AP Chemistry The Gas Laws Basics on Gases

AP Chemistry The Gas Laws

Basics on Gases composition of the atmosphere: ~78% N 2, ~21% O 2 properties of gases: expand to fill container compressible form homogeneous mixtures -- due to gas particles being… 1) far apart 2) in constant, random motion vapors: gases of substances that are normally liquids or solids e. g. , gasoline vapors

Equation for pressure: F N A m 2 P N/m 2 = Pa 1 atm… = 760 mm Hg = 760 torr = 101. 325 k. Pa = 1. 01325 bar (1 bar = 105 Pa) At a depth of 350 m (1150 ft), the hull pressure on a submarine is 3. 4 x 106 Pa (36 tons/ft 2).
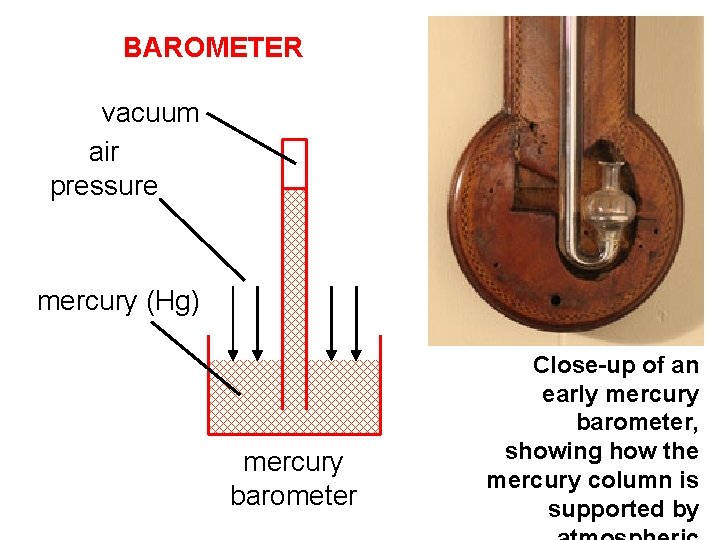
BAROMETER vacuum air pressure mercury (Hg) mercury barometer Close-up of an early mercury barometer, showing how the mercury column is supported by

