AP Chemistry ACIDS BASES Ksp ACIDS AND BASES

AP Chemistry ACIDS & BASES, Ksp

ACIDS AND BASES � "ACID"--Latin word acidus, meaning sour. (lemon) � "ALKALI"--Arabic word for the ashes that come from burning certain plants; � water solutions feel slippery and taste bitter. (soap)

ACID-BASE THEORIES Arrhenius definition � Acid – donates a hydrogen ion (H+) in water � Base – donates a hydroxide ion in water (OH−) � This theory is limited to substances with those “parts”. Ammonia is a MAJOR Acid examples: HCl, H SO 2 4 exception! Base examples: Na. OH, KOH, Ba(OH)2

WHAT IS THE BRONSTED LOWRY THEORY | THE CHEMISTRY JOURNEY � https: //www. youtube. com/watch? v=Ziokq. P 0 a Z 1 E&list=PLW 0 gav. Szh. Ml. Re. KGMVf. Ut 6 Yu. N Qs. O 0 bq. SMV

ACID-BASE THEORIES Bronsted-Lowry Definition � Acid – donates a proton (H+) in water � Base – accepts a proton in water � This theory is better; it explains ammonia as a base! This is the main theory that we will use for our acid/base discussion.

ACID-BASE THEORIES Lewis Definition � Acid – accepts an electron pair � Base – donates an electron pair � This terminology is not as widely used as it used to be because it is too general. Instead, we focus on coordination complexes, which are products of Lewis Acid -Base reactions.

THE BRONSTED-LOWRY CONCEPT OF ACIDS AND BASES Using this theory, you should be able to write weak acid/base dissociation equations and identify acid, base, conjugate acid and conjugate base. � conjugate acid-base pair – A pair of compounds that differ by the presence of one H+ unit
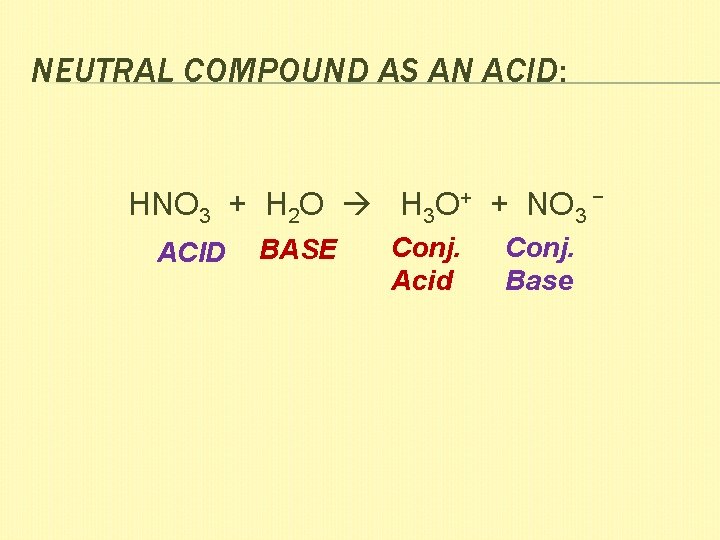
NEUTRAL COMPOUND AS AN ACID: HNO 3 + H 2 O H 3 O+ + NO 3 − ACID BASE Conj. Acid Conj. Base
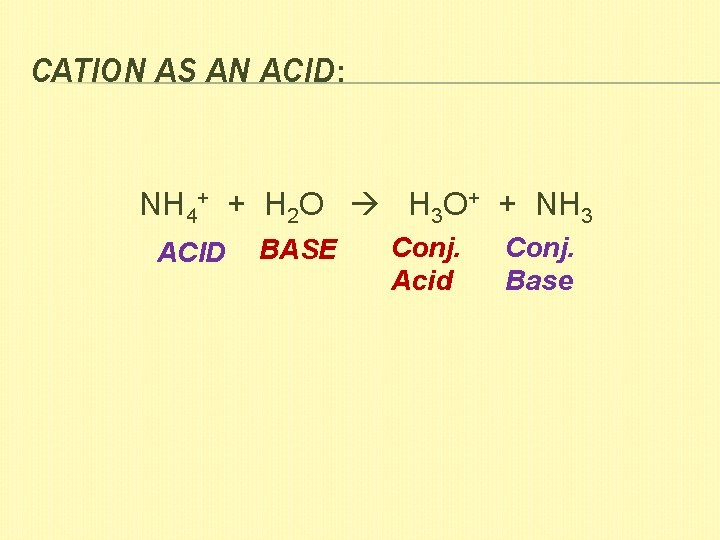
CATION AS AN ACID: NH 4+ + H 2 O H 3 O+ + NH 3 ACID BASE Conj. Acid Conj. Base
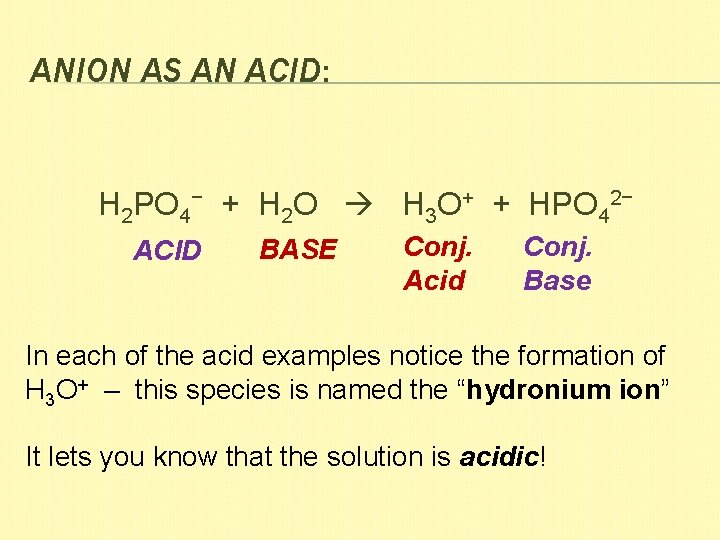
ANION AS AN ACID: H 2 PO 4− + H 2 O H 3 O+ + HPO 42− ACID BASE Conj. Acid Conj. Base In each of the acid examples notice the formation of H 3 O+ – this species is named the “hydronium ion” It lets you know that the solution is acidic!
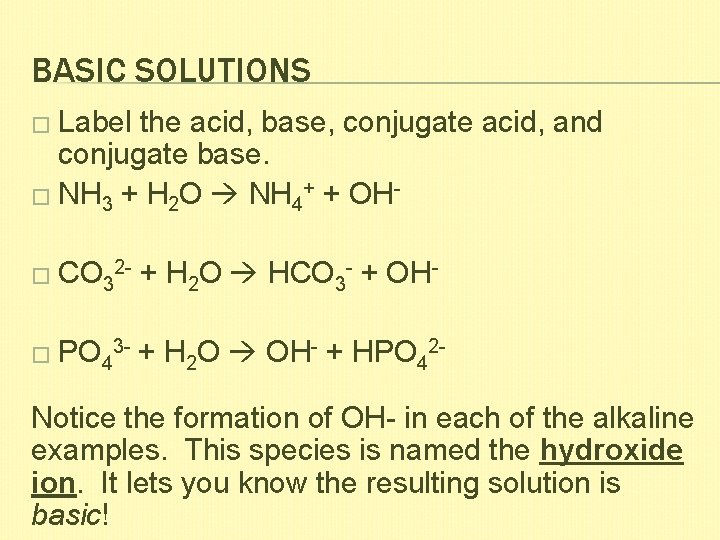
BASIC SOLUTIONS � Label the acid, base, conjugate acid, and conjugate base. � NH 3 + H 2 O NH 4+ + OH� CO 32 - + H 2 O HCO 3 - + OH- � PO 43 - + H 2 O OH- + HPO 42 - Notice the formation of OH- in each of the alkaline examples. This species is named the hydroxide ion. It lets you know the resulting solution is basic!

