AP Chapter 1 Chemical Foundations AP Learning Objectives

AP* Chapter 1 Chemical Foundations

AP Learning Objectives § LO 2. 7 The student is able to explain how solutes can be separated by chromatography based on intermolecular interactions. (Sec 1. 10) § LO 2. 10 The student can design and/or interpret the results of a separation experiment (filtration, paper chromatography, column chromatography, or distillation) in terms of the relative strength of interactions among and between the components. (Sec 1. 10) § LO 3. 10 The student is able to evaluate the classification of a process as a physical change, chemical change, or ambiguous change based on both macroscopic observations and the distinction between rearrangement of covalent interactions and noncovalent interactions. (Sec 1. 10)

Section 1. 1 Chemistry: An Overview § § A main challenge of chemistry is to understand the connection between the macroscopic world that we experience and the microscopic world of atoms and molecules. You must learn to think on the atomic level. 3

Section 1. 1 Chemistry: An Overview Atoms vs. Molecules § Matter is composed of tiny particles called atoms. § Atom: smallest part of an element that is still that element. § Molecule: Two or more atoms joined and acting as a unit. 4

Section 1. 1 Chemistry: An Overview Oxygen and Hydrogen Molecules • Use subscripts when more than one atom is in the molecule. 5

Section 1. 1 Chemistry: An Overview A Chemical Reaction § One substance changes to another by reorganizing the way the atoms are attached to each other. 6

Section 1. 2 The Scientific Method Science § § Science is a framework for gaining and organizing knowledge. Science is a plan of action — a procedure for processing and understanding certain types of information. Scientists are always challenging our current beliefs about science, asking questions, and experimenting to gain new knowledge. Scientific method is needed. 7
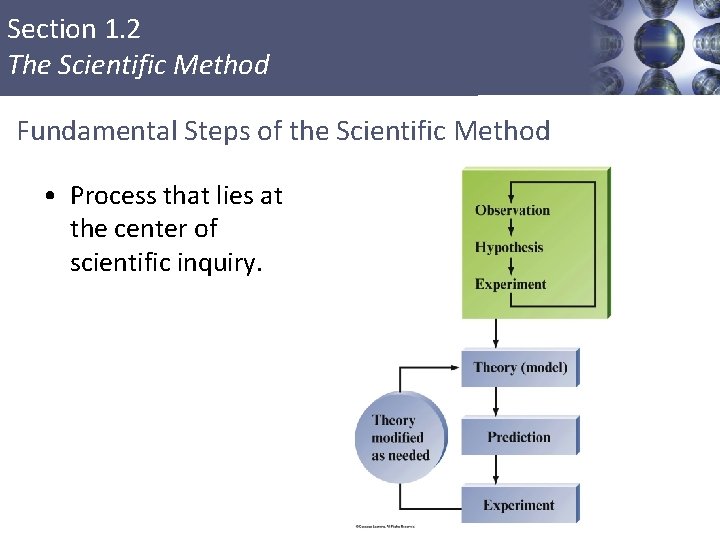
Section 1. 2 The Scientific Method Fundamental Steps of the Scientific Method • Process that lies at the center of scientific inquiry. 8

Section 1. 2 The Scientific Method Fundamental Steps of the Scientific Method § § § § Observation: Hypothesis: Experiment: Theory: Prediction: Experiment: Law:

Section 1. 2 The Scientific Method Fundamental Steps of the Scientific Method § Observation: Something that can be witnessed and recorded § Hypothesis: A possible explanation for an observation. § Experiment: § Theory: tested hypothesis § Prediction: § Experiment: § Law: A summary of repeatable observed (measurable) behavior.

