AP Notes Chapter 9 Hybridization and the Localized

AP Notes Chapter 9 Hybridization and the Localized Electron Model Valence Bond Theory Molecular Orbital Theory Metals & Semiconductors
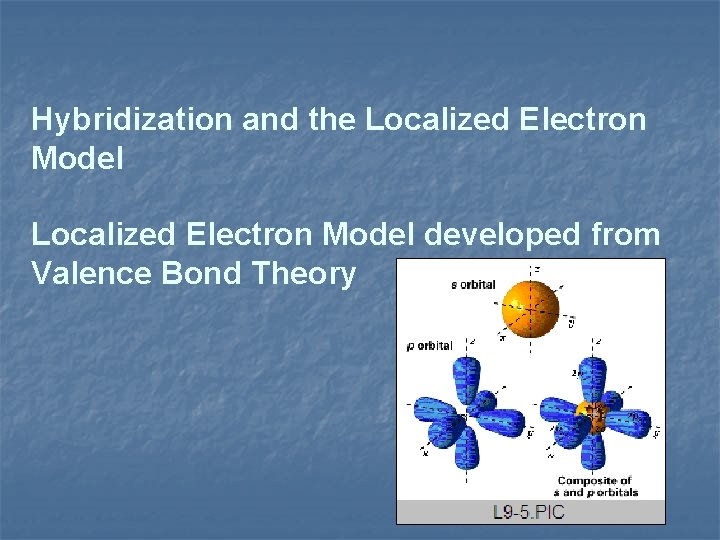
Hybridization and the Localized Electron Model developed from Valence Bond Theory

Why do we need it? n Consider the water species. n. H 1 s 1 1 s ___ 1 H 1 s O 1 s 2 2 p 4 2 s ___ 2 p ___ ___ Gives 2 H’s with no e- and O with full octet.

We Get n. H 1 s 1 1 s ___ 1 H 1 s O 1 s 2 2 p 4 2 s ___ 2 p ___ ___ Gives 2 H’s with no e- and O with full octet.

Hybridization n n Process that changes properties of valence electrons by mixing atomic orbitals to form special orbitals for bonding atomic orbitals AO molecular orbitals MO

Principles 1. Conservation of orbitals 2. Hybrid correlates with molecular geometry 3. Energy level of MO is between that of AO’s 4. All bonded atoms hybridize
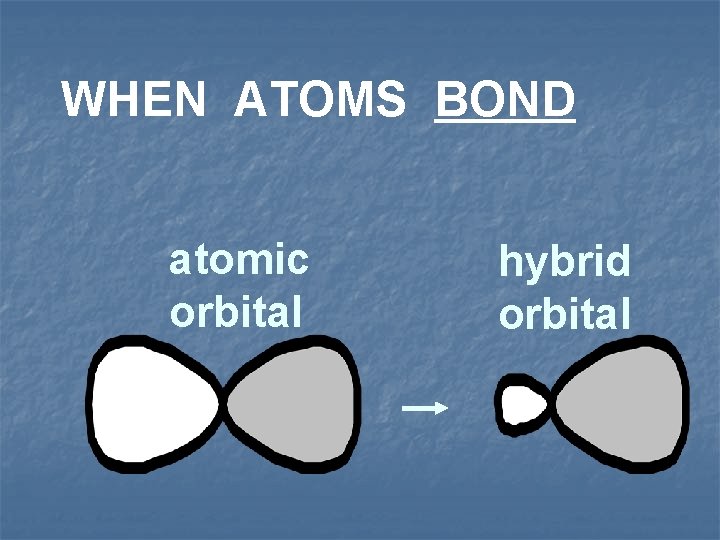
WHEN ATOMS BOND atomic orbital hybrid orbital

All hybrid orbitals of an atom are said to be DEGENERATE (of equal energy)
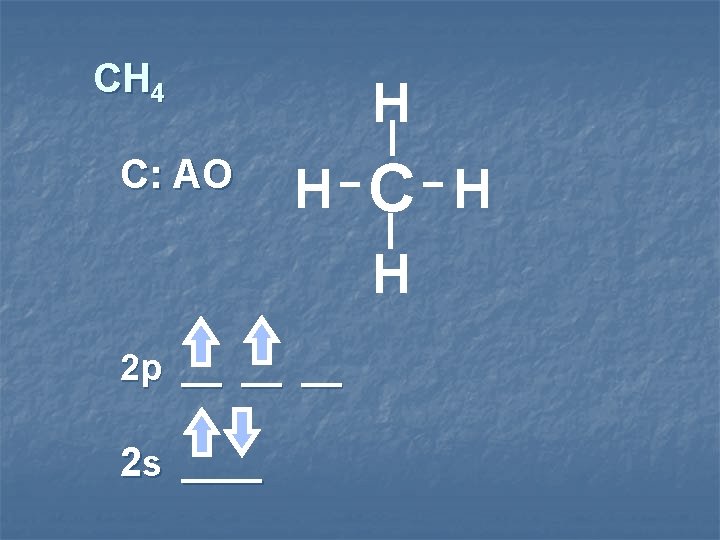
CH 4 C: AO H H C H H 2 p __ __ __ 2 s ____
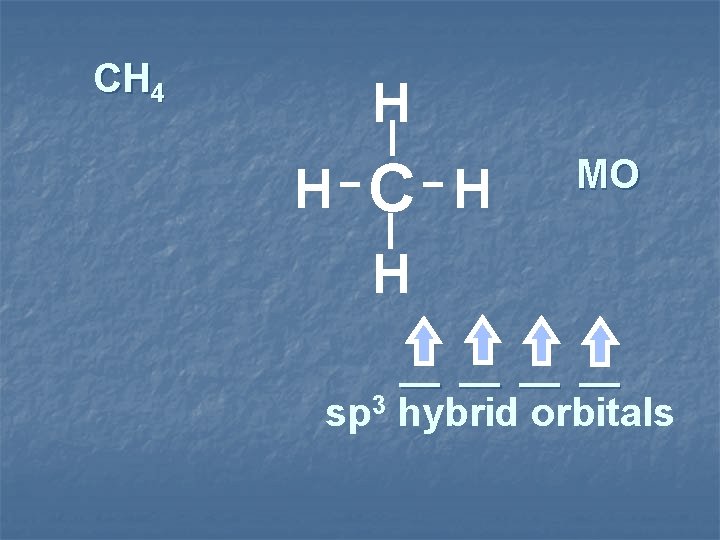
CH 4 H H C H MO H __ __ sp 3 hybrid orbitals

