APNEA OBJECTIVES Understand the control and maturation of

APNEA
OBJECTIVES • Understand the control and maturation of breathing in the fetus and newborn. • Review the basic definitions of abnormal breathing in the newborn. • Discuss evaluation and management of apnea and explore the reasons for home monitoring.

RESPIRATORY CONTROL • Normal Physiology – central generation/maintenance of resp. rhythm – modulation of rhythm by central and peripheral feedback loops – recruitment of respiratory muscles appropriate for the task • Not entirely an “autopilot” process
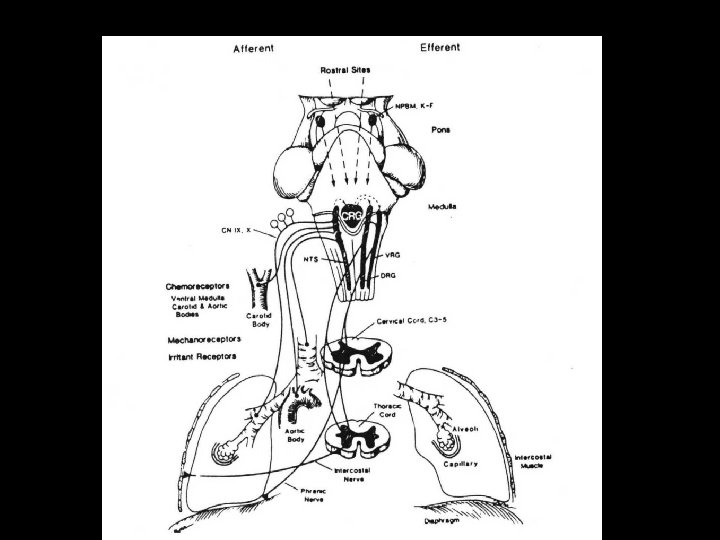

Receptor Response Importance Facial cutaneous receptors inhibitory newborn nasal mucosal receptors inhibitory infants laryngeal receptors inhibitory most potent Airway smooth muscle receptors inhibitory Hering-Breuer reflex Lung parenchyma stretch receptor inhibitory delay inspiration Lung J receptors excitatory pulmonary edema Bronchial irritant receptors inhibitory shorten inspiration Central chemoreceptors (? medulla) excitatory H Pco 2, L p. H (H high, L low) inhibitory L Pco 2, H p. H excitatory L Po 2, H Pco 2, L p. H inhibitory H Po 2, L Pco 2, H p. H Carotid body (peripheral chemoreceptor) Aortic body (peripheral chemoreceptor)

NEUROMODULATION • List goes on and on… – amino acids, neuropeptides, substance P, somatostatin, adenosine, prostaglandins…. . • Effects are most pronounced in least mature infants • Inhibitory effects tend to predominate
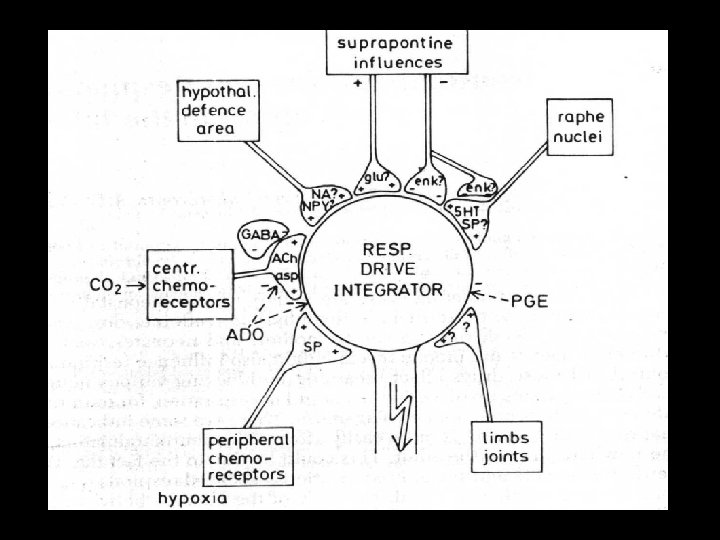
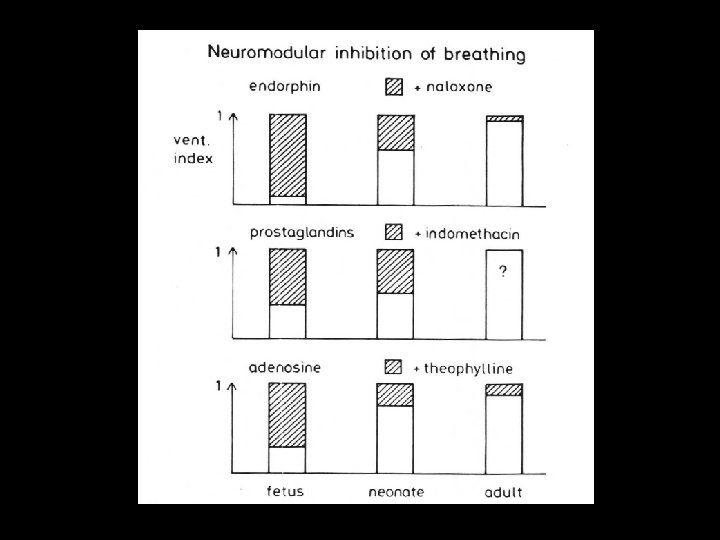