CONJUGATE ACIDS AND BASES | THE CHEMISTRY JOURNEY � https: //www. youtube. com/watch? v=f. Uh. PLe 0 NAv. A

AMPHOTERIC MOLECULES � Amphoteric --molecules or ions that can behave as EITHER acids or bases; water, anions of weak acids (look at the examples above—sometimes water was an acid, sometimes it acted as a base)

NEUTRALIZATION REACTION � Video containing information about water as an amphoteric molecule. � https: //www. youtube. com/watch? v=Hdm. Cagtas. Yg

BELL WORK – 11/29/16 � For the following reactions, identify the acid, base, conjugate acid, and conjugate base. � C 2 H 5 OH + H 2 O H 3 O+ + C 2 H 5 OACID BASE CA CB � HPO 42 - + NH 4+ NH 3 + H 2 PO 4 BASE ACID CB CA

p. H SCALE � https: //www. youtube. com/watch? v=p. FK 16 Gs U 1 e 4
p. H SCALE 0 7 14
![WHY DOES [H+] = [OH-] AT PH 7? WHY DOES [H+] = [OH-] AT PH 7?](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-20.jpg)
WHY DOES [H+] = [OH-] AT PH 7?
![p. H AND p. OH p. H 7 6 3 12 [H+] (M) [OH-] p. H AND p. OH p. H 7 6 3 12 [H+] (M) [OH-]](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-21.jpg)
p. H AND p. OH p. H 7 6 3 12 [H+] (M) [OH-] (M) p. OH

p. H AND p. OH � What is the pattern between p. H and [H+]? � What is the relationship between [H+] and [OH-]? � How is p. H related to p. OH?

CALCULATING p. H � https: //www. youtube. com/watch? v=aw- Y 1 d 2 IIJc
![CALCULATING PH p. H = – log[H+] p. OH = – log[OH–] The p. CALCULATING PH p. H = – log[H+] p. OH = – log[OH–] The p.](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-24.jpg)
CALCULATING PH p. H = – log[H+] p. OH = – log[OH–] The p. H of a solution is 3. 75, what is the Hydrogen ion concentration, [H+]?