Section 1. 3 Units of Measurement Nature of Measurement • Quantitative observation consisting of two parts. § number § scale (unit) • Examples § 20 grams § 6. 63 × 10 -34 joule·second 11

Section 1. 3 Units of Measurement

Section 1. 3 Units of Measurement The Fundamental SI Units Physical Quantity Mass Length Time Temperature Electric current Amount of substance Volume Name of Unit kilogram meter second Abbreviation kg m s kelvin ampere mole Liter K A mol L 13

Section 1. 3 Units of Measurement Prefixes Used in the SI System • Prefixes are used to change the size of the unit. 14

Section 1. 3 Units of Measurement Prefixes Used in the SI System 15

Section 1. 3 Units of Measurement Mass ≠ Weight § Mass is a measure of the resistance of an object to a change in its state of motion. Mass does not vary. § Weight is the force that gravity exerts on an object. Weight varies with the strength of the gravitational field. 16

Section 1. 4 Uncertainty in Measurement § § § A digit that must be estimated in a measurement is called uncertain. A measurement always has some degree of uncertainty. It is dependent on the precision of the measuring device. Record the certain digits and the first uncertain digit (the estimated number). 17

Section 1. 4 Uncertainty in Measurement of Volume Using a Buret § The volume is read at the bottom of the liquid curve (meniscus). § Meniscus of the liquid occurs at about 20. 15 m. L. § Certain digits: 20. 15 § Uncertain digit: 20. 15 18

Section 1. 4 Uncertainty in Measurement Precision and Accuracy • Agreement of a particular value with the true value. Precision • Degree of agreement among several measurements of the same quantity. 19

Section 1. 4 Uncertainty in Measurement Precision and Accuracy 20

Section 1. 5 Accuracy & Precision Significant Figures and Calculations • Consider 3 sets of data that have been recorded after measuring a piece of wood that is exactly 6. 000 m long. Set X Set Y Set Z 5. 864 m 6. 002 m 5. 878 m 6. 004 m 5. 868 m Average length 5. 871 m 6. 003 m 5. 870 m • Which set of data is the most accurate? • Set Y • Which set of data is the most precise? • Set Y

Section 1. 5 Significant Figures and Calculations Rules for Counting Significant Figures 1. Nonzero integers always count as significant figures. § 3456 has ______ sig figs (significant figures). 3456 has 4 sig figs (significant figures). 33456 has 4 sig figs 22

Section 1. 5 Significant Figures and Calculations Rules for Counting Significant Figures 2. There are three classes of zeros. a. Leading zeros are zeros that precede all the nonzero digits. These do not count as significant figures. § 0. 048 has ______ sig figs. 2 sig figs 23

Section 1. 5 Significant Figures and Calculations Rules for Counting Significant Figures b. Captive zeros are zeros between nonzero digits. These always count as significant figures. § 16. 07 has 4 sig figs 24

Section 1. 5 Significant Figures and Calculations Rules for Counting Significant Figures c. Trailing zeros are zeros at the right end of the number. They are significant only if the number contains a decimal point. § 9. 300 has ______ sig figs. § 150 has _______ sig figs. 9. 300 has 4 sig figs. 150 has 2 sig figs. 25

Section 1. 5 Significant Figures and Calculations Rules for Counting Significant Figures 3. Exact numbers have an infinite number of significant figures. § 1 inch = 2. 54 cm, exactly. § 9 pencils (obtained by counting). 26

Section 1. 5 Significant Figures and Calculations Exponential Notation Example § 300. written as 3. 00 × 102 § Contains three significant figures. § Two Advantages § Number of significant figures can be easily indicated. § Fewer zeros are needed to write a very large or very small number. § 27

Section 1. 5 Significant Figures and Calculations Significant Figures in Mathematical Operations 1. For multiplication or division, the number of significant figures in the result is the same as the number in the least precise measurement used in the calculation. 1. 342 × 5. 5 = 7. 381 7. 4 28

Section 1. 5 Significant Figures and Calculations Significant Figures in Mathematical Operations 2. For addition or subtraction, the result has the same number of decimal places as the least precise measurement used in the calculation. 29

Section 1. 5 Significant Figures and Calculations CONCEPT CHECK! You have water in each graduated cylinder shown. You then add both samples to a beaker (assume that all of the liquid is transferred). How would you write the number describing the total volume? Refer to the previous slide for adding 3. 1 m. L What limits the precision of the total volume? 30