Maturation of Breathing • Fetal Breathing – well documented as early as 11 weeks – functions include • exercise • lung development • preparation – reflexes present (e. g. Hering-Breuer) • Sleep State – REM vs NREM

CONTINUOUS BREATHING • Mechanism of transition to continuous breathing is not entirely clear • Not dependent on chemical stimuli • Not dependent on external stimuli • Once established, PO 2 is important in maintaining it.

POSTNATAL MATURATION • Extensive development of synapses and increase in neuronal size • Increase in concentrations of neurotransmitters • Further dendritic elaboration continues for years

DEFINITIONS

APNEA • Cessation of respiratory airflow – Pathologic apnea is a respiratory pause > 20 seconds or of any duration if associated with cyanosis or significant desaturation, pallor, hypotonia, or bradycardia

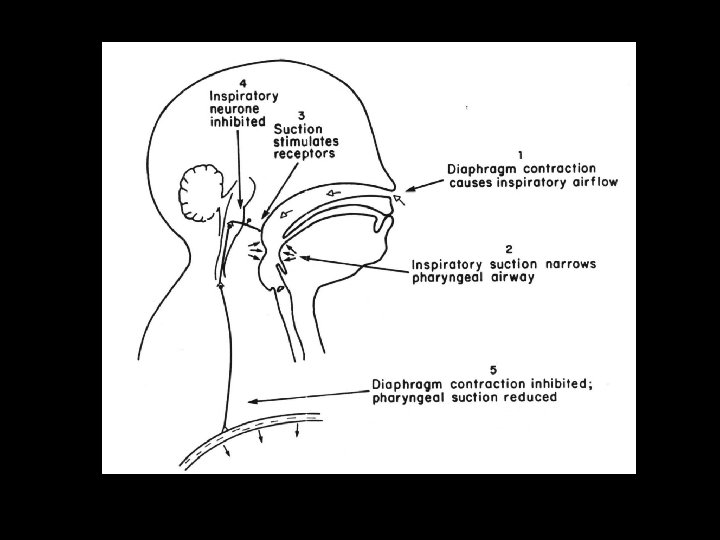
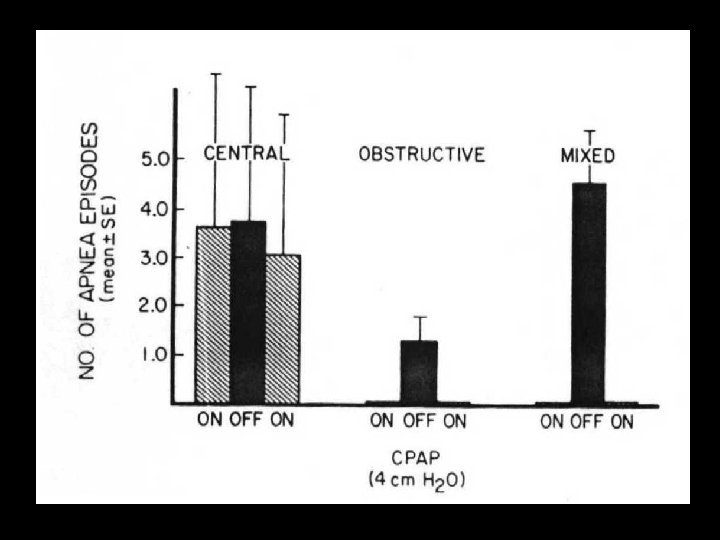
CENTRAL APNEA Apnea due to absent inspiratory effort (10 -25% of all apnea)
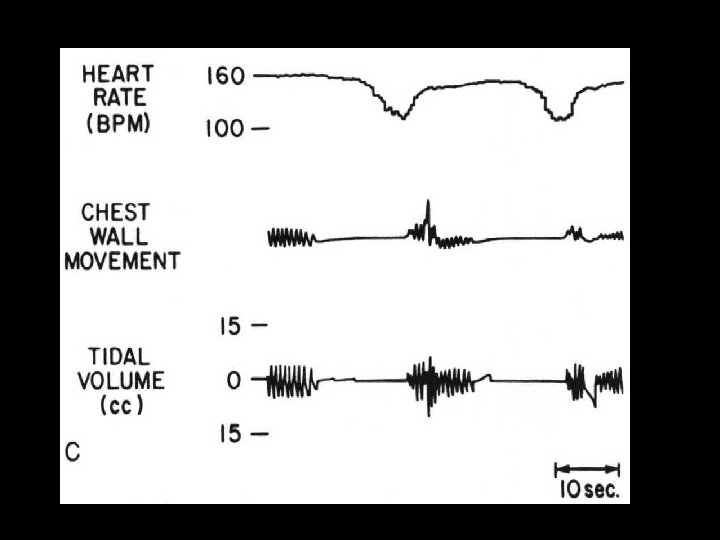
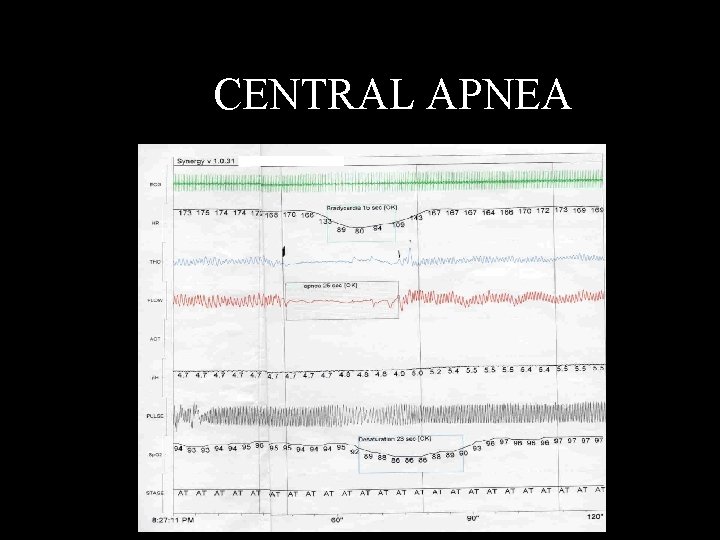
CENTRAL APNEA

