Application of Electrolytic Cells Lesson 11 Electrolysis describes

Application of Electrolytic Cells Lesson 11


Electrolysis describes what happens in an electrolytic cell and means to use electricity to make chemicals. Many elements are made by electrolysis Pb I 2 Al O 2 Zn Na Pb 2+ 2 Cl- + 2 e- → Pb(s) → Cl 2(g) + 2 e- K Li H 2 Cl 2 F 2 This is sometimes called electrowinning- the element is won from its ion

Making Aluminum by Electrolysis Alcan (70, 000 employees in 55 countries) Kitimat B. C. 2. 7 MT 7 % world production

Aluminum

Aluminum Production by Electrolysis Name of the Ore imported from (Guinea and Brazil) Bauxite Al 2 O 3. 3 H 2 O Heating drives off the water Al 2 O 3. 3 H 2 O + Heat → Al 2 O 3 + Melting point of Bauxite is 2045 0 C This is too hot! Cryolite is added Lowers the melting point to 1000 0 C 3 H 2 O

Reduction of water You cannot reduce Aluminum in water! It must be molten!
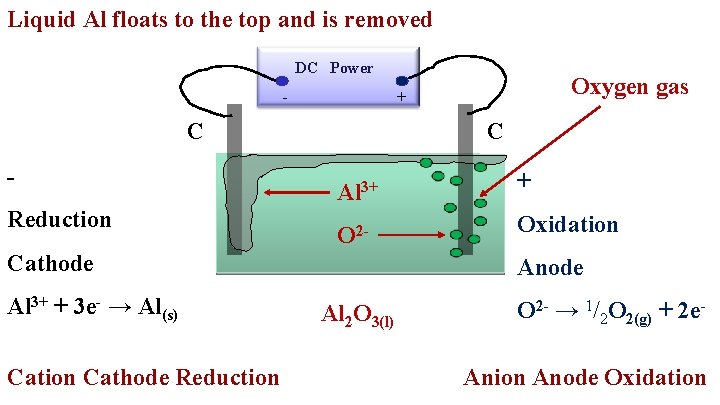
Liquid Al floats to the top and is removed DC - Power C Reduction C Al 3+ + O 2 - Oxidation Cathode Al 3+ + 3 e- → Al(s) Cation Cathode Reduction Oxygen gas + Anode Al 2 O 3(l) O 2 - → 1/2 O 2(g) + 2 e. Anion Anode Oxidation
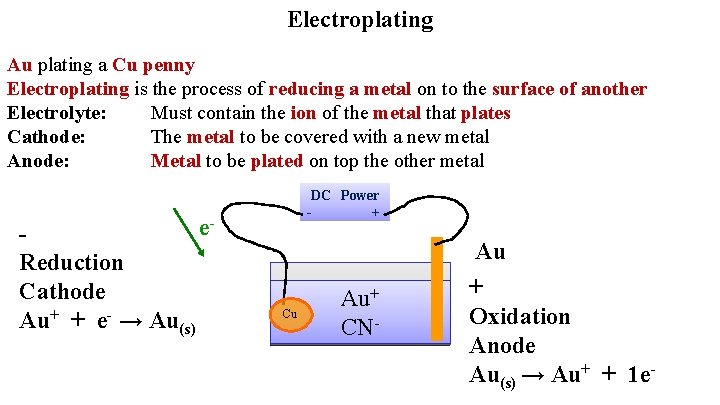
Electroplating Au plating a Cu penny Electroplating is the process of reducing a metal on to the surface of another Electrolyte: Must contain the ion of the metal that plates Cathode: The metal to be covered with a new metal Anode: Metal to be plated on top the other metal DC Power Reduction Cathode Au+ + e- → Au(s) - e- Cu + Au+ CN- Au + Oxidation Anode Au(s) → Au+ + 1 e-

