AP Chemistry The Gas Laws Basics on Gases

AP Chemistry The Gas Laws

Basics on Gases composition of the atmosphere: ~78% N 2, ~21% O 2 properties of gases: expand to fill container compressible form homogeneous mixtures -- due to gas particles being… 1) far apart 2) in constant, random motion vapors: gases of substances that are normally liquids or solids e. g. , gasoline vapors

Equation for pressure: F N A m 2 P N/m 2 = Pa 1 atm… = 760 mm Hg = 760 torr = 101. 325 k. Pa = 1. 01325 bar (1 bar = 105 Pa) At a depth of 350 m (1150 ft), the hull pressure on a submarine is 3. 4 x 106 Pa (36 tons/ft 2).
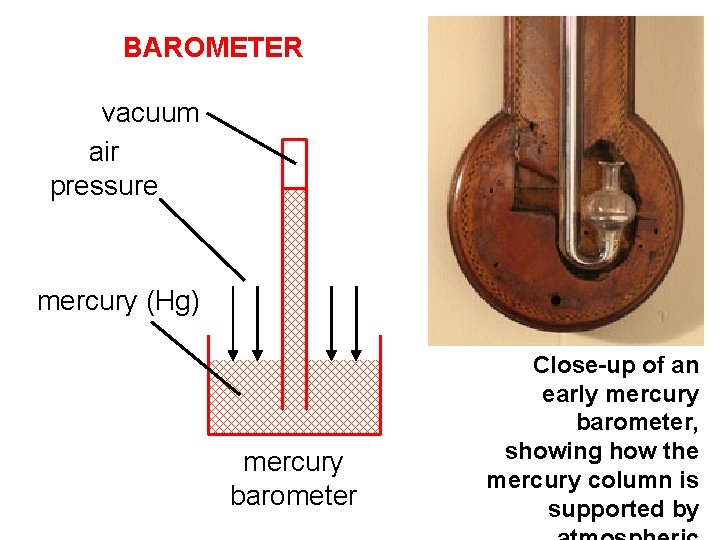
BAROMETER vacuum air pressure mercury (Hg) mercury barometer Close-up of an early mercury barometer, showing how the mercury column is supported by