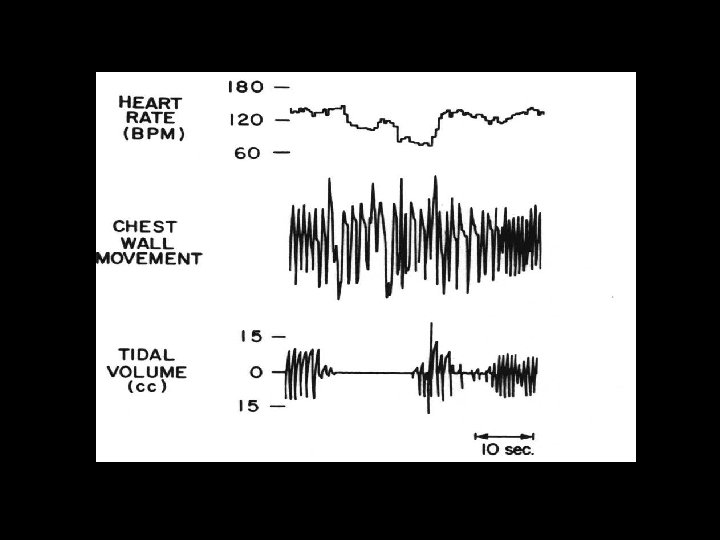
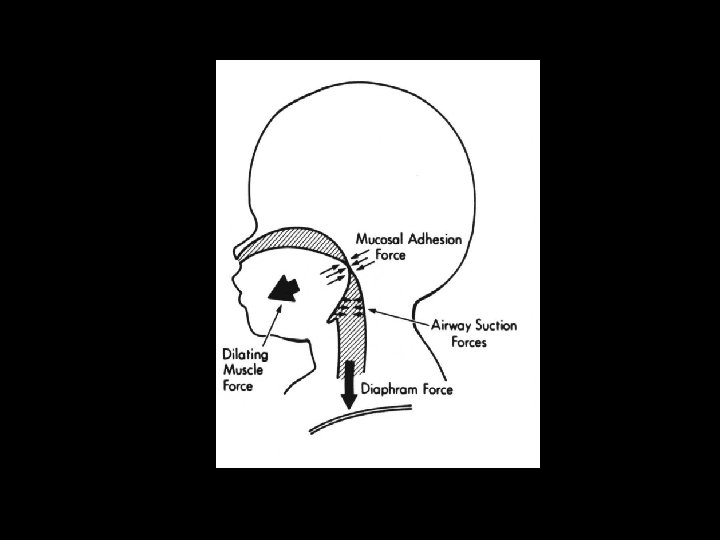
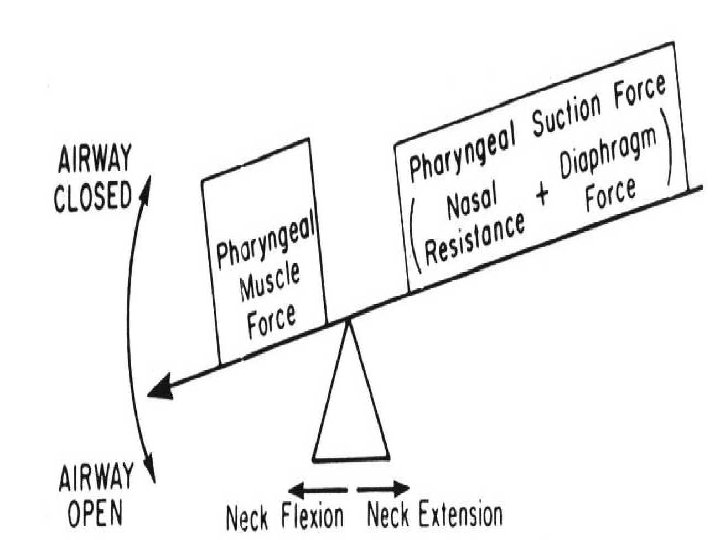
OBSTRUCTIVE APNEA Chest wall motion without air flow (12 -20% of all apnea)

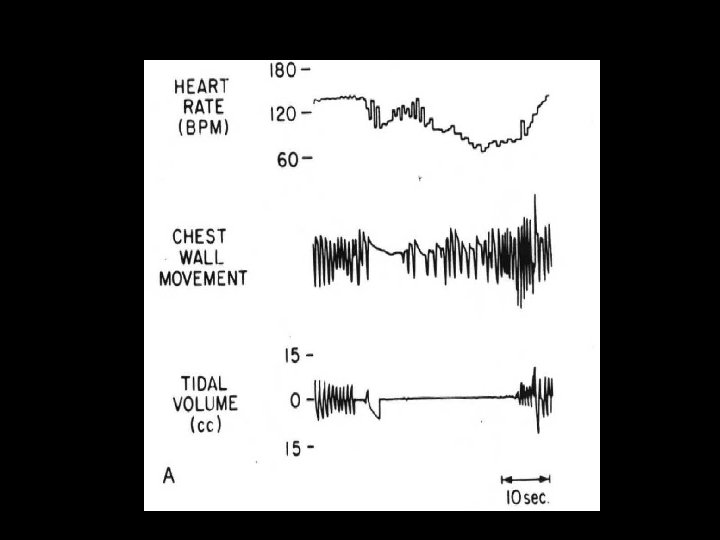
MIXED APNEA Combination of central and obstructive (50 -70% of all apnea in premature)

PERIODIC BREATHING A breathing pattern in which there are > 3 respiratory pauses > 3 seconds in duration with < 20 seconds of respiration between pauses. This is not considered pathologic.

