APLA syndrome Antiphospholipid antibody syndrome Hughes syndrome EPIDEMIOLOGY

APLA syndrome Antiphospholipid antibody syndrome Hughes syndrome

EPIDEMIOLOGY • 15 -20% of DVT is due to APS. • 1/3 rd Strokes below age 50 years. • 5 -15% recurrent fetal loss.

Types of APLA • Primary (Idiopathic) Absence of any associated disease ; genetic marker – HLA DR 7 • Secondary with associated with other autoimmune disorders like SLE, RA, etc. Genetic markers – HLA B 8, HLA DR 2, HLA DR 3 • Catastrophic rapid organ failure due to generalised thrombosis

• Hypercoagulable state • Autoimmune in origin • Antibodies against a cell substancephospholipids esp. cardiolipin & B 2 glycoprotein. leading to……

thrombosis arteries & veins ||| pregnancy-related complications deep vein thrombosis stroke

Mechanisms of Pregnancy complications: 1) APL binds annexin 5 on trophoblast surface with break in anticoagulant shield. 2) An inflammatory reaction due to neutrophils, complement and complement receptors.

pregnancy-related complications • • Miscarriage before 20 weeks Pre-eclampsia after 20 weeks Stillbirth because of placental infarct Mental or developmental retardation in neonate because of failure of trophoblast differenciation

Mechanisms of thrombosis: 1) Endothelial cell activation by complexes of beta 2 glycoprotein and anti- beta 2 GP 1. 2) Platelet activation. 3) Auto antibodies to to annexins 5 and activated protein C. 4) Abnormal fibrinolysis.

Other mechanisms • Anti-Apo. H : inhibits Protein C, (by degradating activated factor V). • LAC antibodies : bind to prothrombin, thus increasing its cleavage to thrombin • antibodies binding to Protein S: which is a co-factor of protein C.

Thrombosis related features • Suspect APS in any young patient with arterial thrombosis or without any obvious risk factors for thrombosis. • arterial thrombotic events Ø CNS: Stroke, TIA, central retinal artery occlusion, amaurosis fugax, Ø Gangrene Ø MI • Venous thrombotic events : Ø DVT Ø CVST

• Thrombocytopenia seen in 1/3 patients, not significant enough to cause bleeding, other causes to be ruled out. • 10 -20% patient’s are Coomb’s positive, but clinical autoimmune haemolytic anaemia is uncommon, and is associated with cardiac valvular lesions. • Evan’s syndrome when AIHA and thrombocytopenia occur simultaneously.
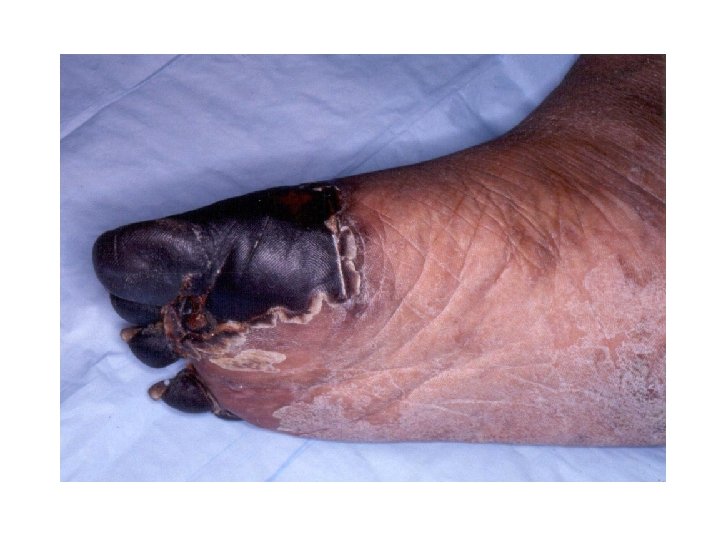
Cardivascular Manifestation: Ø Intracardiac thrombosis Ø Valvular heart lesions (non infectious Libman. Sacks endocarditis) mainly affecting mitral valve in 30 -50% patients. • Skin Manifestations: Ø livido reticularis – 25% patients Ø leg ulcers ØGangrene Ønail fold infarcts.
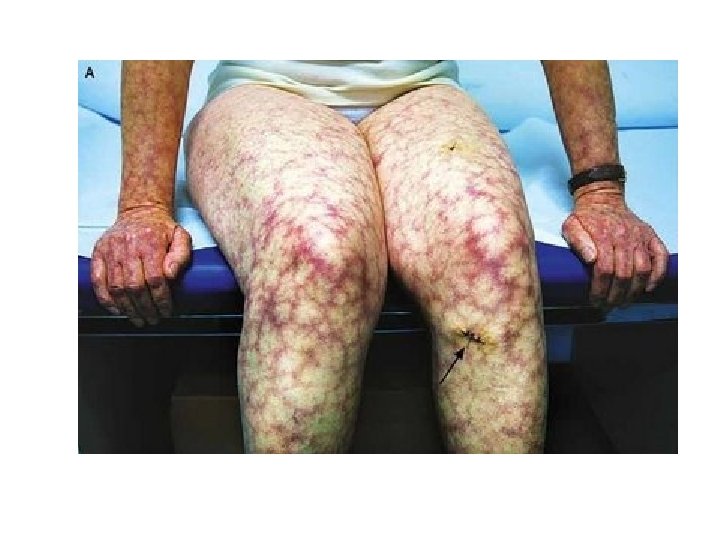
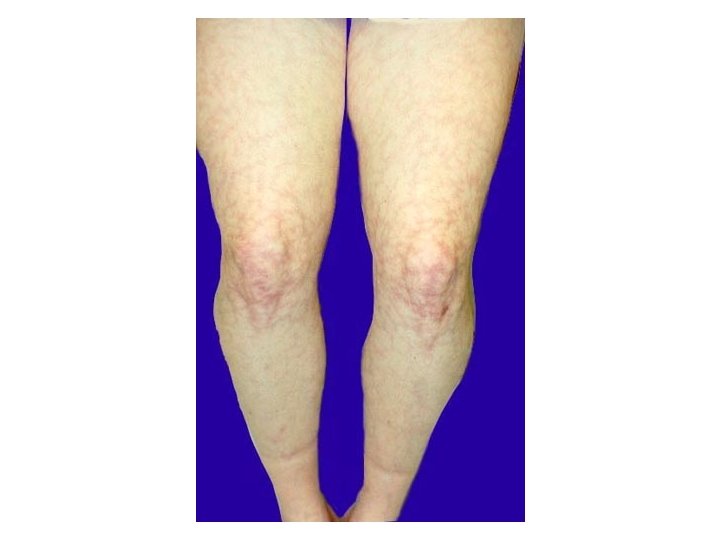

