of dietary n-3 fatty acids, viscous fiber, and plant stanols/sterols intake; and increased physical activity should be recommended to improve the lipid profile and reduce the risk of developing atherosclerotic cardiovascular disease in patients with diabetes. A 10. 16 Intensify lifestyle therapy and optimize glycemic control for patients with elevated triglyceride levels (≥ 150 mg/d. L and/or low HDL cholesterol (<40 mg/d. L for men, <50 mg/d. L] for women). C 10. 18 Obtain a lipid profile at initiation of statins or other lipid-lowering therapy, 4– 12 weeks after initiation or a change in dose, and annually thereafter as it may help to monitor the response to therapy and inform medication adherence. E 12/5/2020 Cardiovascular Disease and Risk Management. Diabetes Care. 2019 Jan; 42(Suppl 1): S 103 -S 123. 89

12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 90

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 91

ASCVD Risk Estimator Plus www. cvriskcalculator. com 12/5/2020 Cardiovascular Disease and Risk Management. Diabetes Care. 2019 Jan; 42(Suppl 1): S 103 -S 123. 92

§ The Risk Calculator. § The American College of Cardiology/American Heart Association § ASCVD risk calculator may be a useful tool to estimate 10 -year ASCVD (http: //my . americanheart. org). 12/5/2020 93

Table 10. 2: Recommendations for statin and combination treatment in adults with diabetes Age <40 years ≥ 40 years ASCVD or 10 -year Recommended statin intensity^ and combination treatment* ASCVD risk >20% No None† Yes High • In patients with ASCVD, if LDL cholesterol ≥ 70 mg/d. L despite maximally tolerated statin dose, consider adding additional LDL-lowering therapy (such as ezetimibe or PCSK 9 inhibitor)# No Moderate‡ Yes High • In patients with ASCVD, if LDL cholesterol ≥ 70 mg/d. L despite maximally tolerated statin dose, consider adding additional LDL-lowering therapy (such as ezetimibe or PCSK 9 inhibitor) ASCVD, atherosclerotic cardiovascular disease; PCSK 9, proprotein convertase subtilisin/kexin type 9. * In addition to lifestyle therapy. ^ For patients who do not tolerate the intended intensity of statin, the maximally tolerated statin dose should be used. † Moderate-intensity statin may be considered based on risk-benefit profile and presence of ASCVD risk factors include LDL cholesterol ≥ 100 mg/d. L , high blood pressure, smoking, CKD, albuminuria, and family history of premature ASCVD. ‡ High-intensity statin may be considered based on risk-benefit profile and presence of ASCVD risk factors. #Adults aged <40 years with prevalent ASCVD were not well represented in clinical trials of non-statin–based LDL reduction. Before initiating combination lipid-lowering therapy, consider the potential for further ASCVD risk reduction, drug-specific adverse effects, and patient preferences. 12/5/2020 Cardiovascular Disease and Risk Management. Diabetes Care. 2019 Jan; 42(Suppl 1): S 103 -S 123. 94
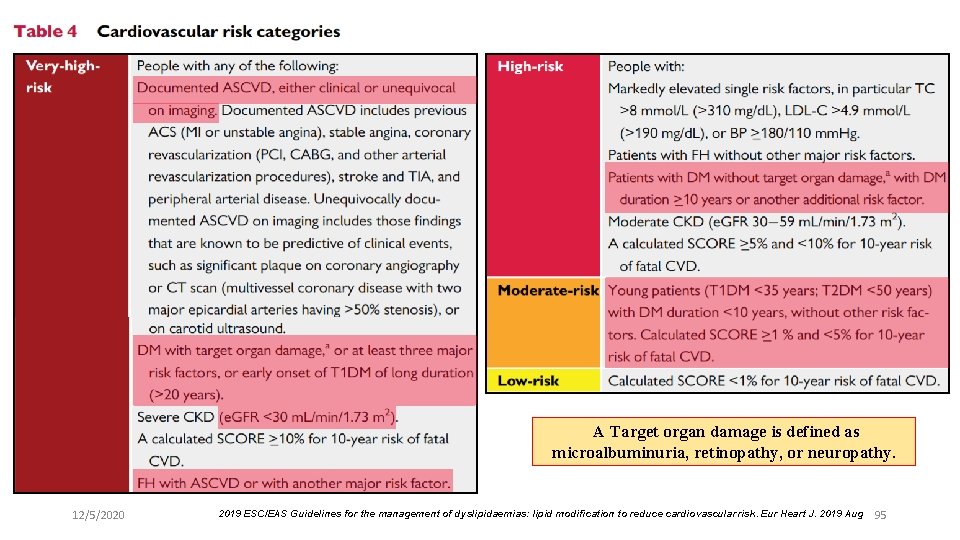
A Target organ damage is defined as microalbuminuria, retinopathy, or neuropathy. 12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 95