PERIODIC BREATHING Age (weeks) Term Infants Preterm Infants 0 - 4 0 - 3. 5 % 0 - 5 % 4 - 8 0 - 2. 5 % 0 - 3. 5 % 8 - 20 0 - 1. 5 % 0 - 2. 5 % > 20 0 - 1. 0 % 0 - 1. 5 %

PERIODIC BREATHING

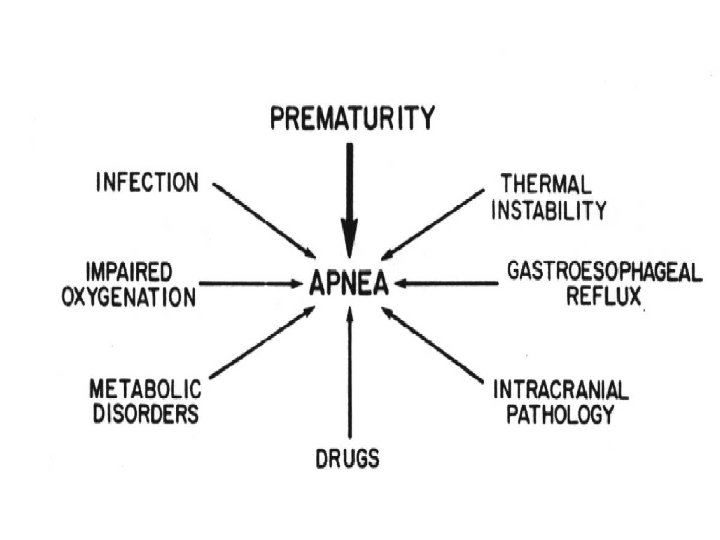
APNEA OF PREMATURITY Pathologic apnea in a premature infant usually ceasing at > 37 weeks gestation • Premature infants (<34 weeks) have immature/inappropriate responses; e. g. apnea in the face of hypoxia. Phrenic neurons are more mature than upper airway neurons leading to persistence of obstructive apnea
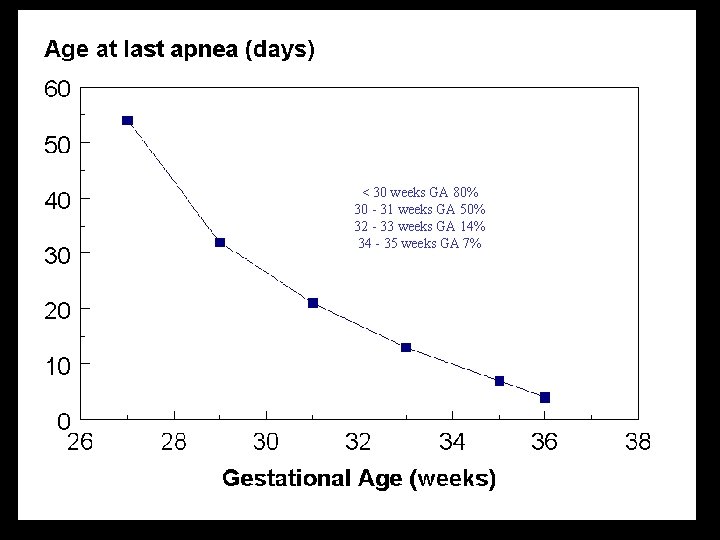
< 30 weeks GA 80% 30 - 31 weeks GA 50% 32 - 33 weeks GA 14% 34 - 35 weeks GA 7%
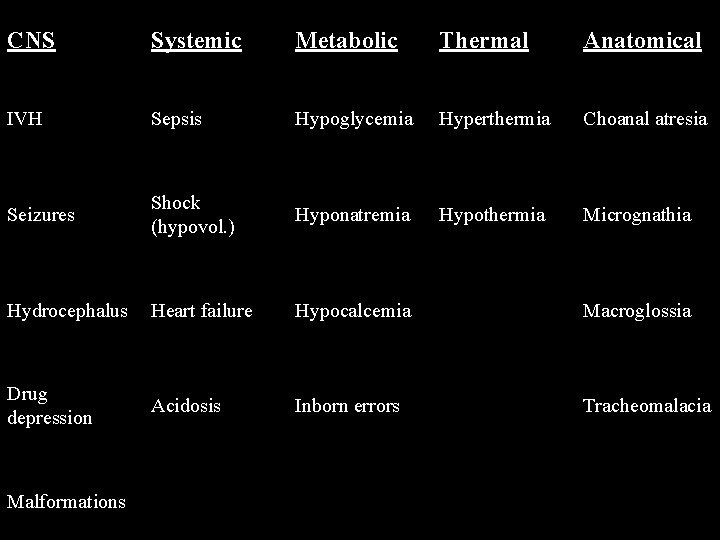
CNS Systemic Metabolic Thermal Anatomical IVH Sepsis Hypoglycemia Hyperthermia Choanal atresia Seizures Shock (hypovol. ) Hyponatremia Hypothermia Micrognathia Hydrocephalus Heart failure Hypocalcemia Macroglossia Drug depression Acidosis Inborn errors Tracheomalacia Malformations

