AP Chemistry 2020 2021 Anthony Petras Day 22















- Slides: 28

AP Chemistry 2020 -2021 Anthony Petras

Day 22 • Attendance • Recording On • Quiz 3. 1 -3. 8 MC Review (avg 9/15) • Quiz 3. 1 -3. 8 FR Review (avg 7/11) • 3. 9 Separation of Solutions and Mixtures Chromatography • Recording Links

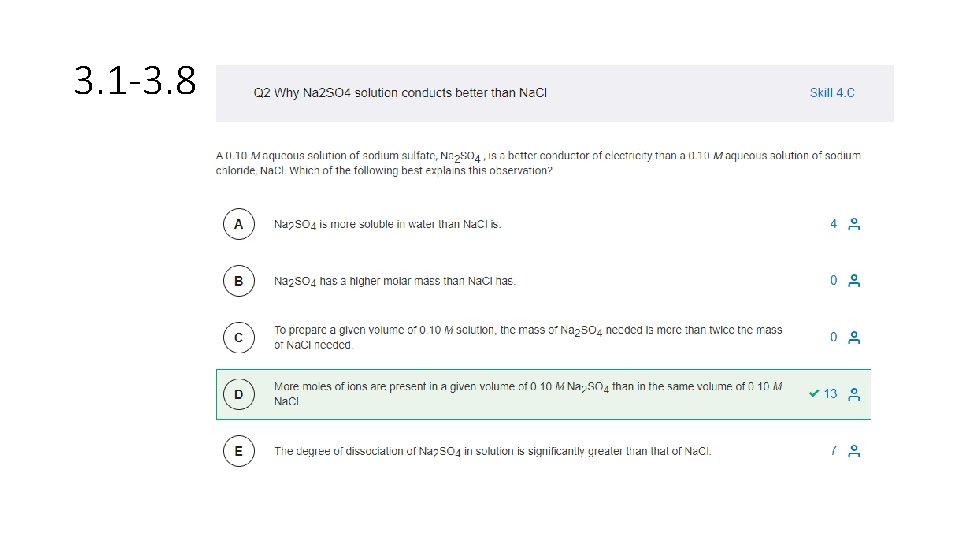
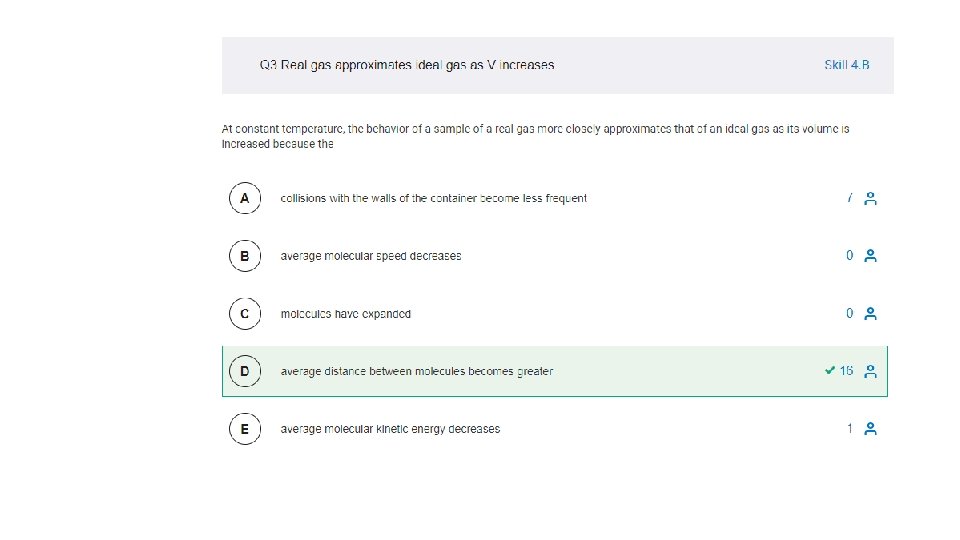
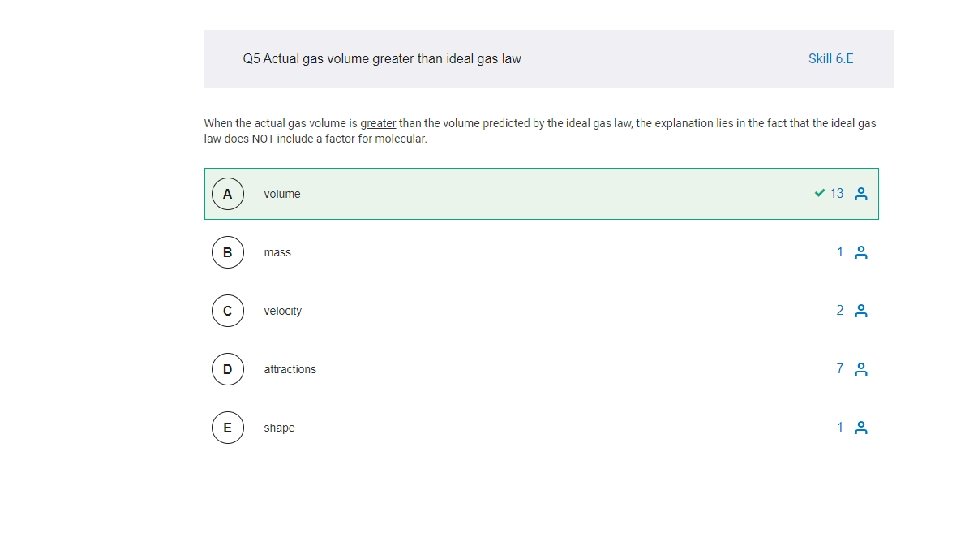
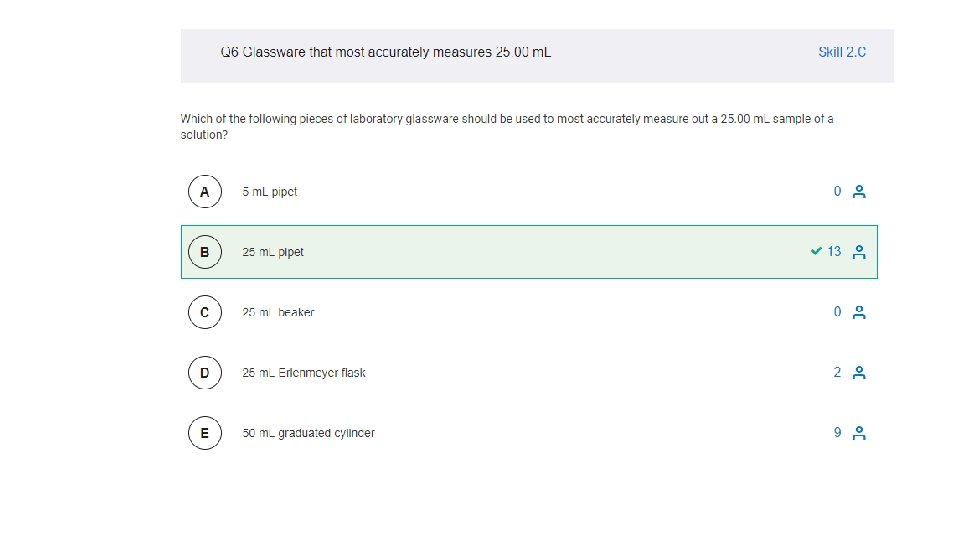
3. 1 -3. 8 MC










