APICOMPLEXA Intestinal coccidian Blood and tissue coccidian Apicomplexans
APICOMPLEXA Intestinal coccidian Blood and tissue coccidian
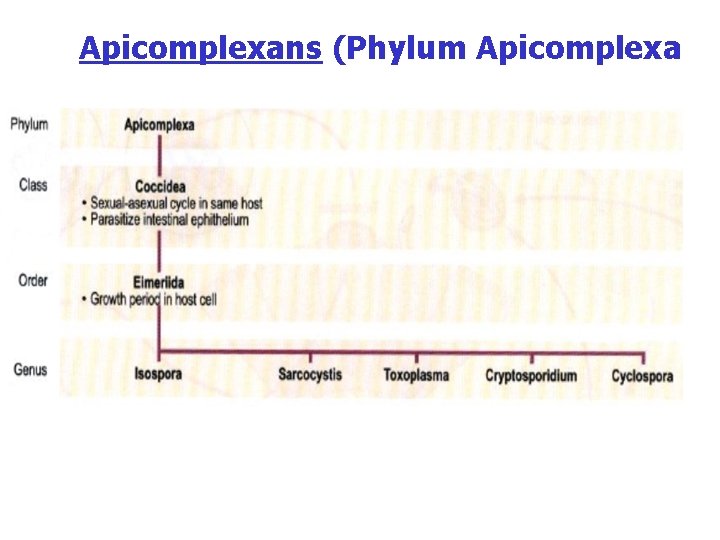
Apicomplexans (Phylum Apicomplexa
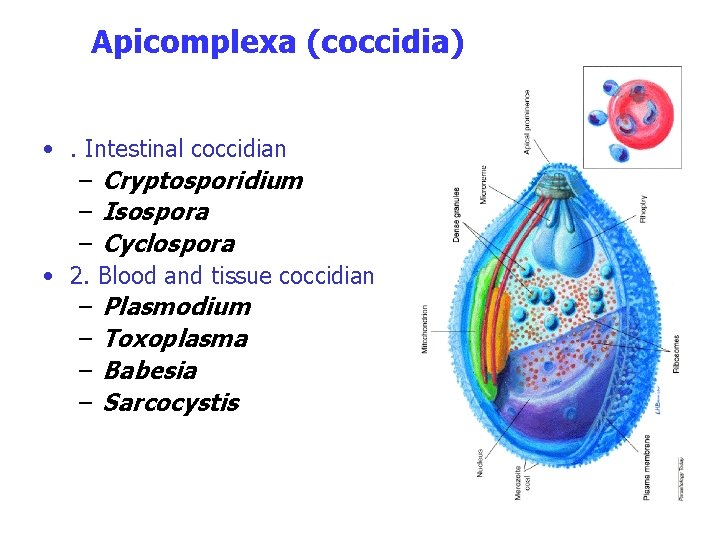
Apicomplexa (coccidia) • . Intestinal coccidian – Cryptosporidium – Isospora – Cyclospora • 2. Blood and tissue coccidian – Plasmodium – Toxoplasma – Babesia – Sarcocystis

Learning Objectives: • After taking this lesson the student will be able to: 1. list common Intestinal coccidian that affects human 2. Describe the common characteristics of. Intestinal coccidian 3. Describe the morphology of the oocyst 4. Discuses the epidemiology, life cycle, and clinical aspects of Cryptosporidium, Isospora and Cyclospora 5. Discuses the pathology of medically important intestinal coccidia. (Cryptosporidium, Isospora and Cyclospora species)

Laboratory Learning Objectives: • Upon completion of this unit of instruction and laboratory experience, the student will be able to: – outline the laboratory technique used to detect and identify the oocyst of intestinal coccida – Describe the morphology of oocyst forms of Cryptosporidium , Isospora and Cyclospora – Detect and identify oocyst forms of Cryptosporidium , Isospora and Cyclospora in stool specimen – Differentiate oocyst forms from other artifacts in stool specimen

Outline • Common feature of intestinal coccidia • Taxonomic Classification of intestinal coccidia • Cryptosporidium, Isospora and Cyclospora – Epidemiology – Morphology – Transmission and life cycle – Pathology and clinical manifestation

Outline • Laboratory diagnosis – Microscopy • Wet mount • Modified acid fast staining – Immunological assay – Molecular methods

Intestinal coccidian Cryptosporidium , Isospora , Cyclospora General characteristics • Considered as opportunistic parasite in immunocompromissed person • Complte entire life cycle in single host • Within the intestinal epithelial cells of the host • Characterized by a thick walled oocyst excreted in faeces • Are transmitted by the fecal-oral route

Cryptosporidium Species: C. parvum Ø A coccidean parasite implicated in intestinal Ø Disease primarily among immunocompromised patient Ø 1985 first reported case in philippines associated with intractable diarrhea in immunocompromised (AIDS) patient Ø Self-limiting diarrhea in immunocompetent persons Ø Profuse, watery diarrhea associated with AIDS (life threatening

• 1976 considered as natural parasite of animal especially calves causing diarrheal diseases • Infect wide range of animal hosts (fish-humans) – C. hominis & C. parvum is species mostly causing disease in humans – C. hominis infects only humans but C. parvum also infects many other mammals – C. felis, C. meleagridis, C. canis, and C. muris infections have also been reported

• Geog. Dist. : = Worldwide distribution • Morphology: = oocyst round or slightly oval-shaped, 4 – 6 um enclosing 4 spindle–shaped sporozoites

Transmission • Mainly through the ingestion and possibly inhalation of sporulated oocysts • mainly through contaminated water & • Occasionally food sources, such as chicken salad • fecal-oral transmission (monoxenous) – anthroponotic transmission – autoinfection – zoonotic transmission
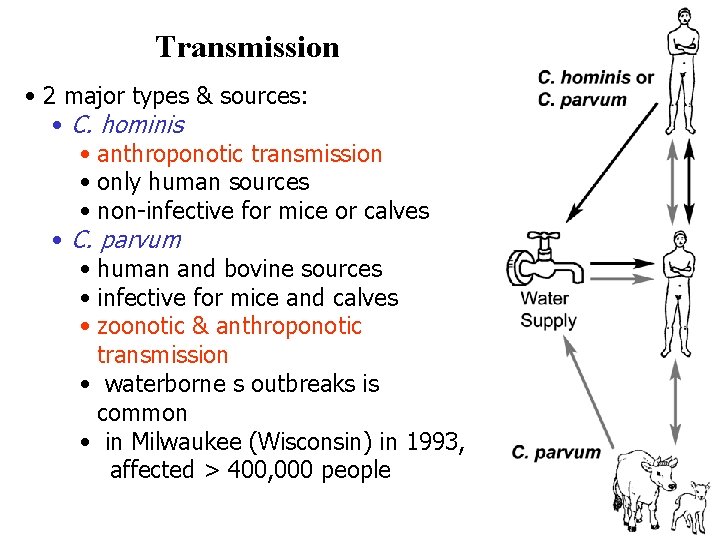
Transmission • 2 major types & sources: • C. hominis • anthroponotic transmission • only human sources • non-infective for mice or calves • C. parvum • human and bovine sources • infective for mice and calves • zoonotic & anthroponotic transmission • waterborne s outbreaks is common • in Milwaukee (Wisconsin) in 1993, affected > 400, 000 people