APNEA OF INFANCY Unexplained episode of pathologic apnea with onset at > 37 weeks gestation

ALTE An episode that is frightening to the observer and that is characterized by some combination of apnea, color change, marked change in muscle tone, choking or gagging, especially when CPR is required.

Digestive (47%) GER, aspiration Infection Malformations Dumping Neurological (29%) Vasovagal response Seizure disorder Infection Subdural hematoma Malformations Respiratory (15%) Infection (RSV, Pertussis) Airway abnormality Alveolar hypoventilation Metabolic (2. 5%) Hypoglycemia Hypocalcemia Food intolerance Inborn errors Miscellaneous (3%) Accidents Sepsis Munchausen by proxy Drug effect Cardiovascular(3. 5% Cardiomyopathy Infection Arrythmia CHD

ALVEOLAR HYPOVENTILATION SYNDROMES • Congenital – CHS, Arnold-Chiari II, Leigh Disease, Pyruvate Dehydrogenase def. , Carnitine def. , Mobius Syndrome • Acquired – CNS injury, infection, asphyxia, tumor, infarction • Transient – Obstructive Sleep Apnea Syndromes

EVALUATION • History – GA; perinatal complications; drug exposure; concern for infection • Physical – complete, but concentrate on cardiorespiratory and neurologic • Laboratory/Radiology – sepsis; sugar; acidosis, hypoxia, hemorrhage

MANAGEMENT • Acute management……. ABC’s • Further management depends on results of evaluation • Goals – prevent further severe episodes – reassure parents

TREATMENT • Pharmacologic – methylxanthines • theophylline • caffeine – doxapram (continuous infusion) • CPAP (3 -5 cm. H 2 O)

METHYLXANTHINES • Proposed mechanisms of effect – increase minute ventilation – shift of CO 2 response curve to left – efficiency of diaphragmatic contraction – improved pulmonary mechanics – decreased hypoxic ventilatory depression • Many potential complications (overdose)