Section 1. 5 Significant Figures and Calculations Rules for Rounding / not as important as Sig Figs § 1. Round at the end of a series of calculations, NOT after each step § 2. Use only the first number to the right of the last sig fig to decide whether or not to round § a. Less than 5, the last significant digit is unchanged § b. 5 or more, the last significant digit is increased by 1

Section 1. 5 Significant Figures and Calculations yes § 1. Significant figures rules will be observed in all calculations throughout the year in this course. You need never ask, "Do we have to watch our sig figs? " § The answer is always "Yes!"
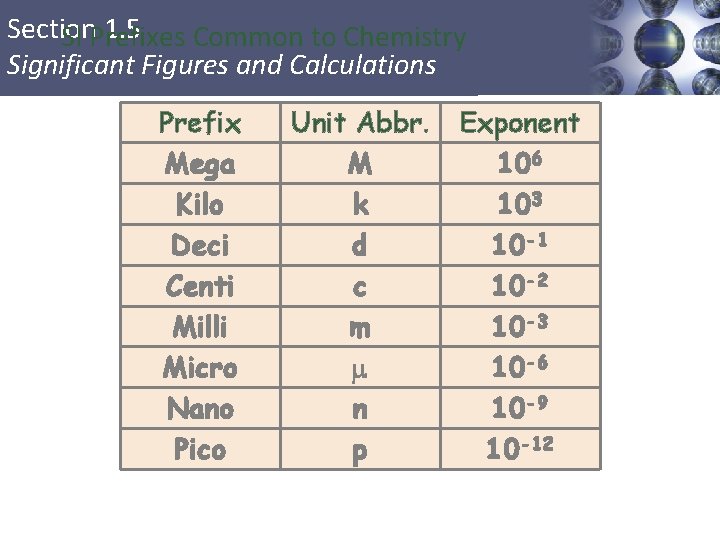
Section 1. 5 SI Prefixes Common to Chemistry Significant Figures and Calculations Prefix Mega Kilo Deci Centi Milli Micro Nano Pico Unit Abbr. M k d c m n p Exponent 106 103 10 -1 10 -2 10 -3 10 -6 10 -9 10 -12

Section 1. 5 Significant Figures and Calculations How many significant figures in each of the following? Sig Fig Practice #1 1. 0070 m 5 sig figs 17. 10 kg 4 sig figs 100, 890 L 5 sig figs 3. 29 x 103 s 3 sig figs 0. 0054 cm 2 sig figs 3, 200, 000 2 sig figs

Rules Section 1. 5 for Significant Figures in Mathematical Significant Figures and Calculations Operations § Multiplication and Division: § # sig figs in the result equals the number in the least precise measurement used in the calculation. § 6. 38 x 2. 0 = § 12. 76 13 (2 sig figs)
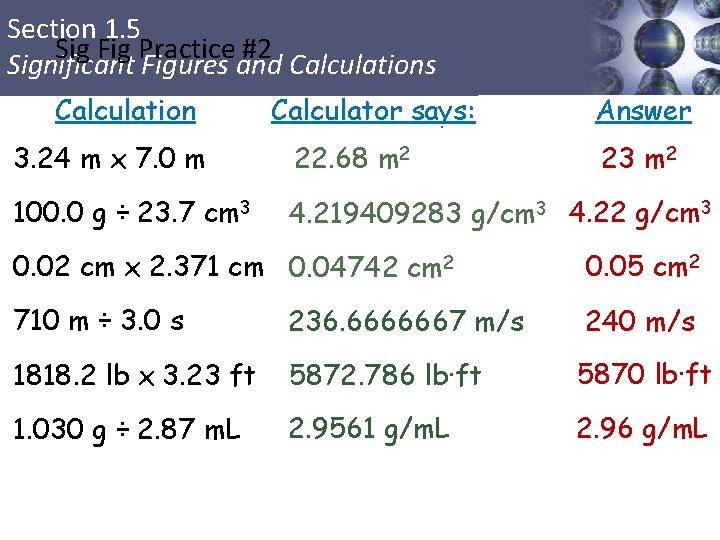
Section 1. 5 Sig Fig Practice #2 Significant Figures and Calculations Calculation Calculator says: Answer 3. 24 m x 7. 0 m 22. 68 m 2 100. 0 g ÷ 23. 7 cm 3 4. 219409283 g/cm 3 4. 22 g/cm 3 23 m 2 0. 02 cm x 2. 371 cm 0. 04742 cm 2 0. 05 cm 2 710 m ÷ 3. 0 s 236. 6666667 m/s 240 m/s 1818. 2 lb x 3. 23 ft 5872. 786 lb·ft 5870 lb·ft 1. 030 g ÷ 2. 87 m. L 2. 9561 g/m. L 2. 96 g/m. L