+ve stainless steel or Au Au. CN -ve



Au plated

Copper Ring Gold Plated

Tanya’s Ring Gold Plated
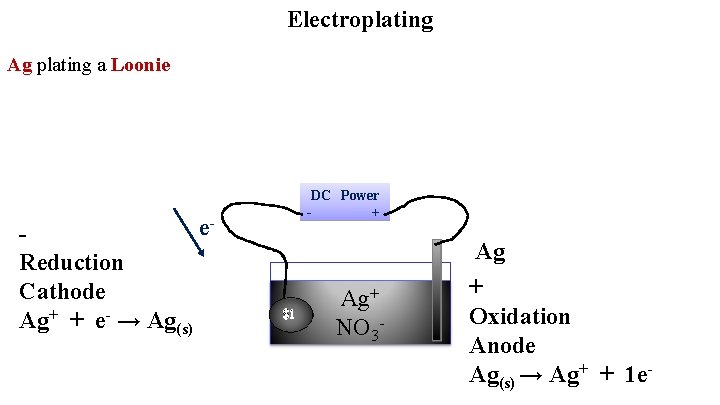
Electroplating Ag plating a Loonie e Reduction Cathode Ag+ + e- → Ag(s) DC Power + - $1 Ag+ NO 3 - Ag + Oxidation Anode Ag(s) → Ag+ + 1 e-

Electrorefinning Lead Trail, B. C. Teck 16 mines in BC Major World Producer of Zn, Cu, Pb, and Coal

Lead Refinery-Trail
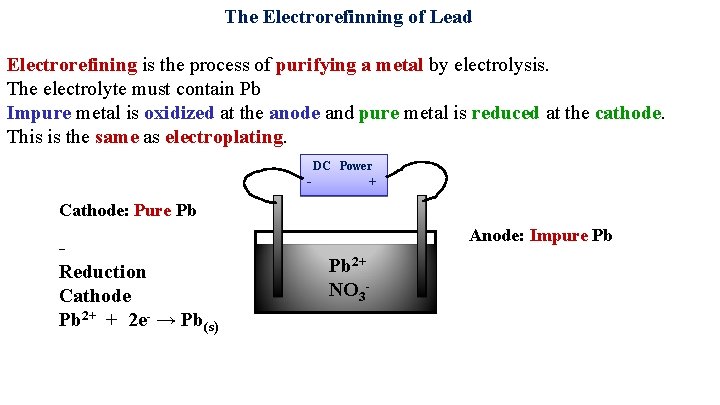
The Electrorefinning of Lead Electrorefining is the process of purifying a metal by electrolysis. The electrolyte must contain Pb Impure metal is oxidized at the anode and pure metal is reduced at the cathode. This is the same as electroplating. DC Power - Cathode: Pure Pb Reduction Cathode 2+ Pb + 2 e- → Pb(s) + Anode: Impure Pb Pb 2+ NO 3 -

The impurities in Pb are: Au Ag Zn

At the Anode Au does not oxidize The voltage is controlled so that: Ag does not oxidize Pb oxidizes Zn oxidizes

At the Cathode The voltage is controlled so that: Pb 2+ reduces Zn 2+ does not reduce
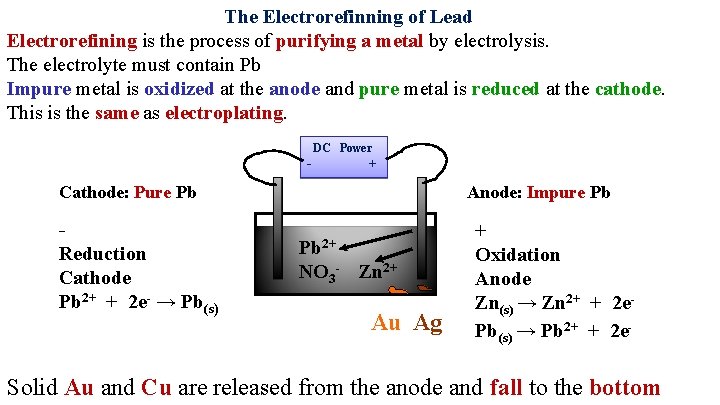
The Electrorefinning of Lead Electrorefining is the process of purifying a metal by electrolysis. The electrolyte must contain Pb Impure metal is oxidized at the anode and pure metal is reduced at the cathode. This is the same as electroplating. DC Power - + Cathode: Pure Pb Reduction Cathode Pb 2+ + 2 e- → Pb(s) Anode: Impure Pb Pb 2+ NO 3 - Zn 2+ Au Ag + Oxidation Anode Zn(s) → Zn 2+ + 2 e- Pb(s) → Pb 2+ + 2 e- Solid Au and Cu are released from the anode and fall to the bottom
Video of thermite reaction used to make Iron Thermite reaction is very exothermic and produces white hot molten iron. Fe 2 O 3 + 2 Al(s) → Al 2 O 3(s) + 2 Fe(l) + energy
- Slides: 24