An aneroid barometer contains a small, pressure-sensitive metal box that has been evacuated of air. The box is prevented from collapsing by being connected to a spring that is also attached to the dial on the barometer. When the air pressure on the walls of the box changes, the box “flexes, ” which moves the spring and the dial.
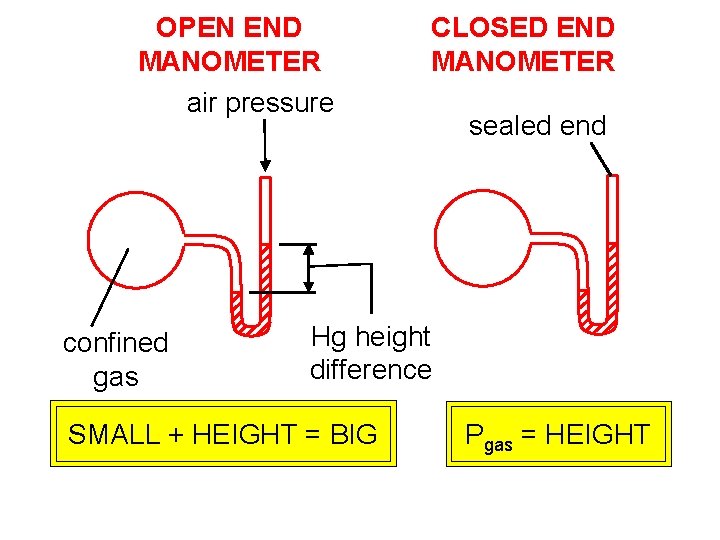
OPEN END MANOMETER air pressure confined gas CLOSED END MANOMETER sealed end Hg height difference SMALL + HEIGHT = BIG Pgas = HEIGHT
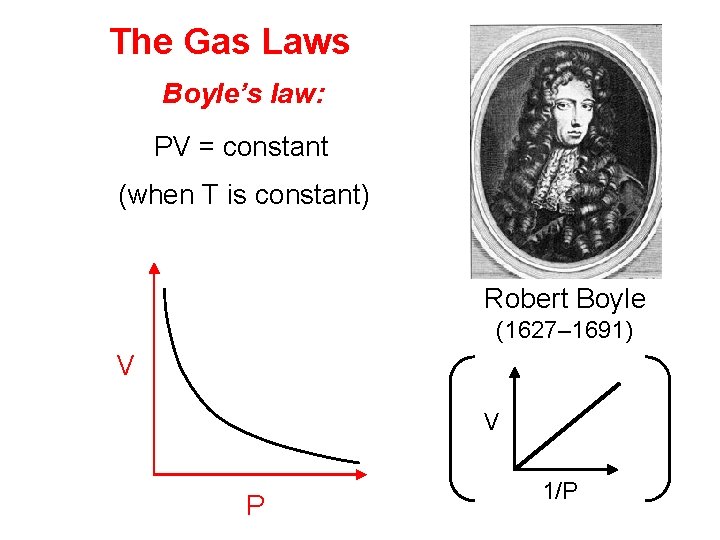
The Gas Laws Boyle’s law: PV = constant (when T is constant) Robert Boyle (1627– 1691) V V P 1/P
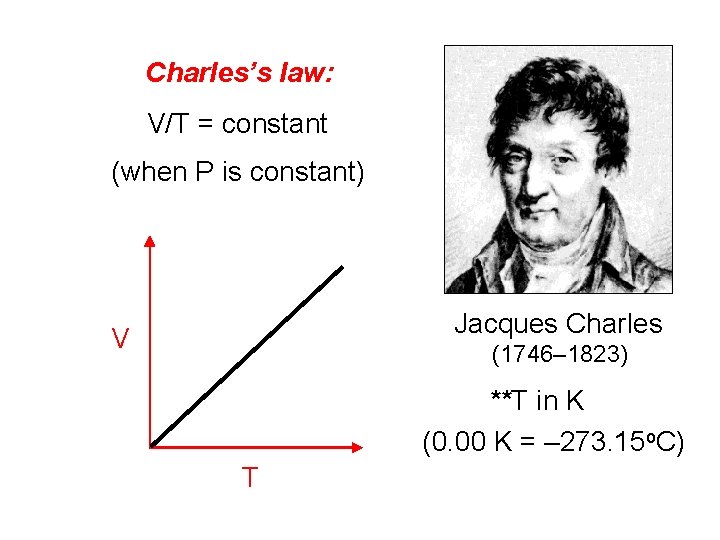
Charles’s law: V/T = constant (when P is constant) Jacques Charles V (1746– 1823) **T in K (0. 00 K = – 273. 15 o. C) T
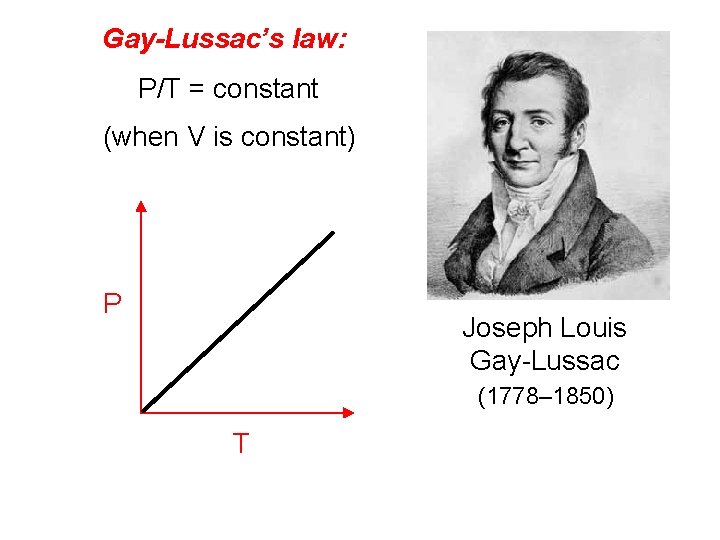
Gay-Lussac’s law: P/T = constant (when V is constant) P Joseph Louis Gay-Lussac (1778– 1850) T