Table 5. Diabetes-Specific Risk Enhancers That Are Independent of Other Risk Factors in Diabetes Mellitus. ADA 2019 ASCVD risk factors include LDL cholesterol ≥ 100 , high blood pressure, smoking and family history of premature ASCVD ABI indicates ankle-brachial index; and e. GFR, estimated glomerular filtration rate. 12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 96

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 97
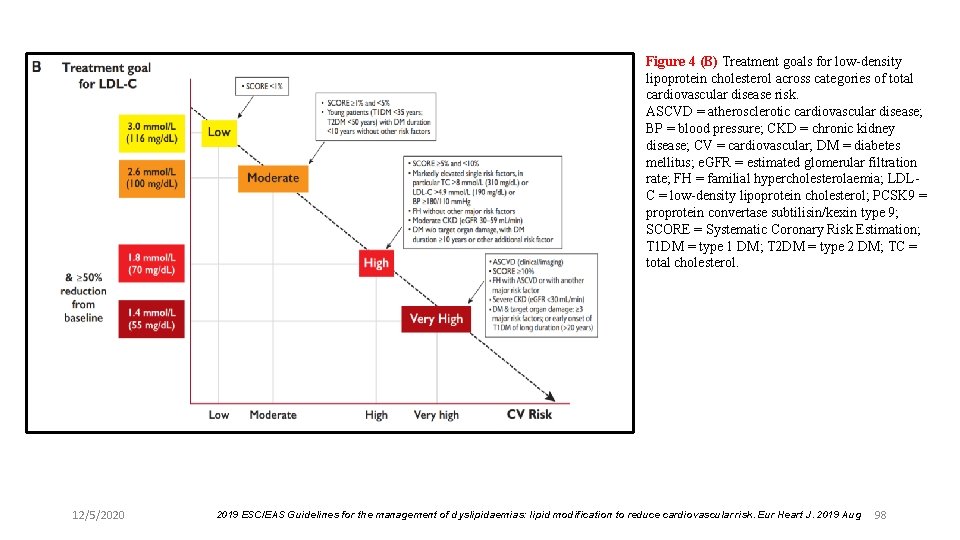
Figure 4 (B) Treatment goals for low-density lipoprotein cholesterol across categories of total cardiovascular disease risk. ASCVD = atherosclerotic cardiovascular disease; BP = blood pressure; CKD = chronic kidney disease; CV = cardiovascular; DM = diabetes mellitus; e. GFR = estimated glomerular filtration rate; FH = familial hypercholesterolaemia; LDLC = low-density lipoprotein cholesterol; PCSK 9 = proprotein convertase subtilisin/kexin type 9; SCORE = Systematic Coronary Risk Estimation; T 1 DM = type 1 DM; T 2 DM = type 2 DM; TC = total cholesterol. 12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 98
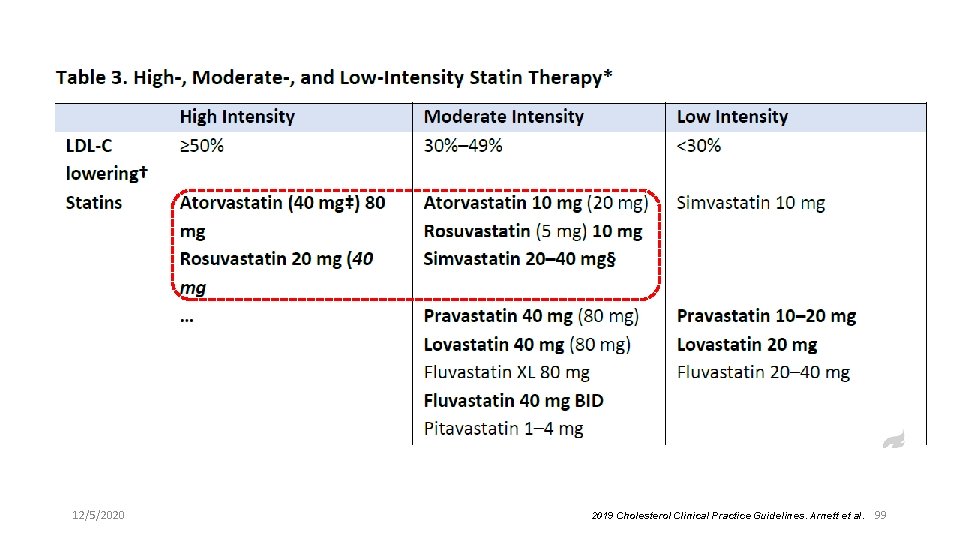
12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 99