3 sp hybridization sp 3 hybrid orbitals tetrahedral species sp 3 shape tetragonal 4 Items Equally Distributed
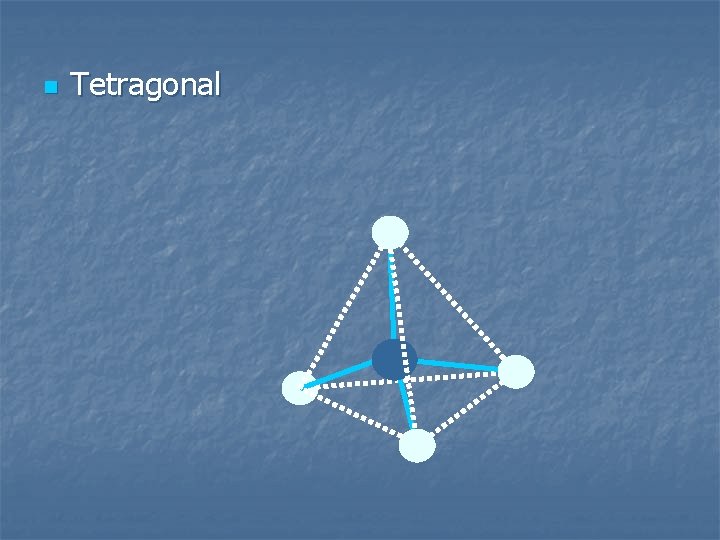
n Tetragonal
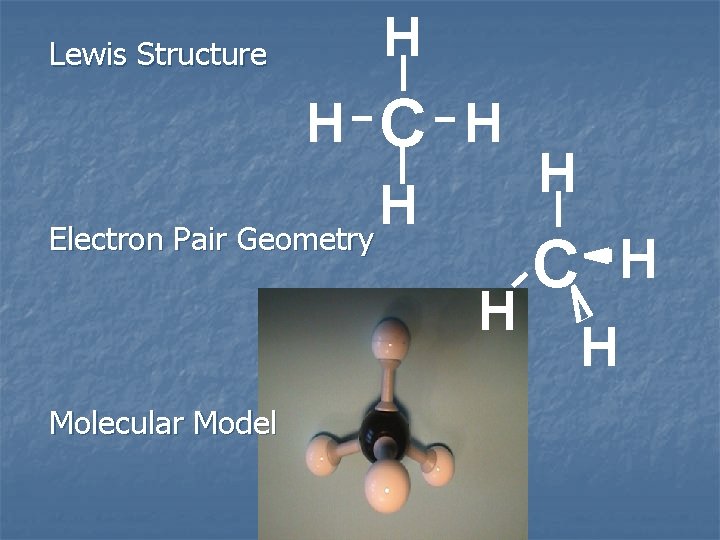
H Lewis Structure H C H H Electron Pair Geometry H Molecular Model H C H H
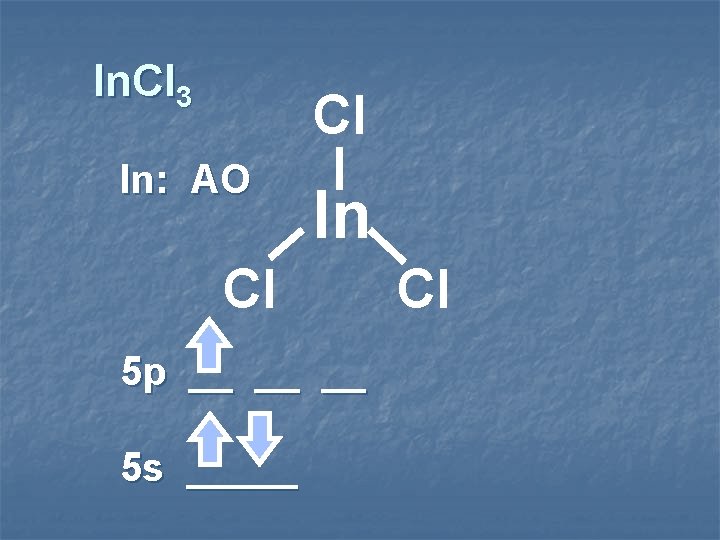
In. Cl 3 Cl In: AO In Cl 5 p __ __ __ 5 s _____ Cl
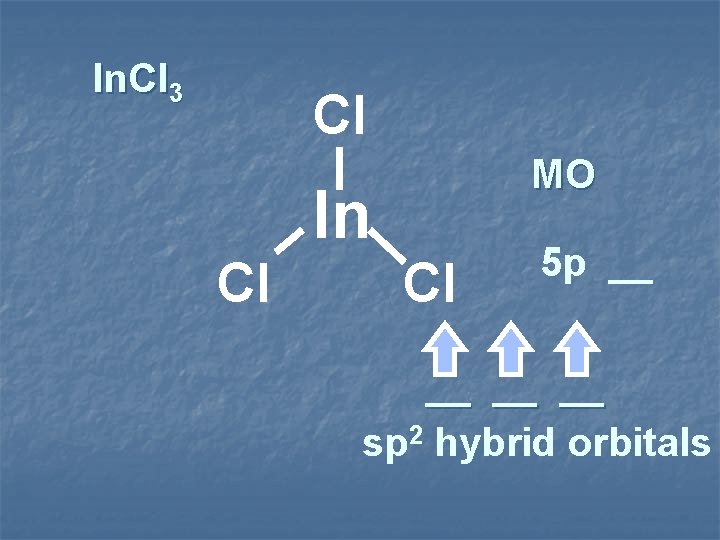
In. Cl 3 Cl MO In Cl Cl 5 p __ __ sp 2 hybrid orbitals

