AP Chemistry Stoichiometry In thermite reaction a ture









- Slides: 14

AP Chemistry Stoichiometry In thermite reaction, a ture of powdered aluminum and powdered iron(III) oxide to yield iron and aluminum xide. The reaction burns hot ough to be useful in underwater welding. 2 Al + Fe 2 O 3 2 Fe + Al 2 O 3 + ener

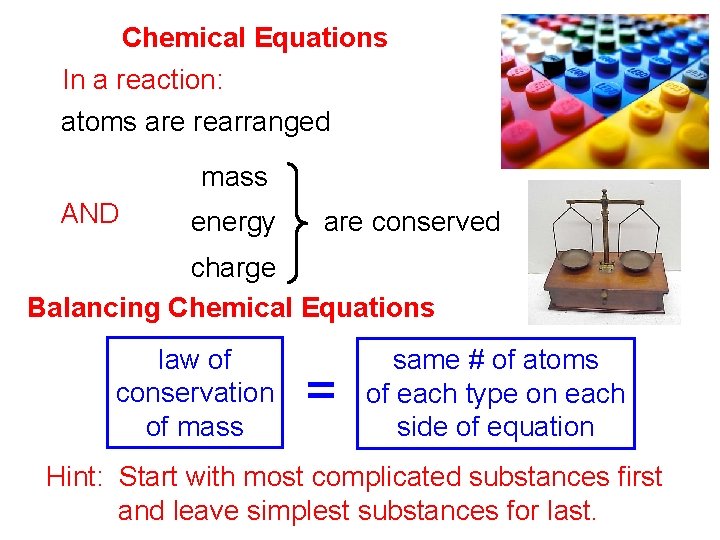
Chemical Equations In a reaction: atoms are rearranged mass AND energy are conserved charge Balancing Chemical Equations law of conservation of mass = same # of atoms of each type on each side of equation Hint: Start with most complicated substances first and leave simplest substances for last.

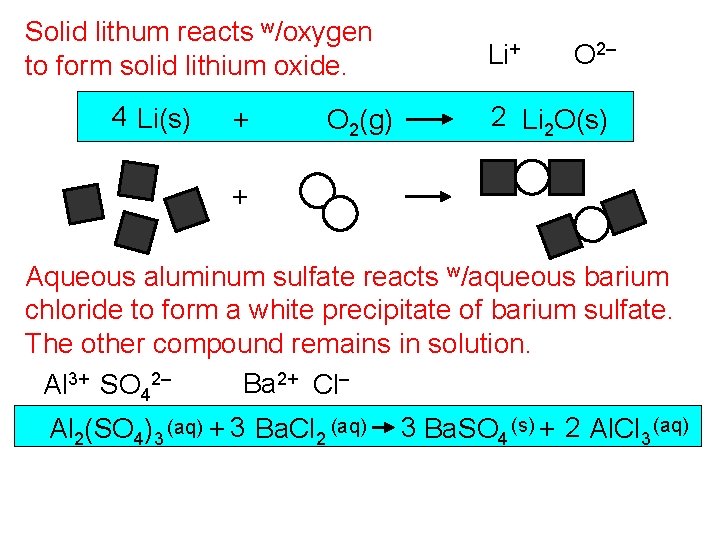
Solid lithum reacts w/oxygen to form solid lithium oxide. 4 Li(s) + O 2(g) Li+ O 2– 2 Li 2 O(s) + Aqueous aluminum sulfate reacts w/aqueous barium chloride to form a white precipitate of barium sulfate. The other compound remains in solution. Ba 2+ Cl– Al 3+ SO 42– Al 2(SO 4)3 (aq) + 3 Ba. Cl 2 (aq) 3 Ba. SO 4 (s) + 2 Al. Cl 3 (aq)

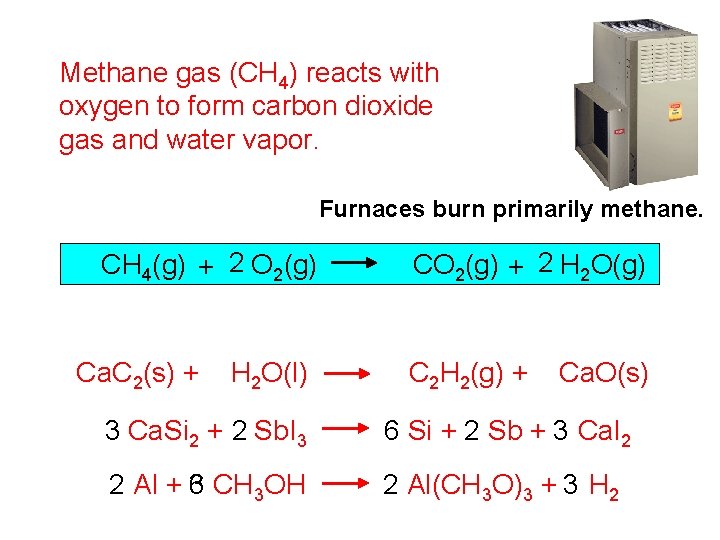
Methane gas (CH 4) reacts with oxygen to form carbon dioxide gas and water vapor. Furnaces burn primarily methane. CH 4(g) + 2 O 2(g) Ca. C 2(s) + H 2 O(l) CO 2(g) + 2 H 2 O(g) C 2 H 2(g) + Ca. O(s) 3 Ca. Si 2 + 2 Sb. I 3 6 Si + 2 Sb + 3 Ca. I 2 2 Al + 3 6 CH 3 OH 2 Al(CH 3 O)3 + 3 H 2