Section 1. 5 Significant Figures and Calculations Rules for Significant Figures in Mathematical Operations § Addition and Subtraction: The number of decimal places in the result equals the number of decimal places in the least precise measurement. § 6. 8 + 11. 934 = § 18. 734 18. 7 (3 sig figs)
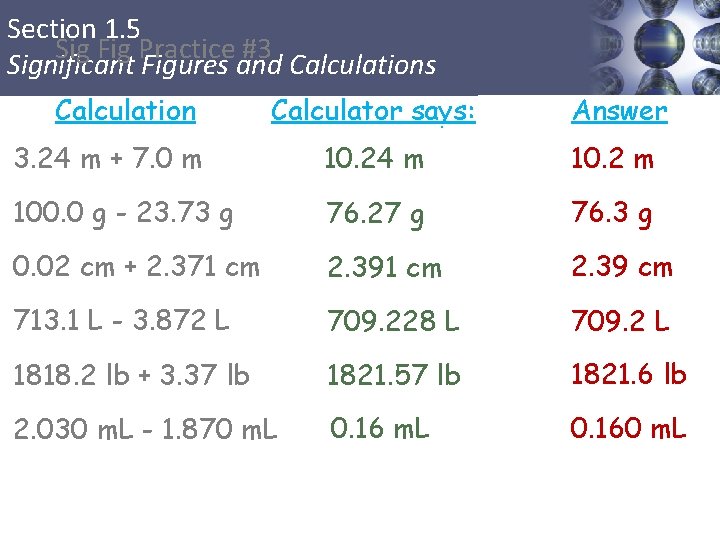
Section 1. 5 Sig Fig Practice #3 Significant Figures and Calculations Calculation Calculator says: Answer 3. 24 m + 7. 0 m 10. 24 m 10. 2 m 100. 0 g - 23. 73 g 76. 27 g 76. 3 g 0. 02 cm + 2. 371 cm 2. 39 cm 713. 1 L - 3. 872 L 709. 228 L 709. 2 L 1818. 2 lb + 3. 37 lb 1821. 57 lb 1821. 6 lb 2. 030 m. L - 1. 870 m. L 0. 160 m. L

Section 1. 5 Uncertainty in Measurement Significant Figures and Calculations § A digit that must be estimated is called uncertain. A measurement always has some degree of uncertainty. v Measurements are performed with instruments v No instrument can read to an infinite number of decimal places

Section 1. 6 Learning to Solve Problems Systematically Questions to ask when approaching a problem § 1. What is my goal? Or you might phrase it as: Where am I going? § 2. Where am I starting? Or you might phrase it as: What do I know? § 3. How do I proceed from where I start to where I want to go? Or you might say: How do I get there?

