AP Biology Unit 1 Chapters 1 7 Introduction

AP Biology Unit 1 Chapters 1 -7 Introduction, Biochemistry and Cells

Chapter One: Overview: Inquiring About Life • An organism’s adaptations to its environment are the result of evolution – For example, the ghost plant is adapted to conserving water; this helps it to survive in the crevices of rock walls • Evolution is the process of change that has transformed life on Earth © 2011 Pearson Education, Inc.

Figure 1. 1

Figure 1. 3 Order Response to the environment Evolutionary adaptation Reproduction Regulation Energy processing Growth and development

Theme: New Properties Emerge at Each Level in the Biological Hierarchy • Life can be studied at different levels, from molecules to the entire living planet • The study of life can be divided into different levels of biological organization © 2011 Pearson Education, Inc.
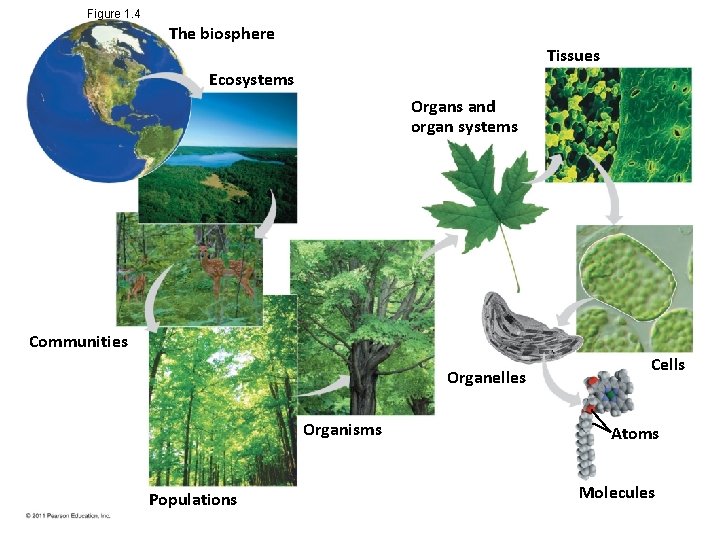
Figure 1. 4 The biosphere Tissues Ecosystems Organs and organ systems Communities Organelles Organisms Populations Cells Atoms Molecules

Theme: Organisms Interact with Other Organisms and the Physical Environment • Every organism interacts with its environment, including nonliving factors and other organisms • Both organisms and their environments are affected by the interactions between them © 2011 Pearson Education, Inc.
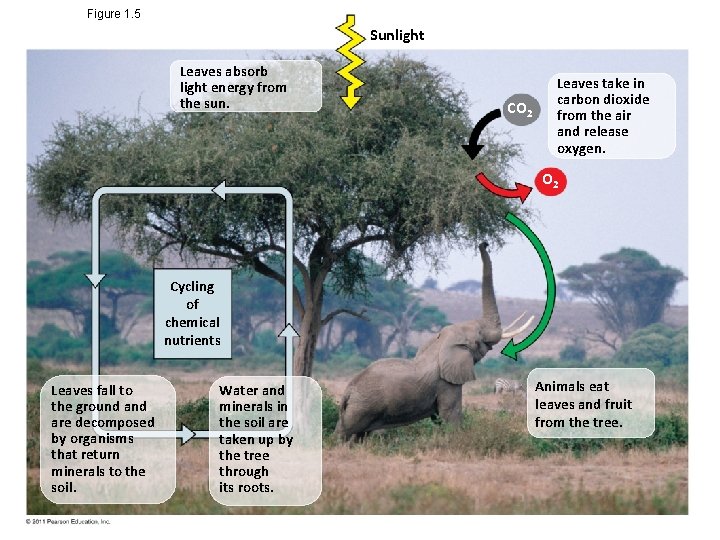
Figure 1. 5 Sunlight Leaves absorb light energy from the sun. CO 2 Leaves take in carbon dioxide from the air and release oxygen. O 2 Cycling of chemical nutrients Leaves fall to the ground are decomposed by organisms that return minerals to the soil. Water and minerals in the soil are taken up by the tree through its roots. Animals eat leaves and fruit from the tree.

Theme: Life Requires Energy Transfer and Transformation • A fundamental characteristic of living organisms is their use of energy to carry out life’s activities • Work, including moving, growing, and reproducing, requires a source of energy • Living organisms transform energy from one form to another – For example, light energy is converted to chemical energy, then kinetic energy • Energy flows through an ecosystem, usually entering as light and exiting as heat © 2011 Pearson Education, Inc.

Figure 1. 6 Sunlight Heat When energy is used to do work, some energy is converted to thermal energy, which is lost as heat. Producers absorb light energy and transform it into chemical energy. An animal’s muscle cells convert chemical energy from food to kinetic energy, the energy of motion. Chemical energy in food is transferred from plants to consumers. (a) Energy flow from sunlight to producers to consumers (b) Using energy to do work A plant’s cells use chemical energy to do work such as growing new leaves.

Theme: Structure and Function Are Correlated at All Levels of Biological Organization • Structure and function of living organisms are closely related – For example, a leaf is thin and flat, maximizing the capture of light by chloroplasts – For example, the structure of a bird’s wing is adapted to flight © 2011 Pearson Education, Inc.
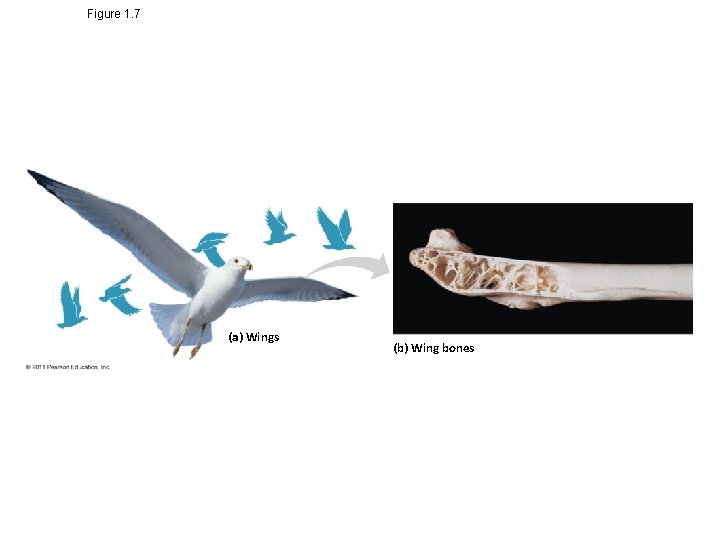
Figure 1. 7 (a) Wings (b) Wing bones

Theme: The Cell Is an Organism’s Basic Unit of Structure and Function • The cell is the lowest level of organization that can perform all activities required for life • All cells – Are enclosed by a membrane – Use DNA as their genetic information © 2011 Pearson Education, Inc.
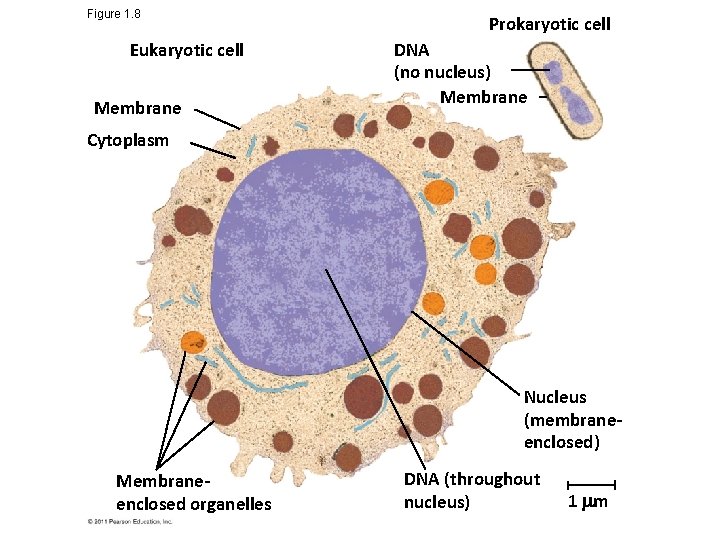
Figure 1. 8 Eukaryotic cell Membrane Prokaryotic cell DNA (no nucleus) Membrane Cytoplasm Nucleus (membraneenclosed) Membraneenclosed organelles DNA (throughout nucleus) 1 m

Theme: The Continuity of Life Is Based on Heritable Information in the Form of DNA • Chromosomes contain most of a cell’s genetic material in the form of DNA (deoxyribonucleic acid) • DNA is the substance of genes • Genes are the units of inheritance that transmit information from parents to offspring • The ability of cells to divide is the basis of all reproduction, growth, and repair of multicellular organisms © 2011 Pearson Education, Inc.
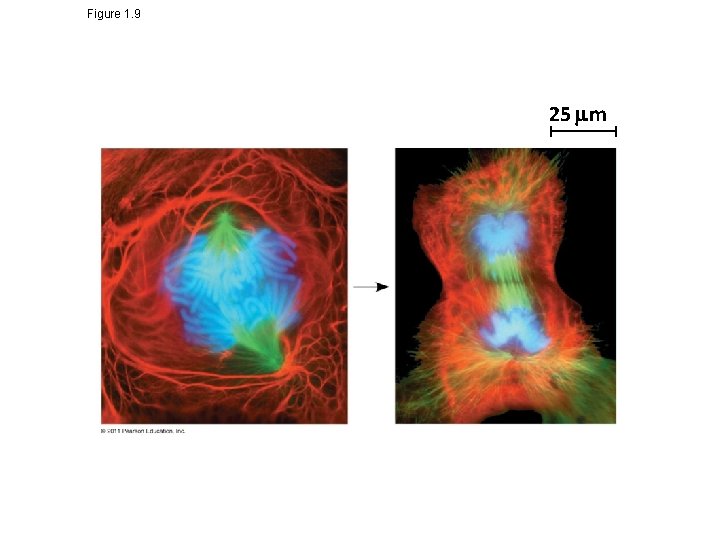
Figure 1. 9 25 m
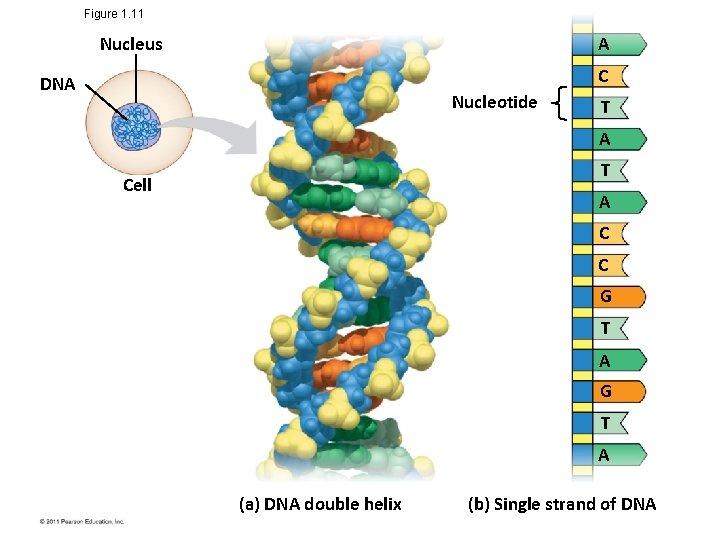
Figure 1. 11 Nucleus A C DNA Nucleotide T A T Cell A C C G T A (a) DNA double helix (b) Single strand of DNA