Avogadro’s hypothesis: Equal volumes of gas at the same temperature and pressure have the same number of particles. e. g. , 22. 4 L of ANY GAS at STP contains 6. 02 x 1023 particles Avogadro’s law: Volume of a gas is proportional to the number of moles of gas. Amedeo Avocado Avogadro (1776– 1856) (1876– 1956) i. e. ,


Combined Gas law: merges Boyle’s, Charles’s, and Gay-Lussac’s laws into one equation. **NOTE: For all gas law calculations, use the absolute temperature (in K).

Other Equations and Constants Ideal Gas law: PV=n. RT R = 8. 314 L-k. Pa/mol-K = 0. 08206 L-atm/mol-K Conditions of standard temp. and pressure (STP): 0 o. C (273. 15 K) 1 atm Equations for gas density: M = molar mass of gas same gas; two sets of conditions

To solve problems involving volumes of gases NOT at STP in chemical reactions: Combine PV = n. RT and stoichiometry.
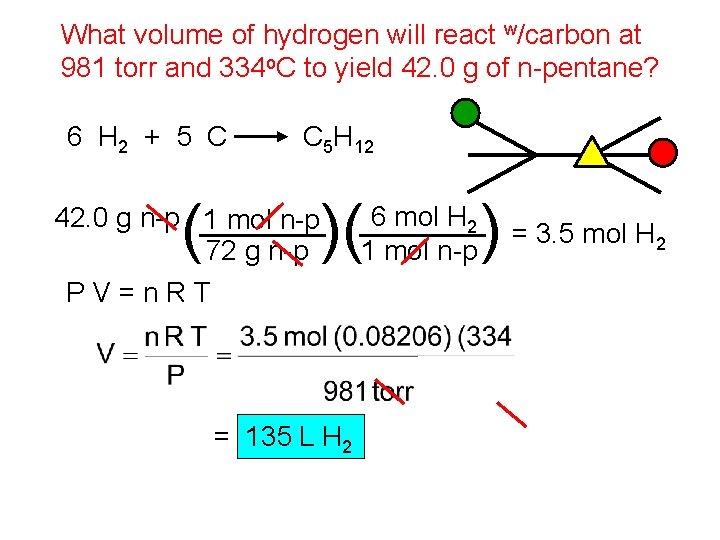
What volume of hydrogen will react w/carbon at 981 torr and 334 o. C to yield 42. 0 g of n-pentane? 6 H 2 + 5 C ( C 5 H 12 42. 0 g n-p 1 mol n-p 72 g n-p PV=n. RT )( = 135 L H 2 6 mol H 2 1 mol n-p ) = 3. 5 mol H 2

Dalton’s law of Partial Pressure Ptot = P 1 + P 2 +. . . partial pressures: total pressure of gaseous mixture Other equations… pressure that each gas would exert by itself Total moles of gas in a mixture: ntot = n 1 + n 2 + … The mole fraction (X) of a gas in a mixture:
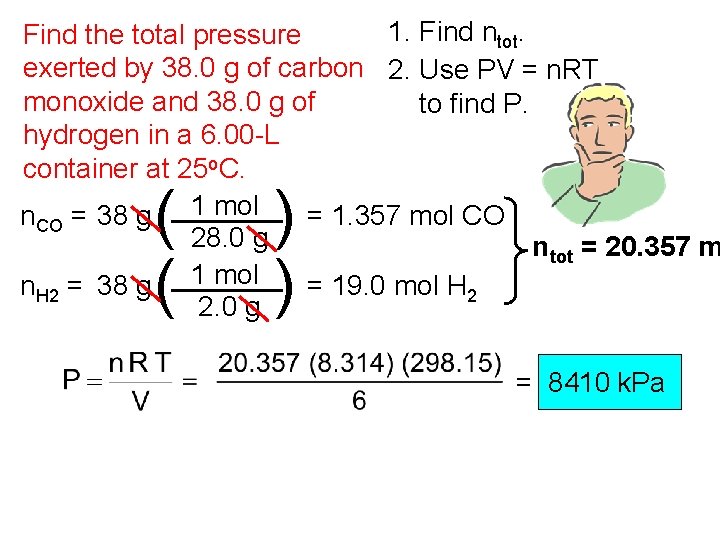
1. Find ntot. Find the total pressure exerted by 38. 0 g of carbon 2. Use PV = n. RT monoxide and 38. 0 g of to find P. hydrogen in a 6. 00 -L container at 25 o. C. n. CO = 38 g 1 mol = 1. 357 mol CO 28. 0 g ntot = 20. 357 m n. H 2 = 38 g 1 mol = 19. 0 mol H 2 2. 0 g ( ( ) ) = 8410 k. Pa

