AP CHEMISTRY Chapter 2 Atoms Molecules and Ions















































- Slides: 65

AP CHEMISTRY Chapter 2 Atoms, Molecules, and Ions

• John Dalton’s Atomic Theory: – Each element is composed of atoms – All atoms of an element are identical. – In chemical reactions, the atoms are not changed. • Compounds are formed when atoms of more than one element combine. • Dalton’s law of multiple proportions: When two elements form different compounds, the mass ratio of the elements in one compound is related to the mass ratio in the other by a small whole number.

Examples of the Law of Multiple Proportions: CO and CO 2 PCl 3 and PCl 5 H 2 O and H 2 O 2 Any others?

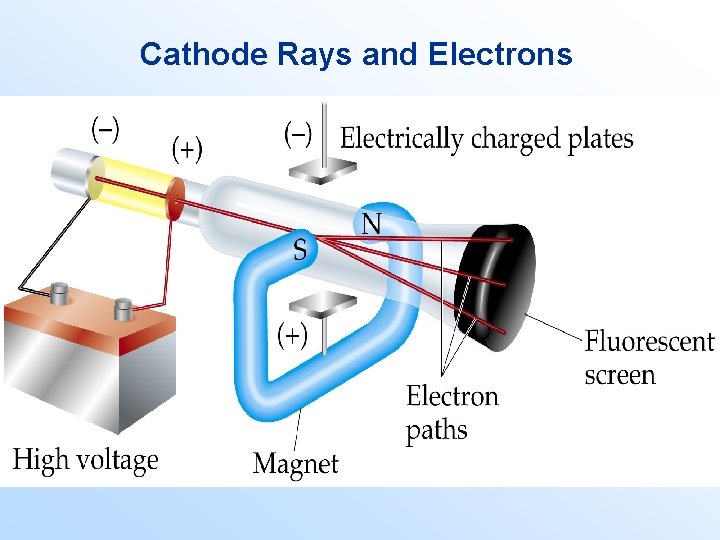
• The ancient Greeks were the first to postulate that matter consists of indivisible constituents. • Later scientists realized that the atom consisted of charged entities. Cathode Rays and Electrons • A cathode ray tube (CRT) is a hollow vessel with an electrode at either end. • A high voltage is applied across the electrodes.

Cathode Rays and Electrons

Cathode Rays and Electrons • The voltage causes negative particles to move from the negative electrode to the positive electrode. • The path of the electrons can be altered by the presence of a magnetic field. • Consider cathode rays leaving the positive electrode through a small hole. – If they interact with a magnetic field perpendicular to an applied electric field, the cathode rays can be deflected by different amounts.

Cathode Rays and Electrons – The amount of deflection of the cathode rays depends on the applied magnetic and electric fields. – In turn, the amount of deflection also depends on the charge to mass ratio of the electron. • In 1897, J. J. Thomson determined the charge to mass ratio of an electron to be 1. 76 108 C/g. • Goal: find the charge on the electron to determine its mass.

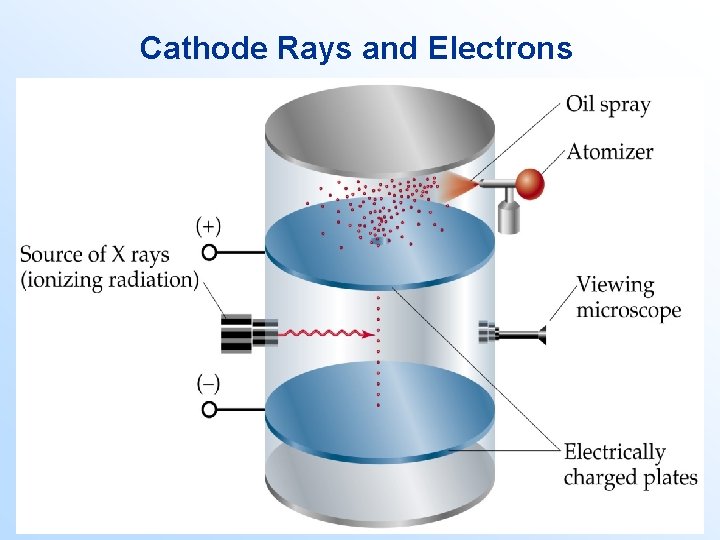
Cathode Rays and Electrons Consider the following experiment: • Oil drops are sprayed above a positively charged plate containing a small hole. • As the oil drops fall through the hole, they are given a negative charge. • Gravity forces the drops downward. The applied electric field forces the drops upward. • When a drop is perfectly balanced, the weight of the drop is equal to the electrostatic force of attraction between the drop and the positive plate.