Theme: Feedback Mechanisms Regulate Biological Systems • Feedback mechanisms allow biological processes to selfregulate • Negative feedback means that as more of a product accumulates, the process that creates it slows and less of the product is produced • Positive feedback means that as more of a product accumulates, the process that creates it speeds up and more of the product is produced Animation: Negative Feedback © 2011 Pearson Education, Inc. Animation: Positive Feedback
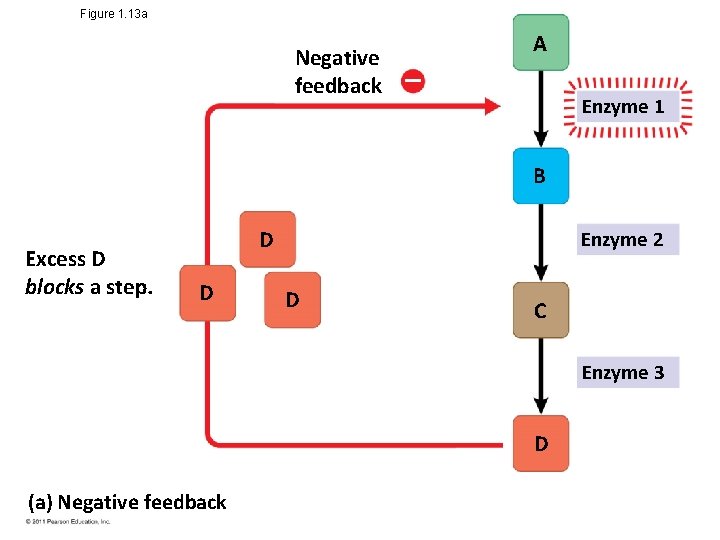
Figure 1. 13 a Negative feedback A Enzyme 1 B Excess D blocks a step. D D Enzyme 2 D C Enzyme 3 D (a) Negative feedback
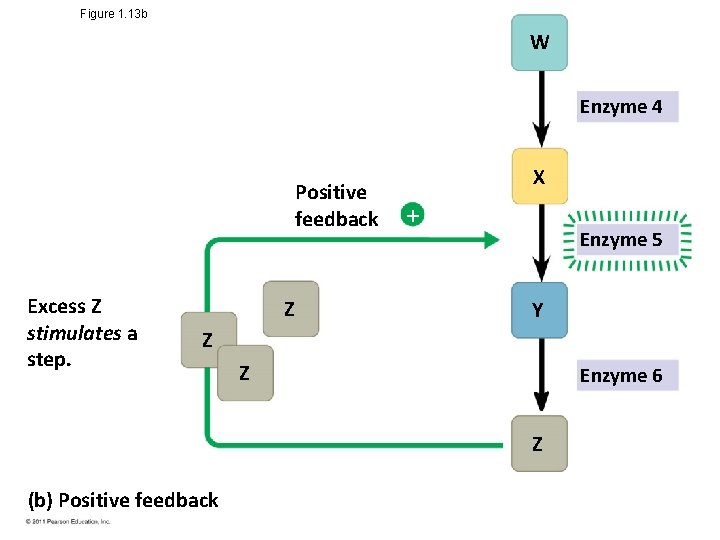
Figure 1. 13 b W Enzyme 4 Positive feedback Excess Z stimulates a step. Z X Enzyme 5 Y Z Z Enzyme 6 Z (b) Positive feedback

Evolution, the Overarching Theme of Biology • Evolution makes sense of everything we know about biology • Organisms are modified descendants of common ancestors © 2011 Pearson Education, Inc.

• Evolution explains patterns of unity and diversity in living organisms • Similar traits among organisms are explained by descent from common ancestors • Differences among organisms are explained by the accumulation of heritable changes © 2011 Pearson Education, Inc.

Figure 1. 15 2 m (b) Domain Archaea 2 m (a) Domain Bacteria (c) Domain Eukarya Kingdom Animalia 100 m Kingdom Plantae Protists Kingdom Fungi

Unity in the Diversity of Life • A striking unity underlies the diversity of life; for example – DNA is the universal genetic language common to all organisms – Unity is evident in many features of cell structure © 2011 Pearson Education, Inc.
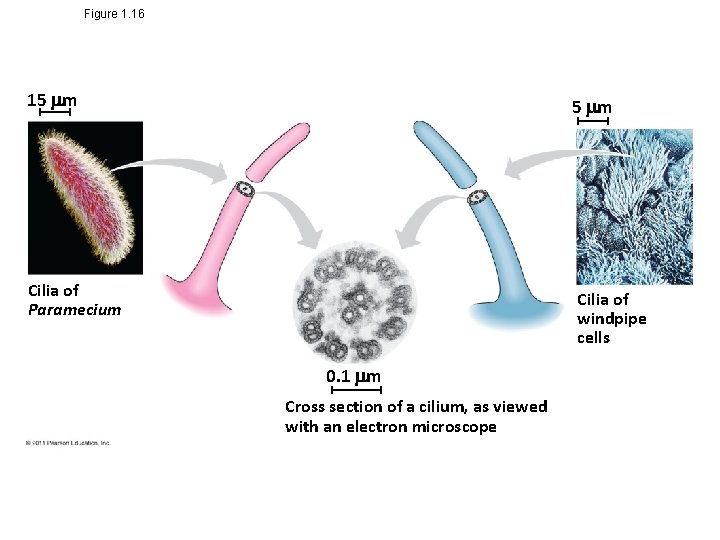
Figure 1. 16 15 m Cilia of Paramecium Cilia of windpipe cells 0. 1 m Cross section of a cilium, as viewed with an electron microscope

Concept 1. 3: In studying nature, scientists make observations and then form and test hypotheses • The word science is derived from Latin and means “to know” • Inquiry is the search for information and explanation • The scientific process includes making observations, forming logical hypotheses, and testing them © 2011 Pearson Education, Inc.

Figure 1. 23

The Role of Hypotheses in Inquiry • A hypothesis is a tentative answer to a well-framed question • A scientific hypothesis leads to predictions that can be tested by observation or experimentation © 2011 Pearson Education, Inc.

• For example, – – Observation: Your flashlight doesn’t work Question: Why doesn’t your flashlight work? Hypothesis 1: The batteries are dead Hypothesis 2: The bulb is burnt out • Both these hypotheses are testable © 2011 Pearson Education, Inc.

• Hypothesis-based science often makes use of two or more alternative hypotheses • Failure to falsify a hypothesis does not prove that hypothesis – For example, you replace your flashlight bulb, and it now works; this supports the hypothesis that your bulb was burnt out, but does not prove it (perhaps the first bulb was inserted incorrectly) © 2011 Pearson Education, Inc.

Questions That Can and Cannot Be Addressed by Science • A hypothesis must be testable and falsifiable – For example, a hypothesis that ghosts fooled with the flashlight cannot be tested • Supernatural and religious explanations are outside the bounds of science © 2011 Pearson Education, Inc.

The Flexibility of the Scientific Method • The scientific method is an idealized process of inquiry • Hypothesis-based science is based on the “textbook” scientific method but rarely follows all the ordered steps © 2011 Pearson Education, Inc.
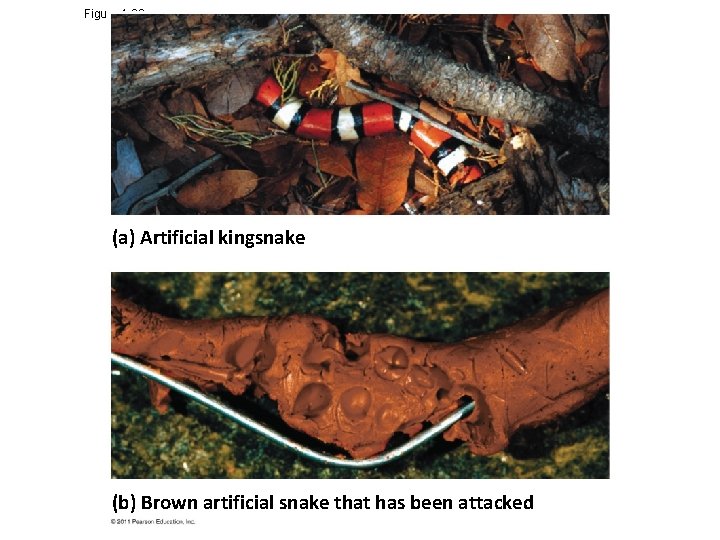
Figure 1. 26 (a) Artificial kingsnake (b) Brown artificial snake that has been attacked

Experimental Controls and Repeatability • A controlled experiment compares an experimental group (the artificial kingsnakes) with a control group (the artificial brown snakes) • Ideally, only the variable of interest (the effect of coloration on the behavior of predators) differs between the control and experimental groups • A controlled experiment means that control groups are used to cancel the effects of unwanted variables • A controlled experiment does not mean that all unwanted variables are kept constant © 2011 Pearson Education, Inc.

• In science, observations and experimental results must be repeatable © 2011 Pearson Education, Inc.

Chapter 2 Chapter TWO: The Chemical Context of Life

• Atoms in a molecule attract electrons to varying degrees • Electronegativity is an atom’s attraction for the electrons in a covalent bond • The more electronegative an atom, the more strongly it pulls shared electrons toward itself © 2011 Pearson Education, Inc.
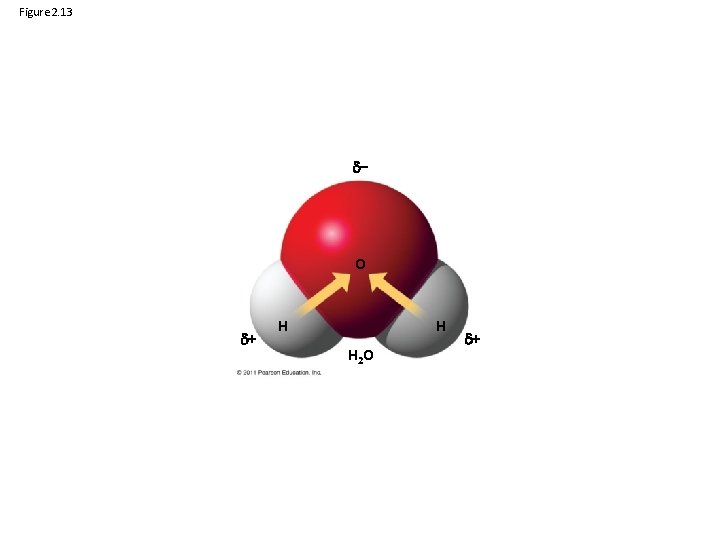
Figure 2. 13 – O + H H H 2 O +

Hydrogen Bonds • A hydrogen bond forms when a hydrogen atom covalently bonded to one electronegative atom is also attracted to another electronegative atom • In living cells, the electronegative partners are usually oxygen or nitrogen atoms © 2011 Pearson Education, Inc.
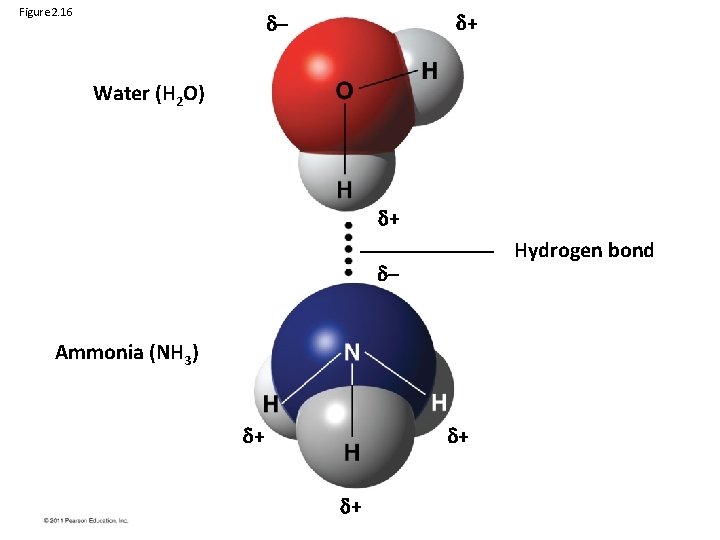
Figure 2. 16 + – Water (H 2 O) + Hydrogen bond – Ammonia (NH 3) + + +

Chapter 3 Chapter THREE: Water and Life

Concept 3. 1: Polar covalent bonds in water molecules result in hydrogen bonding • The water molecule is a polar molecule: the opposite ends have opposite charges • Polarity allows water molecules to form hydrogen bonds with each other Animation: Water Structure © 2011 Pearson Education, Inc.
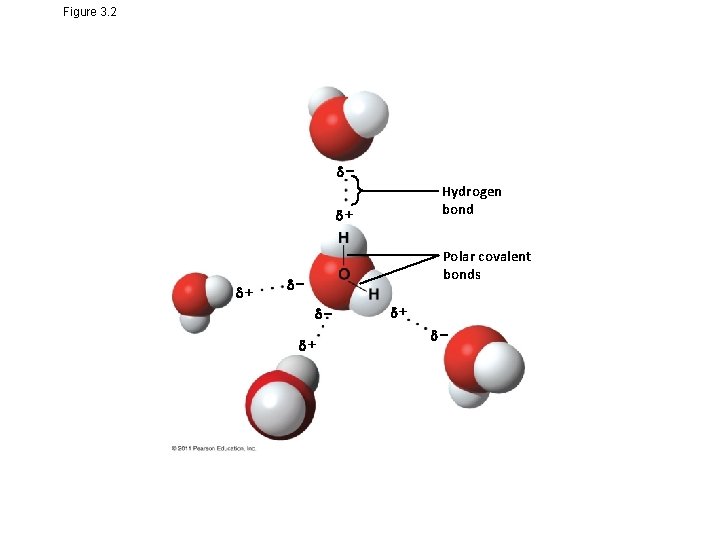
Figure 3. 2 Hydrogen bond + + Polar covalent bonds + +
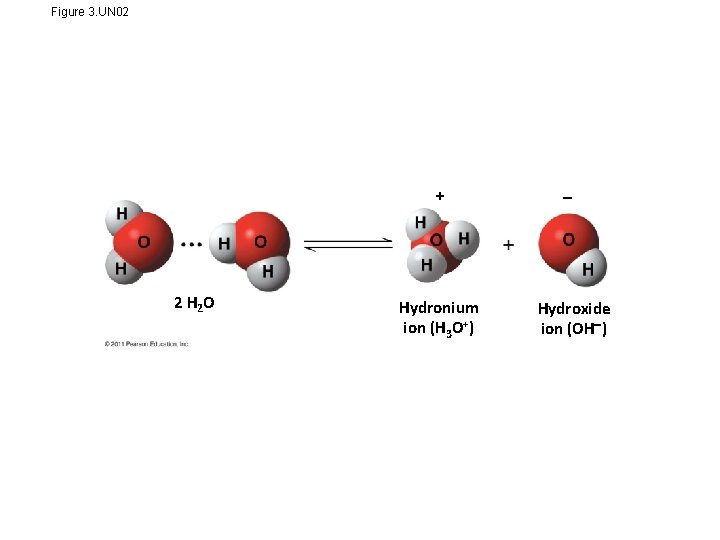
Figure 3. UN 02 + 2 H 2 O Hydronium ion (H 3 O+) Hydroxide ion (OH )

Acidification: A Threat to Water Quality • Human activities such as burning fossil fuels threaten water quality • CO 2 is the main product of fossil fuel combustion • About 25% of human-generated CO 2 is absorbed by the oceans • CO 2 dissolved in sea water forms carbonic acid; this process is called ocean acidification © 2011 Pearson Education, Inc.