EXAMPLES � What is the p. H of a solution with a hydrogen ion concentration of 2. 0 x 1011? 10. 70 � What is the p. OH of a solution with a hydroxide ion concentration of 1. 8 x 10 -4? 3. 74 � The p. H of human blood was measured to be 7. 41 at 25°C. Calculate the p. OH, [H+], and [OH-] for the sample. p. OH = 6. 59, [H+] = 3. 9 x 10 -8 M, [OH-] = 2. 6 x 10 -7 M

ACID AND BASE QUIZ For questions 1 and 2 identify the acid, base, conjugate acid, and conjugate base. 1. OH- + HPO 42 - H 2 O + PO 432. HF + NH 3 NH 4+ + FFor questions 3 -7 Name the acid or base. 3. H 3 PO 4 6. HNO 2 4. HBr 7. Li. OH 5. Mg(OH)2 8. Given a p. H of 9, find the [H+], p. OH, and [OH-]. Is the solution acidic or basic? (show all work) 9. If [H+] = 2. 5 x 10 -5, find the p. H, p. OH, and [OH-]. Is the solution acidic or basic? (show all work)

AFTER QUIZ…… � Predict the products for the following neutralization reactions: � Na. OH + HCl � HBr + Mg(OH)2

CONCENTRATED VS. STRONG � “Concentrated” – refers to the amount dissolved in solution. � “Strong” – refers to the fraction of molecules that ionize. � For example, if you put a lot of ammonia into a little water, you will create a highly concentrated solution. However, since only 0. 5% of ammonia molecules ionize in water, this basic solution will not be very strong.

ACID-BASE TITRATION � Uses a neutralization reaction to determine the concentration of an acid or base. � Standard Solution: the reactant that has a known molarity � Endpoint: the point at which the unknown has been neutralized.
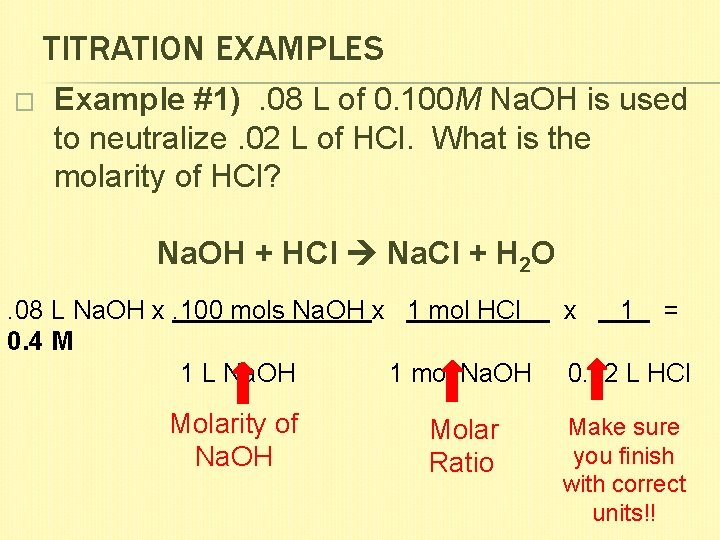
TITRATION EXAMPLES � Example #1). 08 L of 0. 100 M Na. OH is used to neutralize. 02 L of HCl. What is the molarity of HCl? Na. OH + HCl Na. Cl + H 2 O . 08 L Na. OH x. 100 mols Na. OH x 1 mol HCl x 1 = 0. 4 M 1 L Na. OH 1 mol Na. OH 0. 02 L HCl Molarity of Na. OH Molar Ratio Make sure you finish with correct units!!

TITRATION EXAMPLES (CONT’D) � Example #2) A 0. 1 M Mg(OH)2 solution was used to titrate an HBr solution of unknown concentration. At the endpoint, 21. 0 m. L of Mg(OH)2 solution had neutralized 10. 0 m. L of HBr. What is the molarity of the HBr solution?

TITRATION PRACTICE 1) What is the molarity of an Al(OH)3 solution if 30. 0 m. L of the solution is neutralized by 26. 4 m. L of a 0. 25 M HBr solution? 2) A 0. 3 M Ca(OH)2 solution was used to titrate an HCl solution of unknown concentration. At the endpoint, 35. 0 m. L of Ca(OH)2 solution had neutralized 10. 0 m. L of HCl. What is the molarity of the HCl solution? 1) When 34. 2 m. L of a 1. 02 M Na. OH solution is added from a buret to 25. 00 m. L of a phosphoric acid solution that contains phenolphthalein, the solution changes from colorless to pink. What is the molarity of the phosphoric acid?
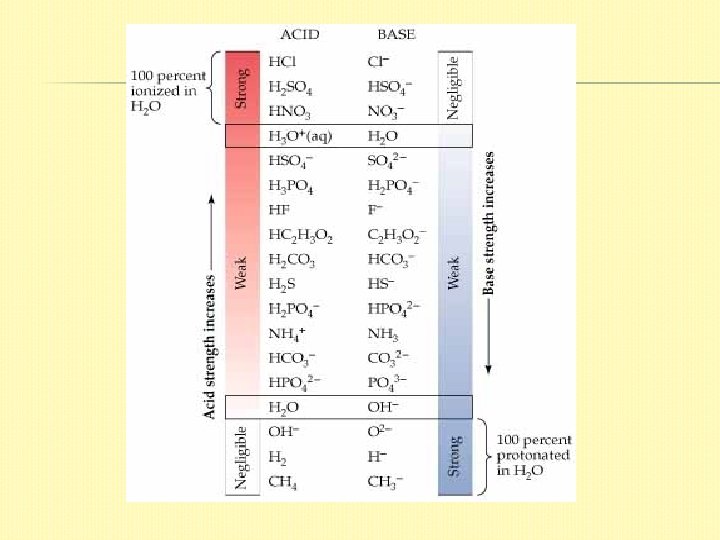