Cathode Rays and Electrons

Cathode Rays and Electrons • Using this experiment, Robert Millikan determined the charge on the electron to be 1. 60 10 -19 C. • Knowing the charge to mass ratio, 1. 76 108 C/g, Millikan calculated the mass of the electron: 9. 10 10 -28 g. • With more accurate numbers, we get the mass of the electron to be 9. 10939 10 -28 g.

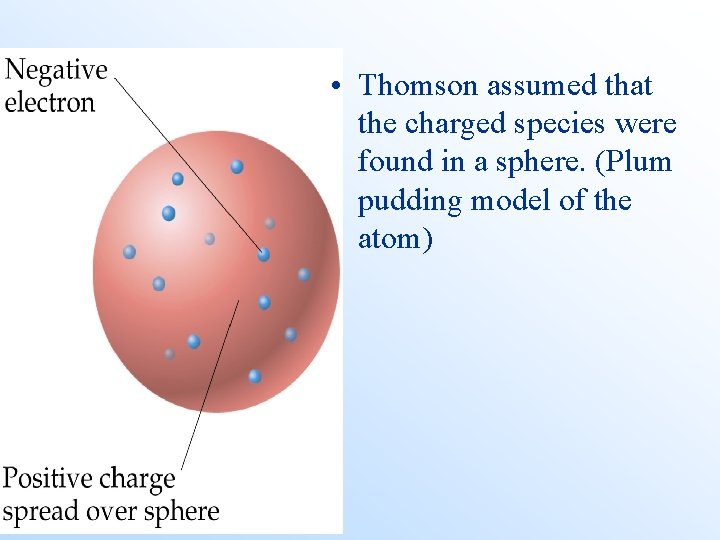
• Thomson assumed that the charged species were found in a sphere. (Plum pudding model of the atom)

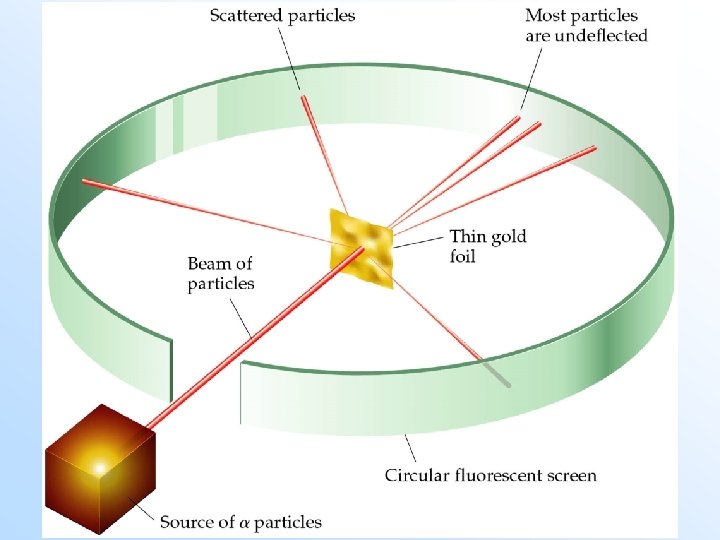
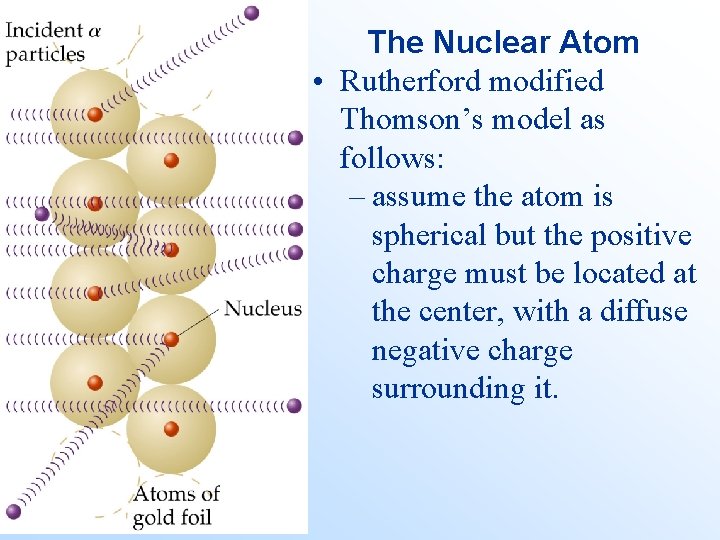
• • • Rutherford’s Gold Foil Experiment Rutherford carried out the following experiment: A radioactive source of -particles ( ) was placed at the mouth of a circular detector. The -particles were shot through a piece of gold foil. Most of the -particles went straight through the foil without deflection. Some -particles were deflected at high angles. If the Thomson model of the atom was correct, then Rutherford’s result was impossible.