Figure 3. 11 CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 H 2 CO 3 H+ + CO 32 + Ca 2+ HCO 3 Ca. CO 3

Chapter 4 Chapter FOUR: Carbon and the Molecular Diversity of Life
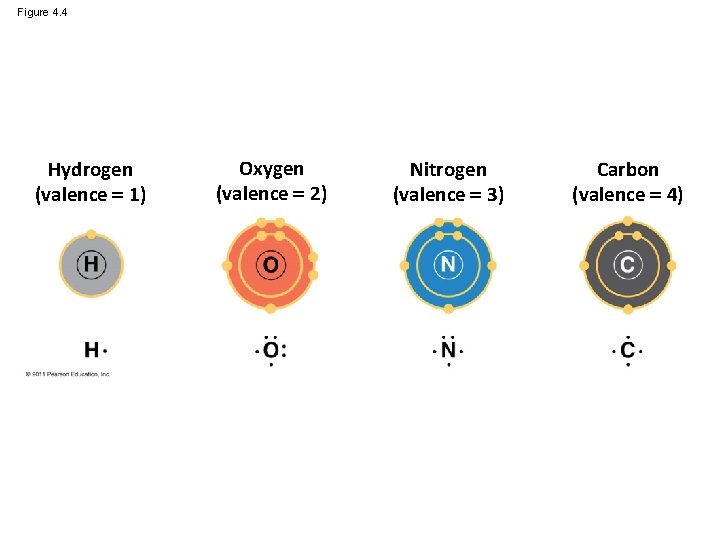
Figure 4. 4 Hydrogen (valence 1) Oxygen (valence 2) Nitrogen (valence 3) Carbon (valence 4)
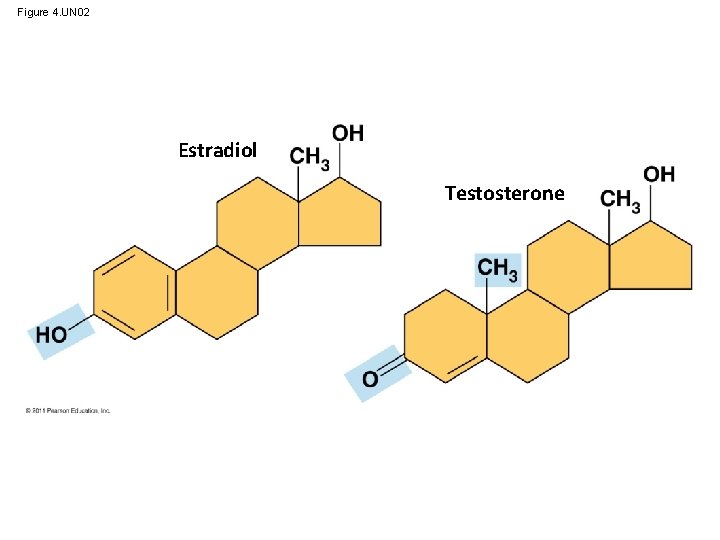
Figure 4. UN 02 Estradiol Testosterone

• The seven functional groups that are most important in the chemistry of life: – – – – Hydroxyl group Carbonyl group Carboxyl group Amino group Sulfhydryl group Phosphate group Methyl group © 2011 Pearson Education, Inc.
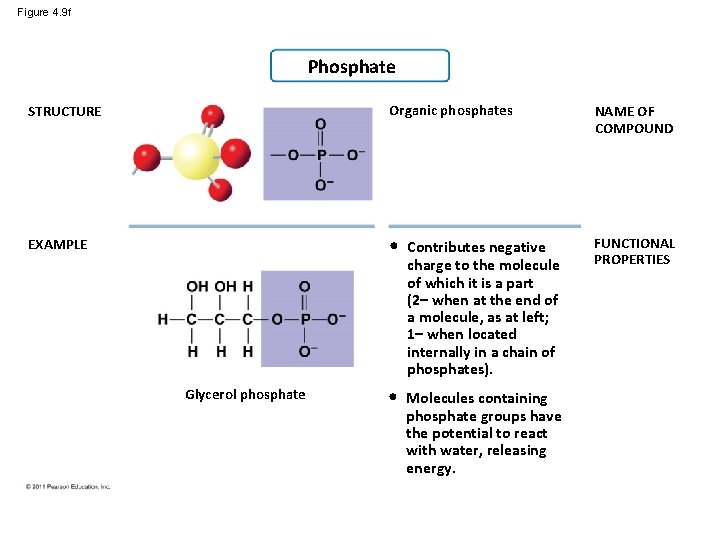
Figure 4. 9 f Phosphate STRUCTURE Organic phosphates NAME OF COMPOUND EXAMPLE • Contributes negative charge to the molecule of which it is a part (2– when at the end of a molecule, as at left; 1– when located internally in a chain of phosphates). FUNCTIONAL PROPERTIES • Molecules containing phosphate groups have the potential to react with water, releasing energy. Glycerol phosphate

ATP: An Important Source of Energy for Cellular Processes • One phosphate molecule, adenosine triphosphate (ATP), is the primary energy-transferring molecule in the cell • ATP consists of an organic molecule called adenosine attached to a string of three phosphate groups © 2011 Pearson Education, Inc.
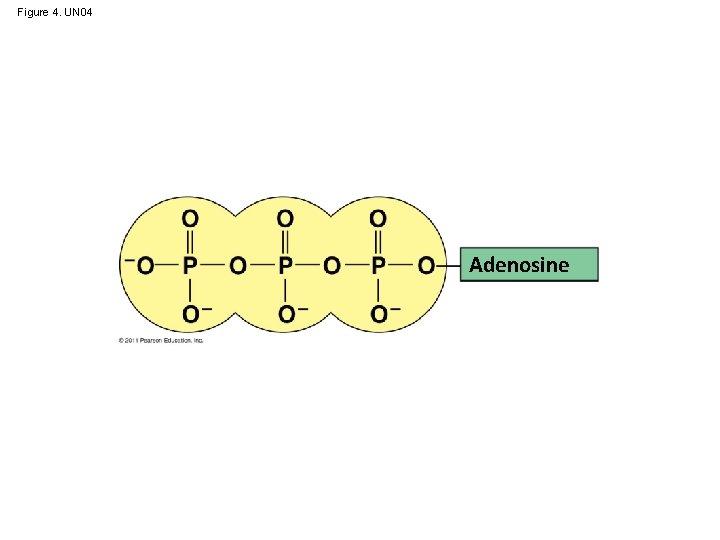
Figure 4. UN 04 Adenosine
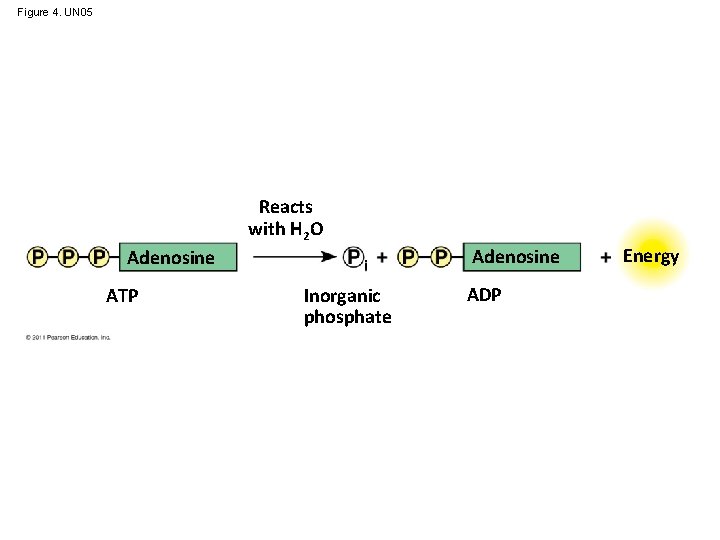
Figure 4. UN 05 Reacts with H 2 O Adenosine ATP Inorganic phosphate ADP Energy