ACID STRENGTH A strong acid completely ionizes in solution � A weak acid only partially ionizes. � The strength of an acid depends on the equilibrium: HA(aq) + H 2 O(l) ⇄ H 3 O+(aq) + A– (aq) � If the equilibrium lies far to the left, the acid is weak (only a small percentage of the acid molecules ionize) If the equilibrium lies far to the right, the acid is strong (completely ionizes)
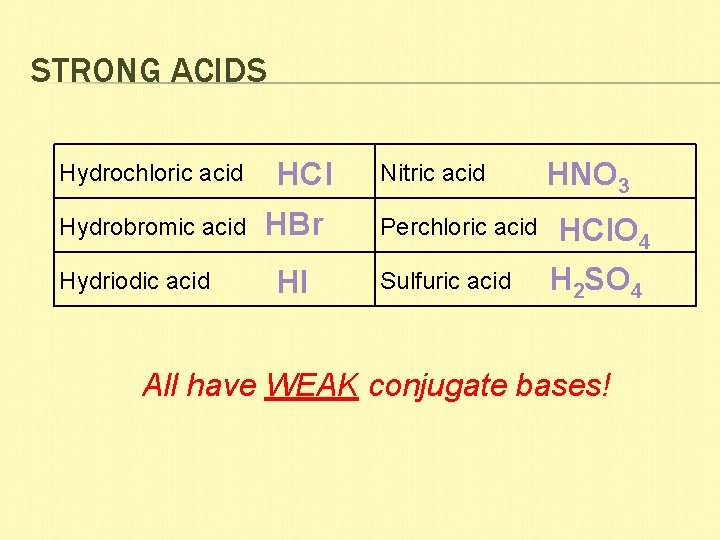
STRONG ACIDS Hydrochloric acid Hydrobromic acid Hydriodic acid HCl HBr HI Nitric acid Perchloric acid Sulfuric acid HNO 3 HCl. O 4 H 2 SO 4 All have WEAK conjugate bases!
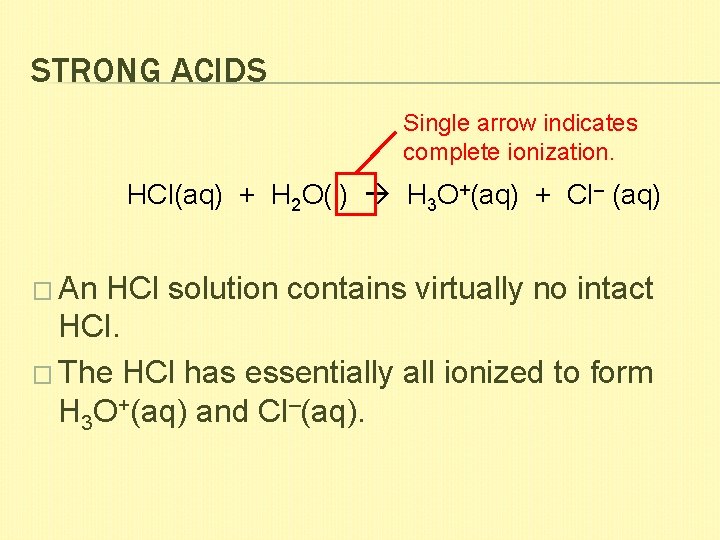
STRONG ACIDS Single arrow indicates complete ionization. HCl(aq) + H 2 O(l) H 3 O+(aq) + Cl– (aq) � An HCl solution contains virtually no intact HCl. � The HCl has essentially all ionized to form H 3 O+(aq) and Cl–(aq).