The Nuclear Atom • In order to get the majority of -particles through a piece of foil to be undeflected, the majority of the atom must consist empty space, containing low mass, diffuse negative charges - the electrons. • To account for the small number of high deflections of the -particles, the center or nucleus of the atom must consist of a very small, dense positive charge (the nucleus).

The Nuclear Atom • Rutherford modified Thomson’s model as follows: – assume the atom is spherical but the positive charge must be located at the center, with a diffuse negative charge surrounding it.

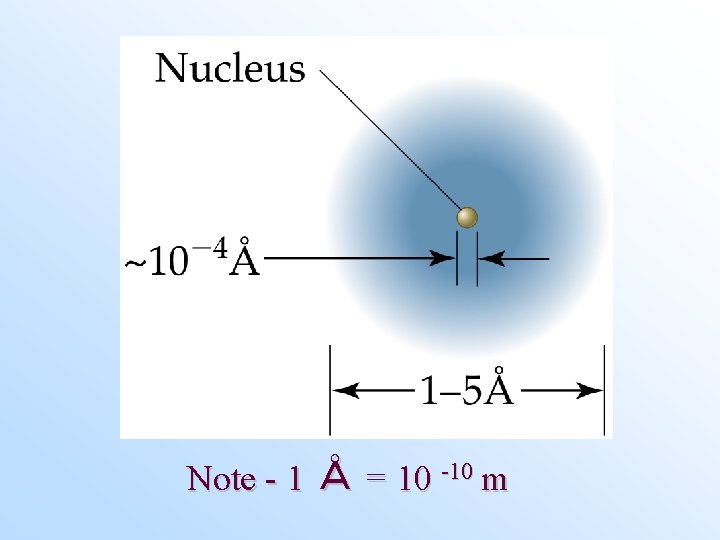
The Nuclear Atom • The atom consists of positive, negative, and neutral entities (protons, electrons, and neutrons). • Protons and neutrons are located in the nucleus of the atom, which is small. Most of the mass of the atom is due to the nucleus. – There can be a variable number of neutrons for the same number of protons. Isotopes have the same number of protons but different numbers of neutrons. • Electrons are located outside of the nucleus. Most of the volume of the atom is due to electrons.

Note - 1 Å = 10 -10 m

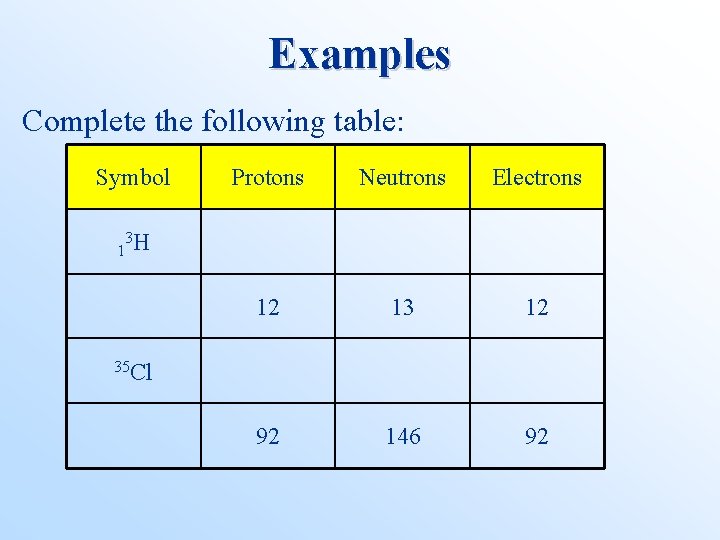
• • • Isotopes, Atomic Numbers, and Mass Numbers Atomic number (Z) = number of protons in the nucleus. Mass number (A) = total number of nucleons in the nucleus (i. e. , protons and neutrons). By convention, for element X, we write ZAX. Isotopes have the same Z but different A. We find Z on the periodic table.