Waterborne Cryptosporidiosis • human infection usually waterborne and acquired by fecal-oral route • highest prevalence of disease in areas with unreliable water and food sanitation

Factors Favoring Waterborne Cryptosporidiosis • Small size of oocysts (4 -5 mm) • Reduced host specificity and monoxenous development • Close associations between human and animal hosts • Large number of oocysts excreted (up to 100 billion per calf per day) • Low infective dose (<30) • Robust oocysts; resistant to chlorine
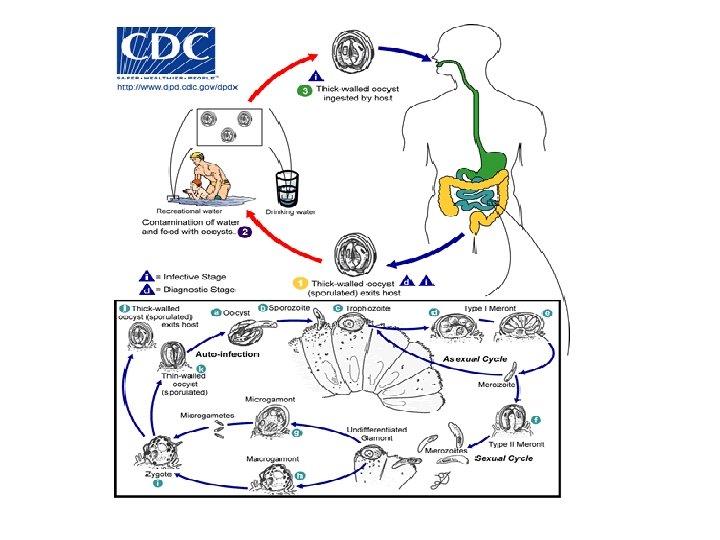
Life Cycle:
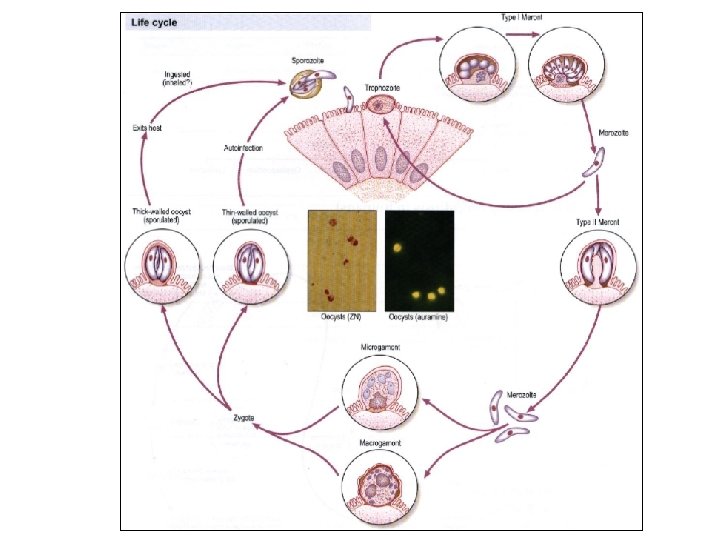
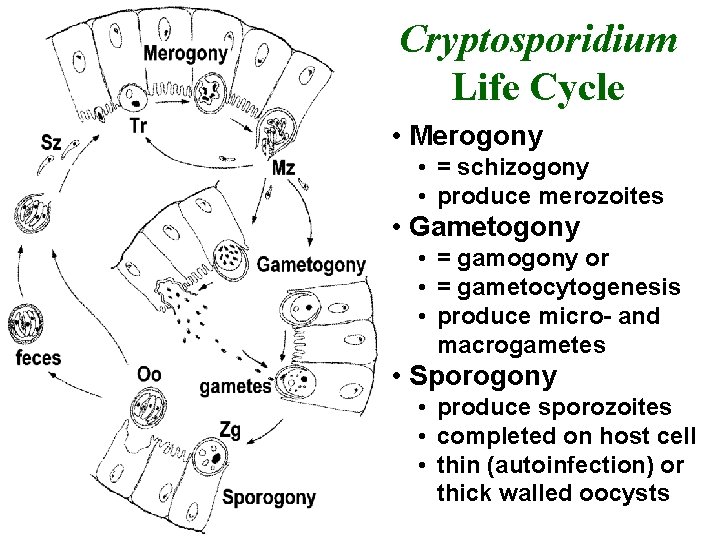
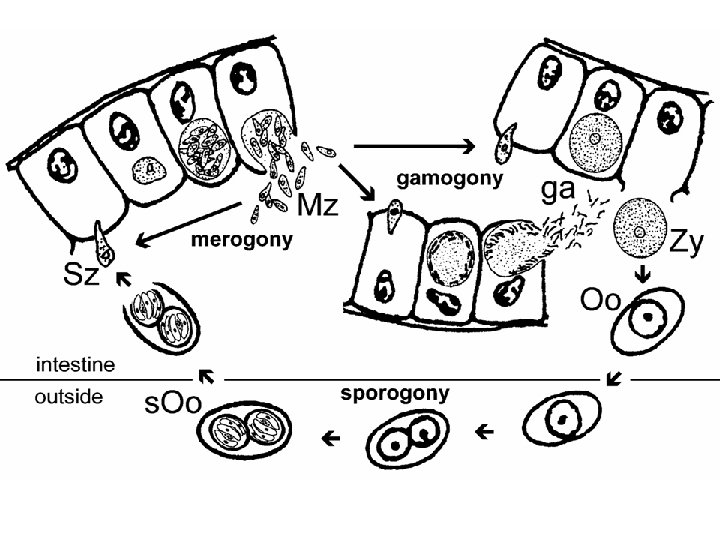
Cryptosporidium Life Cycle • Merogony • = schizogony • produce merozoites • Gametogony • = gamogony or • = gametocytogenesis • produce micro- and macrogametes • Sporogony • produce sporozoites • completed on host cell • thin (autoinfection) or thick walled oocysts
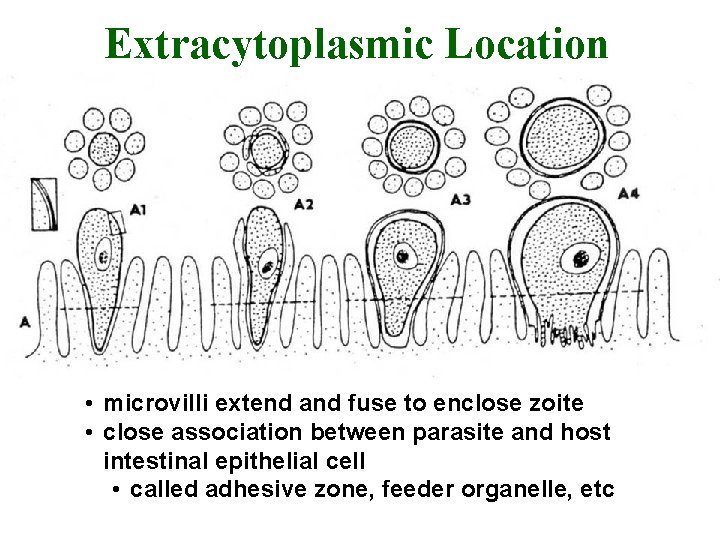
Extracytoplasmic Location • microvilli extend and fuse to enclose zoite • close association between parasite and host intestinal epithelial cell • called adhesive zone, feeder organelle, etc
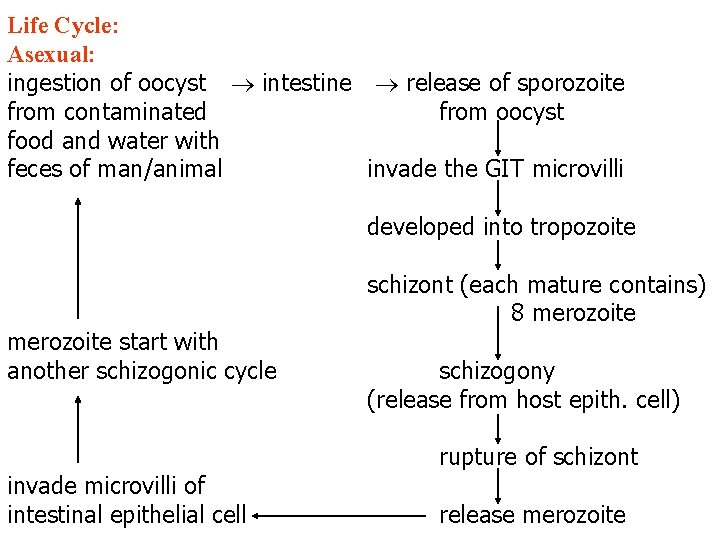
Life Cycle: Asexual: ingestion of oocyst intestine release of sporozoite from contaminated from oocyst food and water with feces of man/animal invade the GIT microvilli developed into tropozoite merozoite start with another schizogonic cycle invade microvilli of intestinal epithelial cell schizont (each mature contains) 8 merozoite schizogony (release from host epith. cell) rupture of schizont release merozoite