Section 1. 6 Learning to Solve Problems Systematically

Section 1. 6 Learning to Solve Problems Systematically

Section 1. 6 Learning to Solve Problems Systematically

Section 1. 7 Dimensional Analysis § Use when converting a given result from one system of units to another. § To convert from one unit to another, use the equivalence statement that relates the two units. § Derive the appropriate unit factor by looking at the direction of the required change (to cancel the unwanted units). § Multiply the quantity to be converted by the unit factor to give the quantity with the desired units. 44

Section 1. 7 Dimensional Analysis Example #1 A golfer putted a golf ball 6. 8 ft across a green. How many inches does this represent? • To convert from one unit to another, use the equivalence statement that relates the two units. 1 ft = 12 in The two unit factors are: 45

Section 1. 7 Dimensional Analysis Example #1 A golfer putted a golf ball 6. 8 ft across a green. How many inches does this represent? § Derive the appropriate unit factor by looking at the direction of the required change (to cancel the unwanted units). 46

Section 1. 7 Dimensional Analysis Example #1 A golfer putted a golf ball 6. 8 ft across a green. How many inches does this represent? § Multiply the quantity to be converted by the unit factor to give the quantity with the desired units. 47

Section 1. 7 Dimensional Analysis Example #2 An iron sample has a mass of 4. 50 lb. What is the mass of this sample in grams? (1 kg = 2. 2046 lbs; 1 kg = 1000 g) 48

Section 1. 7 Dimensional Analysis CONCEPT CHECK! What data would you need to estimate the money you would spend on gasoline to drive your car from New York to Los Angeles? Provide estimates of values and a sample calculation. 49

Section 1. 8 Temperature Three Systems for Measuring Temperature § § § Fahrenheit Celsius Kelvin 50
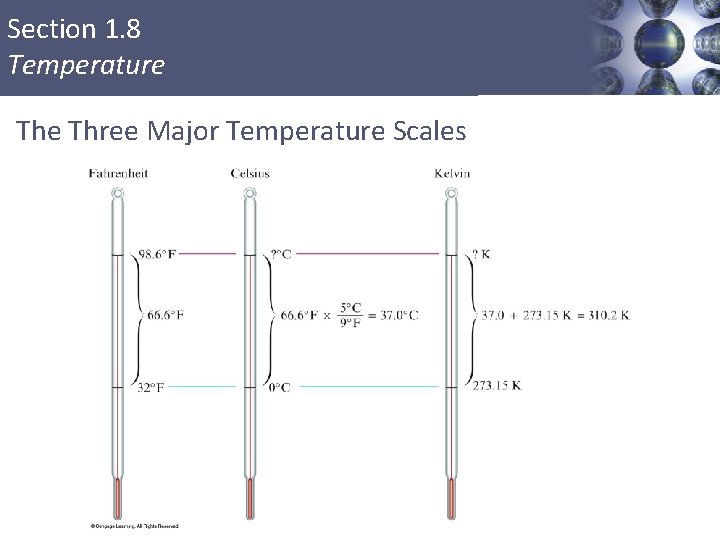
Section 1. 8 Temperature Three Major Temperature Scales

Section 1. 8 Temperature Converting Between Scales 52

Section 1. 8 Temperature EXERCISE! At what temperature does °C = °F? 53

Section 1. 8 Temperature EXERCISE! § Since °C equals °F, they both should be the same value (designated as variable x). Use one of the conversion equations such as: § Substitute in the value of x for both TC and TF. Solve for x. § 54

Section 1. 8 Temperature EXERCISE! 55

Section 1. 9 Density § § Mass of substance per unit volume of the substance. Common units are g/cm 3 or g/m. L. 56

Section 1. 9 Density Example #1 A certain mineral has a mass of 17. 8 g and a volume of 2. 35 cm 3. What is the density of this mineral? 57

Section 1. 9 Density Example #2 What is the mass of a 49. 6 -m. L sample of a liquid, which has a density of 0. 85 g/m. L? 58