Examples Complete the following table: Symbol Protons Neutrons Electrons 12 13 12 92 146 92 3 1 H 35 Cl

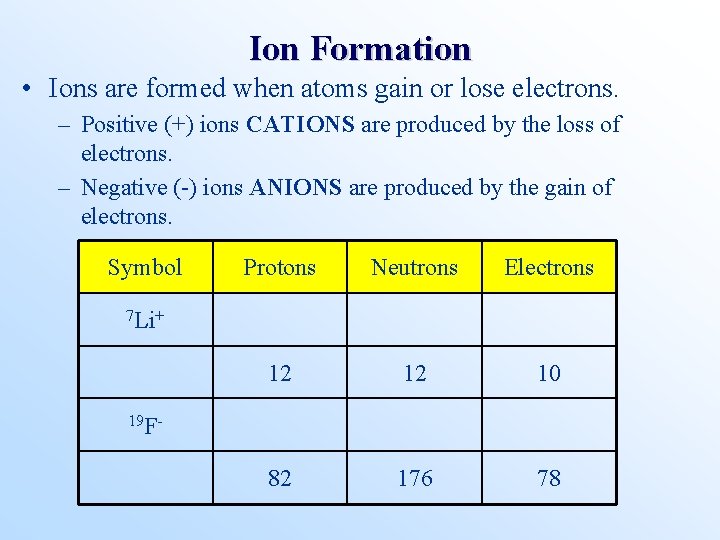
Ion Formation • Ions are formed when atoms gain or lose electrons. – Positive (+) ions CATIONS are produced by the loss of electrons. – Negative (-) ions ANIONS are produced by the gain of electrons. Symbol Protons Neutrons Electrons 12 12 10 82 176 78 7 Li+ 19 F-

The Atomic Mass Scale • 1 H weighs 1. 6735 x 10 -24 g and 16 O 2. 6560 x 10 -23 g. • We define: mass of 12 C = exactly 12 amu. • Using atomic mass units: 1 amu = 1. 66054 x 10 -24 g 1 g = 6. 02214 x 1023 amu

Average Atomic Masses • Relative atomic mass: average masses of isotopes: – Naturally occurring C: 98. 892 % 12 C + 1. 108 % 13 C. • Average mass of C: (0. 98892)(12 amu) + (0. 0108)(13. 00335) = 12. 011 amu. • Average atomic mass is also known as atomic weight (AW). • Atomic weights are listed on the periodic table.

Mass Spectrometer – An instrument used to separate a mixture of samples with different masses. It gives the mass and relative abundance of each sample. The MS can be used to calculate average atomic mass.

Steps in the operation of the mass spectrometer: 1. Vaporization – sample is turned to gas. 2. Ionization – beam of electrons causes sample to lose electrons (some bonds may be broken). 3. Acceleration – electric field causes positive ions to accelerate. 4. Deflection – magnetic field causes moving cations to be deflected (lightest deflected most) 5. Detection – a detector determines the amount of each sample (isotope) present.

The Periodic Table • The Periodic Table is used to organize the 114 elements in a meaningful way. • As a consequence of this organization, there are periodic properties associated with the periodic table.

The Periodic Table

• • • The Periodic Table Columns in the periodic table are called groups (numbered from 1 A to 8 A or 1 to 18). Rows in the periodic table are called periods. Metals are located on the left hand side of the periodic table (most of the elements are metals). Non-metals are located in the top right hand side of the periodic table. Elements with properties similar to both metals and non-metals are called metalloids and are located at the interface between the metals and non-metals.

The Periodic Table • Some of the groups in the periodic table are given special names. • These names indicate the similarities between group members: Group 1: Alkali metals. Group 2: Alkaline earth metals. Group 16: Chalcogens. (not commoly used) Group 17: Halogens. Group 18: Noble gases.

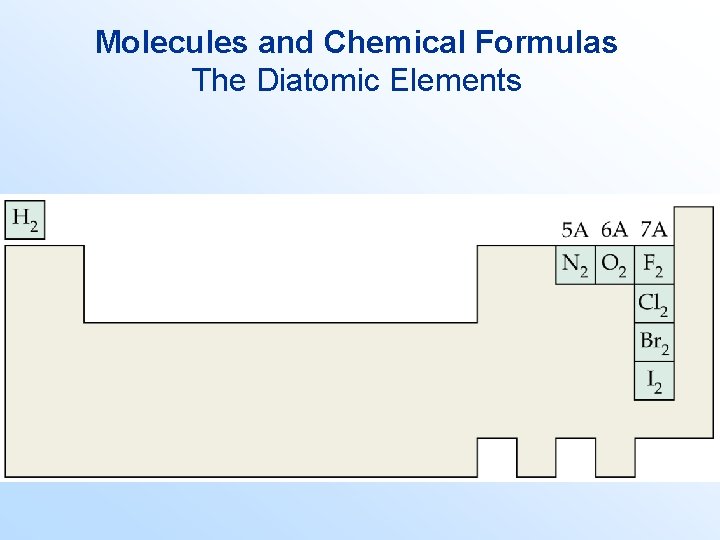
Molecules and Chemical Formulas • Molecules are assemblies of two or more atoms bonded together. • Each molecule has a chemical formula. • The chemical formula indicates – which atoms are found in the molecule, and – in what proportion they are found. • Compounds formed from molecules are molecular compounds. • Molecules that contain two atoms bonded together are called diatomic molecules.