Sexual: = Gametogenesis and Fertilization of male and female gametocytes zygote development of oocyst sporogony 4 sporozoites with in oocyst passed out in feces

Disease: Cryptosporidiosis = human infection usually waterborne and acquired by fecal-oral route = highest prevalence of disease in areas with unreliable water and food sanitation = extraintestinal infection of the respiratory tract, biliary tract and pancreas may occur.

Clinical Features • Varry from asymptomatic to severe, life-threatening illness; • infection usually self-limiting diarrhea of 1 -2 weeks duration charactrized by copious watery diarrhea, vomiting, intense abdominal pain, anorexia and weakness • among immunocompromised patient (AIDS) develops severe chronic diarrhea which may last for months malabsorption

Clinical Features…. • Incubation period ~ 7 days ( 2 to 10 days). • In immunocompetent persons, – Symptoms are usually short lived (1 to 2 weeks) • Persons with AIDS (CD 4 counts <200/µl) – Chronic and more severe lasting for months or even years • Small intestine- most commonly affected – the lungs, and possibly conjunctiva may be affected.

Pathogenesis • Enterocytes damaged or killed – Villus atrophy (blunting) – Enterocyte malfunction – Impaired absorption – Enhanced secretion • Crypt cell hyperplasia • Inflammation in lamina propria – Impaired absorption – Promote secration

Cyclospora Specie: C. cayetanensis = was established to cause human diarrhea in 1990 =1986 cases of prolonged watery diarrhea among immunocompromised (AIDS) patient has been reported worldwide =Species Name C. Cayetanensis was given in 1993 =Initially called ‘cyano-bacteria like body’ (CLB) or large cryptosporidium

Epidemiology: = More common in tropical and sub-tropical areas = 1979 first case of cyclospora infection was reported in papua, new guinea = Subsequent cases has been reported from most part of the world

Epidemology… = Infection caused by cyclospora can be acquired by drinking contaminated water (fecal-oral) = More associated with food-borne outbreaks( social events, weddings, etc) = Parasite infect vertebrates including reptiles, insect & rodents

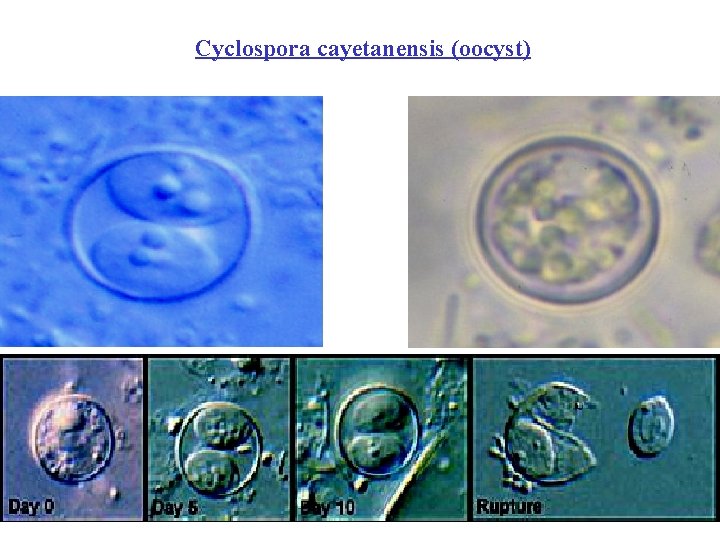
• Morphology: – Oocyst spherical-shaped 8 -10 um dia. – contains membrane bound refractile globules – Mature oocysts contains 2 sporocyst with 2 cresent -shaped sporozoite which fluoresce blue-green under UV light (cryptosporidium & isospora do not fluoresce under UV light)
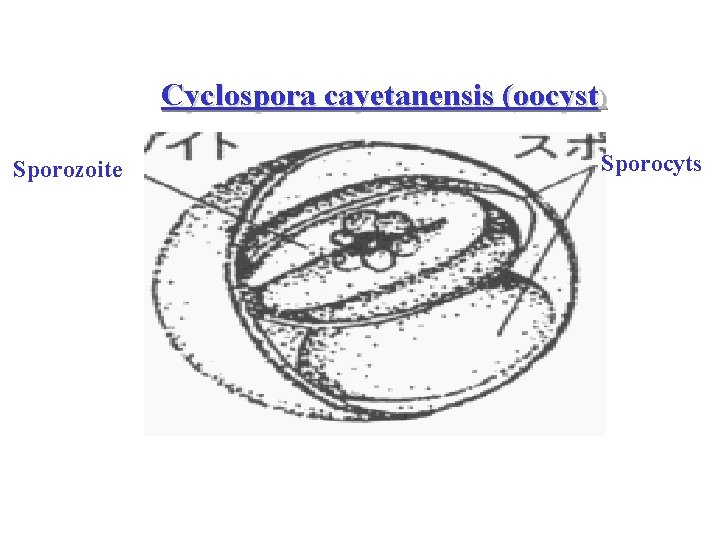
Cyclospora cayetanensis (oocyst) Sporozoite Sporocyts