DESCRIBING CONCENTRATIONS OF STRONG ACIDS �A 1. 0 M HCl solution has an H 3 O+ concentration of 1. 0 M. If [HCl] = 1. 0 M, then [H 3 O+] = 1. 0 M
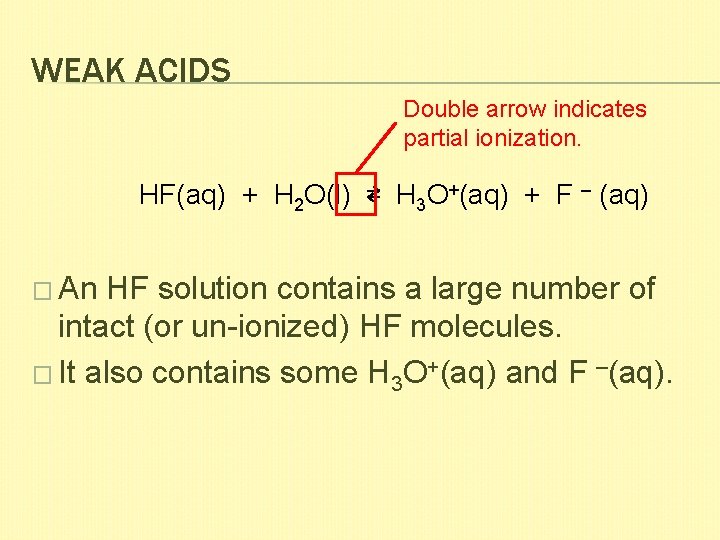
WEAK ACIDS Double arrow indicates partial ionization. HF(aq) + H 2 O(l) ⇄ H 3 O+(aq) + F – (aq) � An HF solution contains a large number of intact (or un-ionized) HF molecules. � It also contains some H 3 O+(aq) and F –(aq).

DESCRIBING CONCENTRATIONS OF WEAK ACIDS �A 1. 0 M HF solution has an [H 3 O+] that is much less than 1. 0 M because only some of the HF molecules ionize to form H 3 O+. [H 3 O+] << [HF]
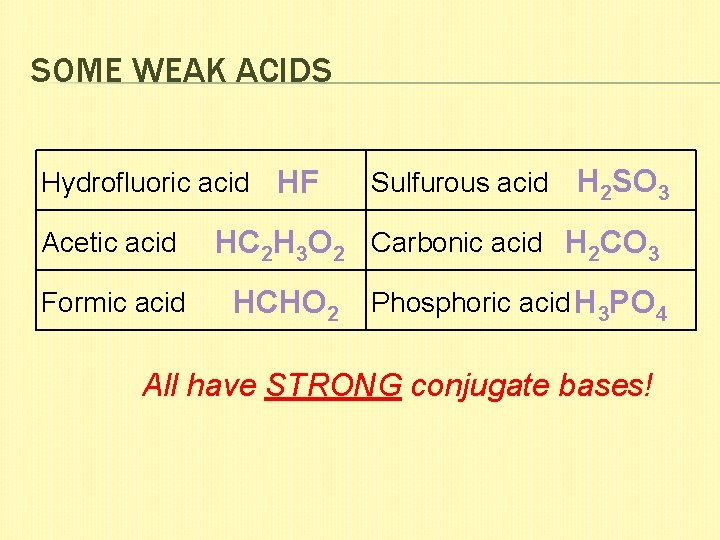
SOME WEAK ACIDS Hydrofluoric acid HF Acetic acid Formic acid Sulfurous acid H 2 SO 3 HC 2 H 3 O 2 Carbonic acid H 2 CO 3 HCHO 2 Phosphoric acid H 3 PO 4 All have STRONG conjugate bases!

CHEMISTRY 12. 4 STRENGTH OF ACIDS AND BASES & INTRO TO KA � https: //www. youtube. com/watch? v=u 93 Dp 0 I Xsak

ACID IONIZATION CONSTANT (Ka) �A way to quantify the relative strength of a weak acid � The equilibrium constant of the weak acid HA(aq) ⇄ H+(aq) + A–(aq)

Ka � Hydrocyanic acid (HCN) Ka = 4. 9 x 10 -10 Nitrous acid (HNO 2) Ka = 4. 6 x 10 -2 Acetic acid (HC 2 H 3 O 2) Ka = 1. 8 x 10 -5 Hydrofluoric acid (HF) Ka = 3. 5 x 10 -4 Chlorous acid (HCl. O 2) Ka = 1. 1 x 10 -2

BASE IONIZATION CONSTANT � The equilibrium expression for a base is known as Kb. � This expression is used to quantify the relative strength of a weak base. B + H 2 O HB+ + OHKb = [HB+] [OH-] [B]

AUTOIONIZATION OF WATER (VIDEO) � https: //www. youtube. com/watch? v=enq 6 e 81 Vv. ZA

AUTOIONIZATION OF WATER � Fredrich Kohlraush, around 1900 found that no matter how pure water is, it still conducts a minute amount of electric current. � This proves that water self-ionizes. � Remember - water is amophoteric! � Only about 2 in a billion water molecules are ionized at any instant!!

AUTOIONIZATION OF WATER

H+/OH- EQUILIBRIUM � The equilibrium expression used here is referred to as the autoionization constant for water, Kw. Kw = [H+] [OH-]

ACIDIC OR BASIC OR NEUTRAL? � It is important to recognize the meaning of Kw. In an aqueous solution at 25°C, no matter what it contains, [H+] x [OH-] = 1. 0 x 10 -14. � There �A are three possible situations: neutral solution, where [H+] = [OH-]. � An acidic solution, where [H+] > [OH-]. � A basic solution, where [H+] < [OH-].