Molecules and Chemical Formulas The Diatomic Elements

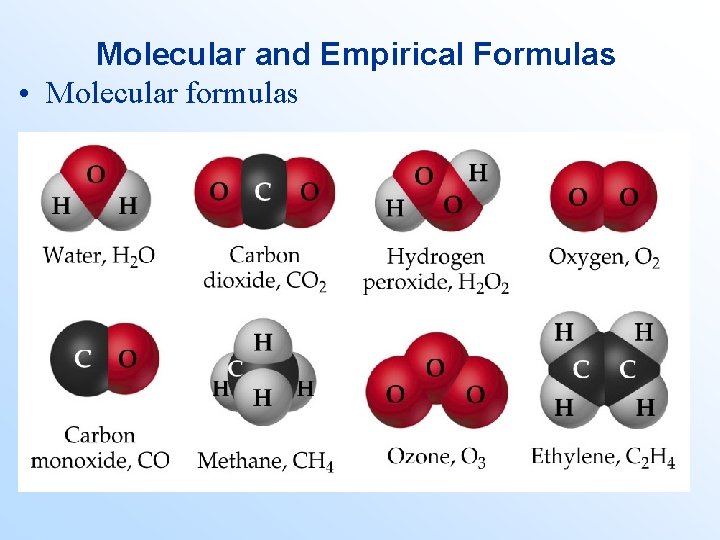
Molecular and Empirical Formulas • Molecular formulas – give the actual numbers and types of atoms in a molecule. – Examples: H 2 O, CO 2, CO, CH 4, H 2 O 2, O 3, and C 2 H 4.

Molecular and Empirical Formulas • Molecular formulas

Molecular and Empirical Formulas • Empirical formulas – give the relative numbers and types of atoms in a molecule. – They give the lowest whole number ratio of atoms in a molecule. – Examples: Give the empirical formula of each: 1. H 2 O 2 2. C 6 H 12 O 6 3. CO 2

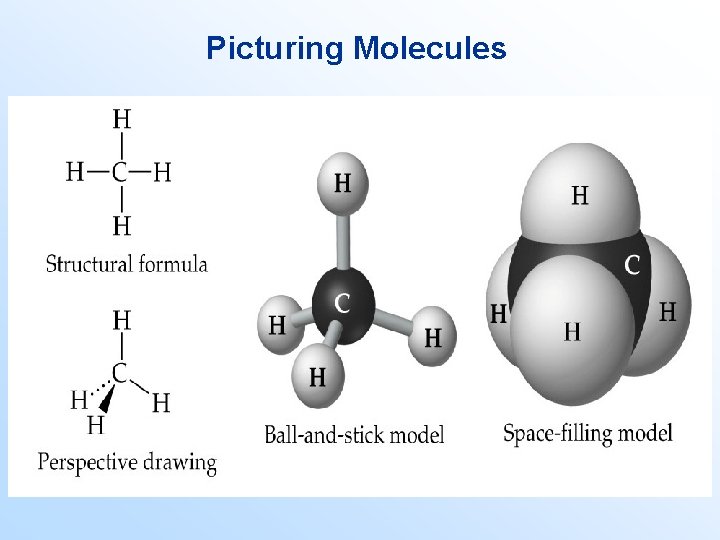
• • • Picturing Molecules occupy three dimensional space. However, we often represent them in two dimensions. The structural formula gives the connectivity between individual atoms in the molecule. The structural formula may or may not be used to show the three dimensional shape of the molecule. If the structural formula does show the shape of the molecule, then either a perspective drawing, ball-and-stick model, or space-filling model is used.

Picturing Molecules

Formation of Compounds • When an atom or molecule loses electrons, it becomes positively charged. – For example, when Na loses an electron it becomes Na+. • Positively charged ions are called cations.

Formation of Compounds • When an atom or molecule gains electrons, it becomes negatively charged. • For example when Cl gains an electron it becomes Cl-. • Negatively charged ions are called anions. • An atom or molecule can lose more than one electron.