Fresh Stool Acid Fast
Cyclospora cayetanensis (oocyst)
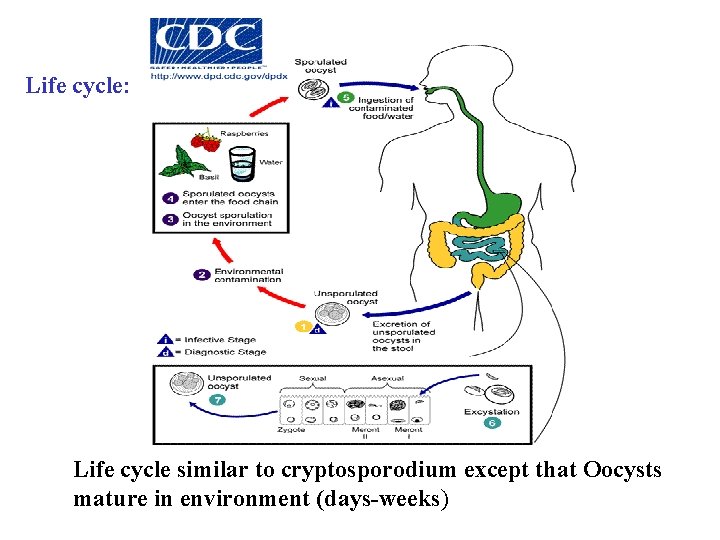
Life cycle: Life cycle similar to cryptosporodium except that Oocysts mature in environment (days-weeks)

Clinical Features Disease: Cyclosporiasis = disease is clinically indistinguishable from cryptosporidiosis and Isosporiasis = self-limiting, characterized by persistent watery diarrhea that ends to recur in a relapsing pattern and last for 3 -4 wks, = associated with abdominal cramps, nausea, vomiting, low grade fever, weight loss and anorexia

Pathogenesis: Ø Infection typically confined to the jejunum Ø characterized by inflammatory changes, villous atrophy and hyperplasia of the jejunal tissue

Isospora Specie: Isospora belli Epidemoogy • wide geographical distribution (higher prevalence in warmer climates) • the least common of the three intestinal coccidia that infect humans • transmitted fecally in contaminated food and drink with oocyst

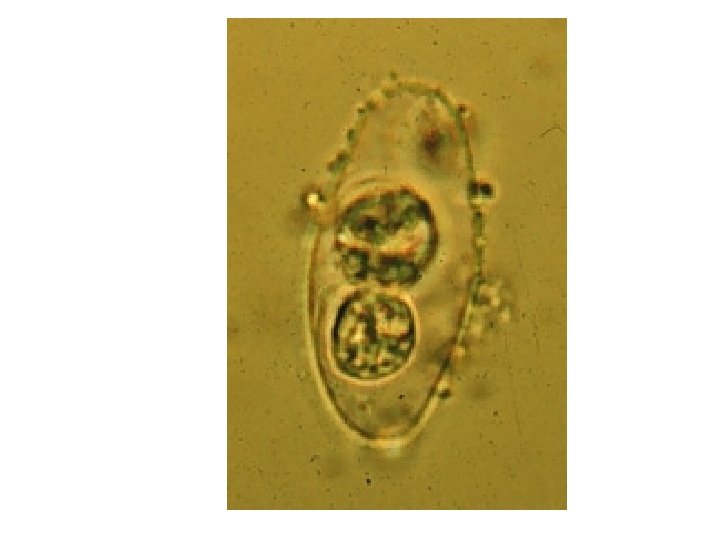
Morphology: =Oocyst elongate ovoidal-shape with moderate constriction in one end giving a charac. “Bottle with short neck” appearance =20 – 33 u L X 10 – 19 u W =Cyst wall double-layered, smooth, thin & colorless =Unsegmented oocyst contains spherical mass of granule with visible nucleus =Mature oocyst has 2 sporocyst and each contains 4 cresent-shaped sporozoites
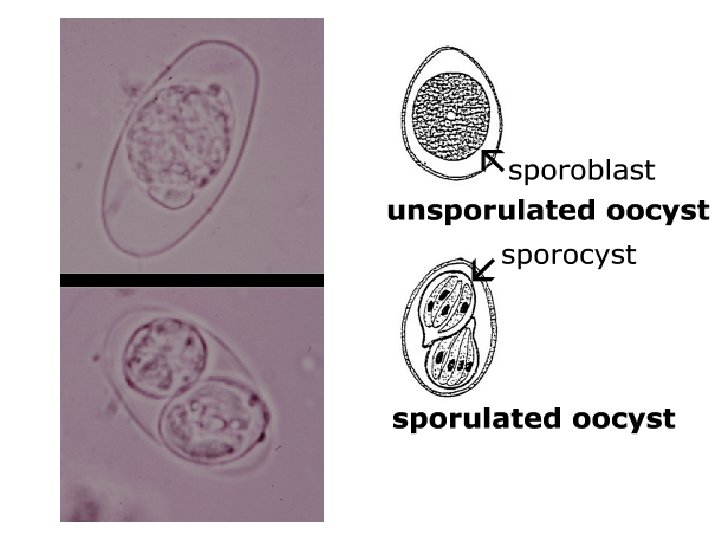
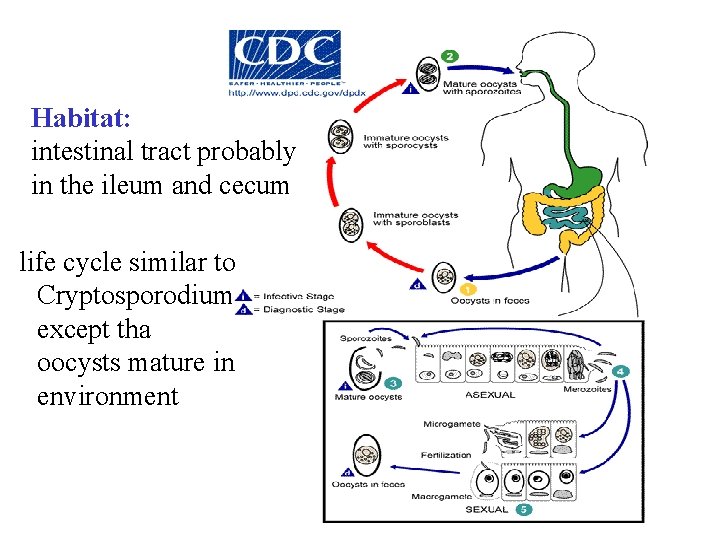
Habitat: intestinal tract probably in the ileum and cecum life cycle similar to Cryptosporodium except tha oocysts mature in environment
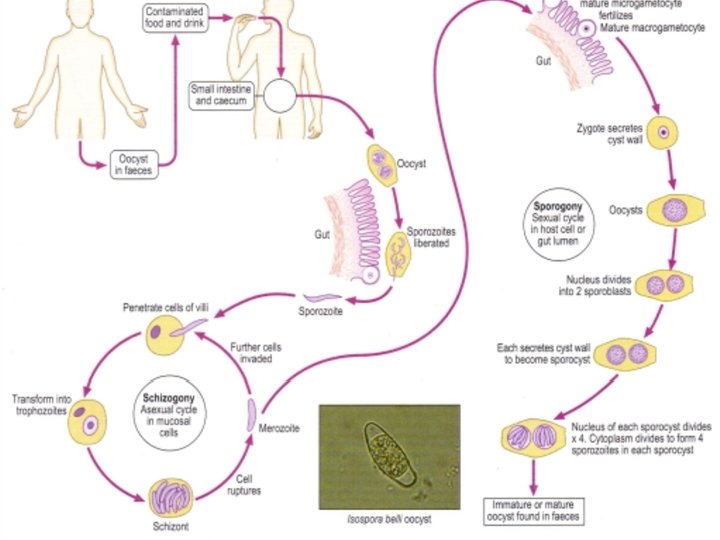