With reference to the previous problem… What is the partial pressure exerted by each gas? The ratio of the partial pressures is the same as the mole ratio. mole fraction ( ) 8410 k. Pa = 560 k. Pa = ( 19. 0 ) 8410 k. Pa = 7850 k. Pa 20. 357 PCO = PH 2 1. 357 20. 357 (38 g CO) (38 g H 2)
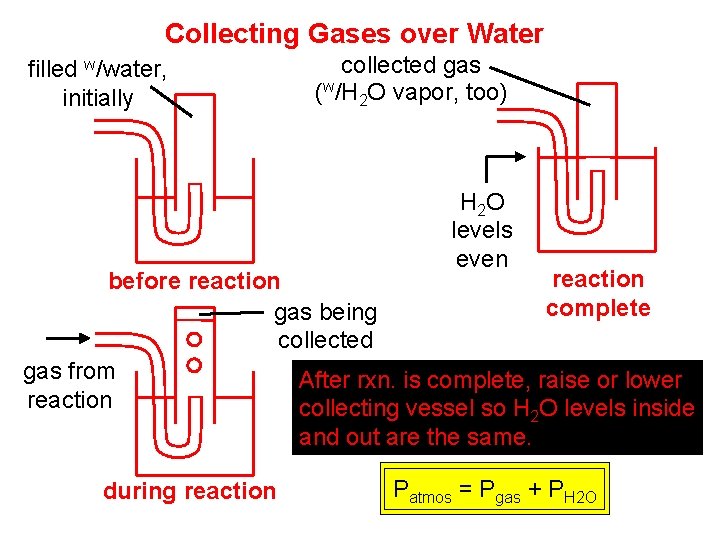
Collecting Gases over Water filled w/water, initially collected gas (w/H 2 O vapor, too) H 2 O levels even reaction before reaction complete gas being collected gas from After rxn. is complete, raise or lower reaction collecting vessel so H O levels inside 2 and out are the same. In this way. . . during reaction Patmos = Pgas + PH 2 O

For the reaction Ca. C 2(s) + H 2 O(l) C 2 H 2(g) + Ca. O(s). . . If 0. 852 L of acetylene are collected over water at 20. 0 o. C, find the moles of acetylene collected and the grams of calcium carbide used. The barometric pressure is 732. 0 torr = PC 2 H 2 + PH 2 O So PC 2 H 2 = 714. 5 torr = 0. 9401 atm 17. 5 torr @ 20. 0 o. C (from p. 1111) = 0. 0333 mol C 2 H 2 From bal. eq. , n. Ca. C 2 = n. C 2 H 2, so g. Ca. C 2 = 0. 0333 mol ( 64. 1 g 1 mol ) = 2. 13 g Ca. C 2
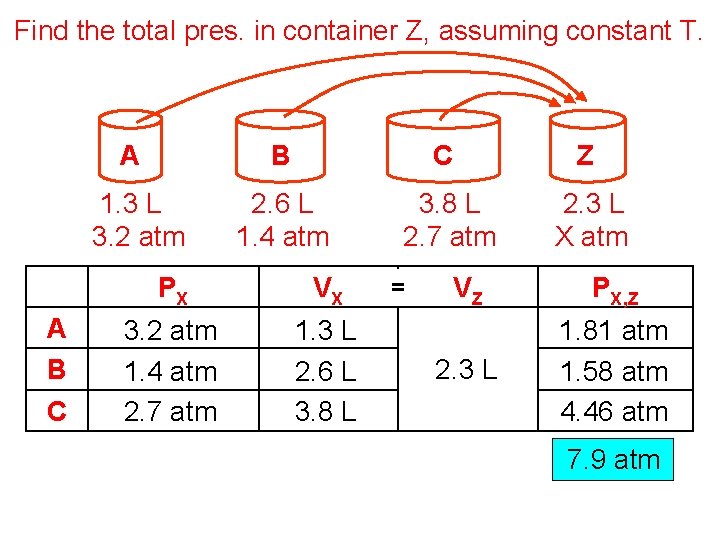
Find the total pres. in container Z, assuming constant T. A B C Z 1. 3 L 3. 2 atm 2. 6 L 1. 4 atm 3. 8 L 2. 7 atm 2. 3 L X atm PX VX 3. 2 atm 1. 4 atm 2. 7 atm 1. 3 L 2. 6 L 3. 8 L = VZ PX, Z 2. 3 L 1. 81 atm 1. 58 atm 4. 46 atm 7. 9 atm