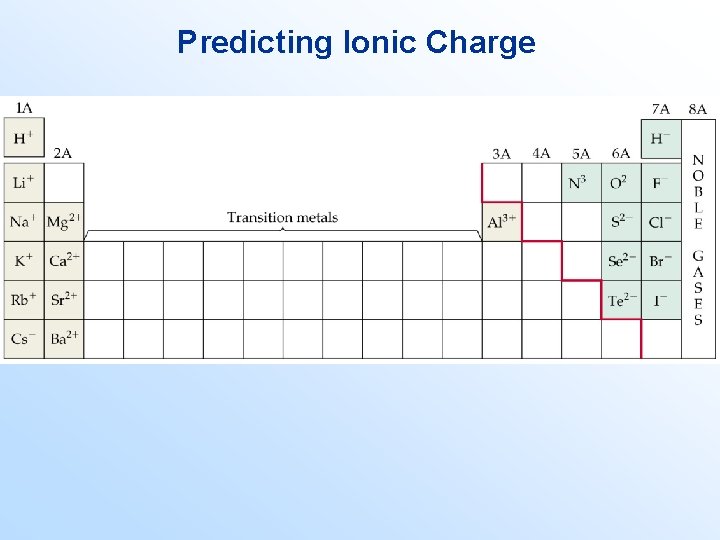
Formation of Compounds • In general: metal atoms tend to lose electrons to become cations; nonmetal ions tend to gain electrons to form anions. Predicting Ionic Charge • The number of electrons an atom loses is related to its position on the periodic table.

Predicting Ionic Charge

Examples: Determine the most likely charge formed by each: 1. 2. 3. 4. 5. 6. Mg Li Cl N Ca S

Ionic Compounds • The majority of chemistry involves the transfer of electrons between species. Example: – To form Na. Cl, the neutral sodium atom, Na, must lose an electron to become a cation: Na+. – The electron cannot be lost entirely, so it is transferred to a chlorine atom, Cl, which then becomes an anion: Cl-. – The Na+ and Cl- ions are attracted to form an ionic Na. Cl lattice which crystallizes.

Ionic Compounds

Ionic Compounds • Important: note that there are no easily identified Na. Cl molecules in the ionic lattice. Therefore, we cannot use molecular formulas to describe ionic substances. • Consider the formation of Mg 3 N 2: • Mg loses two electrons to become Mg 2+; • Nitrogen gains three electrons to become N 3 -. • For a neutral species, the number of electrons lost and gained must be equal. • Overall sum of charges is ZERO

• • Ionic Compounds However, Mg can only lose electrons in twos and N can only accept electrons in threes. Therefore, Mg needs to lose 6 electrons (2 3) and N gain those 6 electrons (3 2). I. e. , 3 Mg atoms need to form 3 Mg 2+ ions (total 3 2+ charges) and 2 N atoms need to form 2 N 3 - ions (total 2 3 - charges). Therefore, the formula is Mg 3 N 2.

Examples: Determine the formula of the compound formed by each: 1. 2. 3. 4. 5. 6. Na and N Ca and Br Al and Cl Al and S K and O Li and F

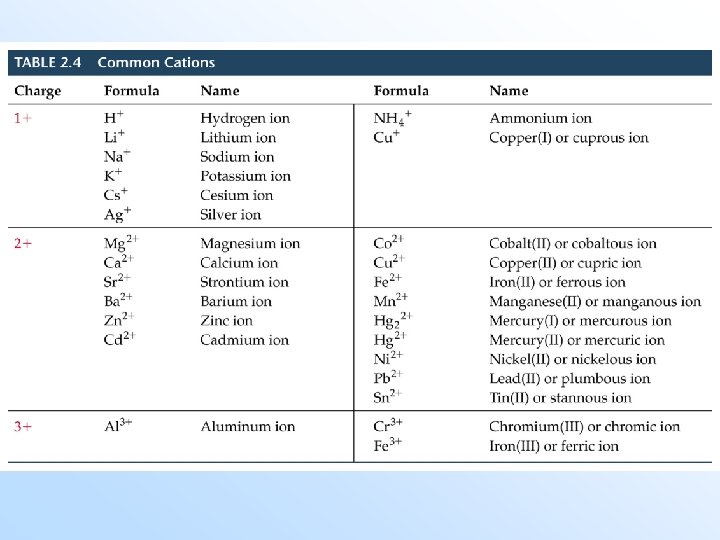
Naming Compounds • Naming of compounds, nomenclature, is divided into organic compounds (those containing C) and inorganic compounds (the rest of the periodic table). • Cations formed from a metal have the same name as the metal. Example: Na+ = sodium ion. • If the metal can form more than one cation, then the charge is indicated in parentheses in the name. Examples: Cu+ = copper(I); Cu 2+ = copper(II). This will also be the treatment for nonmetals.