Clinical feature • Disease: Isosporosis/Intestinal coccidiosis • often asymptomatic (seldom reported) • symptoms range from mild gastro-intestinal distress to severe dysentery • mild self-limiting infection charac. by fever, colicky abdominal pain, severe diarrhea, steatorrhea (fatty stool) and weight loss. • often self-limiting, but can become chronic (wasting, anorexia) • symptoms more severe in AIDS patients

Pathology • intracellular development occurs in the intestinal mucosa (lamina propria) mucosal atrophy • charac. chronic diarrhea (months to years) abdominal discomfort, low grade fever

Genus Sarcocystis Speices: S. hominis S. Suihominis S. lindemanni = parasite of human and domestic animals (cattle, swine and sheep) Geog. Dist. : Cosmopolitan

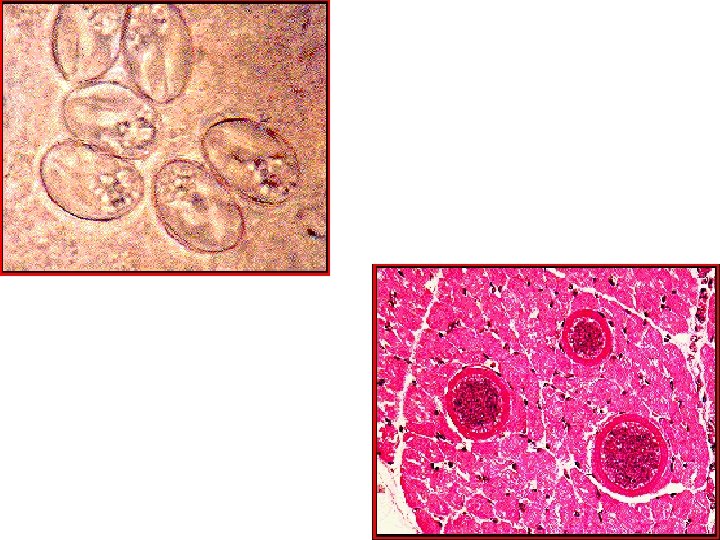
Genus Sarcocystis Morphology: =Oocyst broadly oval lying along infected muscle fibers, hyaline appearance and enclose in a membrane = contains 2 large sporocyst inside tubular mass (Meischer tube) filled up with 4 mature cresent-shaped sporozoites = sporocyst/rainy corpuscles/trophozoite are banana-shape with subspherical nucleus found in the muscle thread extending from end to end
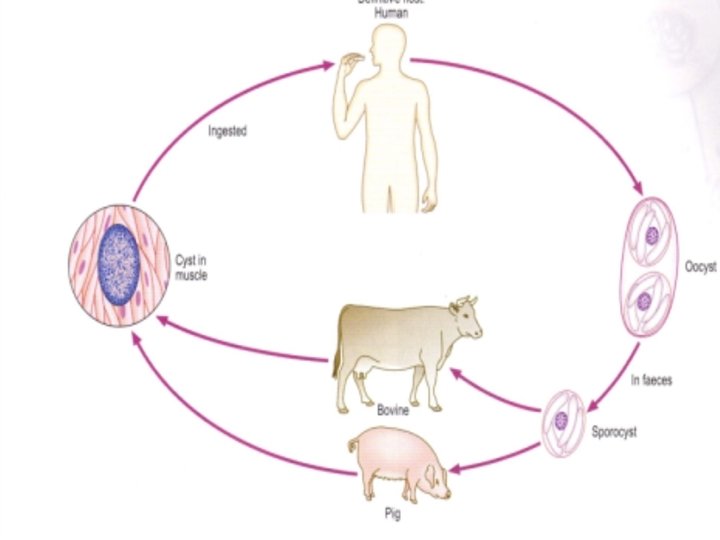

Disease: Sarcocystosis = disease uncommon and rare in human = considered as zoonotic = domestic animals are intermediate host of the parasite that pick up infective cyst while grazing on grasses contaminated with human excreta

Disease: Sarcocystosis = human infection are acquired through ingestion of uncooked meat (beef, pork, lamb) or contaminated food and drink containing the infected sarcocyst = symptoms includes diarrhea, nausea, vomiting, abdominal pain which occurs 1 – 2 days after ingestion and may last for 2 weeks

Lab. Diag. : 1. Demonstration of oocyst in feces / duodenal aspirate 2. Biopsy of tissue of small intestine or colon demonstrate Meischer tube Treatment: = None for tissue infection (Supportive – self-limiting) = For intestinal infection: Trimethoprim + Sulfamethoxazole Pyrimethamine + Sulfadiazine (alternate drug) Prevention: = Avoid contact with infected animal host = Adequate cooking of all meat

Laboratory Diagnosisof Intestinal coccidia • Microscopy • Immunological assay • Molecular methods

General guideline • Safety – C. parvum Oocysts in stool remain infective for extended periods, thus – specimens should be preserved in 10% buffered formalin or (SAF) to render oocysts nonviable( 18 to 24 hours).

Specimen processing: • Fresh Specimens should be submited to the laboratory as rapidly as possible. • If it is not possible it should be preserved. – fixed in 10% formalin (for direct microscopy, concentration procedures, and preparation of stained smears); – fixed in 2. 5% potassium dichromate (for sporulation assays and molecular diagnosis); and – Frozen without fixation (for molecular diagnosis).

Specimen processing…. • Oocysts can be excreted intermittently and vary in numbers. Thus: • A single negative stool specimen does not rule out the diagnosis; • Three or more specimens at 2 - or 3 -day intervals may be required • Concentration procedures should be used – formalin-ethyl acetate sedimentation technique – Sheather’s flotation procedure can also be used.