2 sp hybridization 2 sp hybrid trigonal planar species 2 sp shape 3 Items Equally Distributed
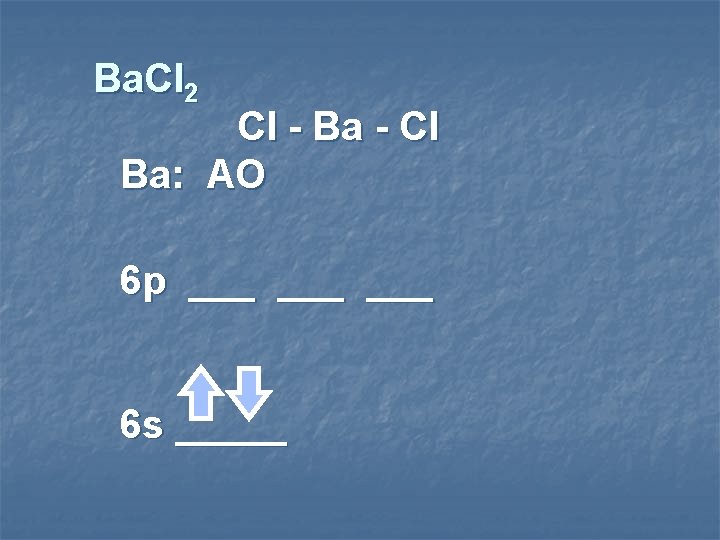
Ba. Cl 2 Cl - Ba - Cl Ba: AO 6 p ___ ___ 6 s _____
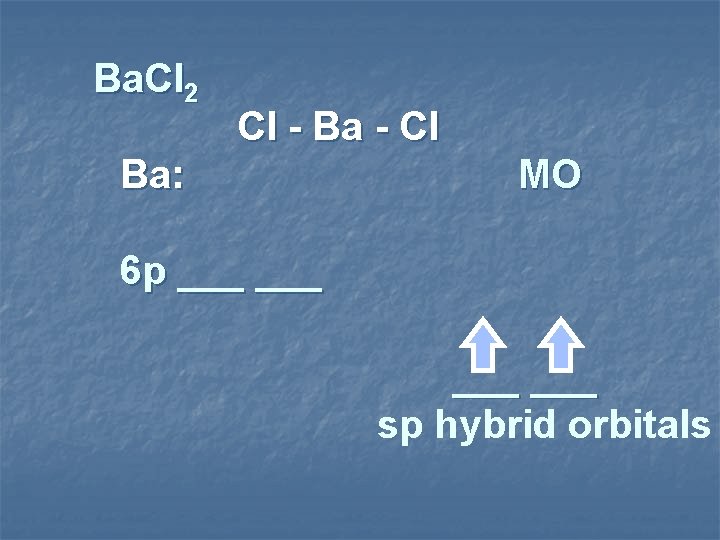
Ba. Cl 2 Cl - Ba - Cl Ba: MO 6 p ___ ___ sp hybrid orbitals

sp hybridization sp hybrid linear species sp shape 2 Items Equally Distributed
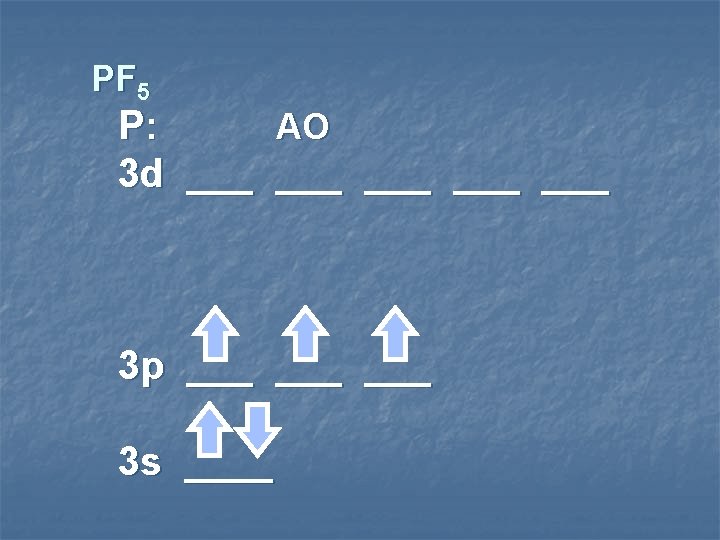
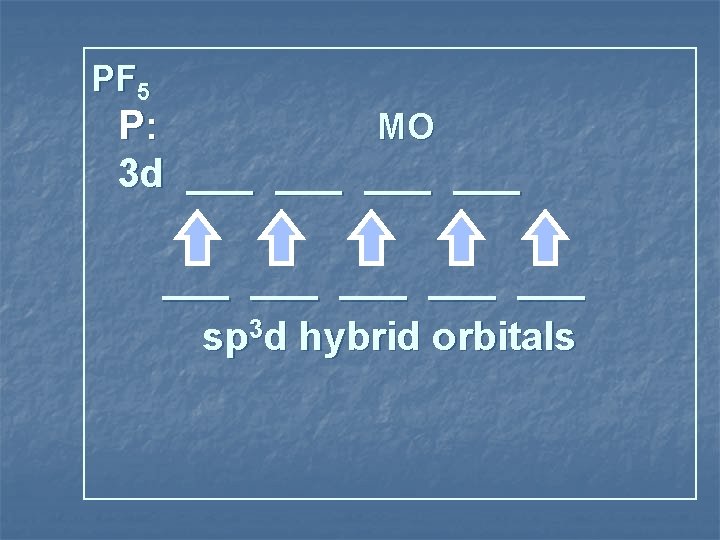
PF 5 P: MO 3 d ___ ___ ___ sp 3 d hybrid orbitals