The Kinetic-Molecular Theory (the theory of moving particles) 1. Gas particles are in constant, random motion. 2. The volume of the particles is negligible compared to the container volume. 3. The attractive-repulsive forces between particles are negligible. 4. Collisions are elastic. (i. e. , no energy is lost). 5. KEavg of particles is proportional to absolute temperature.
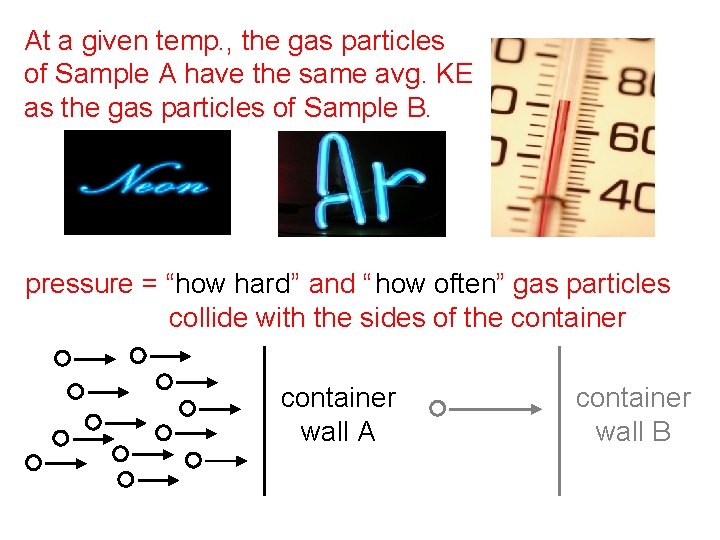
At a given temp. , the gas particles of Sample A have the same avg. KE as the gas particles of Sample B. pressure = “how hard ” and “ how often” gas particles collide with the sides of the container wall A container wall B
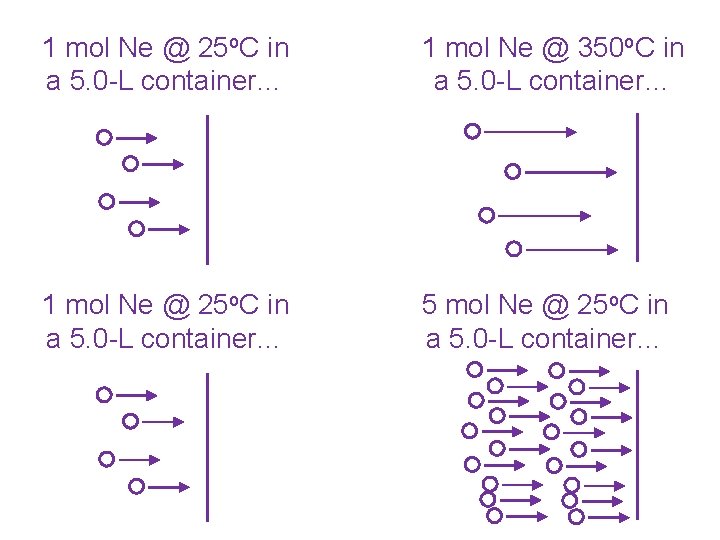
1 mol Ne @ 25 o. C in a 5. 0 -L container… 1 mol Ne @ 350 o. C in a 5. 0 -L container… 1 mol Ne @ 25 o. C in a 5. 0 -L container… 5 mol Ne @ 25 o. C in a 5. 0 -L container…
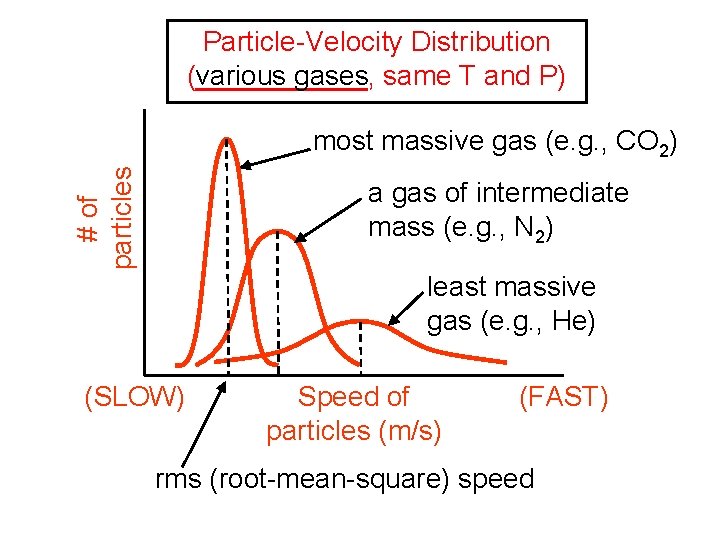
