Applications of Aqueous Equilibria HH Buffered Solutions q

Applications of Aqueous Equilibria HH

Buffered Solutions q A solution that resists a change in p. H when either hydroxide ions or protons are added. q Buffered solutions contain either: Ø A weak acid and its salt Ø A weak base and its salt
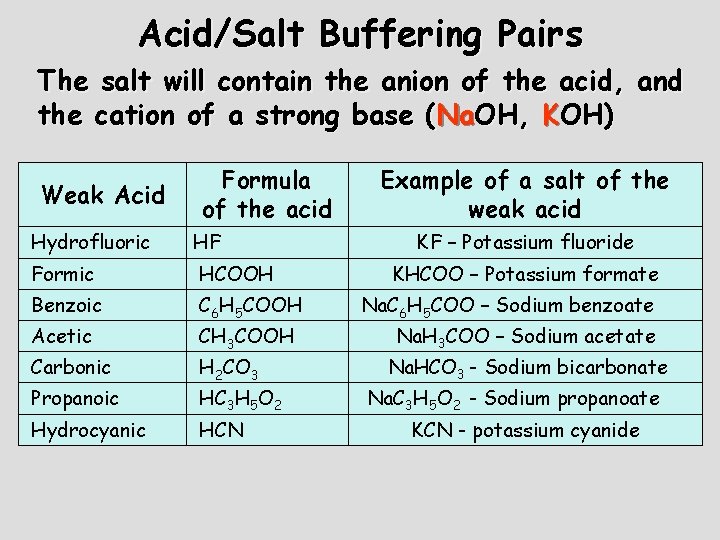
Acid/Salt Buffering Pairs The salt will contain the anion of the acid, and the cation of a strong base (Na. OH, KOH) Weak Acid Formula of the acid Hydrofluoric HF Formic HCOOH Benzoic C 6 H 5 COOH Acetic Carbonic Propanoic Hydrocyanic CH 3 COOH H 2 CO 3 HC 3 H 5 O 2 HCN Example of a salt of the weak acid KF – Potassium fluoride KHCOO – Potassium formate Na. C 6 H 5 COO – Sodium benzoate Na. H 3 COO – Sodium acetate Na. HCO 3 - Sodium bicarbonate Na. C 3 H 5 O 2 - Sodium propanoate KCN - potassium cyanide