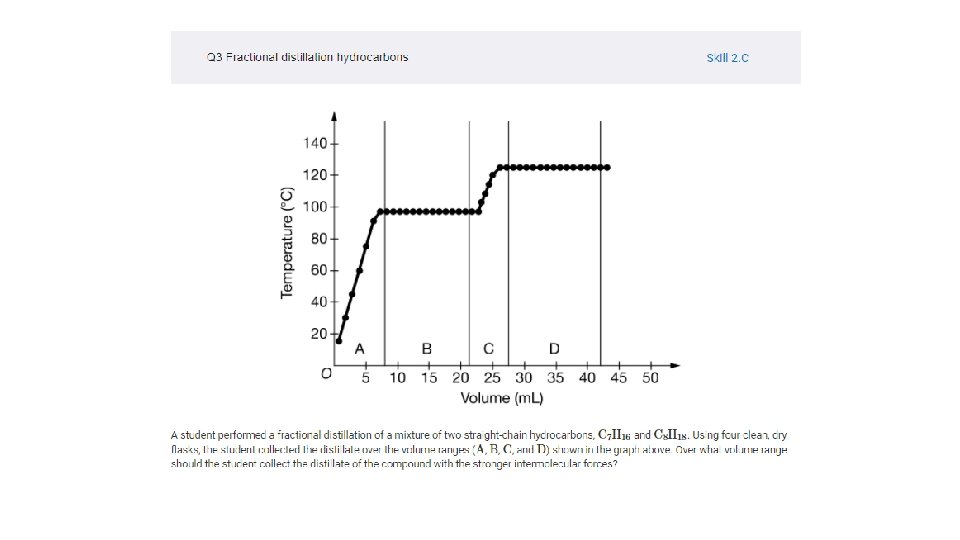
3. 9 Separation of Solutions and Mixtures Chromatography • L. O. : Explain the relationship between the solubility of ionic and molecular compounds in aqueous and nonaqueous solvents, and the intermolecular interactions between particles. • The components of a liquid solution cannot be separated by filtration. They can, however, be separated using processes that take advantage of differences in the intermolecular interactions of the components. a. Chromatography (paper, thin-layer, and column) separates chemical species by taking advantage of the differential strength of intermolecular interactions between and among the components of the solution (the mobile phase) and with the surface components of the stationary phase. b. Distillation separates chemical species by taking advantage of the differential strength of intermolecular interactions between and among the components and the effects these interactions have on the vapor pressures of the components in the mixture.