Chapter 5 Chapter FIVE: The Structure and Function of Large Biological Molecules
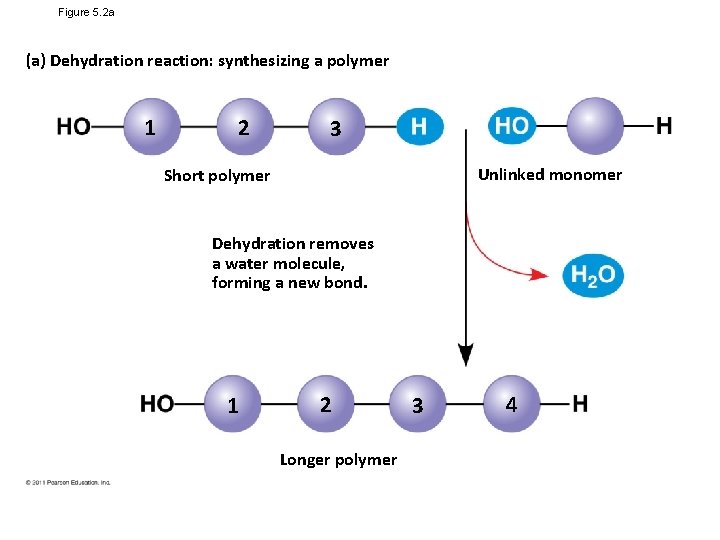
Figure 5. 2 a (a) Dehydration reaction: synthesizing a polymer 1 2 3 Unlinked monomer Short polymer Dehydration removes a water molecule, forming a new bond. 1 2 Longer polymer 3 4
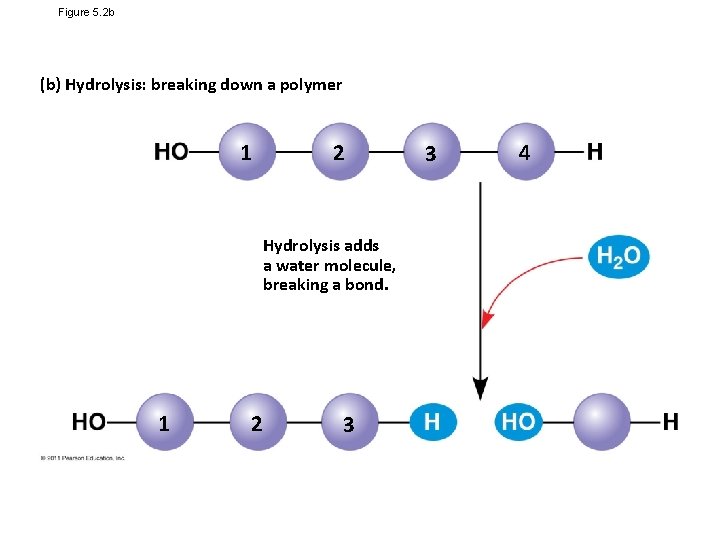
Figure 5. 2 b (b) Hydrolysis: breaking down a polymer 1 2 Hydrolysis adds a water molecule, breaking a bond. 1 2 3 3 4
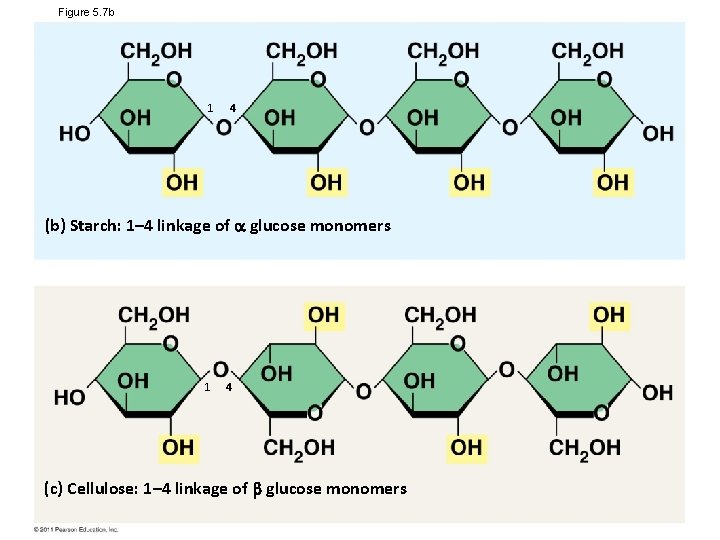
Figure 5. 7 b 1 4 (b) Starch: 1– 4 linkage of glucose monomers 1 4 (c) Cellulose: 1– 4 linkage of glucose monomers
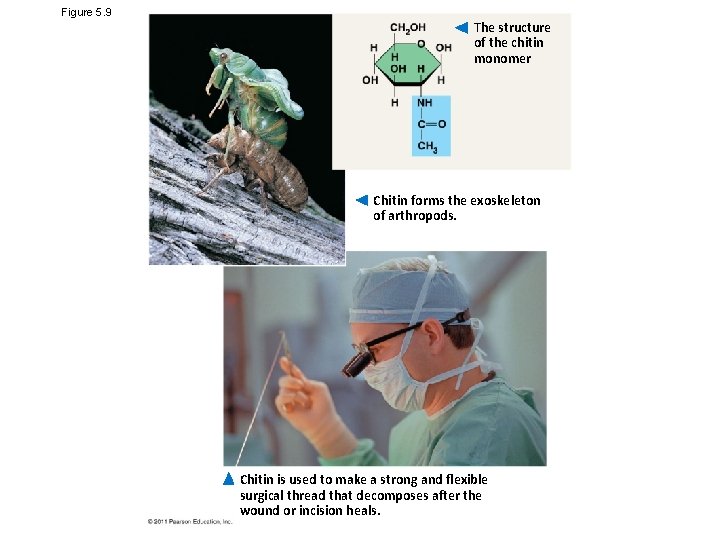
Figure 5. 9 The structure of the chitin monomer Chitin forms the exoskeleton of arthropods. Chitin is used to make a strong and flexible surgical thread that decomposes after the wound or incision heals.
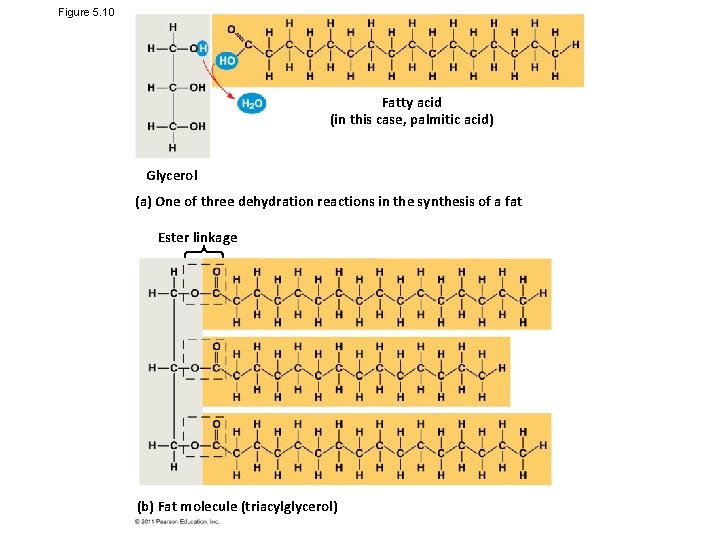
Figure 5. 10 Fatty acid (in this case, palmitic acid) Glycerol (a) One of three dehydration reactions in the synthesis of a fat Ester linkage (b) Fat molecule (triacylglycerol)
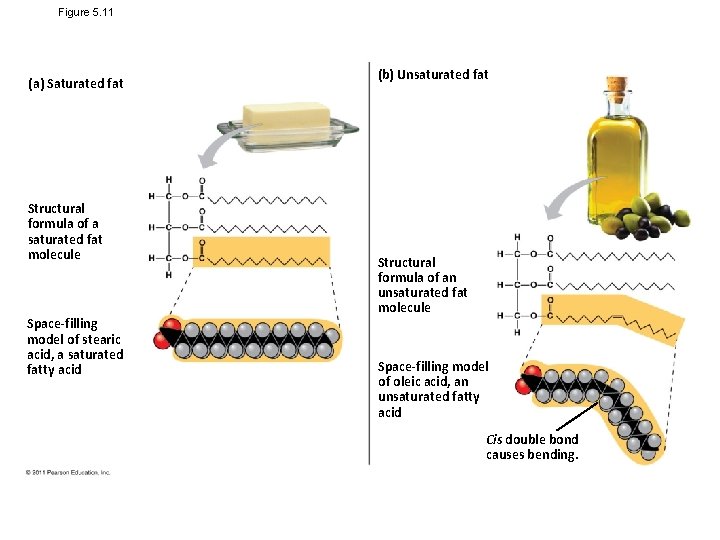
Figure 5. 11 (a) Saturated fat Structural formula of a saturated fat molecule Space-filling model of stearic acid, a saturated fatty acid (b) Unsaturated fat Structural formula of an unsaturated fat molecule Space-filling model of oleic acid, an unsaturated fatty acid Cis double bond causes bending.
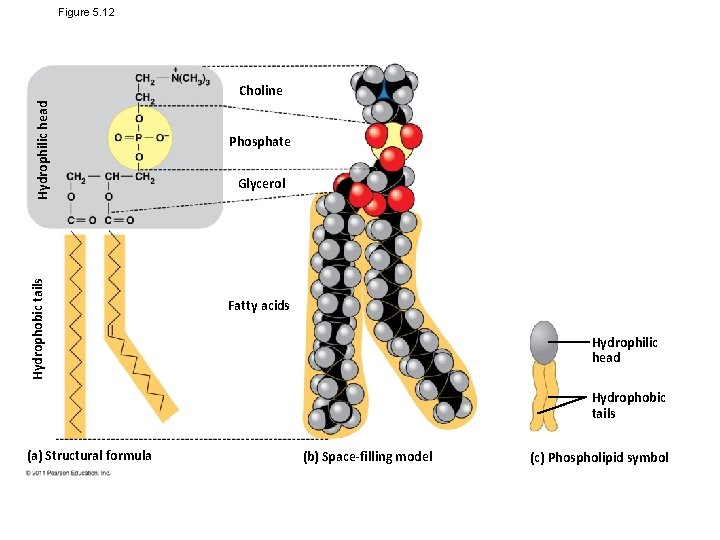
Figure 5. 12 Hydrophobic tails Hydrophilic head Choline Phosphate Glycerol Fatty acids Hydrophilic head Hydrophobic tails (a) Structural formula (b) Space-filling model (c) Phospholipid symbol
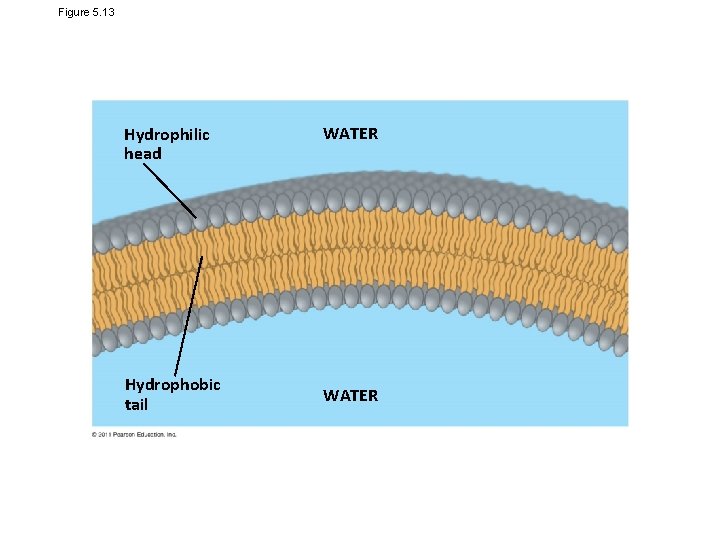
Figure 5. 13 Hydrophilic head Hydrophobic tail WATER
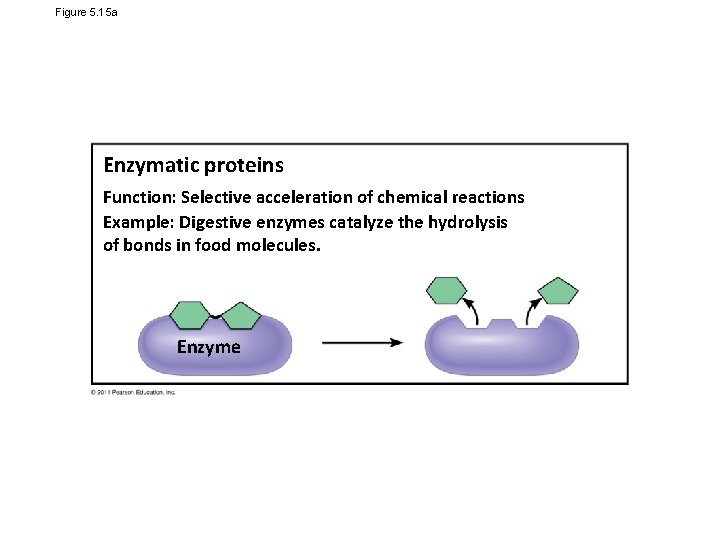
Figure 5. 15 a Enzymatic proteins Function: Selective acceleration of chemical reactions Example: Digestive enzymes catalyze the hydrolysis of bonds in food molecules. Enzyme
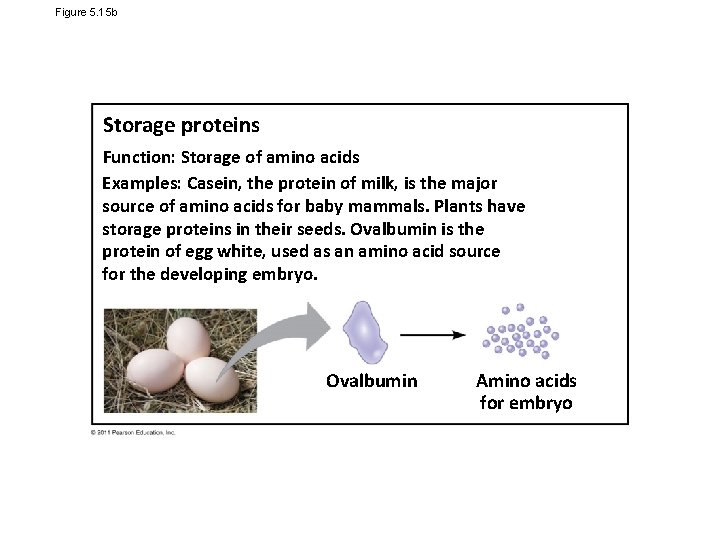
Figure 5. 15 b Storage proteins Function: Storage of amino acids Examples: Casein, the protein of milk, is the major source of amino acids for baby mammals. Plants have storage proteins in their seeds. Ovalbumin is the protein of egg white, used as an amino acid source for the developing embryo. Ovalbumin Amino acids for embryo
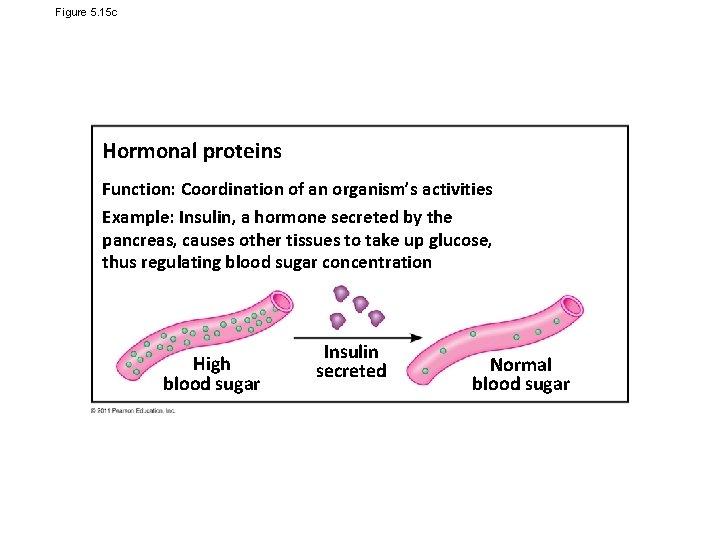
Figure 5. 15 c Hormonal proteins Function: Coordination of an organism’s activities Example: Insulin, a hormone secreted by the pancreas, causes other tissues to take up glucose, thus regulating blood sugar concentration High blood sugar Insulin secreted Normal blood sugar
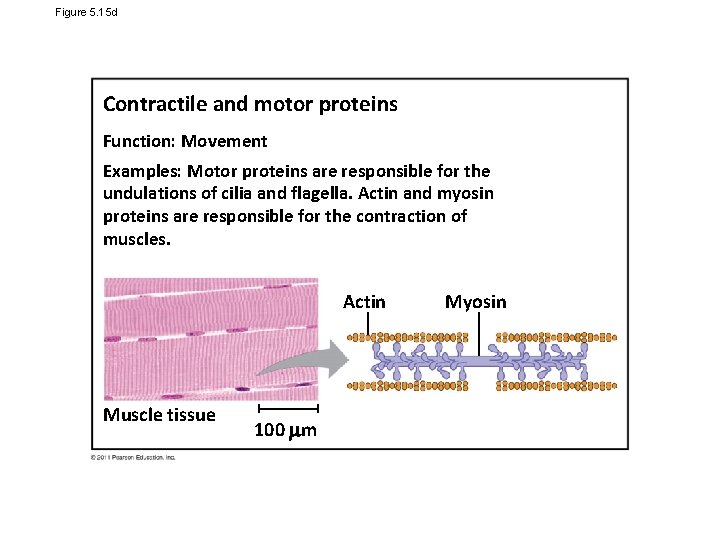
Figure 5. 15 d Contractile and motor proteins Function: Movement Examples: Motor proteins are responsible for the undulations of cilia and flagella. Actin and myosin proteins are responsible for the contraction of muscles. Actin Muscle tissue 100 m Myosin
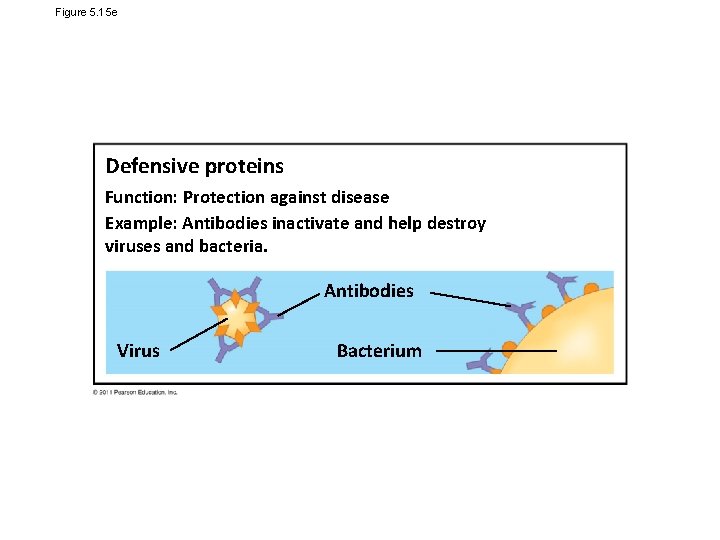
Figure 5. 15 e Defensive proteins Function: Protection against disease Example: Antibodies inactivate and help destroy viruses and bacteria. Antibodies Virus Bacterium
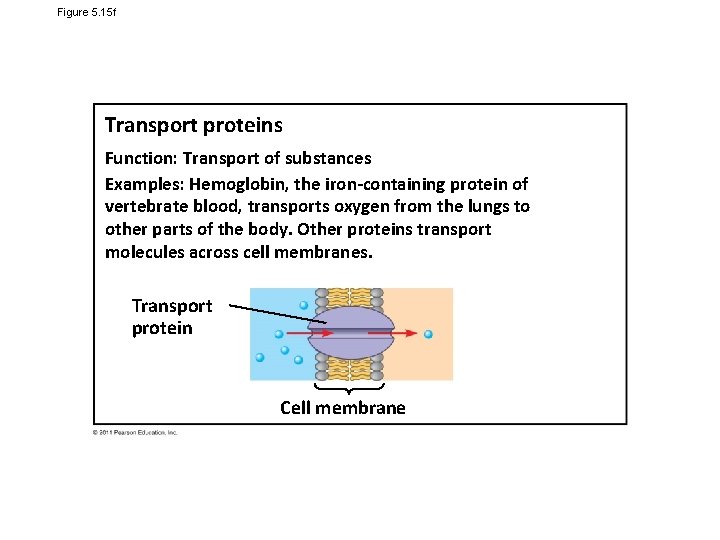
Figure 5. 15 f Transport proteins Function: Transport of substances Examples: Hemoglobin, the iron-containing protein of vertebrate blood, transports oxygen from the lungs to other parts of the body. Other proteins transport molecules across cell membranes. Transport protein Cell membrane
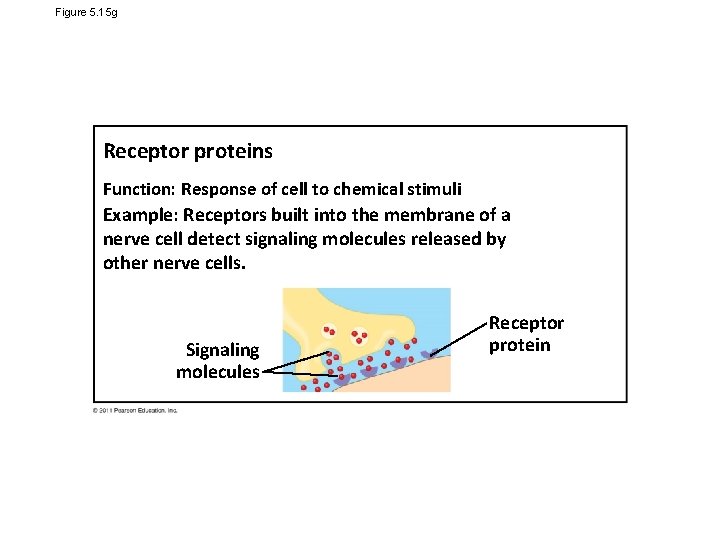
Figure 5. 15 g Receptor proteins Function: Response of cell to chemical stimuli Example: Receptors built into the membrane of a nerve cell detect signaling molecules released by other nerve cells. Signaling molecules Receptor protein
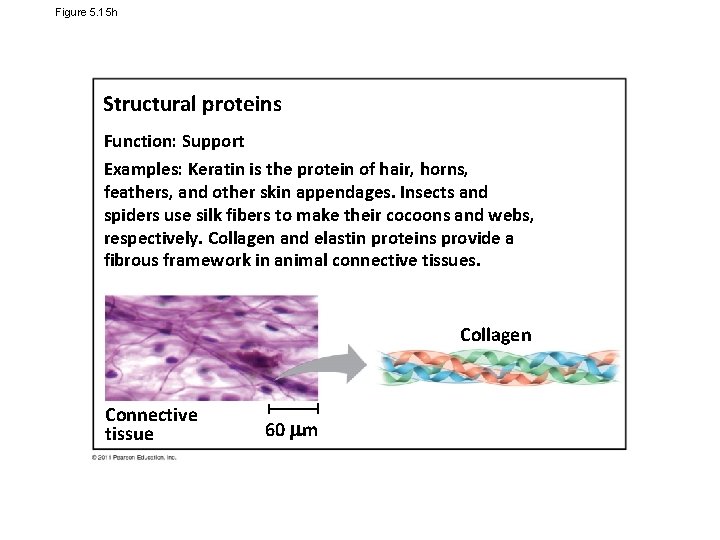
Figure 5. 15 h Structural proteins Function: Support Examples: Keratin is the protein of hair, horns, feathers, and other skin appendages. Insects and spiders use silk fibers to make their cocoons and webs, respectively. Collagen and elastin proteins provide a fibrous framework in animal connective tissues. Collagen Connective tissue 60 m