• Renal related manifestations: Øvessels involvement present with hypertension Ørenal infarction Øhaematuria. • In SLE, presence of APS worsens renal prognosis.
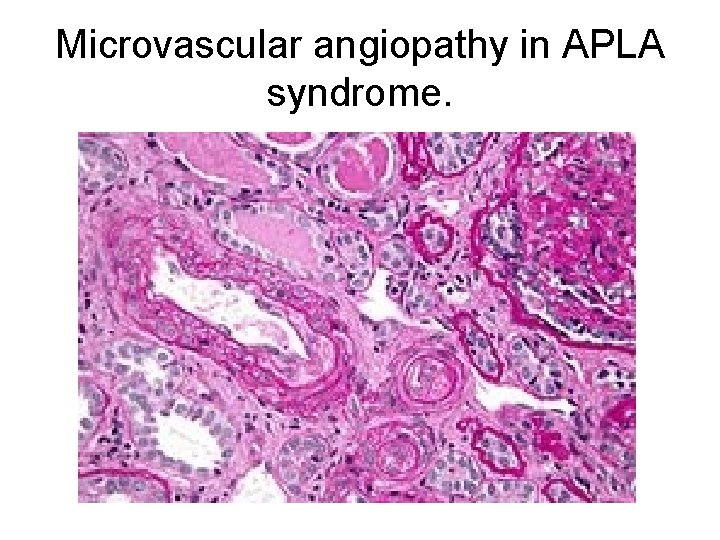
Microvascular angiopathy in APLA syndrome.

Diagnosis of APLA

We need…. One clinical criteria One Laboratory criteria

Clinical criteria to diagnose APLA episode of arterial, venous, or small vessel thrombosis —other than superficial venous thrombosis in any tissue or organ by objective validated criteria with no significant evidence of inflammation in the vessel wall and/or 1 or more unexplained deaths of a morphologically normal fetus (documented by ultrasound or direct examination of the fetus) at or beyond the 10 th week of gestation and/or 3 more unexplained consecutive spontaneous abortions before the 10 th week of gestation, with maternal anatomic or hormonal abnormalities and paternal and maternal chromosomal causes excluded or at least 1 premature birth of a morphologically normal neonate before the 34 th week of gestation due to eclampsia or severe pre-eclampsia according to standard definitions, or recognized features of placental insufficiency