Base/Salt Buffering Pairs The salt will contain the cation of the base, and the anion of a strong acid (HCl, HNO 3) Formula of the base Example of a salt of the weak acid NH 3 NH 4 Cl - ammonium chloride Methylamine CH 3 NH 2 CH 3 NH 3 Cl – methylammonium chloride Ethylamine C 2 H 5 NH 2 C 2 H 5 NH 3 NO 3 - ethylammonium nitrate Aniline C 6 H 5 NH 2 C 6 H 5 NH 3 Cl – aniline hydrochloride Base Ammonia Pyridine C 5 H 5 NHCl – pyridine hydrochloride
![Calculate the [H+] in a solution that is 0. 10 M in Na. F Calculate the [H+] in a solution that is 0. 10 M in Na. F](http://slidetodoc.com/presentation_image_h2/5679832b65c41d966eeab2409d7bef51/image-5.jpg)
Calculate the [H+] in a solution that is 0. 10 M in Na. F and 0. 20 M in HF. (Ka = 7. 2 10 -4) 7. 2 E-4 M 2. 0 M 1. 4 E-3 M 0. 20 M none of these
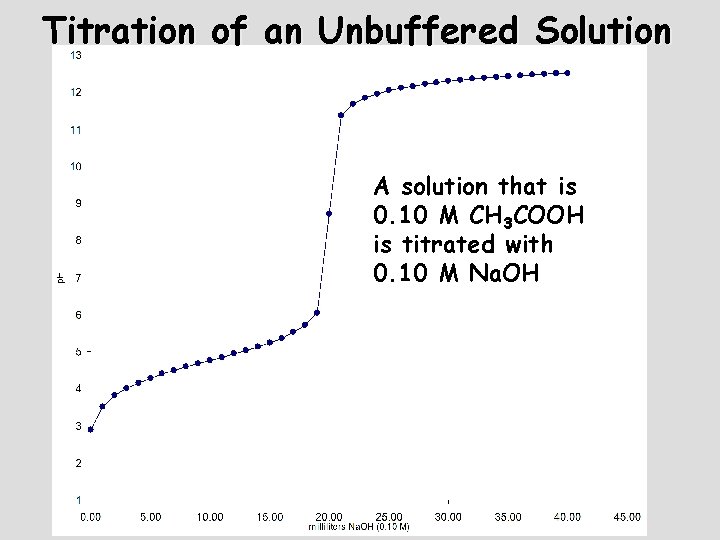
Titration of an Unbuffered Solution A solution that is 0. 10 M CH 3 COOH is titrated with 0. 10 M Na. OH
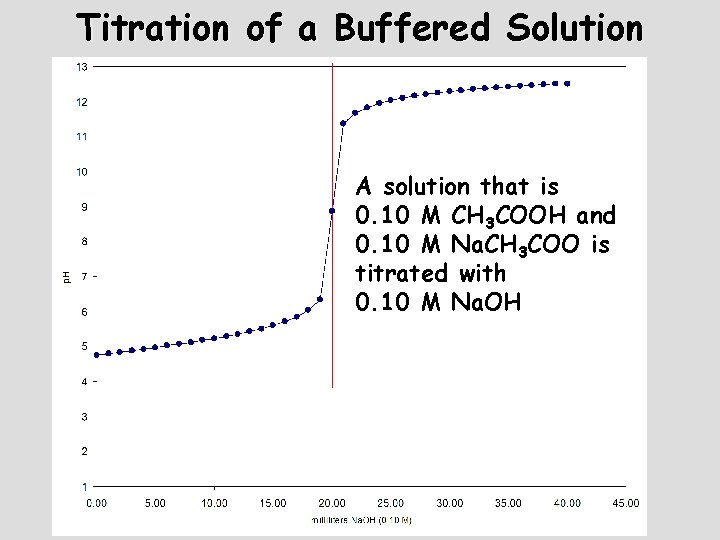
Titration of a Buffered Solution A solution that is 0. 10 M CH 3 COOH and 0. 10 M Na. CH 3 COO is titrated with 0. 10 M Na. OH
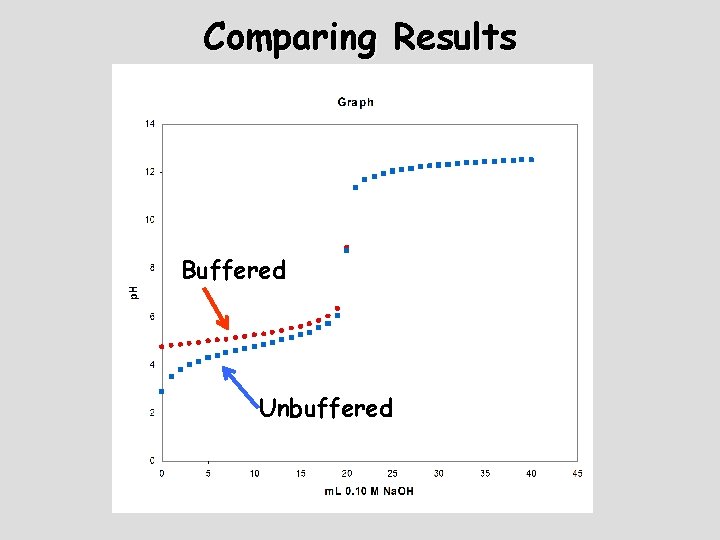
Comparing Results Buffered Unbuffered
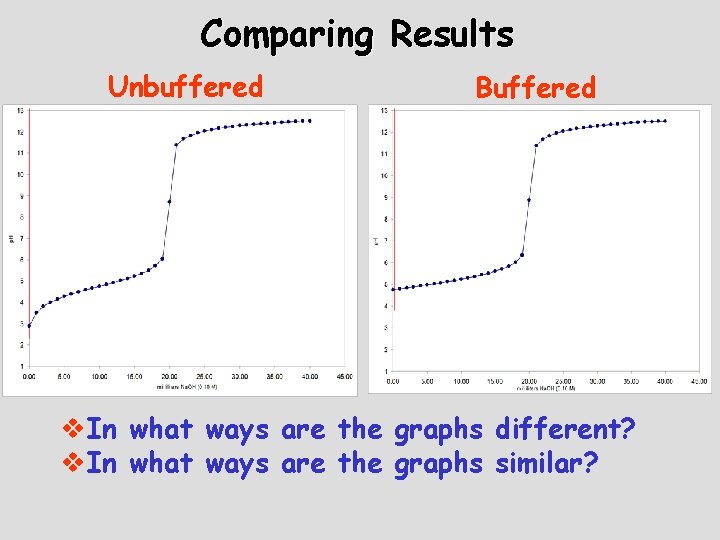
Comparing Results Unbuffered Buffered v. In what ways are the graphs different? v. In what ways are the graphs similar?
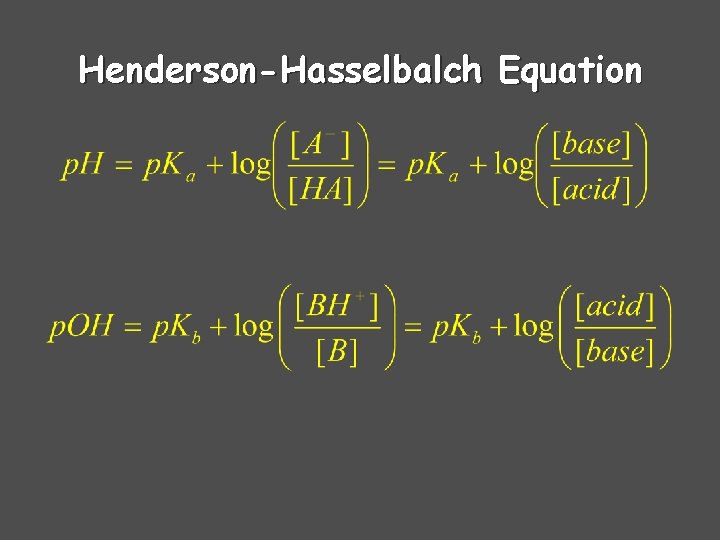
Henderson-Hasselbalch Equation
![Calculate the [H+] in a solution that is 0. 10 M in Na. F Calculate the [H+] in a solution that is 0. 10 M in Na. F](http://slidetodoc.com/presentation_image_h2/5679832b65c41d966eeab2409d7bef51/image-11.jpg)
Calculate the [H+] in a solution that is 0. 10 M in Na. F and 0. 20 M in HF. (Ka = 7. 2 10 -4) 7. 2 E-4 M 2. 0 M 1. 4 E-3 M 0. 20 M none of these
- Slides: 11