Approach to Acid Base Jeff Kaufhold MD FACP

Approach to Acid Base Jeff Kaufhold, MD FACP 2004 Source: The ICU Book Chapter 36 -38

Acid Base Disorders n n n Basic Physiology Approach to A-B disorders Equations Cases Electrolytes

Acid Base Physiology n n n Balance based on Henderson. Hasselbach equation Hydrogen Ion measured in Nano. Equivalents , i. e. very tightly regulated. Buffers for acid: • Bicarbonate, Hemoglobin, Albumin, Bone n Acid excretion: Lungs and Kidneys

Acid - Base Physiology n Sources of Acid: • 70 m. Eq daily as inorganic acids H 2 SO 4, H 3 PO 4 n Excreted by kidney n • 16, 000 m. Mole as CO 2 n Excreted by lungs • 32, 000 m. Eq organic acid which gets metabolised

Acid – Base Physiology n Lung blows off acid as CO 2 • H 2 CO 3 H 2 O + CO 2 n Kidney • Proximal tubule has lumenal carbonic anhydrase to reclaim bicarb from urine • Distal tubule has basolateral CA to pull H+ out of blood, complexed to NH 3 to form NH 4 Cl (ammonium chloride) • Can measure activity by urinary Anion Gap (Na+K – Cl)

ABG reporting n Normals: • p. H 7. 36 – 7. 44 • p. CO 2 36 – 44 mm. Hg • p. O 2 over 60 • HCO 3 (bicarb) 22 – 26 n Reported as p. H / p. CO 2 / p. O 2 / HCO 3 • Example 7. 40 / 80 / 23

Compensatory Mechanisms n Disorder • Resp Acidosis • Resp Alkalosis • Met Acidosis • Met Alkalosis n End point is a constant ratio of: n Primary / Compens • Pco 2 up HCO 3 up • Pco 2 down, HCO down • HCO down, Pco 2 down • HCO up Pco 2 up • PCO 2 / HCO 3

Rules for interpretation n 1. Primary Metabolic disorders: • PCO 2 and p. H move in same direction • Can be quickly identified if last 2 digits of p. H = p. CO 2 n n n i. e 7. 20 / 70 7. 51 / 50 / 120 If 7. 20 / 40 / 70, not simple disorder • Respiratory compensation not adequate

Rules for interpretation n 3. Primary Respiratory disorders: • PCO 2 and p. H move in opposite direction n Resp acidosis: 7. 20 / 58 / 68 n Resp alkalosis: 7. 50 / 25/ 80 7. 25 / 48 / 52 Not a simple disorder n Neither is 7. 40 / 25 / 68 n

Rules for interpretation n Rule 5 Mixed Disorders: • Compensatory responses do not completely correct primary problem • Can have BOTH metabolic acidosis and metabolic alkalosis simultaneously (sepsis plus vomiting is common) • But can only hyperventilate (resp alk) OR hypoventilate (resp acidosis). • Usually due to failure to adequately lower p. CO 2 (resp fatigue)

Expected Changes Rules of Thumb n Met Acidosis • Pco 2 = 1. 5 * bicarb + 8 +/- 2 • 7. 20 / 68 / HCO 3 = 8 n Met Alkalosis • Pco 2 = 0. 7 * HCO 3 + 21 +/- 2 • 7. 50 / 120 / HCO 3 = 40

Expected Changes n Acute Resp Acidosis • Change in p. H = 0. 008 * change p. CO 2 • 7. 30 / 50 / 68 / HCO 3 = 22 n Acute Resp Alkalosis • Change in p. H = 0. 008 * change p. CO 2 • 7. 50 / 20 / 120 / HCO 3 = 20 n Shortcut: D p. H = 0. 08 for each 10 mm. Hg change in p. CO 2

Expected Changes n Chronic Resp Acidosis • Change in p. H = 0. 003 * change p. CO 2 • 7. 33 / 50 / 68 / HCO 3 = 32 n Chronic Resp Alkalosis • Change in p. H = 0. 0017 * change p. CO 2 • 7. 48 / 25 / 120 / HCO 3 = 20 n Note handout wrong chronic resp alkalosis use 0. 0017, not 0. 017.

Expected Changes n Note that the chronic respiratory conditions induce chronic changes in bicarbonate handling by the kidney, which takes time.