Animation: Structural Proteins Animation: Storage Proteins Animation: Transport Proteins Animation: Receptor Proteins Animation: Contractile Proteins Animation: Defensive Proteins Animation: Hormonal Proteins Animation: Sensory Proteins Animation: Gene Regulatory Proteins © 2011 Pearson Education, Inc.

Polypeptides • Polypeptides are unbranched polymers built from the same set of 20 amino acids • A protein is a biologically functional molecule that consists of one or more polypeptides © 2011 Pearson Education, Inc.

Four Levels of Protein Structure • The primary structure of a protein is its unique sequence of amino acids • Secondary structure, found in most proteins, consists of coils and folds in the polypeptide chain • Tertiary structure is determined by interactions among various side chains (R groups) • Quaternary structure results when a protein consists of multiple polypeptide chains Animation: Protein Structure Introduction © 2011 Pearson Education, Inc.
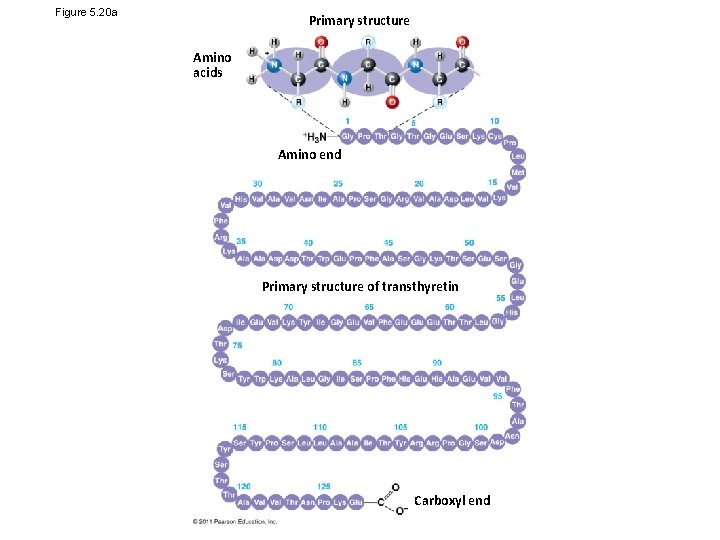
Figure 5. 20 a Primary structure Amino acids Amino end Primary structure of transthyretin Carboxyl end

• Primary structure, the sequence of amino acids in a protein, is like the order of letters in a long word • Primary structure is determined by inherited genetic information Animation: Primary Protein Structure © 2011 Pearson Education, Inc.
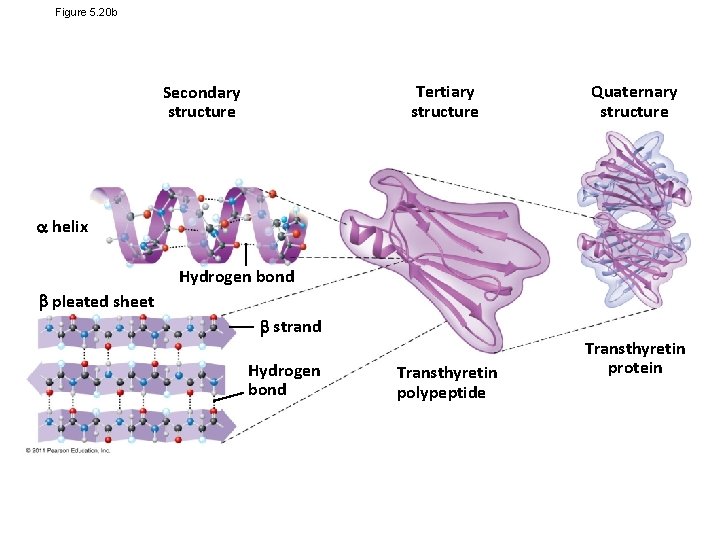
Figure 5. 20 b Tertiary structure Secondary structure Quaternary structure helix Hydrogen bond pleated sheet strand Hydrogen bond Transthyretin polypeptide Transthyretin protein

• The coils and folds of secondary structure result from hydrogen bonds between repeating constituents of the polypeptide backbone • Typical secondary structures are a coil called an helix and a folded structure called a pleated sheet Animation: Secondary Protein Structure © 2011 Pearson Education, Inc.

• Tertiary structure is determined by interactions between R groups, rather than interactions between backbone constituents • These interactions between R groups include hydrogen bonds, ionic bonds, hydrophobic interactions, and van der Waals interactions • Strong covalent bonds called disulfide bridges may reinforce the protein’s structure Animation: Tertiary Protein Structure © 2011 Pearson Education, Inc.