Chromatography • Physical method used to separate mixtures. Mixture is distributed into two phases; a stationary phase and a mobile phase. The mobile phase moves through the stationary phase carrying the mixture. Each component in the mixture will have a different affinity for the stationary phase. A high affinity for the stationary phase means a component in the mixture will not travel far. Less affinity causes a component to travel further. The differing distances traveled, results in separation. The resulting separated bands are called a chromatogram. • Rf values can be determined for each component; Df = Distance traveled by component; Ds = Distance traveled by solvent. • R f = Df ÷ Ds

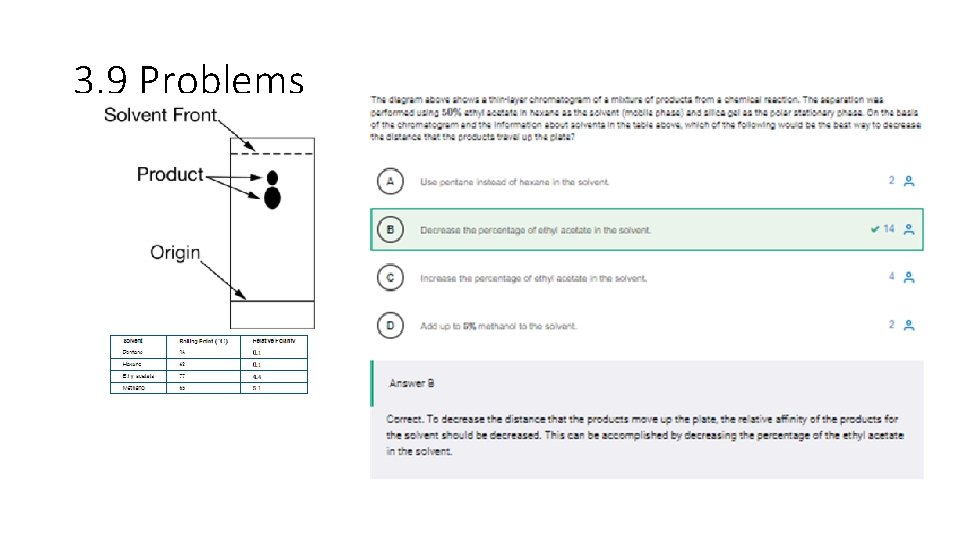
3. 9 Problems

3. 9 • A student obtains a liquid sample of green food coloring that is known to contain a mixture of two solid pigments, one blue and one yellow, dissolved in an aqueous solution of ethanol. Which of the following laboratory setups is most appropriate for the student to use in order to separate and collect a substantial sample of each of the two pigments? • Answer D • Correct. The molecules of the two pigments will most likely have different affinities for a liquid phase and a stationary phase that could be used in a chromatography column. The different times it takes each pigment to pass through the column allow for collection of each pigment separately.