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 100

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 101
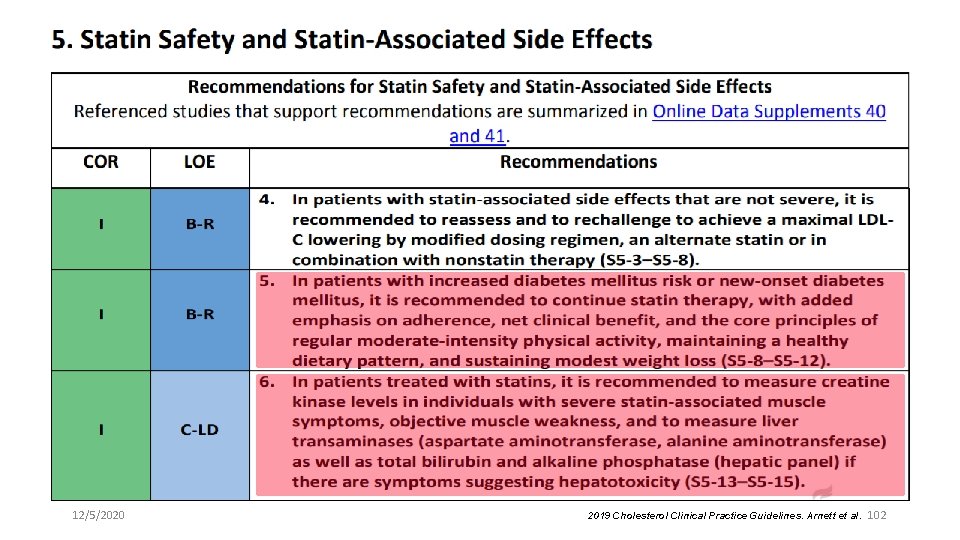
12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 102

12/5/2020 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2019 Aug 103

Returning to the case • The present case patient is at very high risk of recurrent ASCVD owing to a background of T 2 DM, HTN, CKD, and premature ASCVD. • In considering therapeutic strategies to reduce his residual risk, recent data from the IMPROVE-IT (ezetimibe) and FOURIER and ODYSSEY Outcomes (proprotein convertase subtilisin/kexin type 9 inhibitor) trials have shown further reductions in ASCVD of between 6% and 15% to 20%, respectively after co-administration with statins (Lowering LDL-C to less than 55 mg/dl using PCSK 9 inhibitors, COST!!) • Assessing secondary causes of hypertriglyceridemia , more adherence on diet and physical activity. 12/5/2020 104

Returning to the case • Although fibrates reduce TG levels, they were associated with inconclusive effects when added to statins in the ACCORD-Lipid trial. • In patients with TG levels of 200 to 499 mg/dl, OM 3 FA have been shown to improve dyslipidemia and inflammatory biomarkers. With the recent data on the potential pleotropic effects of OM 3 FA on plaque stabilization, REDUCE-IT: 2 g of icosapent ethyl twice daily 12/5/2020 105

Take Home Message l Statin therapy is recommended as the first-line lipid-lowering drug therapy for the management of dyslipidemia in individuals with diabetes mellitus, reinforcing patient compliance and using appropriate dosage according to presence or absence of CVD , traditional risk factors and diabetes-specific risk enhancers l In major statin trials, significant residual cardiovascular risk remains even after acceptable reduction of LDL-C, especially in patients with diabetes 12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 106

Take Home Message l Non statin therapy including Ezetimibe and PCSK 9 inhibitors have some beneficial effects to address the residual risk l Based on recent outcome based trials, PCSK 9 inhibitors provide significant LDL-C lowering in individuals with hyperlipidemia and diabetes mellitus on top of maximally tolerated statin therapy and can prevent or reduce further cardiovascular events l The results of REDUCE-IT trial have introduced a new tool to address the residual risk 12/5/2020 2019 Cholesterol Clinical Practice Guidelines. Arnett et al. 107

Thanks for your patience, dear colleagues! 12/5/2020 108
- Slides: 108