Particle-Velocity Distribution (______, various gases same T and P) # of particles most massive gas (e. g. , CO 2) a gas of intermediate mass (e. g. , N 2) least massive gas (e. g. , He) (SLOW) Speed of particles (m/s) (FAST) rms (root-mean-square) speed
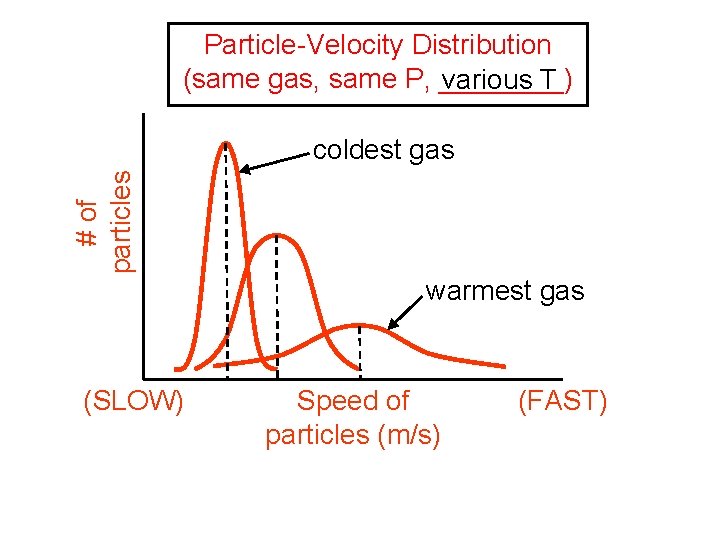
Particle-Velocity Distribution (same gas, same P, ____) various T # of particles coldest gas warmest gas (SLOW) Speed of particles (m/s) (FAST)
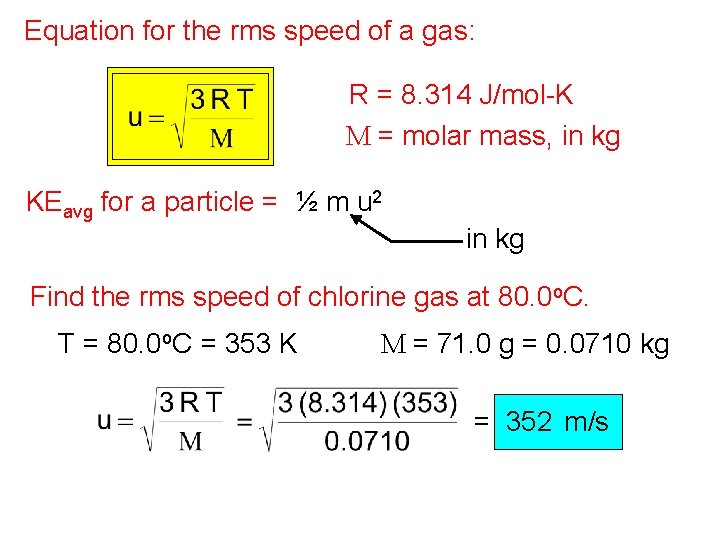
Equation for the rms speed of a gas: R = 8. 314 J/mol-K M = molar mass, in kg KEavg for a particle = ½ m u 2 in kg Find the rms speed of chlorine gas at 80. 0 o. C. T = 80. 0 o. C = 353 K M = 71. 0 g = 0. 0710 kg = 352 m/s
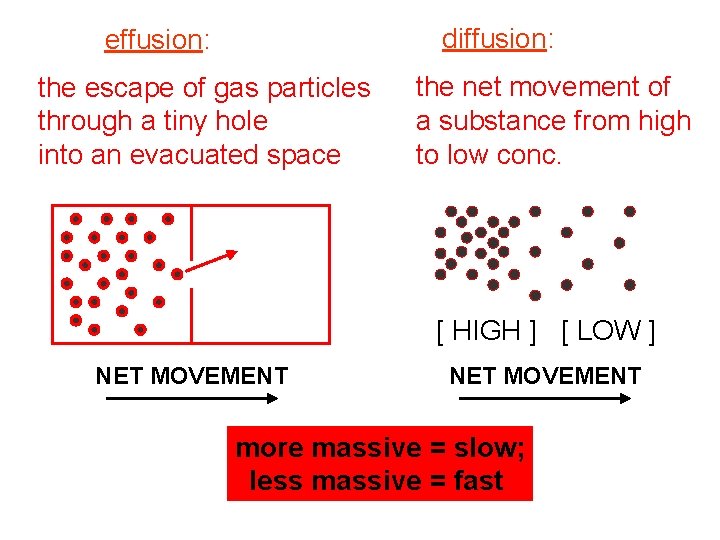
diffusion: effusion: the escape of gas particles through a tiny hole into an evacuated space the net movement of a substance from high to low conc. [ HIGH ] [ LOW ] NET MOVEMENT more massive = slow; less massive = fast