Section 1. 10 Classification of Matter AP Learning Objectives, Margin Notes and References § Learning Objectives § § § LO 2. 7 The student is able to explain how solutes can be separated by chromatography based on intermolecular interactions. LO 2. 10 The student can design and/or interpret the results of a separation experiment (filtration, paper chromatography, column chromatography, or distillation) in terms of the relative strength of interactions among and between the components. LO 3. 10 The student is able to evaluate the classification of a process as a physical change, chemical change, or ambiguous change based on both macroscopic observations and the distinction between rearrangement of covalent interactions and noncovalent interactions. § AP Margin Notes § § § Solutions do not contain components large enough to scatter visible light. See Tyndall effect in Chapter 11. The components of a solution can be separated by distillation. The components of a solution are so small that they cannot be separated by filtration. § Additional AP References § § LO 2. 10 (see APEC Lab 5, "Thin-Layer Chromatography”) LO 3. 10 (see APEC Lab 9, "Actions, Reactions, and Interactions”)

Section 1. 10 Classification of Matter § § Anything occupying space and having mass. Matter exists in three states. § Solid § Liquid § Gas 60

Section 1. 10 Classification of Matter The Three States of Water 61

Section 1. 10 Classification of Matter Solid § § Rigid Has fixed volume and shape. 62

Section 1. 10 Classification of Matter Structure of a Solid To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE 63

Section 1. 10 Classification of Matter Liquid § § Has definite volume but no specific shape. Assumes shape of container. 64

Section 1. 10 Classification of Matter Structure of a liquid To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE 65

Section 1. 10 Classification of Matter Gas § § Has no fixed volume or shape. Takes on the shape and volume of its container. 66

Section 1. 10 Classification of Matter Structure of a gas To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE 67

Section 1. 10 Classification of Matter Mixtures § Have variable composition. Homogeneous Mixture § Having visibly indistinguishable parts; solution. Heterogeneous Mixture § Having visibly distinguishable parts. 68
Section 1. 10 Classification of Matter Homogeneous Mixtures To play movie you must be in Slide Show Mode PC Users: Please wait for content to load, then click to play Mac Users: CLICK HERE Copyright © Cengage Learning. All rights reserved 69

Section 1. 10 Classification of Matter CONCEPT CHECK! Which of the following is a homogeneous mixture? § § § Pure water Gasoline Jar of jelly beans Soil Copper metal 70

Section 1. 10 Classification of Matter Physical Change in the form of a substance, not in its chemical composition. § Example: boiling or freezing water § Can be used to separate a mixture into pure compounds, but it will not break compounds into elements. § Distillation § Filtration § Chromatography § 71

Section 1. 10 Classification of Matter Chemical Change § A given substance becomes a new substance or substances with different properties and different composition. § Example: Bunsen burner (methane reacts with oxygen to form carbon dioxide and water) 72

Section 1. 10 Classification of Matter CONCEPT CHECK! Which of the following are examples of a chemical change? § § Pulverizing (crushing) rock salt Burning of wood Dissolving of sugar in water Melting a popsicle on a warm summer day 73
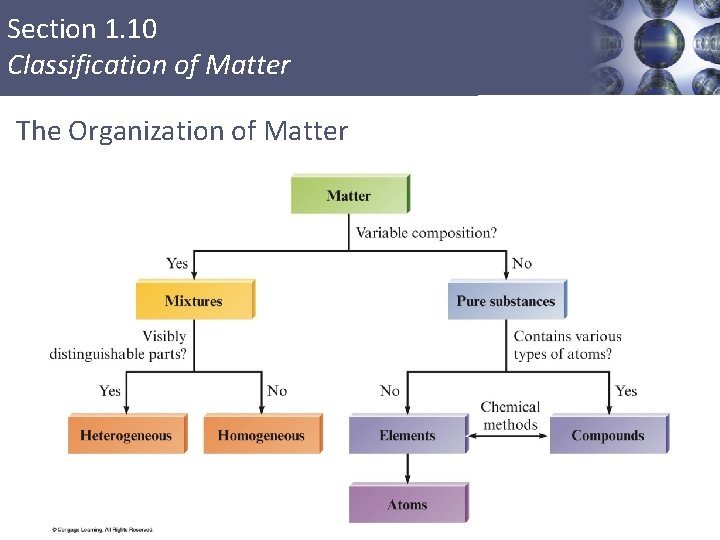
Section 1. 10 Classification of Matter The Organization of Matter 74
- Slides: 74