OTHER TEMPERATURES… � If at a temperature OTHER THAN 25°C, the Kw will be a different value. � Remember that the numerical value of Keq is dependent on temperature!
![EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-52.jpg)
EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for each of the following solutions at 25°C, and state whether the solution is neutral, acidic, or basic. Ø 1. 0 x 10 -5 M OH- Ø 1. 0 x 10 -7 M OH-
![EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-53.jpg)
EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for each of the following solutions at 25°C, and state whether the solution is neutral, acidic, or basic. Ø 10. 0 M H+
![AT 10°C, THE AUTOIONIZATION OF WATER EQUALS 2. 9 X 10 -15. CALCULATE [H+] AT 10°C, THE AUTOIONIZATION OF WATER EQUALS 2. 9 X 10 -15. CALCULATE [H+]](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-54.jpg)
AT 10°C, THE AUTOIONIZATION OF WATER EQUALS 2. 9 X 10 -15. CALCULATE [H+] AND [OH-] AS NECESSARY UNDER EACH OF THE FOLLOWING CONDITIONS: � Calculate [H+] if [OH-] = 9. 3 x 10 -4 M. Is the solution acidic or basic? � Calculate [H+] and [OH-] for a neutral solution. � Calculate [OH-] if [H+] = 6. 7 x 10 -11 M. Is the solution acidic or basic?

CALCULATING THE PH OF A WEAK ACID 1. 2. 3. Write the balanced equation Write the equilibrium expression Use the ICE Box method I – Initial C – Change E - Equilibrium

CALCULATING THE PH OF A WEAK ACID Ex. 1 Calculate the p. H of a 1. 00 x 10 -4 M solution of acetic acid. (Ka = 1. 8 x 10 -5) HC 2 H 3 O 2 ⇌ H+ + C 2 H 3 O 2 Ka = I 1. 00 x 10 -4 0 0 [H+][C 2 H 3 O 2] C -x +x +x E 1. 00 x 10 -4 –x x x [HC 2 H 3 O 2 ] Ka = x 2 we can ignore the “- x” because 1. 00 x 10 -4 –x it is so small. 1. 8 x 10 -5 = x 2 1. 00 x 10 -4 4. 24 x 10 -5 = x p. H = -log (4. 24 x 10 -5 ) p. H = 4. 37

CALCULATING THE PH OF A WEAK ACID � The hypochlorite ion (OCl-) is a strong oxidizing agent found in household bleaches and disinfectants. It also has a relatively high affinity for protons (it is a much stronger base than Cl- for example) and forms the weakly acidic hypochlorous acid (HOCl, Ka = 3. 5 x 10 -8). Calculate the p. H of a 0. 100 M aqueous solution of hypochlorous acid.

CALCULATING THE PH OF A WEAK ACID

CALCULATING THE PH OF A WEAK BASE � The same steps are used to calculate weak bases and weak acids. (Make and ICE box). � Remember! You are now calculating p. OH and [OH-]!!

CALCULATING THE PH OF A WEAK BASE � Calculate the p. H for a 15 M solution of NH 3 (Kb = 1. 8 x 10 -5)

PERCENT IONIZATION OF A WEAK ACID Concentration of ionized acid (at equilibrium) Concentration of acid (initially)

EXAMPLES OF PERCENT IONIZATION � What is the percent ionization in a 0. 200 M solution nitrous acid if the [H 3 O+] is 0. 0096 M? � If a 2. 5 M solution of nitrous acid is 1. 4% ionized, what is the hydronium concentration in the solution? What is the p. H of the solution?

EXAMPLES OF PERCENT IONIZATION � What is the percent ionization in a 0. 200 M solution nitrous acid if the [H 3 O+] is 0. 0096 M? � If a 2. 5 M solution of nitrous acid is 1. 4% ionized, what is the % ionization hydronium concentration in the solution? What is the p. H of the DECREASES solution? with increasing concentration of acid

PERCENT IONIZATION OF A WEAK ACID Concentration of ionized acid (at equilibrium) Concentration of acid (initially) 1. 2. 3. Determine equilibrium concentrations for all species in the ionization reaction (Use ICE table if needed). Use Ka and the equilibrium constant expression to find [H+]. Use [H+]eq and the initial acid concentration, [HA]0, to solve for percent ionization.

1. 2. 3. Determine equilibrium concentrations for all species in the ionization reaction (Use ICE table if needed). Use Ka and the equilibrium constant expression to find [H+]. Use [H+]eq and the initial acid concentration, [HA]0, to solve for percent ionization. Find the percent ionization of a 2. 5 M HNO 2 solution. The Ka of nitrous acid is 4. 6 x 10 -4. HNO 2 ⇄ H+ + NO 2[HNO 2]eq = _________ x [H+] = _____ eq x [NO 2 -]eq = _____

1. 2. 3. Determine equilibrium concentrations for all species in the ionization reaction (Use ICE table if needed). Use Ka and the equilibrium constant expression to find [H+]. Use [H+]eq and the initial acid concentration, [HA]0, to solve for percent ionization. Find the percent ionization of a 2. 5 M HNO 2 solution. The Ka of nitrous acid is 4. 6 x 10 -4. HNO 2 ⇄ H+ + NO 2(x is small) Solve for x, which equals [H+]

ACID-BASE PROPERTIES OF SALTS � Salts are ionic compounds (cation + anion) � When dissolved in water, some salts are p. Hneutral, some are acidic, and some are basic � IN GENERAL, anions tend to form basic or neutral solutions. � IN GENERAL, cations tend to form acidic or neutral solutions.