For gases, rates of diffusion & effusion obey Graham’s law: ** To use Graham’s Law, both gases must be at the… same temp. r = rates M = molar masses t = times The rate of diffusion of gases is slower than the molecular speeds because of. . . collisions. -- ~10 x 109 collisions/sec per particle The mean free path is the average distance traveled by a particle between collisions. -- it is shorter when the pressure is…high
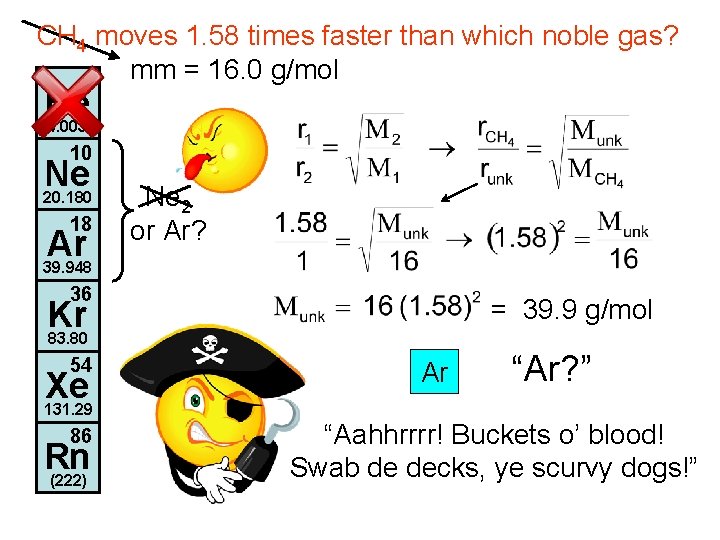
CH 4 moves 1. 58 times faster than which noble gas? mm = 16. 0 g/mol 2 He 4. 003 10 Ne 20. 180 18 Ar 39. 948 Ne 2 or Ar? 36 = 39. 9 g/mol Kr 83. 80 54 Xe 131. 29 86 Rn (222) Ar “Ar? ” “Aahhrrrr! Buckets o’ blood! Swab de decks, ye scurvy dogs!”