An aneroid barometer contains a small, pressure-sensitive metal box that has been evacuated of air. The box is prevented from collapsing by being connected to a spring that is also attached to the dial on the barometer. When the air pressure on the walls of the box changes, the box “flexes, ” which moves the spring and the dial.
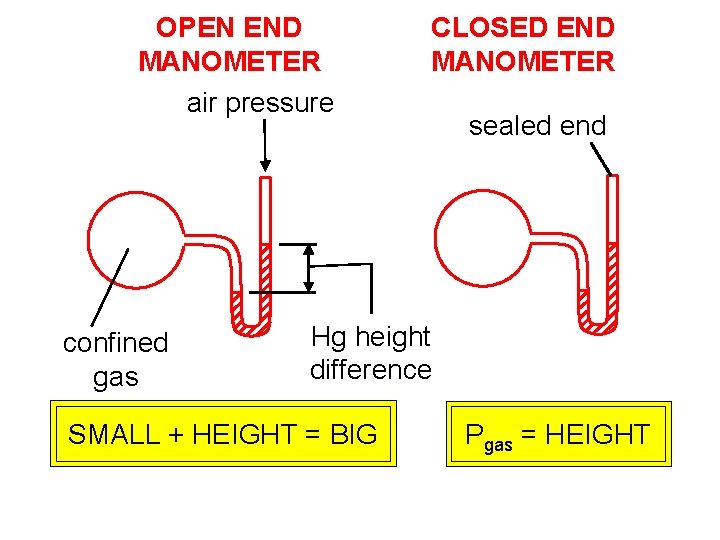
OPEN END MANOMETER air pressure confined gas CLOSED END MANOMETER sealed end Hg height difference SMALL + HEIGHT = BIG Pgas = HEIGHT
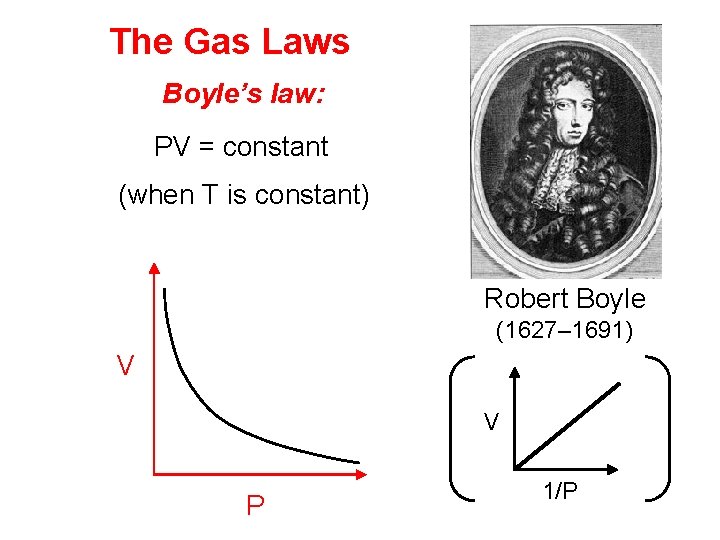
The Gas Laws Boyle’s law: PV = constant (when T is constant) Robert Boyle (1627– 1691) V V P 1/P
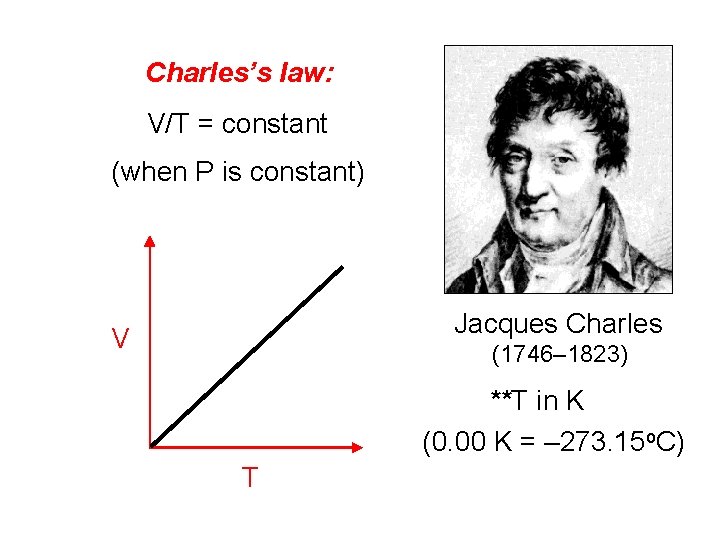
Charles’s law: V/T = constant (when P is constant) Jacques Charles V (1746– 1823) **T in K (0. 00 K = – 273. 15 o. C) T
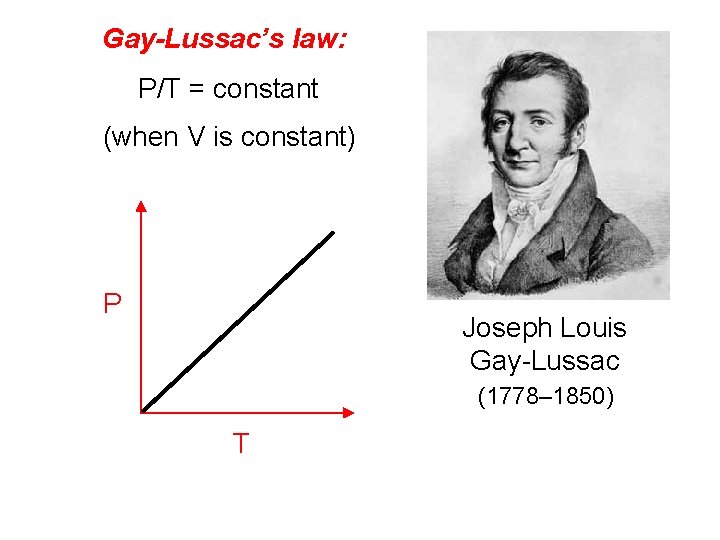
Gay-Lussac’s law: P/T = constant (when V is constant) P Joseph Louis Gay-Lussac (1778– 1850) T

Avogadro’s hypothesis: Equal volumes of gas at the same temperature and pressure have the same number of particles. e. g. , 22. 4 L of ANY GAS at STP contains 6. 02 x 1023 particles Avogadro’s law: Volume of a gas is proportional to the number of moles of gas. Amedeo Avocado Avogadro (1776– 1856) (1876– 1956) i. e. ,


Combined Gas law: merges Boyle’s, Charles’s, and Gay-Lussac’s laws into one equation. **NOTE: For all gas law calculations, use the absolute temperature (in K).

Other Equations and Constants Ideal Gas law: PV=n. RT R = 8. 314 L-k. Pa/mol-K = 0. 08206 L-atm/mol-K Conditions of standard temp. and pressure (STP): 0 o. C (273. 15 K) 1 atm Equations for gas density: M = molar mass of gas same gas; two sets of conditions
- Slides: 13