• Quaternary structure results when two or more polypeptide chains form one macromolecule • Collagen is a fibrous protein consisting of three polypeptides coiled like a rope • Hemoglobin is a globular protein consisting of four polypeptides: two alpha and two beta chains Animation: Quaternary Protein Structure © 2011 Pearson Education, Inc.
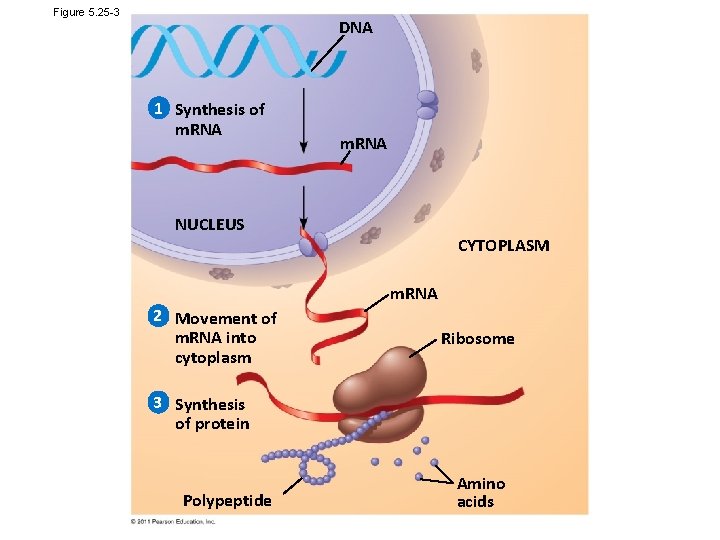
Figure 5. 25 -3 DNA 1 Synthesis of m. RNA NUCLEUS CYTOPLASM m. RNA 2 Movement of m. RNA into cytoplasm Ribosome 3 Synthesis of protein Polypeptide Amino acids
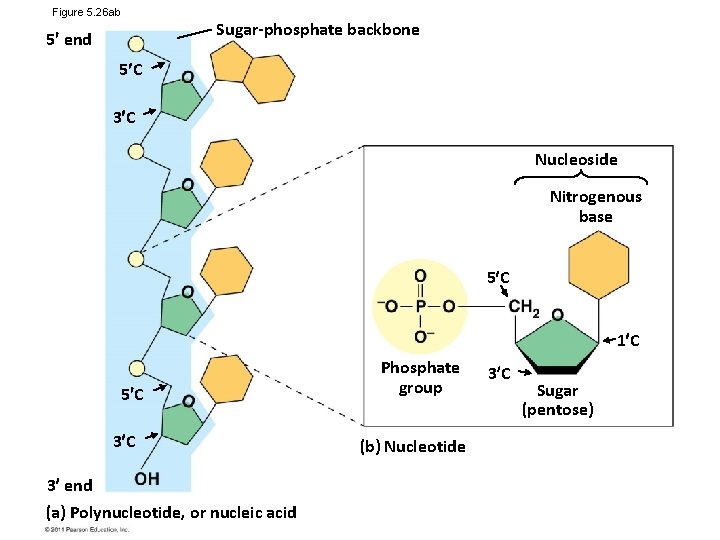
Figure 5. 26 ab 5 end Sugar-phosphate backbone 5 C 3 C Nucleoside Nitrogenous base 5 C 1 C 5 C 3 end (a) Polynucleotide, or nucleic acid Phosphate group (b) Nucleotide 3 C Sugar (pentose)

Chapter 6 Chapter SIX: A Tour of the Cell
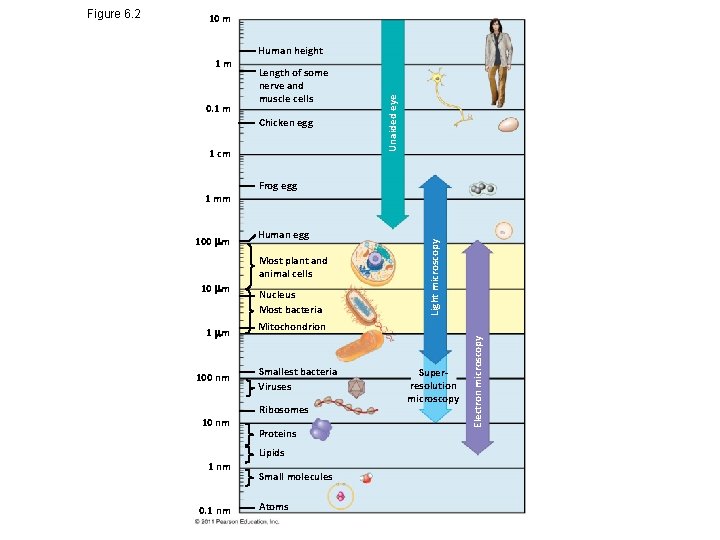
10 m Length of some nerve and muscle cells Chicken egg 1 cm 1 mm 100 m Frog egg Human egg Most plant and animal cells 10 m 100 nm 10 nm Nucleus Most bacteria Mitochondrion Smallest bacteria Viruses Ribosomes Proteins Lipids 1 nm 0. 1 nm Small molecules Atoms Superresolution microscopy Electron microscopy 0. 1 m Human height Light microscopy 1 m Unaided eye Figure 6. 2
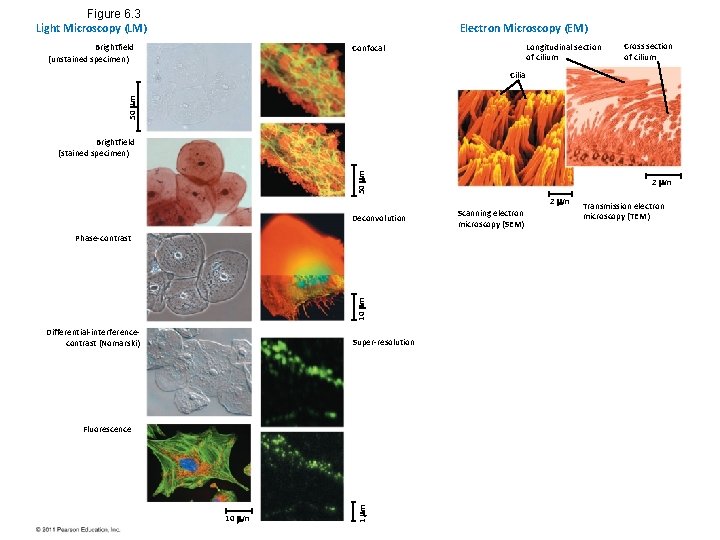
Figure 6. 3 Light Microscopy (LM) Electron Microscopy (EM) Brightfield (unstained specimen) Longitudinal section of cilium Confocal Cross section of cilium 50 m Cilia 50 m Brightfield (stained specimen) 2 m Deconvolution 10 m Phase-contrast Differential-interferencecontrast (Nomarski) Super-resolution 10 m 1 m Fluorescence Scanning electron microscopy (SEM) Transmission electron microscopy (TEM)

Concept 6. 2: Eukaryotic cells have internal membranes that compartmentalize their functions • The basic structural and functional unit of every organism is one of two types of cells: prokaryotic or eukaryotic • Only organisms of the domains Bacteria and Archaea consist of prokaryotic cells • Protists, fungi, animals, and plants all consist of eukaryotic cells © 2011 Pearson Education, Inc.

• Metabolic requirements set upper limits on the size of cells • The surface area to volume ratio of a cell is critical • As the surface area increases by a factor of n 2, the volume increases by a factor of n 3 • Small cells have a greater surface area relative to volume © 2011 Pearson Education, Inc.
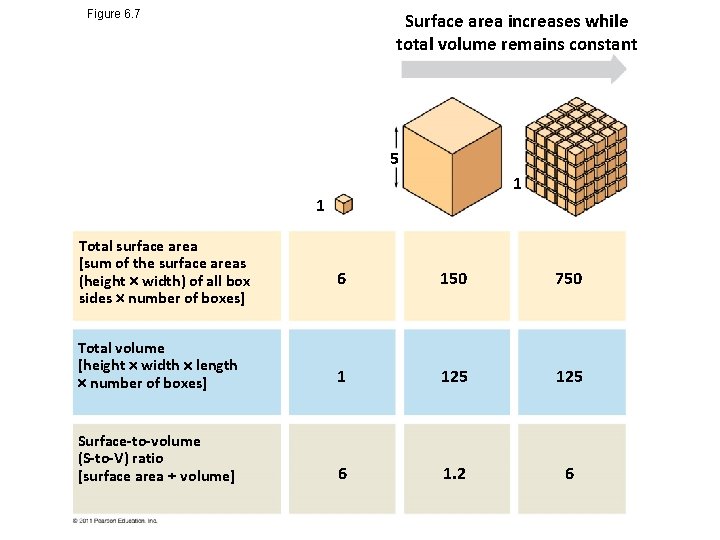
Figure 6. 7 Surface area increases while total volume remains constant 5 1 1 Total surface area [sum of the surface areas (height width) of all box sides number of boxes] 6 150 750 Total volume [height width length number of boxes] 1 125 Surface-to-volume (S-to-V) ratio [surface area volume] 6 1. 2 6

A Panoramic View of the Eukaryotic Cell • A eukaryotic cell has internal membranes that partition the cell into organelles • Plant and animal cells have most of the same organelles Bio. Flix: Tour of an Animal Cell Bio. Flix: Tour of a Plant Cell © 2011 Pearson Education, Inc.
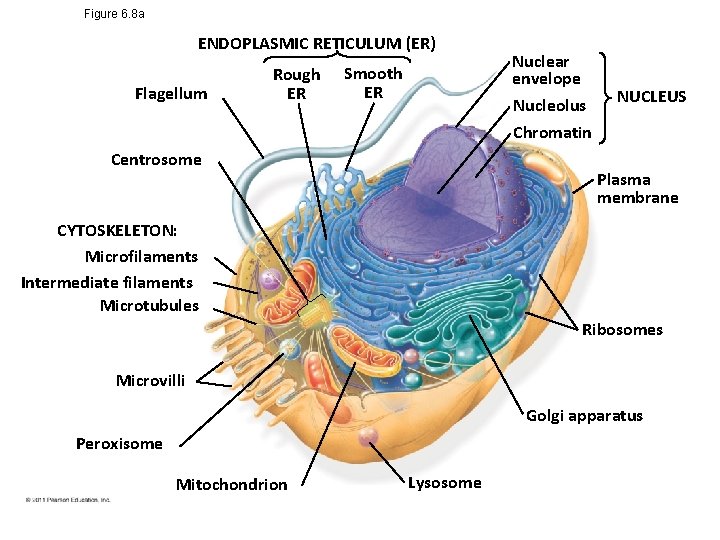
Figure 6. 8 a ENDOPLASMIC RETICULUM (ER) Flagellum Rough ER Smooth ER Centrosome Nuclear envelope Nucleolus Chromatin NUCLEUS Plasma membrane CYTOSKELETON: Microfilaments Intermediate filaments Microtubules Ribosomes Microvilli Golgi apparatus Peroxisome Mitochondrion Lysosome
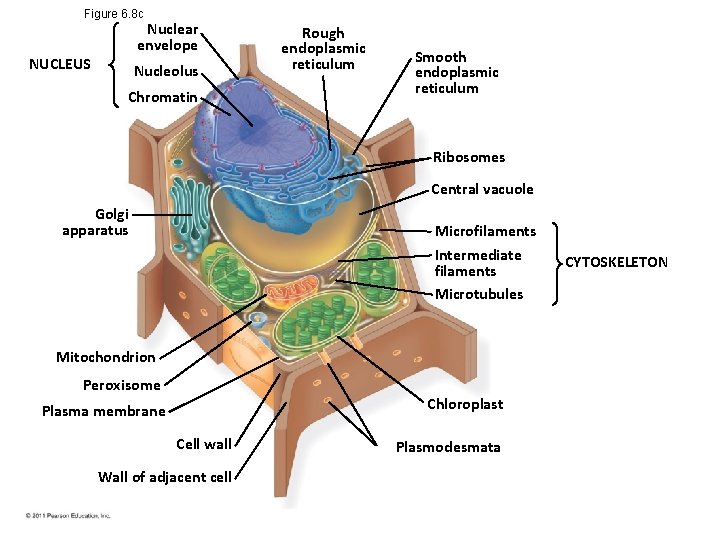
Figure 6. 8 c Nuclear envelope NUCLEUS Nucleolus Chromatin Rough endoplasmic reticulum Smooth endoplasmic reticulum Ribosomes Central vacuole Golgi apparatus Microfilaments Intermediate filaments Microtubules Mitochondrion Peroxisome Chloroplast Plasma membrane Cell wall Wall of adjacent cell Plasmodesmata CYTOSKELETON

