Application of the colligative properties Determination of Molecular









- Slides: 10

Application of the colligative properties Determination of Molecular Masses (Session 22) Ciencias de la Tierra II

Colligative Properties • With the colligative properties of the solutions we can determine the molar masses or molecular of solutes dissolved in them. • This is an important application when we want to identify a unknown solute because from the molecular mass and percentage composition can determine the chemical formula of a solute. Ciencias de la Tierra II

• The colligative properties decrease od the freezing point and elevation of the boiling point are used to determine the molar or molecular masses with the methods know as: • Cryoscopy: decrease of the freezing point • Ebullioscopy: increase of the boiling point Ciencias de la Tierra II

Decrease of the freezing point: Cryoscopy Δ Tc = Kc * m Where: Δ Tc= Kc * solute grams /PM solvent kilograms PM = Kc * solute grams Δ Tc * solvent grams Ciencias de la Tierra II

Elevation of the boiling point: Ebullioscopy Δ T e = Ke * m Where: Δ Te= Ke * solute grams/PM solvent kilograms PM = Ke * solute grams Δ Te * solvent kilograms Ciencias de la Tierra II

Problem #1 When in 150 g of water 0. 946 g of fructose (sugar of fruits) are dissolved, we can observe in the resulting solution has a freezing point of– 0. 0651 0 C. What is the molecular mass of the fructose? Ciencias de la Tierra II

Problem #2 A solution that contains 22. 0 g of ascorbic acid (vitamin C) in 100 g of water freezes at -2. 33 0 C. Which is molecular mass of ascorbic acid? Ciencias de la Tierra II

Problem #3 A solution has 22. 0 g ascorbic acid (vitamin C) in 100 g of water it freezes at -2. 33 0 C. What is the molecular mass of ascorbic acid? Ciencias de la Tierra II

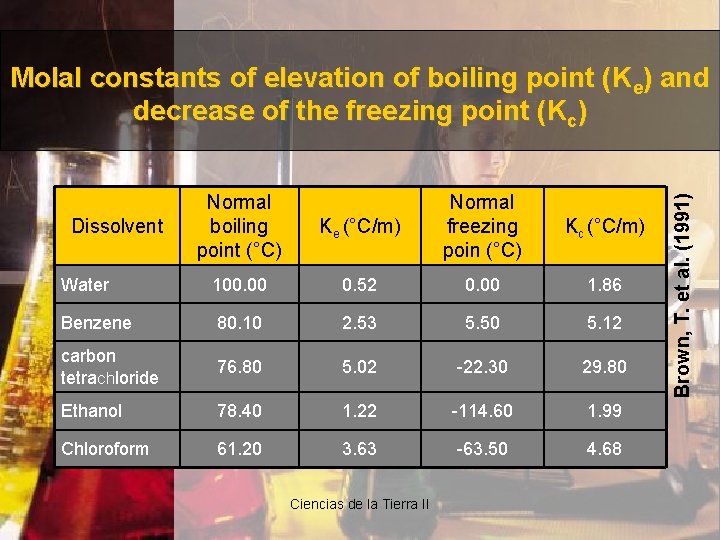
Normal boiling point (°C) Ke (°C/m) Normal freezing poin (°C) Kc (°C/m) Water 100. 00 0. 52 0. 00 1. 86 Benzene 80. 10 2. 53 5. 50 5. 12 carbon tetrachloride 76. 80 5. 02 -22. 30 29. 80 Ethanol 78. 40 1. 22 -114. 60 1. 99 Chloroform 61. 20 3. 63 -63. 50 4. 68 Dissolvent Ciencias de la Tierra II Brown, T. et al. (1991) Molal constants of elevation of boiling point (Ke) and decrease of the freezing point (Kc)

Bibliography • Brown, T. et al. (1991) Chemistry: The Central Science. 5 th ed. United States of America: Prentice Hall. • Burns, R. (1996) Chemistry Fundaments. 2 nd. ed. United States of America: Prentice Hall. • Chang, R. (1992). Chemistry. 4 th ed. United States of America: Mc. Graw Hill. Ciencias de la Tierra II