3 sp d hybridization sp 3 d shape trigonal bipyramid species 5 Items Equally Distributed
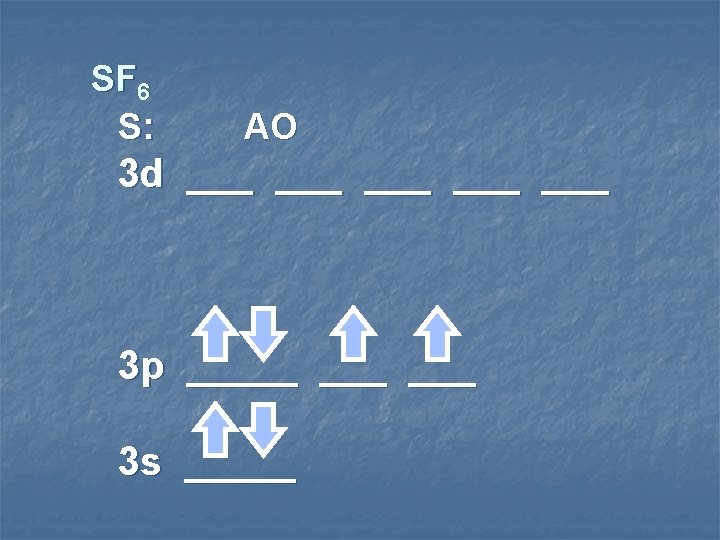
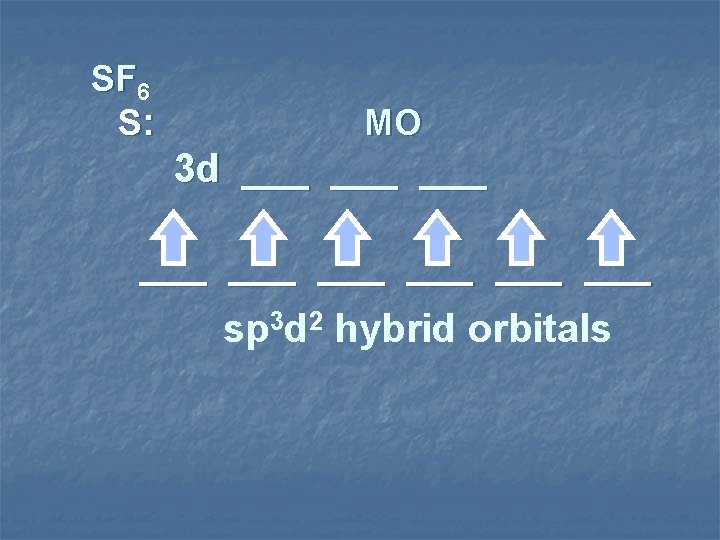
SF 6 S: MO 3 d ___ ___ ___ sp 3 d 2 hybrid orbitals

3 2 sp d hybridization sp 3 d 2 shape octahedral species 6 Items Equally Distributed

Multiple Bonds sigma bonds ( ) pi bonds ( )

n O 2 EXAMPLES O 1 s 2 2 p 4 2 s 2 1 s 2 O 2 s ___ 2 p ___ ___ ___ 2 s ___ ___ 2 p-orbitals touching end to end sigma - σ 2 p-electrons reaching over and under pi - π

Valence Bond Theory Multiple Bond Examples n. C 2 H 4 (ethylene) 3 n (sp hybridization) n ( n both bonding)
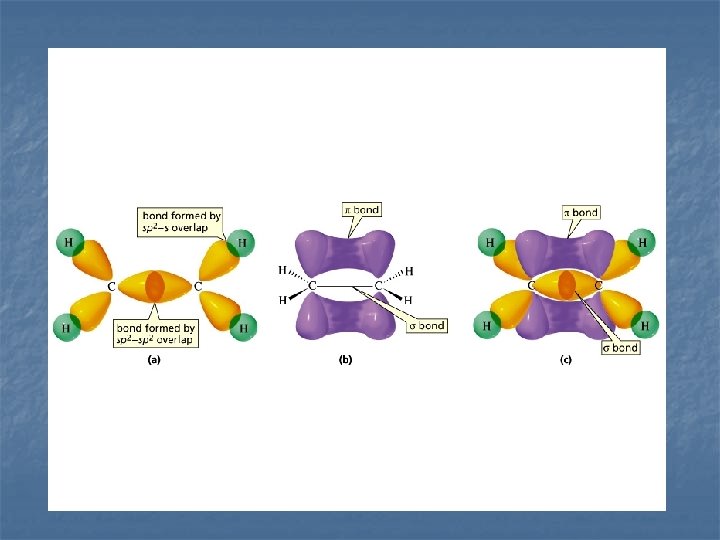

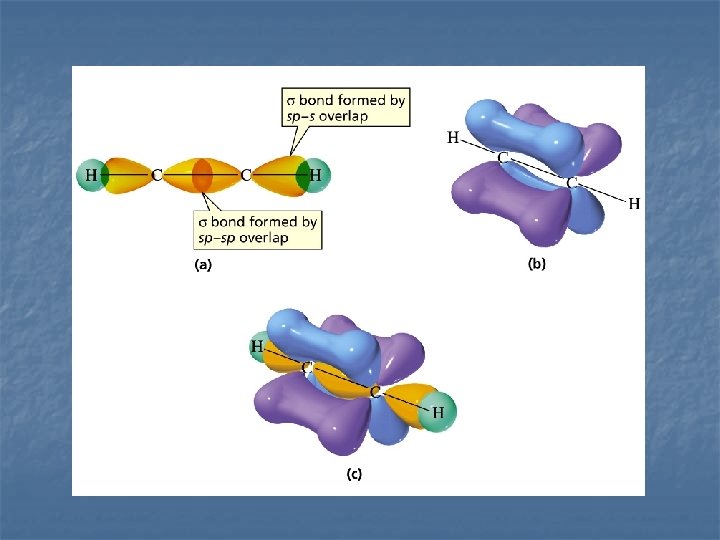
EXAMPLES n C 2 H 2

EXAMPLES n. CH 3 COOH

MOLECULAR ORBITAL MODEL Valence Bond Theory concentrates on individual bonds in a molecule and tends to ignore electrons not used in bonding.

Molecular Orbital Theory assumes ALL the orbitals of the atoms are able to take part in bonding.