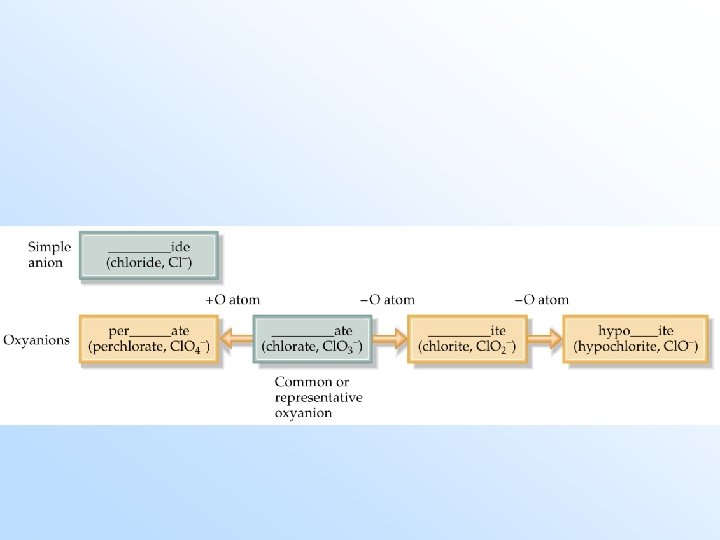
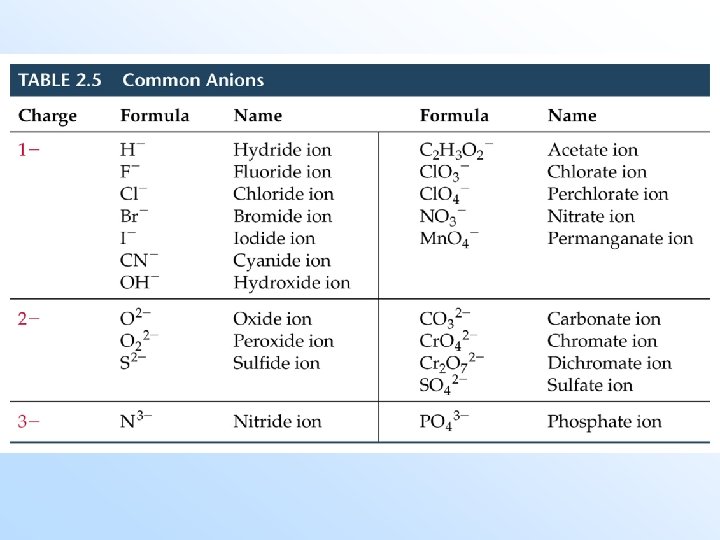
Anions (Negative Ions) • Monatomic anions (with only one atom) are called -ide. Example: Cl- is ______. Exceptions: hydroxide (OH-), cyanide (CN-), peroxide (O 22 -). Polyatomic anions (with many atoms) containing oxygen end in -ate or -ite. (The one with more oxygen is called -ate. ) Examples: NO 3 - is nitrate, NO 2 - is ___.

Polyatomic Ions • Polyatomic anions containing oxygen with more than two members in the series are named as follows (in order of decreasing oxygen): per-…. -ate -ite hypo-…. -ite


Example: For ions of Iodine: I 1 - = IO 31 - = iodate = iodite = hypoiodite = periodate

Polyatomic Ions with Hydrogen • Polyatomic anions containing oxygen with additional hydrogens are named by adding hydrogen or bi- (one H), dihydrogen (two H), etc. , to the name as follows: CO 32 - is the carbonate anion HCO 3 - is the hydrogen carbonate (or bicarbonate) anion. H 2 PO 4 - is the dihydrogen phosphate anion.

Examples - Name each: 1. HSO 42. H 2 PO 33. HSO 3 -


Naming Ionic Compounds • Name the cation then anion for the ionic compound. Example: Ba. Br 2 = barium bromide. Examples – Name each: 1. Ca(HSO 3)2 2. Ba(Cl. O 3)2 3. Au 3 N 4. Al(CN)3 5. (NH 4)3 PO 4 6. Cu. NO 3

The Nomeclature of Hydrates • Hydrates are substances that include water into their formula. • The water is not actually part of the chemical substance and this is reflected in the way the formula is written. • Here is the example: Cu. SO 4 · 5 H 2 O • This formula means that for every one Cu. SO 4 in the piece of this substance you are holding, there also five water molecules. No, the substance is not wet, it appears dry. There are some hydrates that have a wet appearance, but most appear perfectly dry to the eye and to the touch. • The dot IS NOT a multiplication sign.