Cryptosporidium species Macroscopy • Stool spesimen –Have offensive smell –Usually watery and do not contain pus cell
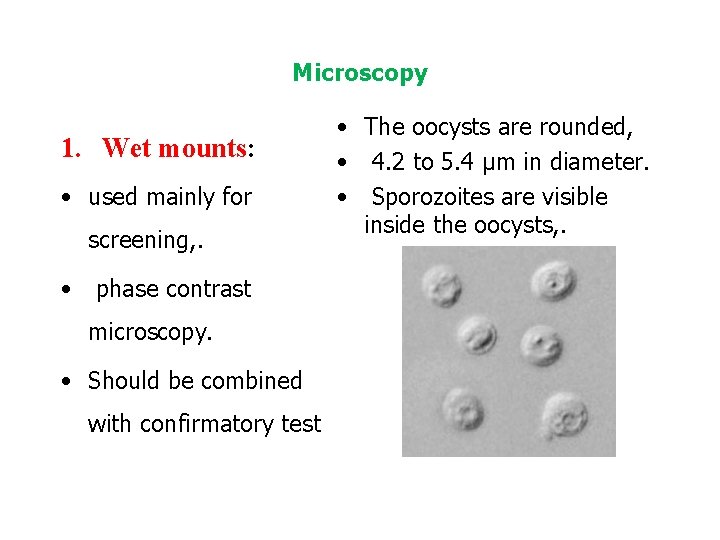
Microscopy 1. Wet mounts: • used mainly for screening, . • phase contrast microscopy. • Should be combined with confirmatory test • The oocysts are rounded, • 4. 2 to 5. 4 µm in diameter. • Sporozoites are visible inside the oocysts, .
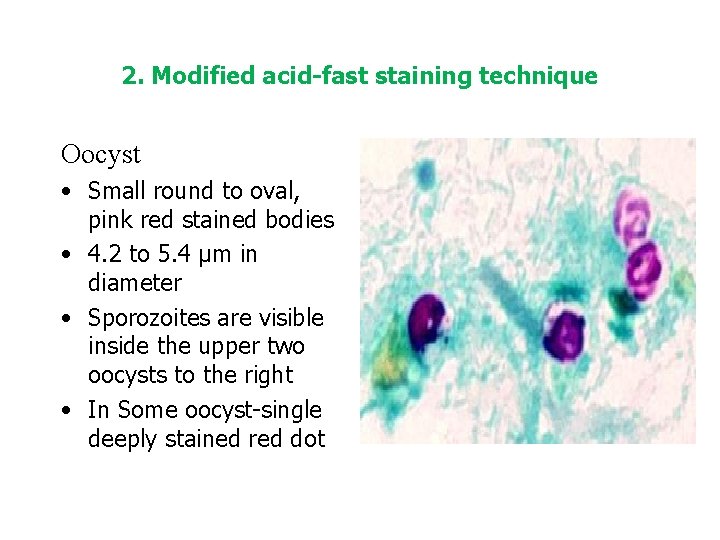
2. Modified acid-fast staining technique Oocyst • Small round to oval, pink red stained bodies • 4. 2 to 5. 4 µm in diameter • Sporozoites are visible inside the upper two oocysts to the right • In Some oocyst-single deeply stained red dot
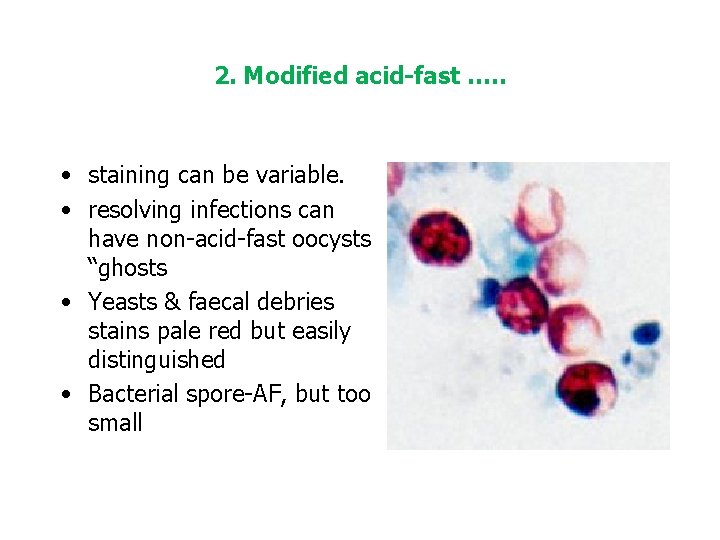
2. Modified acid-fast …. . • staining can be variable. • resolving infections can have non-acid-fast oocysts “ghosts • Yeasts & faecal debries stains pale red but easily distinguished • Bacterial spore-AF, but too small

• 4 -5 mm oocysts
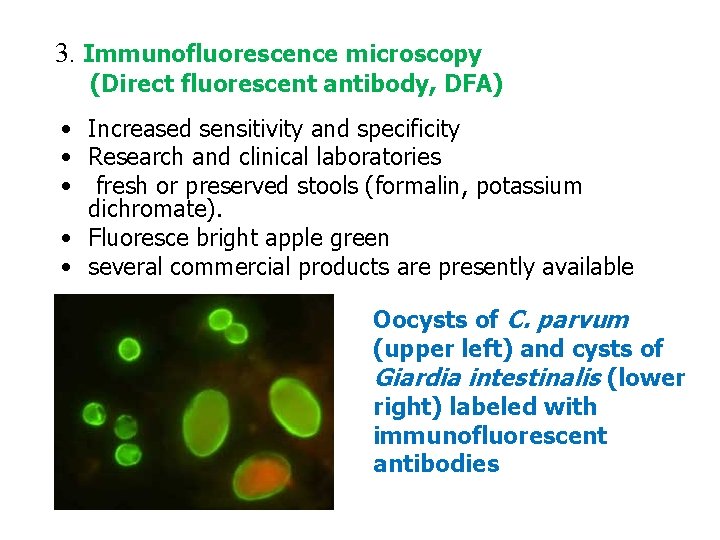
3. Immunofluorescence microscopy (Direct fluorescent antibody, DFA) • Increased sensitivity and specificity • Research and clinical laboratories • fresh or preserved stools (formalin, potassium dichromate). • Fluoresce bright apple green • several commercial products are presently available Oocysts of C. parvum (upper left) and cysts of Giardia intestinalis (lower right) labeled with immunofluorescent antibodies

4) additional methods for microscopic detection of oocysts include • Alternate bright-field stains – Hot safranin-methylene blue stain, – Fluorescent stains (including auramine O, auraminerhodamine, auramine-carbolfuchsin, acridine orange, ) • Higher sensetivity but non specific • Used for screening Oocysts of C. parvum stained with the fluorescent stain auramine-rhodamine

Antigen detection • Enzyme immunoassays (EIA) – EIA test for the detection of cryptosporidial antigens in stool samples. – Highly sensitive and specific method – are reportedly superior to conventional microscopic
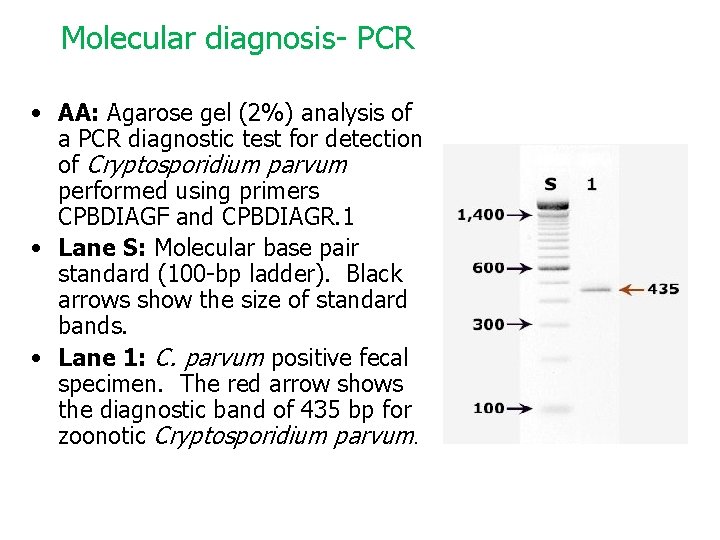
Molecular diagnosis- PCR • AA: Agarose gel (2%) analysis of a PCR diagnostic test for detection of Cryptosporidium parvum performed using primers CPBDIAGF and CPBDIAGR. 1 • Lane S: Molecular base pair standard (100 -bp ladder). Black arrows show the size of standard bands. • Lane 1: C. parvum positive fecal specimen. The red arrow shows the diagnostic band of 435 bp for zoonotic Cryptosporidium parvum.