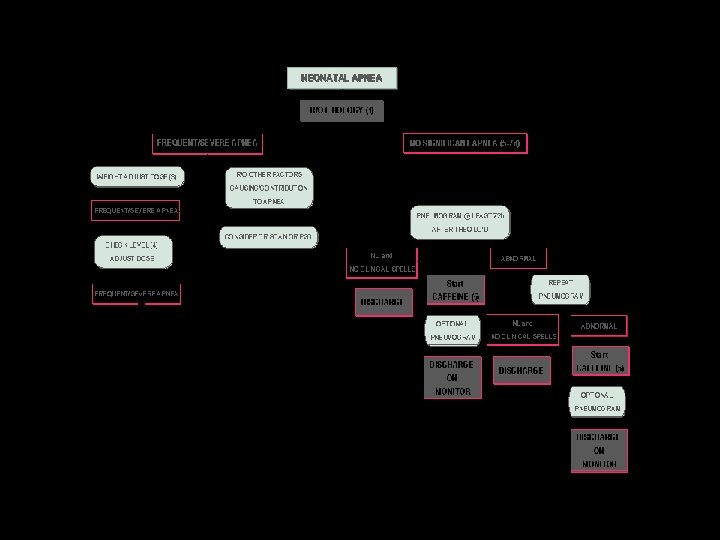
Management: In absence of any underlying problems, these are some helpful guidelines a) Infants born at <35 weeks of GA (and not on ventilatory support) should be monitored for the first week of life, at least. b) Infants with less than 10 apneic episodes a day, without profound hypoxemia or bradycardia and with quick response to stimulation should be simply watched. c) Infants with several episodes during the day, with prolonged episodes associated with hypoxemia and/or bradycardia, with slow response to stimulation, requiring bag-and-mask ventilation to recover, should go through a trial of xanthines (caffeine, theophylline). d) Infants who fail on xanthines could be tried on CPAP. IV Doxapram infusion has been advocated by some, with variable success. e) Infants with refractory apnea and severe episodes should be considered for long term, low-setting mechanical ventilation

HOME MONITORING

INDICATIONS • Persistent episodes of significant apnea/bradycardia when otherwise ready for discharge (5 day rule) • Sibling/Twin of SIDS • Technology-dependent infant

REQUIREMENTS FOR HOME MONITORING • • • Parental understanding of plan Parental/guardian knowledge of infant CPR Knowledge of monitoring capabilities Technical support Documented monitoring Physician follow-up

PROBLEMS • • False alarms Monitor malfunction Alternate caretaker difficulties Skin irritation/breakdown Sibling jealousy Overdependence/difficulty weaning Lack of compliance

DISCONTINUATION • Depends on reason for monitoring • Most can be weaned within 2 -3 months of discharge • No indication for monitoring of a normal child > 1 year of age.

AAP RECOMMENDATIONS • Home cardiorespiratory monitoring should not be prescribed to prevent SIDS.

• Home cardiorespiratory monitoring may be warranted for premature infants who are at high risk of recurrent episodes of apnea, bradycardia, and hypoxemia after hospital discharge. The use of home cardiorespiratory monitoring in this population should be limited to approximately 43 weeks' postmenstrual age or after the cessation of extreme episodes, whichever comes last.

• Home cardiorespiratory monitoring may be warranted for infants who are technology dependent (tracheostomy, continuous positive airway pressure), have unstable airways, have rare medical conditions affecting regulation of breathing, or have symptomatic chronic lung disease. • If home cardiorespiratory monitoring is prescribed, the monitor should be equipped with an event recorder.

• Parents should be advised that home cardiorespiratory monitoring has not been proven to prevent sudden unexpected deaths in infants. • Pediatricians should continue to promote proven practices that decrease the risk of SIDS—supine sleep position, safe sleeping environments, and elimination of prenatal and postnatal exposure to tobacco smoke

NICHD CONSENSUS CONFERENCE 2005 • • • No consensus has been reached regarding the definition, diagnosis, or treatment of AOP. Systematic research has not been conducted to investigate the value of different interventions for AOP. Available technology is not routinely used to document real-time events associated with apnea. The time required to demonstrate an improvement in AOP with a specific treatment has not been established. The observational period needed after therapy for AOP is unknown, and an appropriate duration of surveillance off therapy is needed to reasonably prevent acute lifethreatening events. Important confounding conditions that influence the occurrence of AOP are poorly recognized and/or integrated into care. The relationship between gastroesophageal reflux (GER) and AOP requires additional investigation because current knowledge suggests an infrequent association. Improved characterization of the effects of AOP on neurodevelopment during infancy and childhood is needed. Other confounders associated with brain injury in preterm infants are difficult to separate from AOP as meaningful causes of abnormal child development.

- Slides: 54