Every atom has a complete set of orbitals, but not all of them contain electrons

Remember that orbitals are really the solutions of Schrodinger’s equation, and that they are called wave-functions
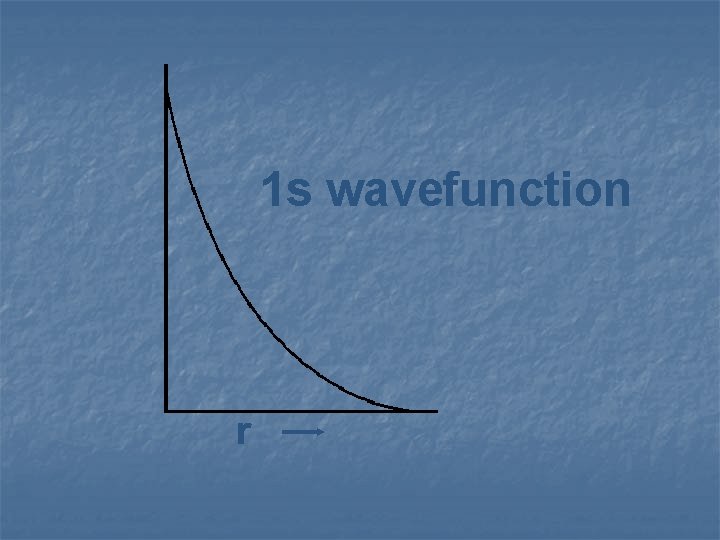
1 s wavefunction r
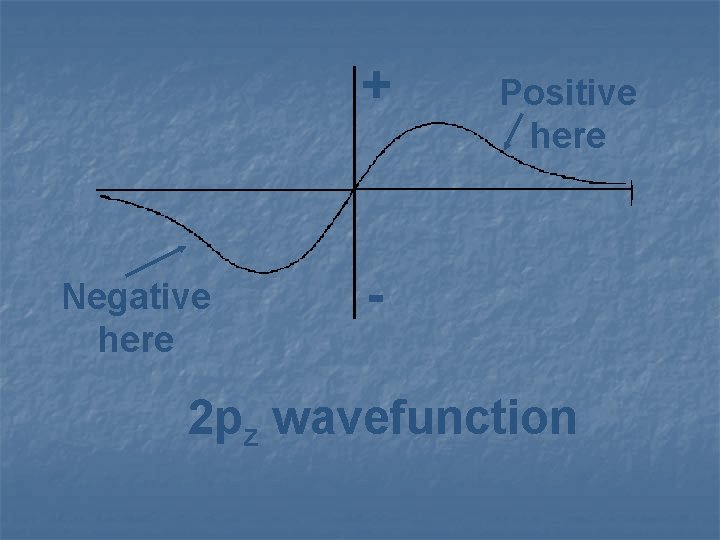
+ Negative here Positive here - 2 pz wavefunction
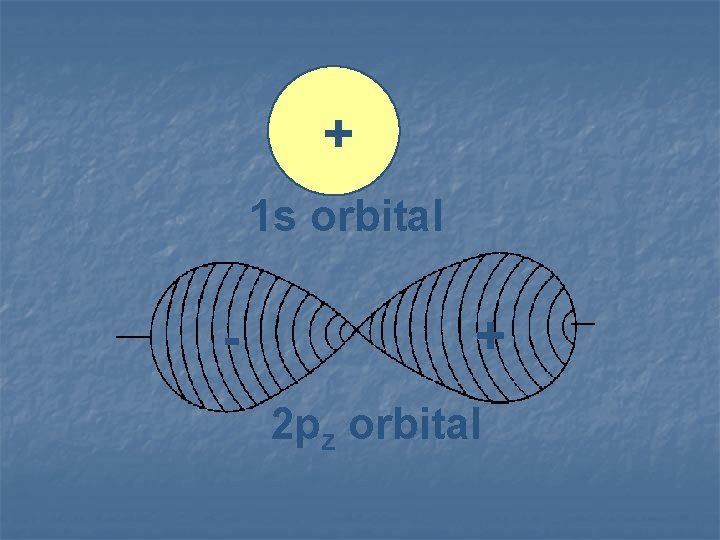
+ 1 s orbital - + 2 pz orbital

While wave functions can be positive or negative, probabilities can only be positive.

Wave functions, like waves, can overlap with one another. They can reinforce each other, or they cancel each other out.
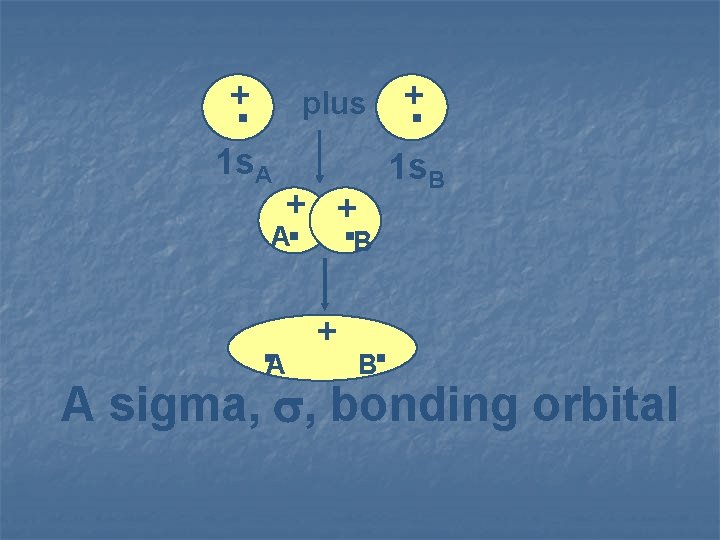
. + plus 1 s. A 1 s. B + + . . B A . A + . B A sigma, , bonding orbital
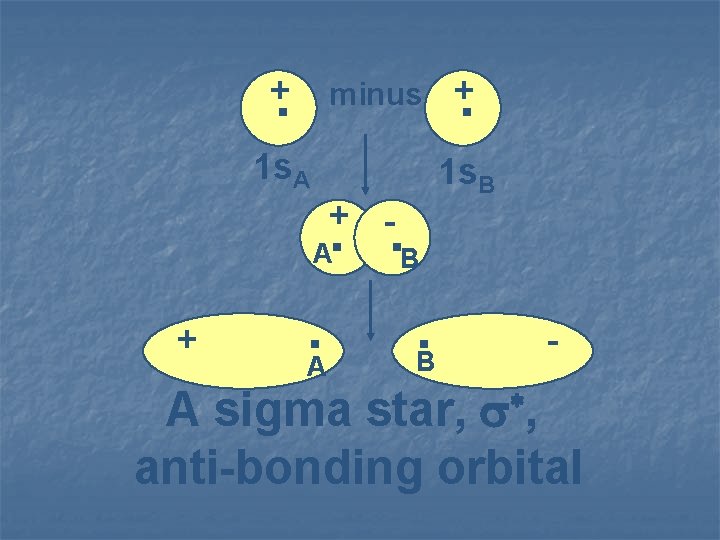
. . + minus + 1 s. A + - . . B 1 s. B A + . A . B * , A sigma star, anti-bonding orbital