Real Gases: Deviations from Ideal Behavior All real gases deviate, to some degree, from PV = n. RT. The deviations are most pronounced at. . . high P and/or low T. Real gas particles. . . 1) DO occupy space 2) DO experience attractive forces
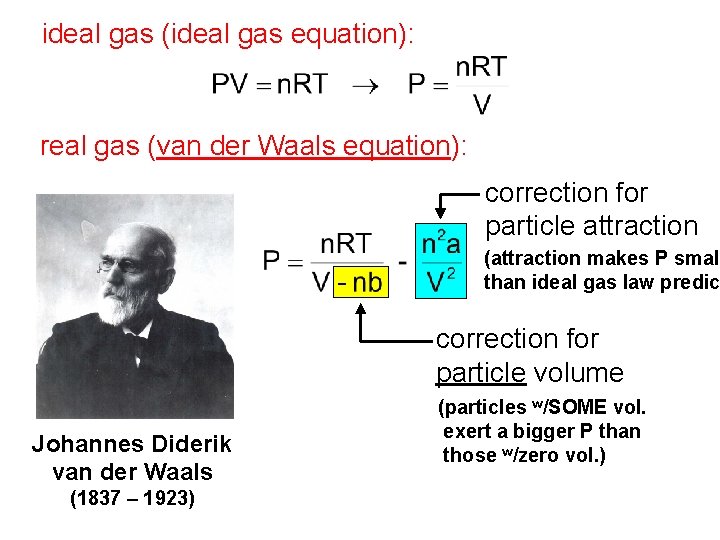
ideal gas (ideal gas equation): real gas (van der Waals equation): correction for particle attraction (attraction makes P small than ideal gas law predic correction for particle volume Johannes Diderik van der Waals (1837 – 1923) (particles w/SOME vol. exert a bigger P than those w/zero vol. )
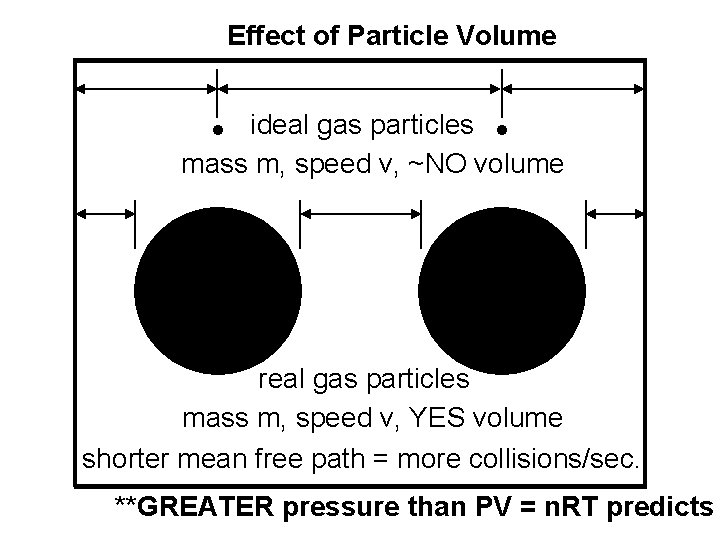
Effect of Particle Volume ideal gas particles mass m, speed v, ~NO volume real gas particles mass m, speed v, YES volume shorter mean free path = more collisions/sec. **GREATER pressure than PV = n. RT predicts

The constants a and b are unique for each gas. -- a is large when interparticle attractions are large -- b is large for large gas particles a and b both tend to increase with mass e. g. , both are larger for Xe than for Ne
- Slides: 34