Common Causes of A-B Disorders n n Met Acidosis MUD PILES Met Alkalosis Vomiting, NG suction, IV infusion of bicarb or Acetate (TPN), Diuretics Resp Acidosis : respiratory failure, COPD exacerbation, Narcotics Resp Alkalosis: Hyperventilation/anxiety, Pregnancy, Early sepsis, early stage asthma

Anion Gap n n n Useful for evaluation of metabolic acidosis. Calculate AG every time you see electrolytes, esp on exams. AG = Anions – Cations • • AG = Na + K – (CL + HCO 3) AG = 140 + 4 – (102 + 25) = 8 to 16 > 18 nephrologist gets excited

Anion Gap n n Low AG suggests extra plasma proteins, such as Myeloma Non Anion Gap acidosis suggests loss of bicarbonate through either: • Diarrhea • Renal Tubular Acidosis (RTA) • Or Infusion of acid via NS, TPN n High AG means accumulation of acid • MUDD PILES or other nemonic

Anion Gap n High AG means accumulation of acid • Organic acid can be metabolised back to bicarbonate • So AG over 16 or so can represent “potential Bicarb” or “delta gap”. • Calculating the potential bicarb is useful for identifying mixed disorders, since both met acidosis and met alkalosis can be present at same time. • Delta Gap = AG – 12 (extra anions) • Potential bicarb = HCO 3 + delta gap
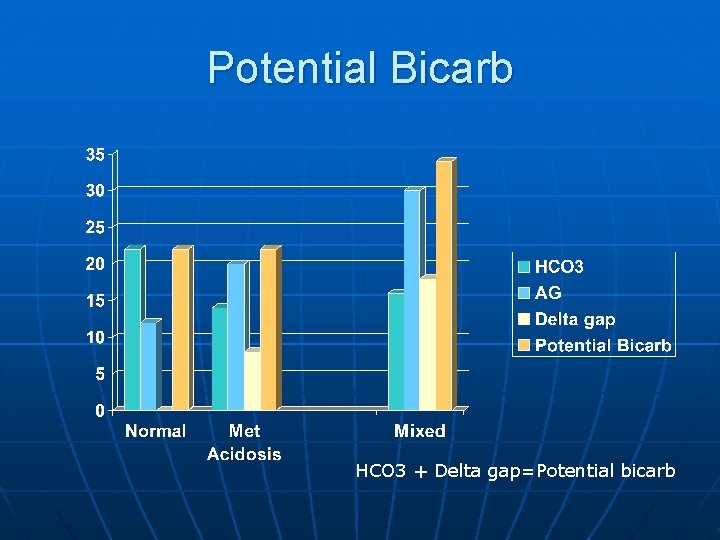
Potential Bicarb HCO 3 + Delta gap=Potential bicarb

Gapped Metabolic Acidosis n n n n n Methanol (wood alcohol) Uremia/acute renal failure DKA D lactate (Metformin/blind loop in bowel) Paraldehyde Isopropyl alcohol Lactic acidosis (hypoxia/hypoperfusion) Ethanol Starvation Ketosis

Osmolal Gap n n Used to help identify presence of exogenous acids which may be causing gapped metabolic acidosis Measured Osm – calculated Osm calc = 2(Na) + Glucose/18 + BUN/30 OG > 20 suggests Ethanol, Methanol

Clinical Problems in A-B n n n 1. Calculate AG and potential bicarb 2. Is p. H acidic or alkalotic 3. Is p. CO 2 alkalotic or acidotic • n > 40 4. Is the bicarb measured by the ABG machine same as that on lytes? n n < 40 If not the samples are not simultaneous 5. Apply the rules of thumb n n If values are consistent, Simple disorder If values are not consistent: Mixed.

Case 1 n n 38 y. o. male with chronic GN. Admitted with weakness, fatigue. Labs: Na 134, K 5. 6, Cl 100, CO 2 14. ABG: 7. 26 / 27 / 72 / 14 What is A-B disturbance? • • • AG = Delta gap/Potential bicarb = Acidosis or alkalosis ? Direction of CO 2 ? MA rule: p. CO 2 = 1. 5*HCO 3 + 8 +/-2

Case 1 n n n Labs: Na 134, K 5. 6, Cl 100, CO 2 14. ABG: 7. 26 / 27 / 72 / 14 What is A-B disturbance? • • • AG = 20 Delta gap/Potential bicarb = 8 / 22 Acidosis or alkalosis ? acidosis Direction of CO 2 ? alkalosis MA rule: p. CO 2 = 1. 5*HCO 3 + 8 +/-2 works So simple metabolic acidosis with appropriate respiratory compensation