The work on molecular orbitals can be generalized to p-orbitals.
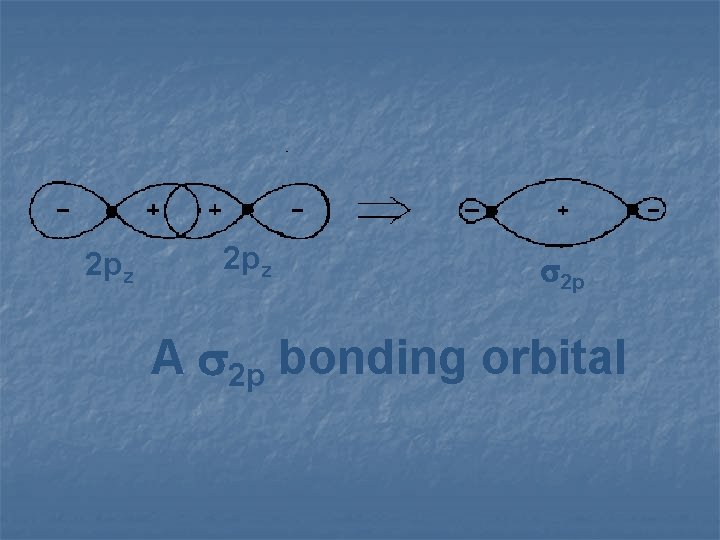
2 pz 2 p A 2 p bonding orbital
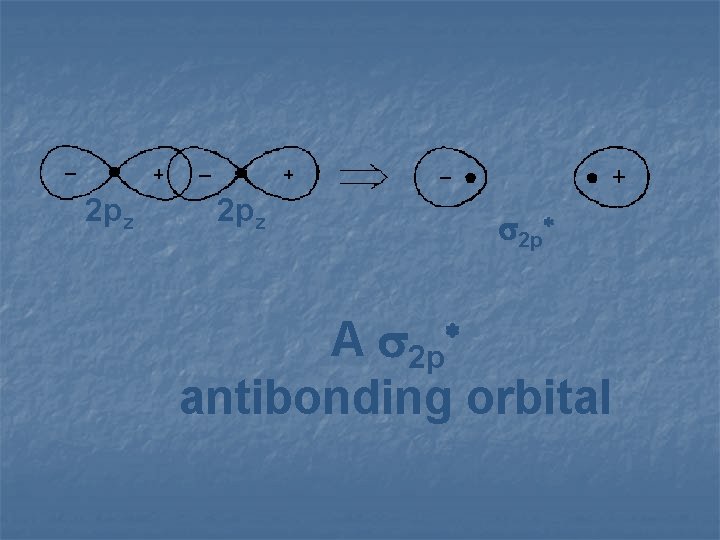
2 pz 2 p* A 2 p antibonding orbital *
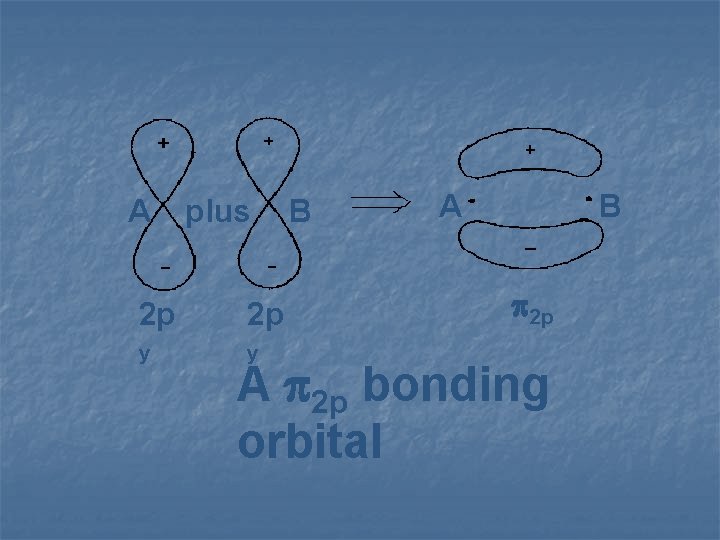
A plus 2 p 2 p y y B A B 2 p A 2 p bonding orbital
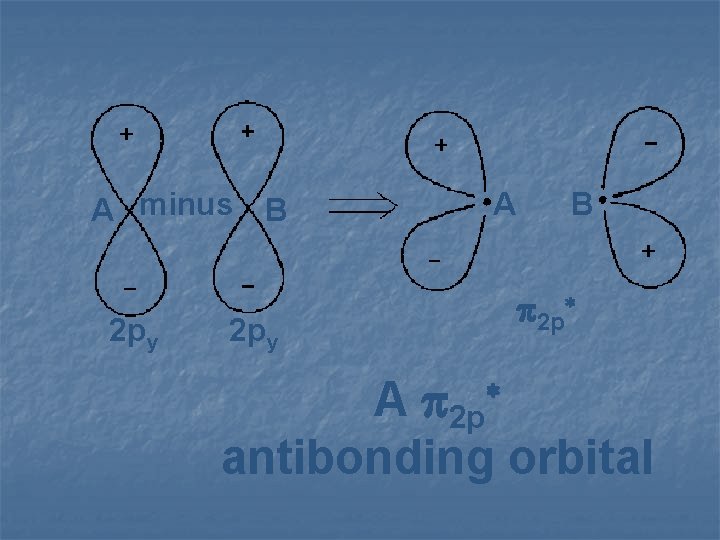
A minus B 2 py A B 2 p* 2 py A 2 p antibonding orbital *

Many combinations of orbitals can produce bonding and antibonding molecular orbitals, s with p, d with p, etc.