3. 9

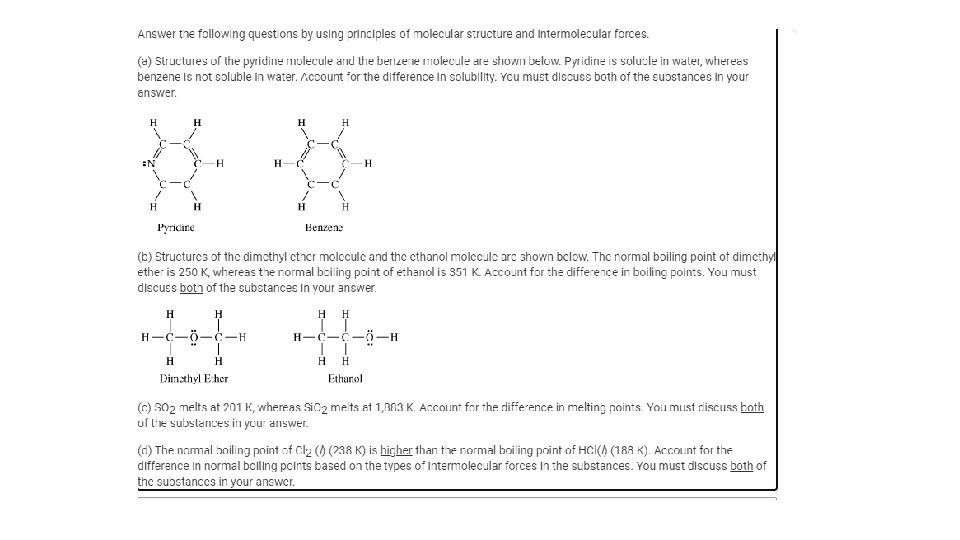
3. 10 Solubility • L. O. : Explain the relationship between the solubility of ionic and molecular compounds in aqueous and nonaqueous solvents, and the intermolecular interactions between particles. • Substances with similar intermolecular interactions tend to be miscible or soluble in one another.

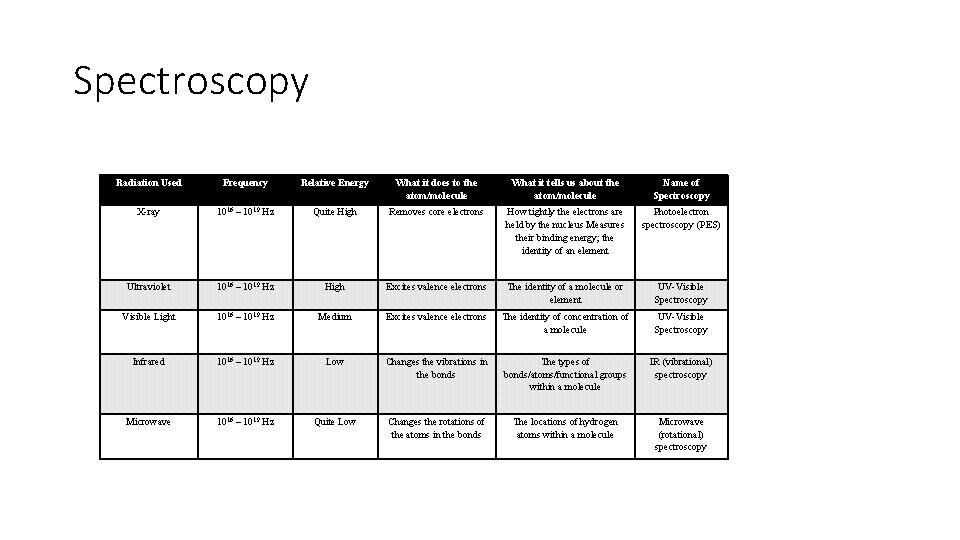
3. 11 Spectroscopy and the Electromagnetic Spectrum • Explain the relationship between a region of the electromagnetic spectrum and the types of molecular or electronic transitions associated with that region. • Differences in absorption or emission of photons in different spectral regions are related to the different types of molecular motion or electronic transition: • a. Microwave radiation is associated with transitions in molecular rotational levels. • b. Infrared radiation is associated with transitions in molecular vibrational levels. • c. Ultraviolet/visible radiation is associated with transitions in electronic energy levels.