ANIONS AS WEAK BASES � Anions can be thought as the conjugate base of an acid (the acid lost its proton, H+) This anion is the conjugate base of this acid Cl- HCl F- HF NO 3 - HNO 3 C 2 H 3 O 2 - HC 2 H 3 O 2

ANIONS AS WEAK BASES This anion Cl. FNO 3 C 2 H 3 O 2 - � In is the conjugate base of this acid HCl HF HNO 3 HC 2 H 3 O 2 general, the anion A- is the conjugate base of HA. � However, not every anion will act as a base… � Conjugate base of a weak acid, it’s a weak base � Conjugate base of a strong acid, it’s p. H-neutral

CLASSIFY THE ANION AS BASIC OR p. H-NEUTRAL: � NO 3� NO 2� C 2 H 3 O 2 -

CATIONS AS WEAK ACIDS � Cations that are the counterions of strong bases � Na. OH, KOH, and Ca(OH)2 are examples of strong bases, and form OH- in solution. � Cations Na+, K+, and Ca 2+ do not contribute to acidity � THEREFORE, they are p. H-neutral

Cations that are the conjugate acids of weak bases � A cation can be formed from any non-ionic weak base by adding a proton, H+. � Cations can be thought as the conjugate acid of an base (the base gained a proton, H+) � This cation acid of NH 4+ C 2 H 5 NH 3+ CH 3 NH 3+ is the conjugate this weak base NH 3 C 2 H 5 NH 2 CH 3 NH 2

This cation acid of is the conjugate this weak base NH 3 NH 4+ C 2 H 5 NH 2 C 2 H 5 NH 3+ CH 3 NH 2 CH 3 NH 3+ � Cations with the general formula BH+ will act as a weak acid according to the equation: BH+(aq) + H 2 O(aq) ⇄ H 3 O+(aq) + B(aq) � Conjugate acid of a weak base, it’s a weak acid

CLASSIFY THE CATION AS ACIDIC OR p. H-NEUTRAL: � C 5 H 5 NH+ � Ca 2+ � Cr 3+


STRONG ACID – STRONG BASE � Reacts completely � Yields neutral solution (p. H = 7) � Example: HCl + Na. OH H 2 O(l) + Na. Cl � Net ionic equation: H+(aq) + OH-(aq) H 2 O(l)

STRONG ACID – WEAK BASE � Reacts completely � Yields a weak acid (p. H < 7) � Weak acid is a product of the reaction (it’s the conj. acid of the strong base) � Example: HCl + CH 3 COONa(aq) CH 3 COOH(aq) + Na. Cl(aq) � Net Ionic Equation: H+(aq) + CH 3 COO-(aq) CH 3 COOH

WEAK ACID – STRONG BASE � Reacts completely � Yields a weak base (p. H > 7) � Weak base is a product of the reaction (it’s the conj. base of the strong acid) � Example: CH 3 COOH(aq) + Na. OH(aq) � Net Ionic Equation: CH 3 COOH(aq) + OH-(aq) CH 3 COO-(aq) + H 2 O(l)

WEAK ACID – WEAK BASE � Does not react well � Yields a buffer solution (described later) � Both a weak acid & weak base exist together � Example: CH 3 COOH(aq) + CH 3 COO-(aq) No net reaction

L acid X mol acid (H+) 1 L L base Y mol base (OH-) 1 L 1 mol base (OH-) 1 mol acid (H+) 1 mol base (OH-) 1 L Y mol base (OH-) 1 L X mol acid (H+)

� Equilibrium and base lies to the side of the weaker acid

Acids Donate ONE proton at a time! � monoprotic--acids donating one H+ (ex. HC 2 H 3 O 2) � diprotic--acids donating two H+'s (ex. H 2 C 2 O 4) � polyprotic--acids donating many H+'s (ex. H 3 PO 4) � � polyprotic bases--accept more than one H+; anions with − 2 and − 3 charges (ex. PO 43− ; HPO 42−) � Amphiprotic or amphoteric --molecules or ions that can behave as EITHER acids or bases; water, anions of weak acids (look at the examples above— sometimes water was an acid, sometimes it acted as a base) �

AUTOIONIZATION OF WATER (VIDEO) � https: //www. youtube. com/watch? v=enq 6 e 81 Vv. ZA � Add notes to page 7 using the video link above. Include examples given in the video.