Lab crieteria to diagnose APLA • Anti-cardiolipin Ig. G and/or Ig. M measured by standardized, • • non-cofactor dependent ELISA medium or high titre (i. e. , > 40 GPL or MPL, or > the 99 th percentile) and/or Anti-β 2 glycoprotein I Ig. G and/or Ig. M measured by standardized ELISA medium or high titre (> the 99 th percentile) and/or Lupus anticoagulant detected according to the guidelines of the International Society of Thrombosis and Hemostasis. All are done on 2 or more occasions, not less than 12 weeks apart;

Laboratory Diagnosis • • Routine Laboratory investigations: Ø CBC Ø Blood Sugars Ø ECG Ø RFT Other Diagnosis: Ø Ø • Thrombophilia Profile ANA, anti ds DNA, ANA blot Blood cultures and Procalcitonin Sr. Homocysteine Specific Diagnosis: Ø Positive ACL/ antibeta 2 GP-1/LA separately at least 12 weeks apart. Ø LA positivity is a best marker for risk of thrombosis. Ø Increased risk for thrombosis and pregnancy morbidity with multiple antibody positivity.

Lab investigations PT a. PTT TCT DRVVT Factor VIII Factor IX Factor XI Bethesda titer Platelets Factor X cntue….

• Mixing studies of a. PTT • D-dimer • Kaolin clotting time

Guidelines from International Society on Thrombosis and Haemostasis Scientific Subcommittee • Demonstration of a prolonged phospholipid-dependent coagulation assay • Failure to correct the prolonged clotting assay in mixing studies using normal platelet-poor plasma • Shortening or correction of the prolonged coagulation assay by addition of excess phospholipid • Exclusion of other coagulopathies

Classification of APLA for research purposes • l : more than one laboratory criterion present in any combination; • IIa: lupus anticoagulant present alone • IIb: anti-cardiolipin Ig. G and/or Ig. M present alone in medium or high titers • IIc: anti-β 2 glycoprotein I Ig. G and/or Ig. M present alone in a titer greater than 99 th percentile

• Functional assay or clot based assay: inhibition of phospholipid dependent clotting testsfor lupus anti-coagulant (it may be present in absence of SLE, hence a misnomer ) • Immunoassay : Detection of antibody to specific protein phospholipid complexesfor anticardiolipin & beta 2 gycoprotein 1.

The panel consists of 21 antiphospholipid antibodies that include Ig. G, Ig. M and Ig. A antibodies to Øcardiolipin, Øphosphatidylserine, Øphosphatidylinositol, Øphosphatidylethanolamine, Ø phosphatidylcholine, Øphosphatidylglycerol, Øphosphatidic acid.

MANAGEMENT • For venous thrombosis and thromboembolic phenomena: 1. Unfractionated heparin 2. LMWH and 3. Later addition of anti-coagulants • Target INR 2 to 3 • Lifelong anticoagulation • Increased incidence of recurrence during first 6 months of discontinuation of anticoagulant. • Recurrence usually occurs in same type of vessel.

• Arterial thrombosis 1) Low Dose Aspirin 2) Non cerebral thrombosis – uncertain management 3) In MI patient – long anticoagulation is required. • Pregnancy 1) Unfractionated heparin 5000 units BD/ LMWH to reach anti factor Xa level of 0. 10. 3 u/ml with low dose aspirin. 2) Aspirin is started at time of conception. 3) Heparin at time of confirmation of pregnancy. 4) Till 3 rd trimester of pregnancy.

CAPS

Mechanism of Rapid organ failure Antigen: endothelium of vessel wall + Autoantibody ll Antigen-antibody reaction at endothelium ↓ Platelet aggregation ↓ Initiation of coagulation cascade

Crieteria to diagsose CAPS • a) Vascular thrombosis in three or more organs or tissues and • b) Development of manifestations simultaneously or in less than a week 'and • c) Evidence of small vessel thrombosis in at least one organ or tissue and • d) Laboratory confirmation of the presence of a. PL.

CATASTROPHIC APS • 35% cases develop CAPS after infections. • Widespread micro vascular thrombosis. • Resultant multi-organ failure-Kidneys commonly affected followed by lungs , CNS and skin. • Thrombocytopenia and DIC are common findings. • Mortality is upto 50%, recurrence in 26% of survivors. • Managed aggressively with heparin, plasmapheresis, Iv. Ig and high dose steroids.
- Slides: 34