Cu. SO 4 · 5 H 2 O • Here is the name: copper(II) sulfate pentahydrate. Notice penta meaning five and hydrate meaning water. You would use this name both when writing the name or speaking it. • That means that when you hear "pentahydrate, " you have to know to write the dot and then the 5 H 2 O.

Hydrate Practice Problems • 1) Suppose you heard "trihydrate. " What would you write? • 2) Suppose you heard "octahydrate. " What would you write? • 3) Name this substance: Mg. SO 4 · 9 H 2 O • 4) Write the formula for: barium chloride dihydrate

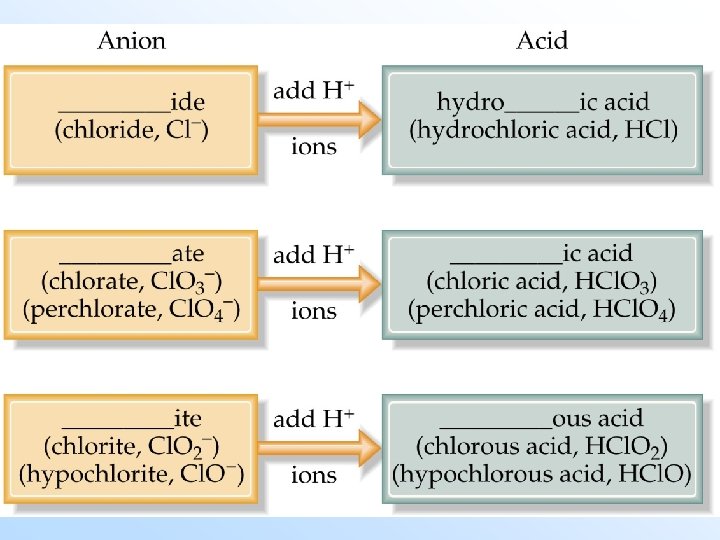
Names and Formulas for Acids • The names of acids are related to the names of anions: -ide becomes hydro-…. -ic acid; -ate becomes -ic acid; -ite becomes -ous acid.


Examples – Name each acid: 1. HCl. O 2. HI 3. H 2 SO 4 4. HC 2 H 3 O 2 5. HBr. O 3

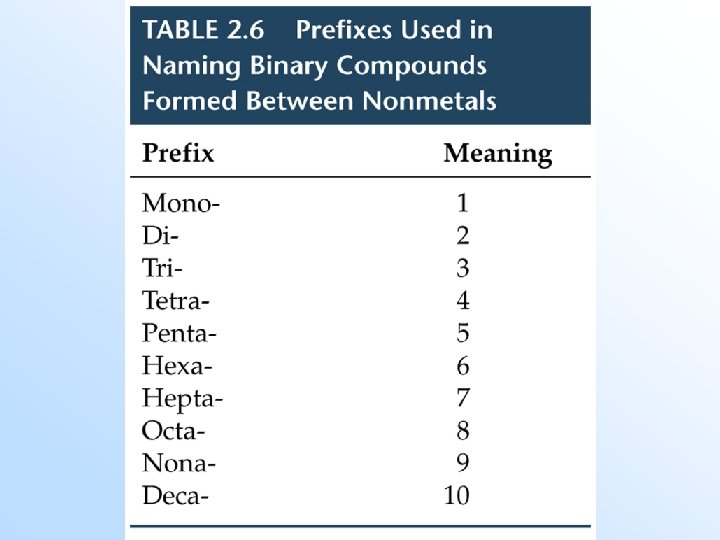
Names and Formulas of Binary Molecular Compounds (Old Method) • Binary molecular compounds have two elements. • The most metallic element is usually written first (i. e. , the one to the farthest left on the periodic table). Exception: NH 3. (N has a -3 oxidation state and H has a +1 oxidation state. ) • If both elements are in the same group, the lower one is written first. • Greek prefixes are used to indicate the number of atoms. (No mono on first atom)


Examples – Name the following: 1. CCl 4 2. P 2 O 7 3. SO 3 4. N 2 O 5 5. CO 2 6. CO

Powerpoint based on Jeff Venables Northwestern High School