Spectroscopy Radiation Used Frequency Relative Energy What it does to the atom/molecule What it tells us about the atom/molecule Name of Spectroscopy X-ray 1016 – 1019 Hz Quite High Removes core electrons How tightly the electrons are held by the nucleus Measures their binding energy; the identity of an element Photoelectron spectroscopy (PES) Ultraviolet 1016 – 1019 Hz High Excites valence electrons The identity of a molecule or element UV-Visible Spectroscopy Visible Light 1016 – 1019 Hz Medium Excites valence electrons The identity of concentration of a molecule UV-Visible Spectroscopy Infrared 1016 – 1019 Hz Low Changes the vibrations in the bonds The types of bonds/atoms/functional groups within a molecule IR (vibrational) spectroscopy Microwave 1016 – 1019 Hz Quite Low Changes the rotations of the atoms in the bonds The locations of hydrogen atoms within a molecule Microwave (rotational) spectroscopy

3. 12 Photoelectric Effect • L. O. : Explain the properties of an absorbed or emitted photon in relationship to an electronic transition in an atom or molecule • When a photon is absorbed (or emitted) by an atom or molecule, the energy of the species is increased (or decreased) by an amount equal to the energy of the photon. • The wavelength of the electromagnetic wave is related to its frequency and the speed of light by the equation: EQN: c = λν. • The energy of a photon is related to the frequency of the electromagnetic wave through Planck’s equation (E = ℎν).

3. 12 Photoelectric Effect • The Photoelectric Effect is the observation that photons short-wave (visible or ultraviolet) light can cause it to emit electrons. This is an example of ionization, with the photons providing the ionization energy. When visible or ultraviolet light is shone on a substance, the energy from the photons of light excites electrons in the substance. If the energy exceeds the ionization energy of an atom in the substance, the electron is emitted. The emitted electrons are called photoelectrons.

3. 13 Beer-Lambert Law • L. O. : Explain the amount of light absorbed by a solution of molecules or ions in relationship to the concentration, path length, and molar absorptivity. • The Beer-Lambert law relates the absorption of light by a solution to three variables according to the equation: EQN: A = εbc. • The molar absorptivity ε describes how intensely a sample of molecules or ions absorbs light of a specific wavelength. The path length b and concentration c are proportional to the number of absorbing species. • In most experiments the path length and wavelength of light are held constant. In such cases, the absorbance is proportional only to the concentration of absorbing molecules or ions. • On-line demo: http: //phet. colorado. edu/sims/html/beers-lawlab/latest/beers-law-lab_en. html

Beer’s Law Lab • Purpose: The primary objective of this experiment is to determine the concentration of an unknown copper (II) sulfate solution and to prepare a solution of copper(II) sulfate pentahydrate of desired molarity. The Cu. SO 4 solution used in this experiment has a blue color, so Colorimeter users will be instructed to use the red LED. Spectrometer users will determine an appropriate wavelength based on the absorbance spectrum of the solution. A higher concentration of the colored solution absorbs more light (and transmits less) than a solution of lower concentration. • You will prepare five copper (II) sulfate solutions of known concentration (standard solutions). Each solution is transferred to a small, rectangular cuvette that is placed into the Colorimeter or Spectrometer. The amount of light that penetrates the solution and strikes the photocell is used to compute the absorbance of each solution. When you graph absorbance vs. concentration for the standard solutions, a direct relationship should result. The direct relationship between absorbance and concentration for a solution is known as Beer’s law. • You will determine the concentration of an unknown Cu. SO 4 solution by measuring its absorbance. By locating the absorbance of the unknown on the vertical axis of the graph, the corresponding concentration can be found on the horizontal axis. The concentration of the unknown can also be found using the slope of the Beer’s law curve.