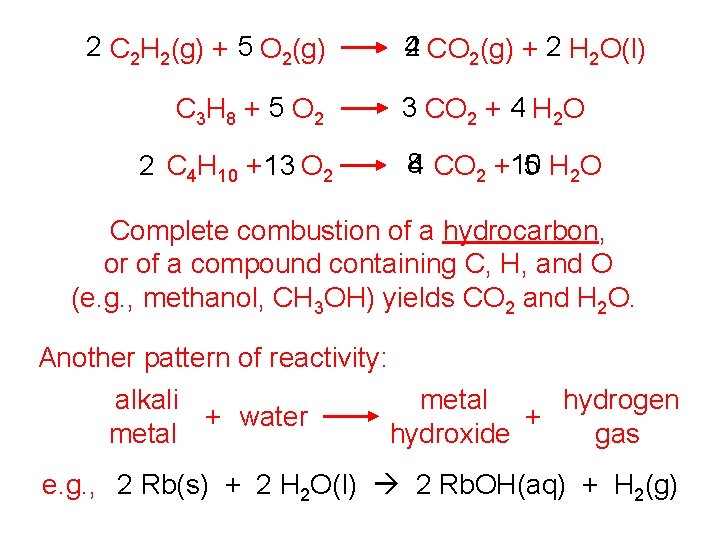
2 C 2 H 2(g) + 5 O 2(g) C 3 H 8 + 5 O 2 2 C 4 H 10 + 13 O 2 2 CO 2(g) + 2 H 2 O(l) 4 3 CO 2 + 4 H 2 O 8 4 CO 2 +10 5 H 2 O Complete combustion of a hydrocarbon, or of a compound containing C, H, and O (e. g. , methanol, CH 3 OH) yields CO 2 and H 2 O. Another pattern of reactivity: alkali metal hydrogen + water + metal hydroxide gas e. g. , 2 Rb(s) + 2 H 2 O(l) 2 Rb. OH(aq) + H 2(g)

Two (of the several) Types of Reactions combination (synthesis): simpler substances combine to form more complex substances -- form: A + B AB AB + C ABC sodium + chlorine gas 2 Na + Cl 2 A + B + C ABC sodium chloride 2 Na. Cl

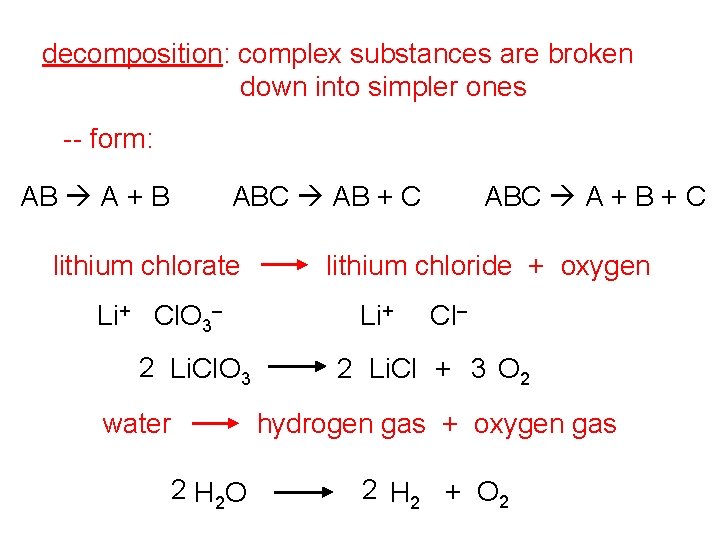
decomposition: complex substances are broken down into simpler ones -- form: AB A + B ABC AB + C lithium chlorate Li+ Cl. O 3– 2 Li. Cl. O 3 water ABC A + B + C lithium chloride + oxygen Li+ Cl– 2 Li. Cl + 3 O 2 hydrogen gas + oxygen gas 2 H 2 O 2 H 2 + O 2

formula weight: the mass of all of the atoms in a chemical formula (unit is amu) -- If the substance is a molecular substance (e. g. , C 3 H 8), then the term molecular weight is also used. molar mass: the mass of one mole of a substance (unit is usually grams) -- recall that 1 mole of any = 6. 02 x 1023 particles substance of that substance

Find the molar mass and formula weight of ammonium phosphate. NH 4+ PO 43– (NH 4)3 PO 4 N: 3 (14. 0 g) = 42. 0 g H: 12 (1. 0 g) = 12. 0 g P: 1 (31. 0 g) = 31. 0 g O: 4 (16. 0 g) = 64. 0 g m. m. = 149. 0 g f. w. = 149. 0 amu

percentage composition: the mass % of each element in a compound equation: g element x 100 % of element = molar mass of compound Find the percentage of oxygen, by mass, in calcium nitrate. Ca(NO 3)2 %O= = 58. 5% O

Empirical Formula and Molecular Formula lowest-terms formula shows the true number and type of atoms in a m’cule Compound Molecular Formula Empirical Formula glucose C 6 H 12 O 6 CH 2 O propane C 3 H 8 butane C 4 H 10 C 2 H 5 naphthalene C 10 H 8 C 5 H 4 sucrose C 12 H 22 O 11 octane C 8 H 18 C 4 H 9

Finding an Empirical Formula from Experimental Data 1. Find # of g of each element. 2. Convert each g to mol. 3. Divide each “# of mol” by the smallest “# of mol. ” 4. Use ratio to find formula. “What’s your flavor of ice cream? ”

A ruthenium/sulfur compound is 67. 7% Ru. Find its empirical formula. Ru. S 1. 5 Ru 2 S 3

A sample of a compound contains 4. 63 g lead, 1. 25 g nitrogen, and 2. 87 g oxygen. Name the compound. ? Pb. N 4 O 8 ? Pb(NO 2)4 Pb? 4 NO 2– lead(IV) nitrite