Orbitals on the two bonding atoms must meet 2 conditions n They must be similar in energy n They must have the right symmetry

plus 2 pz 2 py Orbitals pointing in different directions cannot overlap to form molecular orbitals.

Molecular Orbital Theory 1. Molecular orbitals are made from atomic orbitals 2. Orbitals are conserved 3. Molecular orbitals form in pairs: bonding & antibonding

Bonding Molecular Orbital n. Geometry overlap favorable to

When a bonding orbital is formed, the energy of the orbital is lower than those of its parent atomic orbitals.

Anti-bonding Molecular Orbital n. Geometry not favorable to overlap

Similarly, when an antibonding orbital is formed, the energy of the orbital is higher than those of its parent atomic orbitals.

Molecular Orbital Diagrams Bond Order

Examine some homonuclear diatomic molecules n Hydrogen n Helium
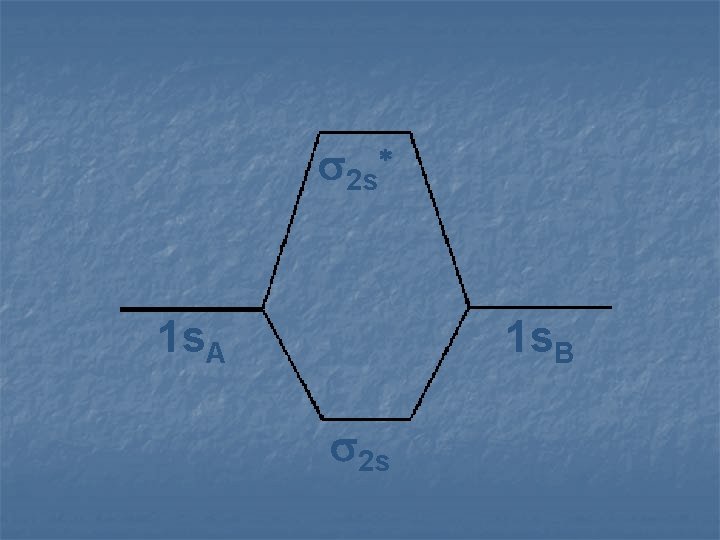
2 s* 1 s. A 1 s. B 2 s

Paramagnetic 1. Responds to magnetic field 2. Has unpaired electrons

Diamagnetic 1. Does not respond to magnetic field 2. All electron paired
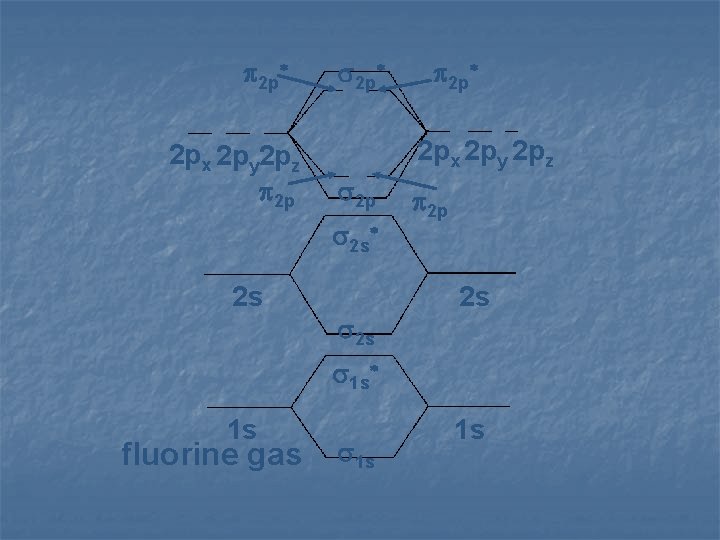
2 p* 2 px 2 py 2 pz 2 p 2 s* 2 s 2 p* 2 s 2 p 2 s 1 s* 1 s fluorine gas 1 s 1 s
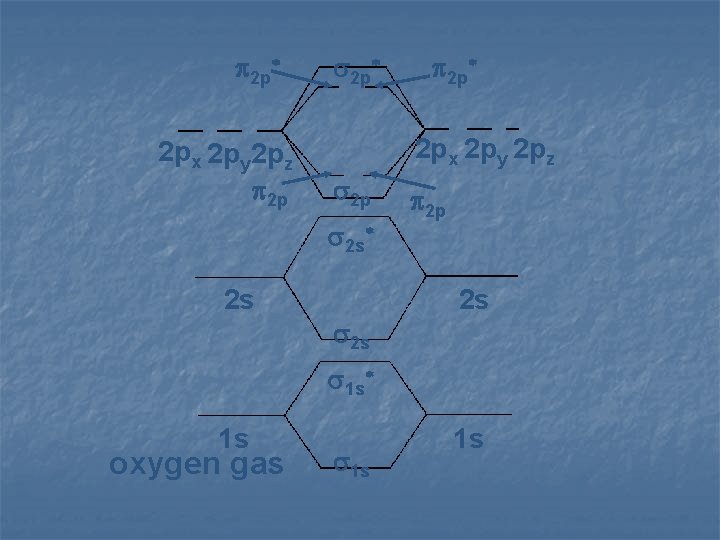
2 p* 2 px 2 py 2 pz 2 p 2 s* 2 s 2 p* 2 s 2 p 2 s 1 s* 1 s oxygen gas 1 s 1 s
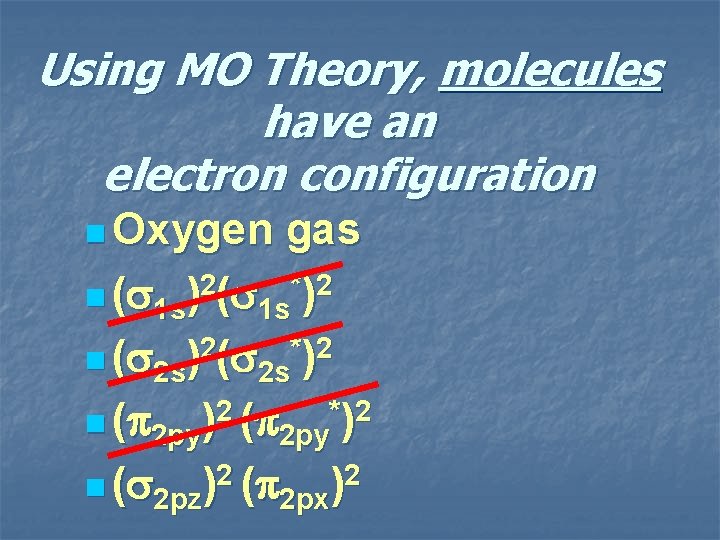
Using MO Theory, molecules have an electron configuration n Oxygen gas n ( 1 s)2( 1 s*)2 n ( 2 s)2( 2 s*)2 n ( 2 py)2 ( 2 py*)2 n ( 2 pz)2 ( 2 px)2
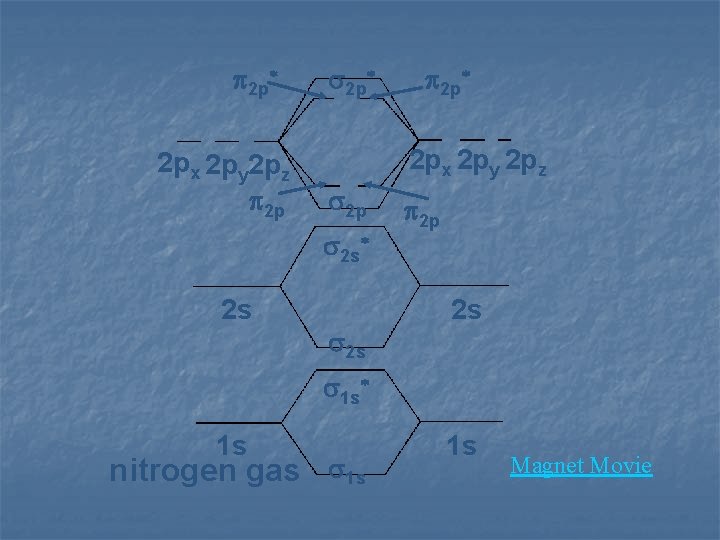
2 p* 2 px 2 py 2 pz 2 p 2 s* 2 s 2 p* 2 s 2 p 2 s 1 s* 1 s nitrogen gas 1 s 1 s Magnet Movie