Cyclospora cayetanensis • Laboratory Diagnosis: – Microscopy – Sporulation assay – Molecular methods – Microscopic examination: • wet mounts (by conventional light microscopy, UV fluorescence microscopy or differential interference contrast (DIC) • stained smears (using modified acid-fast stain or a modified safranin stain)

Microscopy • Wet mounts – Phase contrast (DIC) microscope • oocysts as nonrefractile spheres • contain undifferentiated cytoplasm or • refractile globules (D) • 8 to 10 µm in diameter

• UV fluorescence microscopy – the oocysts autofluoresce under ultraviolet light (E) – The walls of the oocysts fluoresce brightly, while their interiors do not, – differentiates Cyclospora from other objects of similar size and shape. – UV fluorescence microscopy is a sensitive technique for rapidly examining stool sediments.

Stained smear • Modified acid fast stains – Round irregularly stained – 8 -10µm – Offen contain red staining granules – Some may appear unstained glassy wrinkled spheres
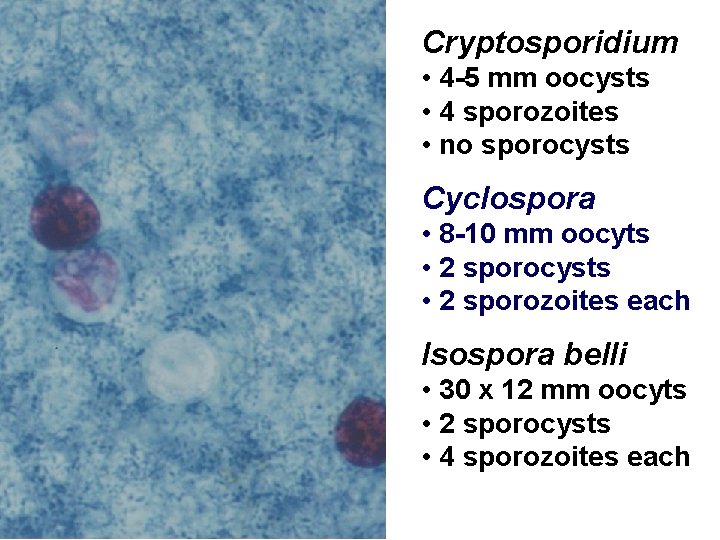
Cryptosporidium • 4 -5 mm oocysts • 4 sporozoites • no sporocysts Cyclospora • 8 -10 mm oocyts • 2 sporocysts • 2 sporozoites each Isospora belli • 30 x 12 mm oocyts • 2 sporocysts • 4 sporozoites each
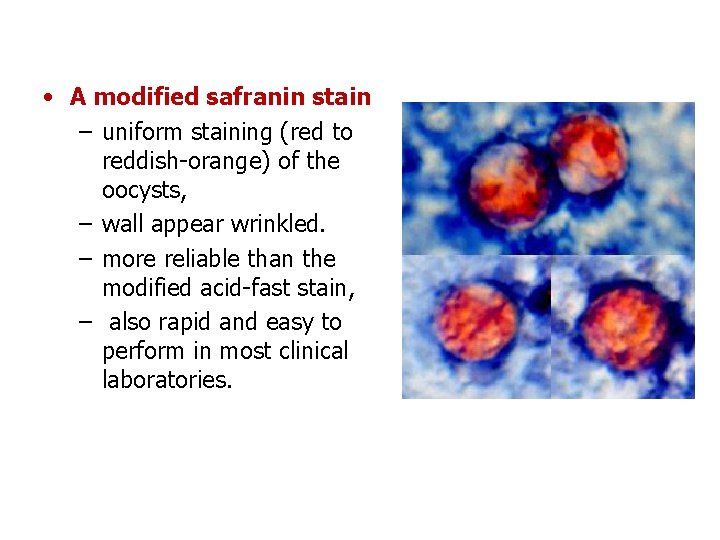
• A modified safranin stain – uniform staining (red to reddish-orange) of the oocysts, – wall appear wrinkled. – more reliable than the modified acid-fast stain, – also rapid and easy to perform in most clinical laboratories.
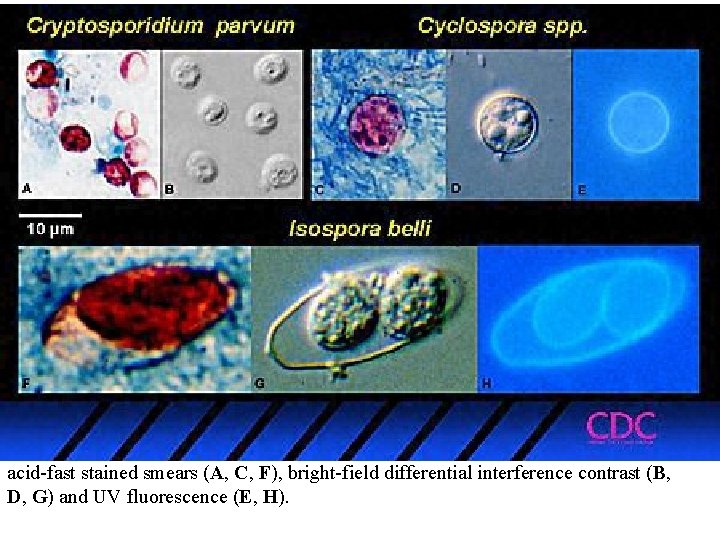
acid-fast stained smears (A, C, F), bright-field differential interference contrast (B, D, G) and UV fluorescence (E, H).