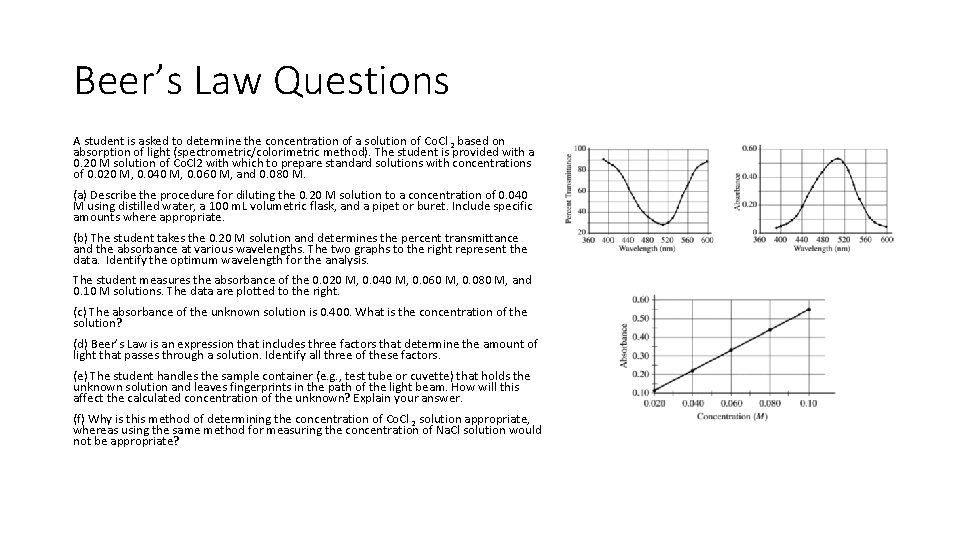
Beer’s Law Questions A student is asked to determine the concentration of a solution of Co. Cl 2 based on absorption of light (spectrometric/colorimetric method). The student is provided with a 0. 20 M solution of Co. Cl 2 with which to prepare standard solutions with concentrations of 0. 020 M, 0. 040 M, 0. 060 M, and 0. 080 M. (a) Describe the procedure for diluting the 0. 20 M solution to a concentration of 0. 040 M using distilled water, a 100 m. L volumetric flask, and a pipet or buret. Include specific amounts where appropriate. (b) The student takes the 0. 20 M solution and determines the percent transmittance and the absorbance at various wavelengths. The two graphs to the right represent the data. Identify the optimum wavelength for the analysis. The student measures the absorbance of the 0. 020 M, 0. 040 M, 0. 060 M, 0. 080 M, and 0. 10 M solutions. The data are plotted to the right. (c) The absorbance of the unknown solution is 0. 400. What is the concentration of the solution? (d) Beer’s Law is an expression that includes three factors that determine the amount of light that passes through a solution. Identify all three of these factors. (e) The student handles the sample container (e. g. , test tube or cuvette) that holds the unknown solution and leaves fingerprints in the path of the light beam. How will this affect the calculated concentration of the unknown? Explain your answer. (f) Why is this method of determining the concentration of Co. Cl 2 solution appropriate, whereas using the same method for measuring the concentration of Na. Cl solution would not be appropriate?

Homework – Day 22 • Review this Power. Point • Thursday: Finish Power. Point, Review 3. 9 -3. 13 from AP Classroom • Prepare for test on Unit 3 - 12/8 • Review Videos & Complete 3. 10, 3. 11, 3. 12 & 3. 13 (due 11/30)

Recording Links – New Quarter – New Links • Period 1: • 23. https: //us. bbcollab. com/recording/6 a 8 a 868073 bd 4 f 82 bb 3462 f 671 dde 095 • 22. https: //us. bbcollab. com/recording/eec 12 a 527 f 2742 a 5 beb 1 d 73 ef 467 ead 0 • 21. https: //us. bbcollab. com/recording/30 af 65 c 5 dd 64401386035 b 1 c 6104 f 7 c 6 • 20. https: //us. bbcollab. com/recording/d 6063 a 9 fe 6 f 14971 bf 77 cd 7 b 196 b 66 c 2 • 19. https: //us. bbcollab. com/recording/f 04 eac 7 ad 13 c 40 c 99949 ed 4 c 2 ed 2 bc 3 d • 18. https: //us. bbcollab. com/recording/4 ced 3 a 4 c 8 a 9 c 46828 dcd 40 d 6 d 06 f 7 a 66 • 17. https: //us. bbcollab. com/recording/cd 877 eb 7702145 bda 62 cfea 20 f 6 ad 4 eb • 16. https: //us. bbcollab. com/recording/ae 785 f 0803644 cd 38558 d 22 eced 5 e 785