Bond Strength Bond Length

Strengths of Localized Electron Model 1. Simple 2. Easy to understand 3. Predicts geometry of molecule

Limitations of Localized Electron Model 1. Does not address concept of resonance or unpaired e 2. Cannot explain color in transition metal compounds

Strengths of Molecular Orbital Model 1. Better represents actual molecular system 2. Provides basis for explaining properties of molecular systems

Limitations of Molecular Orbital Model 1. MO diagrams are complex. 2. MO diagrams are difficult for molecules with more than two atoms. 3. No prediction of geometry

Combining the Localized Electron and Molecular Orbital Models

Draw the Lewis structure of benzene C 6 H 6 Lewis Structure
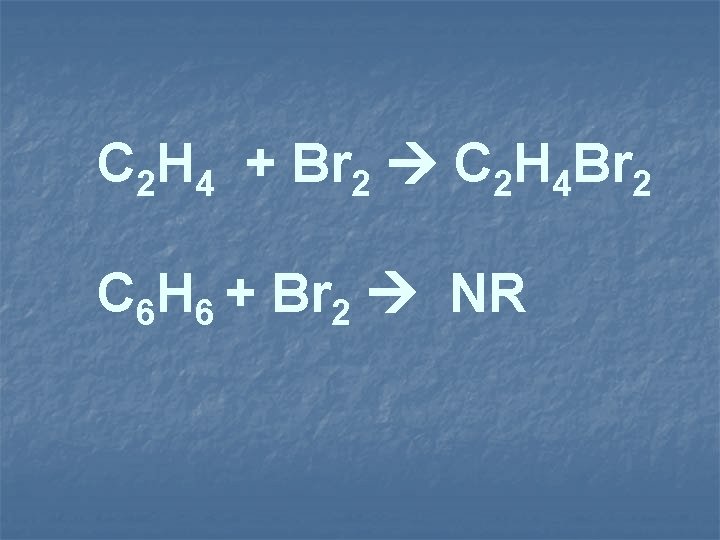
C 2 H 4 + Br 2 C 2 H 4 Br 2 C 6 H 6 + Br 2 NR

bonds in benzene

Isomerism n n Isomers – two or more compounds with same molecular formula but different arrangements of atoms Cis – Trans Isomerism (NOT mirror images of each other NOT super imposable. Cis Trans
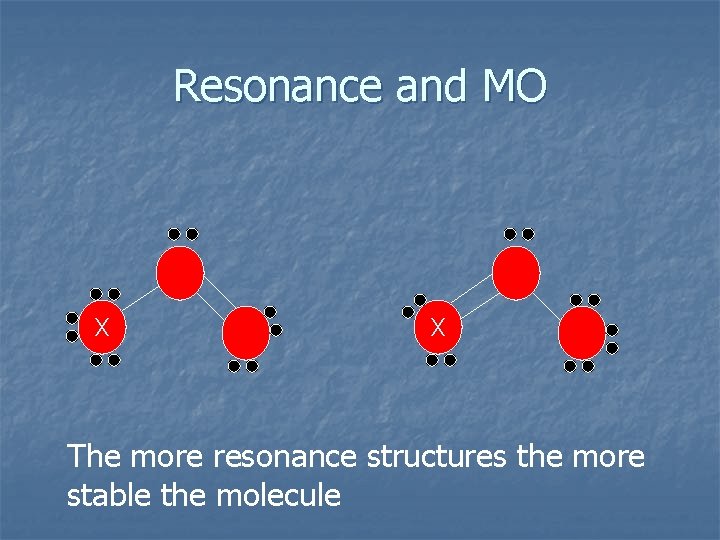
Resonance and MO X X The more resonance structures the more stable the molecule

Metals & Semiconductors Read pg 657 -669 n Study Figures 1 -23 Know n Insulators Conductors, Semiconductors – intrinsic, extrinsic Dopants
- Slides: 77