Sporulation Assay • Unfixed oocysts is examined over a 2 - to 3 - week period for evidence of sporulation • Accomplished by placing an aliquot of fresh stool in 2. 5% potassium dichromate • Used to differentiate between freshly passed, unsporulated oocysts and blue-green algae • to confirm the diagnosis of cyclosporiasis,
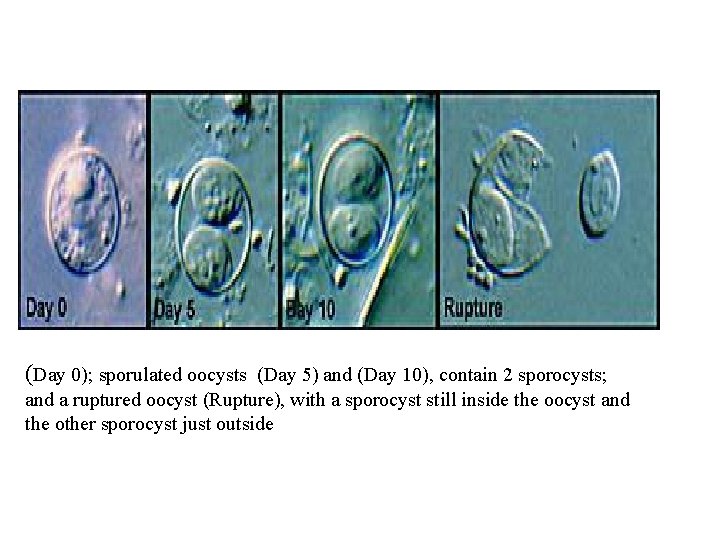
(Day 0); sporulated oocysts (Day 5) and (Day 10), contain 2 sporocysts; and a ruptured oocyst (Rupture), with a sporocyst still inside the oocyst and the other sporocyst just outside
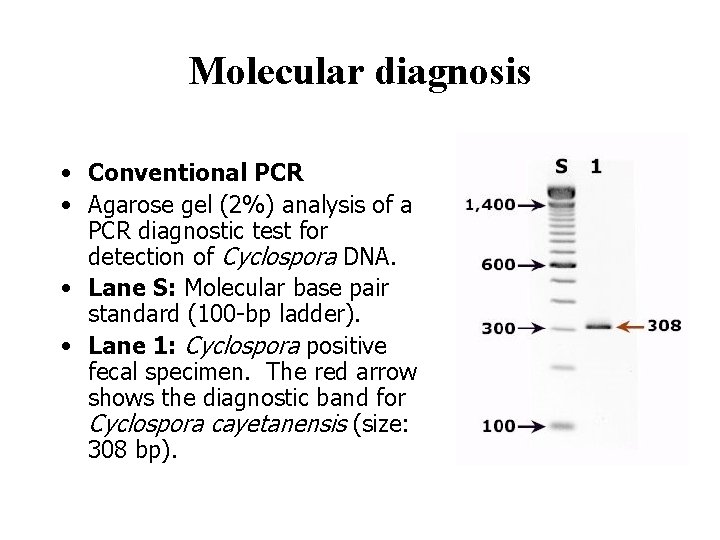
Molecular diagnosis • Conventional PCR • Agarose gel (2%) analysis of a PCR diagnostic test for detection of Cyclospora DNA. • Lane S: Molecular base pair standard (100 -bp ladder). • Lane 1: Cyclospora positive fecal specimen. The red arrow shows the diagnostic band for Cyclospora cayetanensis (size: 308 bp).
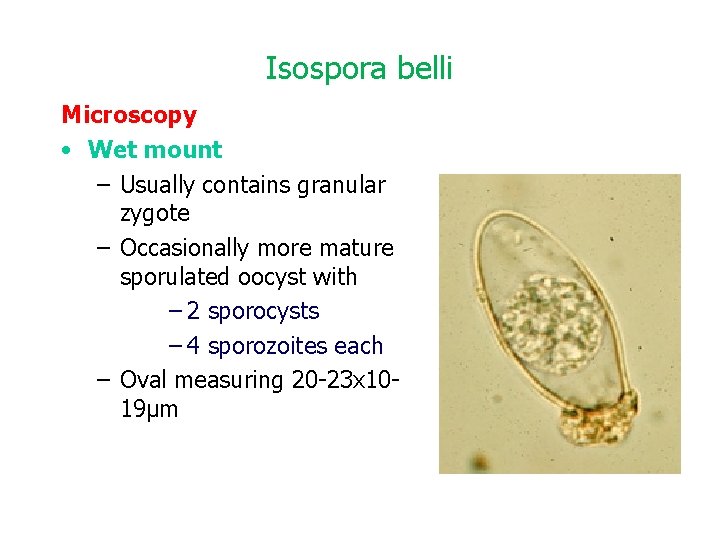
Isospora belli Microscopy • Wet mount – Usually contains granular zygote – Occasionally more mature sporulated oocyst with – 2 sporocysts – 4 sporozoites each – Oval measuring 20 -23 x 1019µm
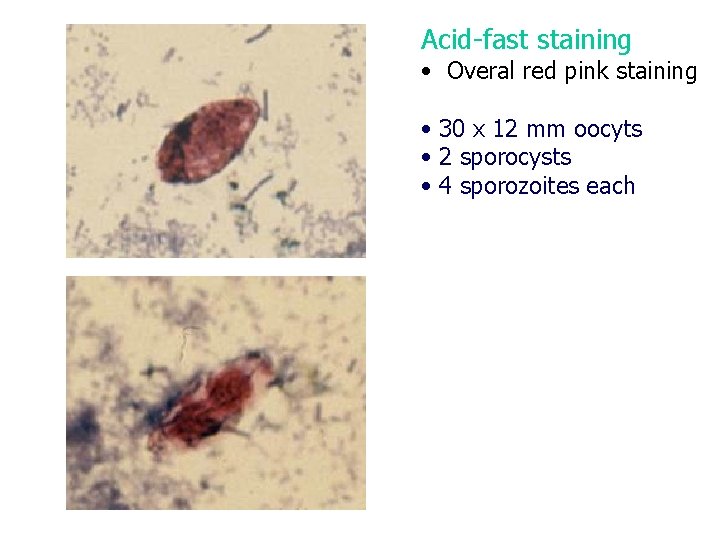
Acid-fast staining • Overal red pink staining • 30 x 12 mm oocyts • 2 sporocysts • 4 sporozoites each
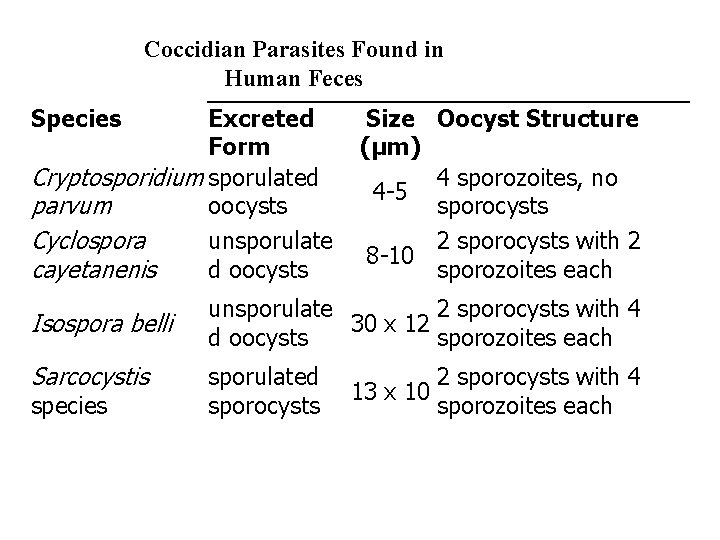
Coccidian Parasites Found in Human Feces Species Excreted Form Cryptosporidium sporulated parvum oocysts Cyclospora unsporulate cayetanenis d oocysts Isospora belli Sarcocystis species Size Oocyst Structure (µm) 4 sporozoites, no 4 -5 sporocysts 2 sporocysts with 2 8 -10 sporozoites each unsporulate 2 sporocysts with 4 30 x 12 d oocysts sporozoites each sporulated sporocysts 13 x 10 2 sporocysts with 4 sporozoites each

Intestinal Coccidia Treatment • paromomycin for Cryptosporidium • modest benefit • lowers parasitemia in AIDS • trimethoprim-sulfamethoxazole for Cyclospora and Isospora

Intestinal coccidian • Summary – – – are opportunistic parasite in immunocompromissed person Have both sexual and asexual reproduction Require single host to comlte their entire life cycle characterized by a thick walled oocyst excreted in faeces Are transmitted by the fecal-oral route Laboratory diagnosis is by finding the oocyst in faeces

Intestinal Coccidia- Summary • Obligate intracellular • Got medical attention after the emergence of HIV/AIDS • Fecal-oral or contaminated food and water are means of transmission • Simple lifecycle and reproduce Sexually and asexually • Infection leads to acute, watery diarrhea; self limiting in • immunocompetent individuals but significant, chronic illness in the immunosuppressed may occur • oocyst stage : Infective and diagnostic stage • Laboratory dx; finding oocyct in stool specimen
- Slides: 80