Concept 6. 4: The endomembrane system regulates protein traffic and performs metabolic functions in the cell • Components of the endomembrane system – Nuclear envelope – Endoplasmic reticulum – Golgi apparatus – Lysosomes – Vacuoles – Plasma membrane • These components are either continuous or connected via transfer by vesicles © 2011 Pearson Education, Inc.
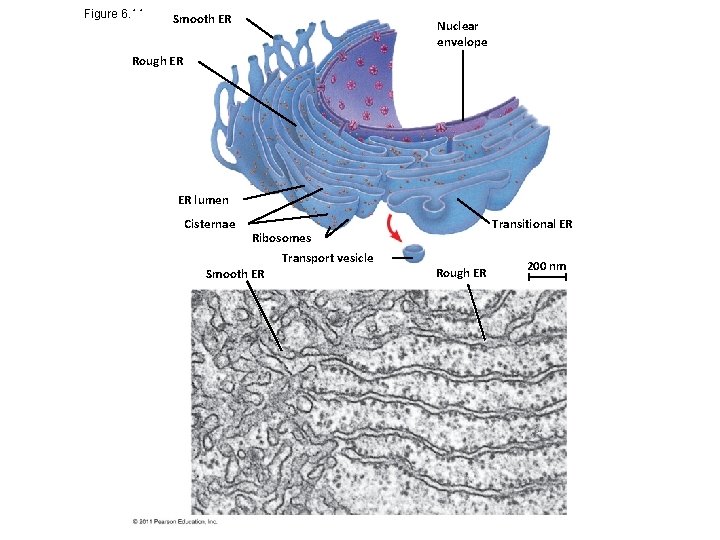
Figure 6. 11 Smooth ER Nuclear envelope Rough ER ER lumen Cisternae Ribosomes Transport vesicle Smooth ER Transitional ER Rough ER 200 nm
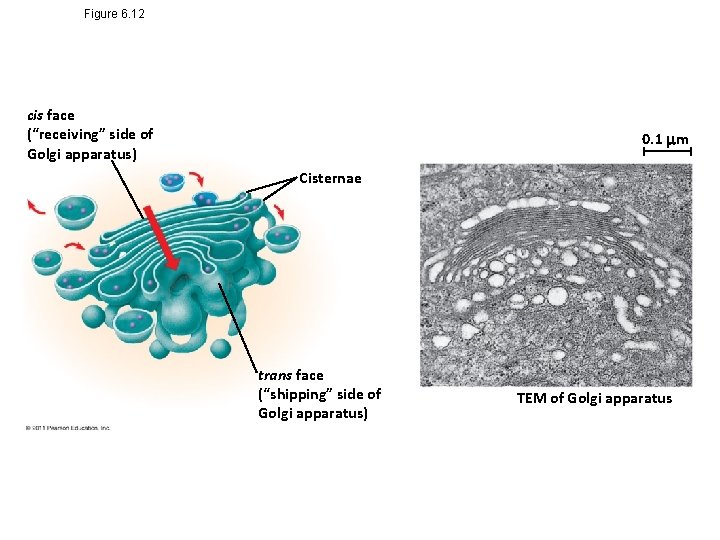
Figure 6. 12 cis face (“receiving” side of Golgi apparatus) 0. 1 m Cisternae trans face (“shipping” side of Golgi apparatus) TEM of Golgi apparatus
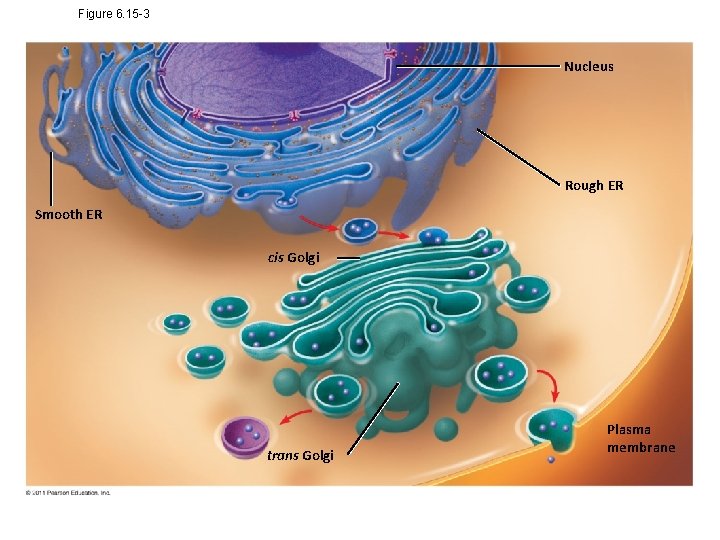
Figure 6. 15 -3 Nucleus Rough ER Smooth ER cis Golgi trans Golgi Plasma membrane
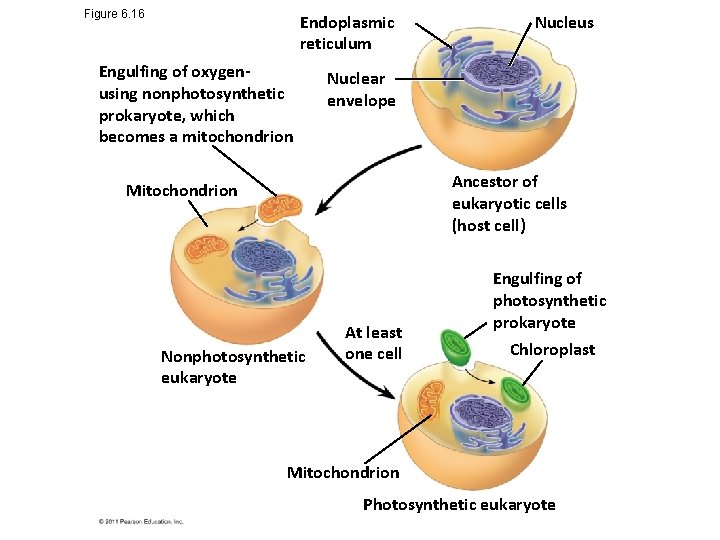
Figure 6. 16 Endoplasmic reticulum Engulfing of oxygenusing nonphotosynthetic prokaryote, which becomes a mitochondrion Nucleus Nuclear envelope Ancestor of eukaryotic cells (host cell) Mitochondrion Nonphotosynthetic eukaryote At least one cell Engulfing of photosynthetic prokaryote Chloroplast Mitochondrion Photosynthetic eukaryote
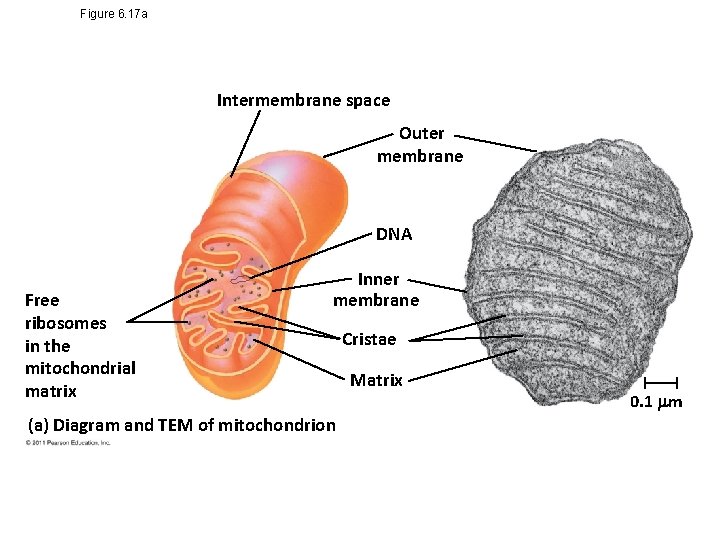
Figure 6. 17 a Intermembrane space Outer membrane DNA Free ribosomes in the mitochondrial matrix Inner membrane (a) Diagram and TEM of mitochondrion Cristae Matrix 0. 1 m
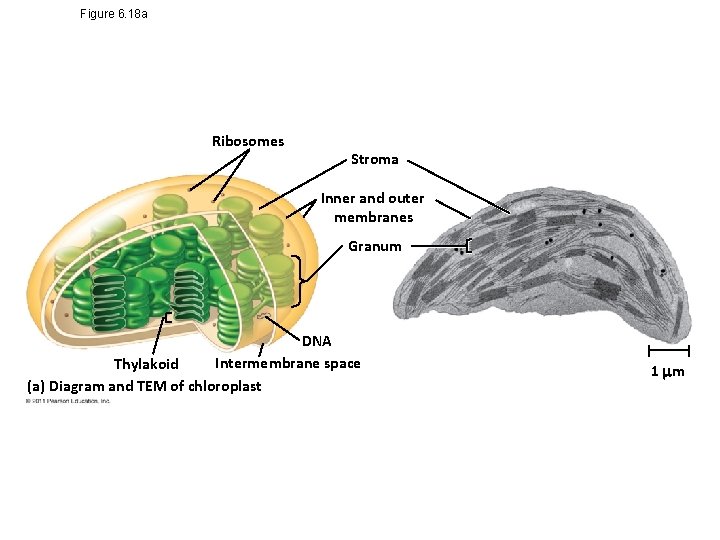
Figure 6. 18 a Ribosomes Stroma Inner and outer membranes Granum DNA Intermembrane space Thylakoid (a) Diagram and TEM of chloroplast 1 m

Peroxisomes: Oxidation • Peroxisomes are specialized metabolic compartments bounded by a single membrane • Peroxisomes produce hydrogen peroxide and convert it to water • Peroxisomes perform reactions with many different functions • How peroxisomes are related to other organelles is still unknown © 2011 Pearson Education, Inc.
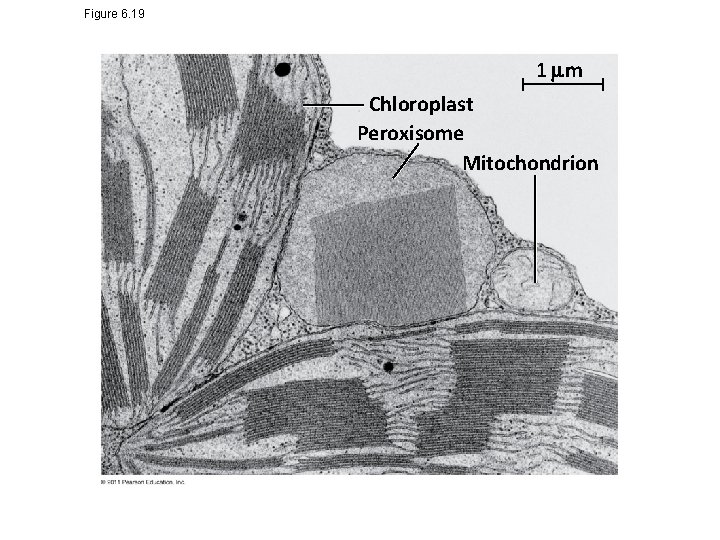
Figure 6. 19 1 m Chloroplast Peroxisome Mitochondrion

Centrosomes and Centrioles • In many cells, microtubules grow out from a centrosome near the nucleus • The centrosome is a “microtubule-organizing center” • In animal cells, the centrosome has a pair of centrioles, each with nine triplets of microtubules arranged in a ring © 2011 Pearson Education, Inc.
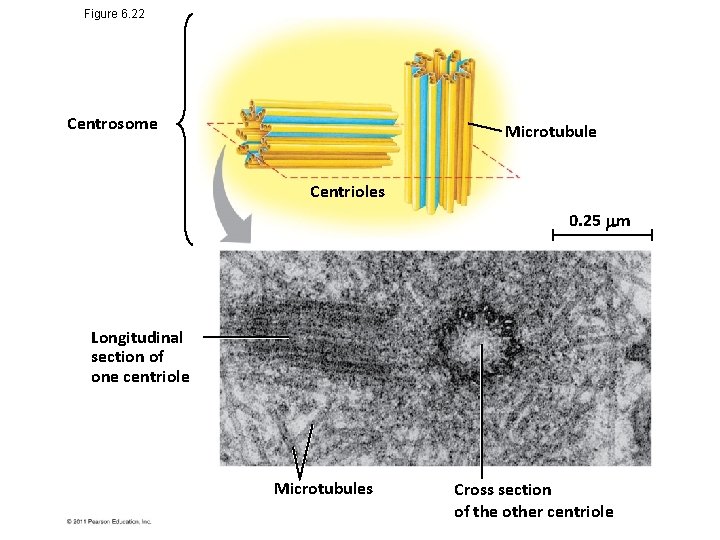
Figure 6. 22 Centrosome Microtubule Centrioles 0. 25 m Longitudinal section of one centriole Microtubules Cross section of the other centriole
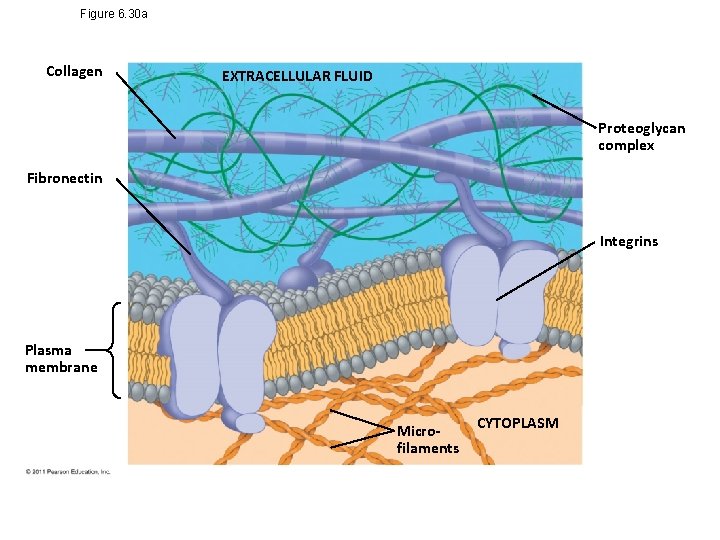
Figure 6. 30 a Collagen EXTRACELLULAR FLUID Proteoglycan complex Fibronectin Integrins Plasma membrane Microfilaments CYTOPLASM

• Functions of the ECM – Support – Adhesion – Movement – Regulation © 2011 Pearson Education, Inc.