Case 2 n n 23 y. o. garage mechanic presents with acute confusion after drinking antifreeze. Lab: Na 137 K 5. 4 Cl 95 HCO 5 ABG: 6. 95 / 15 / 80 / 4 What is A-B disturbance? • • • AG = Delta gap/Potential bicarb = Acidosis or alkalosis ? Direction of CO 2 ? MA rule: p. CO 2 = 1. 5*HCO 3 + 8 +/-2

Case 2 n n n Lab: Na 137 K 5. 4 Cl 105 HCO 5 ABG: 6. 95 / 15 / 80 / 4 What is A-B disturbance? • • • AG = 27 Delta gap/Potential bicarb = 15 / 20 Acidosis or alkalosis ? acidosis Direction of CO 2 ? alkalosis MA rule: p. CO 2 = 1. 5*HCO 3 + 8 +/-2 works so simple met acidosis with appropriate resp compensation.

Case 3 n n 65 y. o. recently discharged after pneumonia. Presents with C dif Diarrhea. Lab: Na 132 K 2. 4 Cl 105 HCO 15 ABG: 7. 30 / 80 / 14 What is A-B disturbance? • • • AG = Delta gap/Potential bicarb = Acidosis or alkalosis ? Direction of CO 2 ? MA rule: p. CO 2 = 1. 5*HCO 3 + 8 +/-2

Case 3 n n n Lab: Na 132 K 2. 4 Cl 105 HCO 15 ABG: 7. 30 / 80 / 14 What is A-B disturbance? • AG = 12 • Delta gap/Potential bicarb = 0 / 15 • Acidosis • Direction of CO 2 ? alkalotic • MA rule: p. CO 2 = 1. 5*HCO 3 + 8 +/-2 n Works so Non Gap Metabolic Acidosis

Case 4 n n 40 y. o. with status asthmaticus. Lab: Na 135 K 3. 4 Cl 100 HCO 21 ABG: 7. 50 / 30 / 80 / 20 What is A-B disturbance? • AG = • Delta gap/Potential bicarb = • Acidosis or alkalosis ? • Direction of CO 2 ? • Acute RA rule: Dp. H = 0. 008 * Dp. CO 2

Case 4 n n 40 y. o. with status asthmaticus. Lab: Na 135 K 3. 4 Cl 100 HCO 21 ABG: 7. 50 / 30 / 80 / 20 What is A-B disturbance? • • • AG = 14 Delta gap/Potential bicarb = alkalosis Direction of CO 2 ? alkalosis Acute RA rule: Dp. H = 0. 008 * Dp. CO 2 n works

Case 5 n n 32 y. o. chronic pyelo, CRF, pleural effusion. Admitted for thoracentesis Lab: Na 130 K 5. 0 Cl 94 HCO 15 ABG: 7. 32 / 55 / 15 Attempted thoracentesis unsuccessful, became dyspneic and coded as blood drawn: • Na 131/k 7. 8/cl 92/CO 9/ 6. 9/50/40

Case 5 CRF with pneumothorax n Admission AG = 20 • Appropriate resp compensation • Uremic metabolic acidosis n After thoracentesis: • Resp acidosis, plus met acidosis • Met acidosis worse, gap up to 30. • Due to uremia plus lactic acidosis.

Case 6 n n 44 y. o. alcoholic admitted with abd pain and N/V/Fever/chills. SOB and CXR shows pneumonia with effusion. Lab: Na 142 K 3. 4 Cl 98 HCO 20 ABG: 7. 28 / 41 / 58 / 20 What is A-B disturbance? • • AG = Delta gap/Potential bicarb = Acidosis or alkalosis ? Direction of CO 2 ?

Case 6 n n 44 y. o. alcoholic admitted with abd pain and N/V/Fever/chills. SOB and CXR shows pneumonia with effusion. Lab: Na 142 K 3. 4 Cl 98 HCO 20 ABG: 7. 28 / 41 / 58 / 20 What is A-B disturbance? • • • AG = 24 Delta gap/Potential bicarb = 12 / 32 Acidosis or alkalosis ? Acidosis Direction of CO 2 ? Not appropriate What is going on?
- Slides: 34