AUTOIONIZATION OF WATER H 2 O(l) ⇄ H+(aq) + OH-(aq) 2 H 2 O ⇄ OH- + H 3 O+ � See video for notes

H+/OH- EQUILIBRIUM � See video for notes Kw =
![WHAT IS THE ION-PRODUCT CONSTANT? HOW DOES IT RELATE TO [H+] AND [OH-]? � WHAT IS THE ION-PRODUCT CONSTANT? HOW DOES IT RELATE TO [H+] AND [OH-]? �](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-86.jpg)
WHAT IS THE ION-PRODUCT CONSTANT? HOW DOES IT RELATE TO [H+] AND [OH-]? � See video for notes

ACIDIC OR BASIC OR NEUTRAL? � It is important to recognize the meaning of Kw. In an aqueous solution at 25°C, no matter what it contains, [H+] x [OH-] = 1. 0 x 10 -14. � There �A are three possible situations: neutral solution, where [H+] = [OH-]. � An acidic solution, where [H+] > [OH-]. � A basic solution, where [H+] < [OH-].

OTHER TEMPERATURES… (ADD TO p. 7) � If at a temperature OTHER THAN 25°C, the Kw will be a different value. � Remember that the numerical value of Keq is dependent on temperature!
![EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-89.jpg)
EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for each of the following solutions at 25°C, and state whether the solution is neutral, acidic, or basic. Ø 1. 0 x 10 -5 M OH- Ø 1. 0 x 10 -7 M OH-
![EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-90.jpg)
EXAMPLE 1 (since it’s at 25°C) � Calculate [H+] or [OH-] as required for each of the following solutions at 25°C, and state whether the solution is neutral, acidic, or basic. Ø 10. 0 M H+

EXAMPLE 2 AT 60°C, THE VALUE OF KW IS 1 X 10 -13. � Using Le Chatelier’s principle, predict whether the reaction is exothermic or endothermic 2 H 2 O ⇄ H 3 O+ + OH- At 25°C, Kw = 1 x 10 -14 At a higher temperature, Kw = 1 x 10 -13. Kw increases with temperature, energy is a reactant, and so the process must be endothermic.

EXAMPLE 2 AT 60°C, THE VALUE OF KW IS 1 X 10 -13. � Calculate [H+] and [OH-] in a neutral solution at 60°C. 2 H 2 O ⇄ H 3 O+ + OH-
![AT 10°C, THE AUTOIONIZATION OF WATER EQUALS 2. 9 X 10 -15. CALCULATE [H+] AT 10°C, THE AUTOIONIZATION OF WATER EQUALS 2. 9 X 10 -15. CALCULATE [H+]](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-93.jpg)
AT 10°C, THE AUTOIONIZATION OF WATER EQUALS 2. 9 X 10 -15. CALCULATE [H+] AND [OH-] AS NECESSARY UNDER EACH OF THE FOLLOWING CONDITIONS: � Calculate [H+] if [OH-] = 9. 3 x 10 -4 M. Is the solution acidic or basic? � Calculate [H+] and [OH-] for a neutral solution. � Calculate [OH-] if [H+] = 6. 7 x 10 -11 M. Is the solution acidic or basic?

p. H SCALE � https: //www. youtube. com/watch? v=p. FK 16 Gs U 1 e 4 � Add notes to page 9 using the video link above. Include examples given in the video.

p. H SCALE � See 0 video for notes 7 14
![WHY DOES [H+] = [OH-] AT PH 7? � See video for notes WHY DOES [H+] = [OH-] AT PH 7? � See video for notes](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-96.jpg)
WHY DOES [H+] = [OH-] AT PH 7? � See video for notes

p. H AND p. OH � See video for notes p. H 7 6 3 12 [H+] (M) [OH-] (M) p. OH

p. H AND p. OH � What is the pattern between p. H and [H+]? � What is the relationship between [H+] and [OH-]? � How is p. H related to p. OH?

CALCULATING p. H � https: //www. youtube. com/watch? v=aw- Y 1 d 2 IIJc � Add notes to page 10 using the video link above. Include examples given in the video.
![� See video for notes p. H = – log[H+] p. OH = – � See video for notes p. H = – log[H+] p. OH = –](http://slidetodoc.com/presentation_image_h2/8905fcabf16b8bc7d086d51a472f7ad1/image-100.jpg)
� See video for notes p. H = – log[H+] p. OH = – log[OH–] The p. H of a solution is 3. 75, what is the Hydrogen ion concentration, [H+]?

EXAMPLES � What is the p. H of a solution with a hydrogen ion concentration of 2. 0 x 1011? 10. 70 � Calculate the p. H of a 1. 0 x 10 -3 M OH- solution. (Hint: Use Kw to first find 11. 00 + [H ]. Assume 25°C. ) � What is the p. OH of a solution with a hydroxide ion concentration of 1. 8 x 10 -4? 3. 74 14. 00 solution. (Hint: Use Kw to first find [OH-]. H+ � Calculate the p. OH of a 1. 0 M Assume 25°C. ) � The p. H of p. OH human blood was to be 7. 41 at-] 25°C. Calculate the +]measured -8 M, -7 M = 6. 59, [H = 3. 9 x 10 [OH = 2. 6 x 10 p. OH, [H+], and [OH-] for the sample.
- Slides: 101