Chapter 7 Chapter SEVEN: Membrane Structure and Function
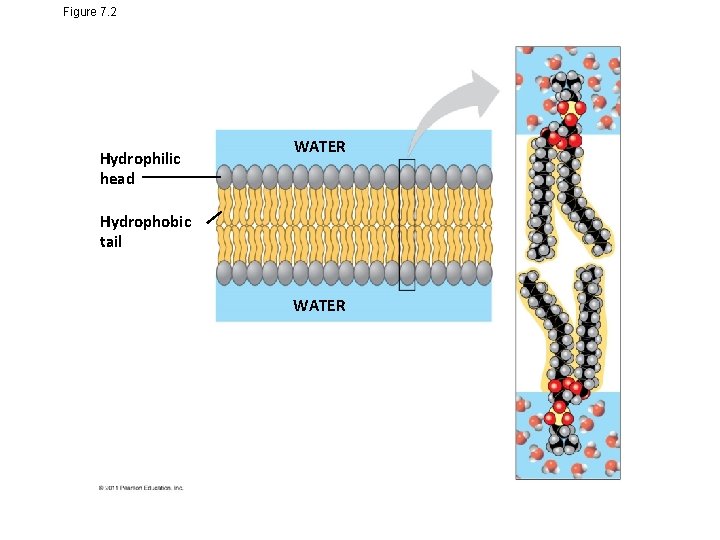
Figure 7. 2 Hydrophilic head WATER Hydrophobic tail WATER
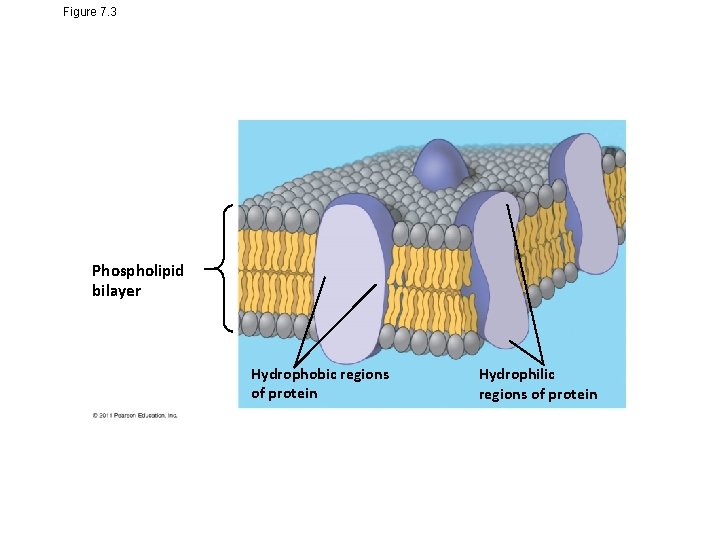
Figure 7. 3 Phospholipid bilayer Hydrophobic regions of protein Hydrophilic regions of protein
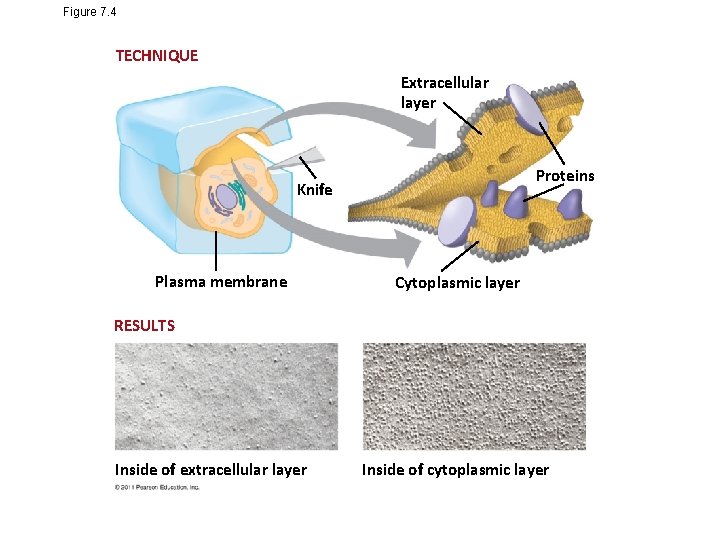
Figure 7. 4 TECHNIQUE Extracellular layer Proteins Knife Plasma membrane Cytoplasmic layer RESULTS Inside of extracellular layer Inside of cytoplasmic layer
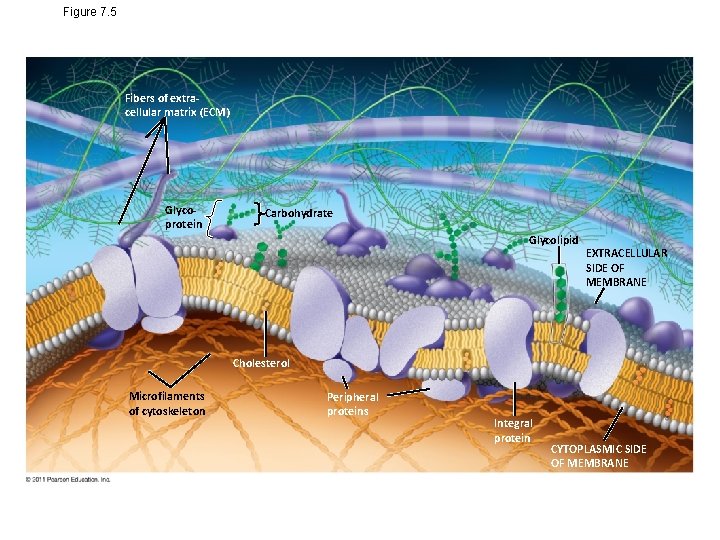
Figure 7. 5 Fibers of extracellular matrix (ECM) Glycoprotein Carbohydrate Glycolipid EXTRACELLULAR SIDE OF MEMBRANE Cholesterol Microfilaments of cytoskeleton Peripheral proteins Integral protein CYTOPLASMIC SIDE OF MEMBRANE
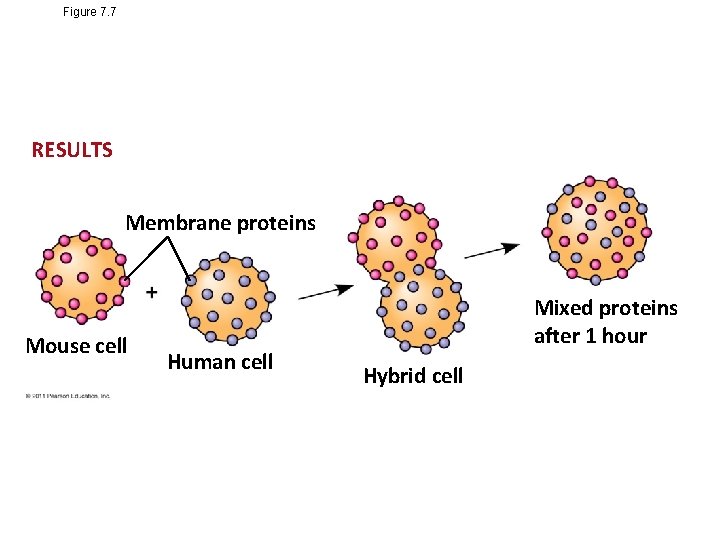
Figure 7. 7 RESULTS Membrane proteins Mouse cell Human cell Mixed proteins after 1 hour Hybrid cell
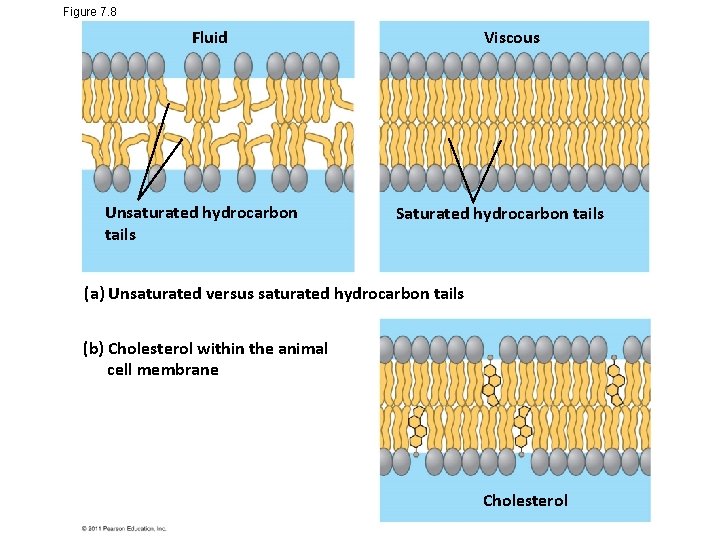
Figure 7. 8 Viscous Fluid Unsaturated hydrocarbon tails Saturated hydrocarbon tails (a) Unsaturated versus saturated hydrocarbon tails (b) Cholesterol within the animal cell membrane Cholesterol
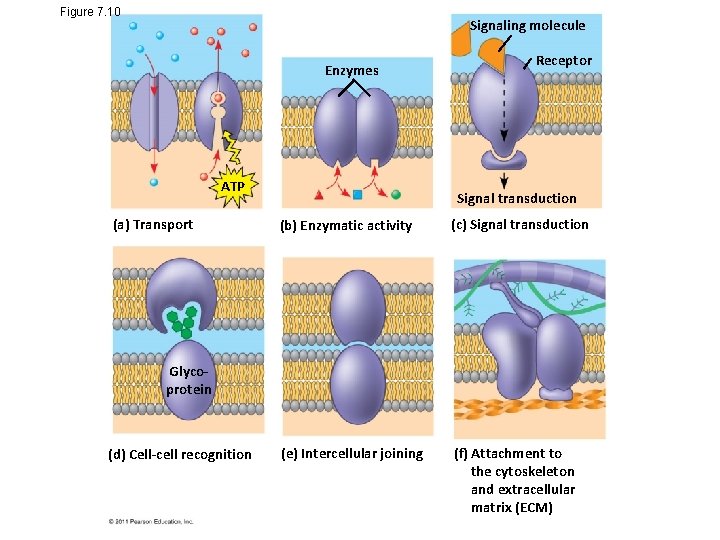
Figure 7. 10 Signaling molecule Enzymes ATP (a) Transport Receptor Signal transduction (b) Enzymatic activity (c) Signal transduction (e) Intercellular joining (f) Attachment to the cytoskeleton and extracellular matrix (ECM) Glycoprotein (d) Cell-cell recognition
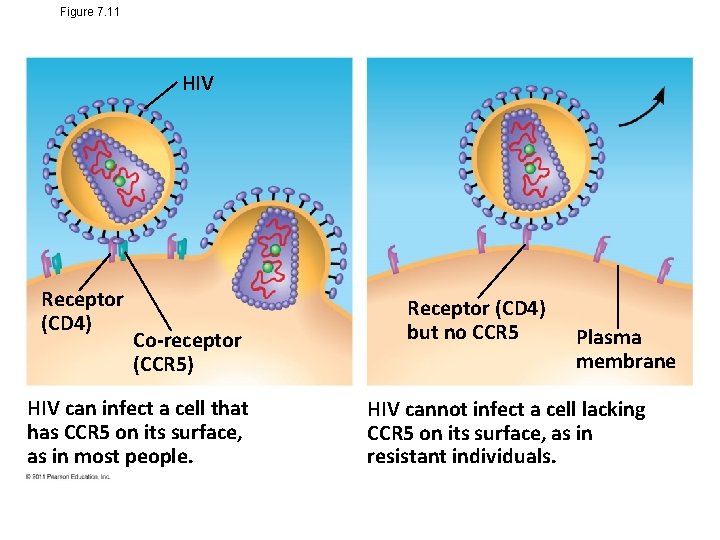
Figure 7. 11 HIV Receptor (CD 4) Co-receptor (CCR 5) HIV can infect a cell that has CCR 5 on its surface, as in most people. Receptor (CD 4) but no CCR 5 Plasma membrane HIV cannot infect a cell lacking CCR 5 on its surface, as in resistant individuals.
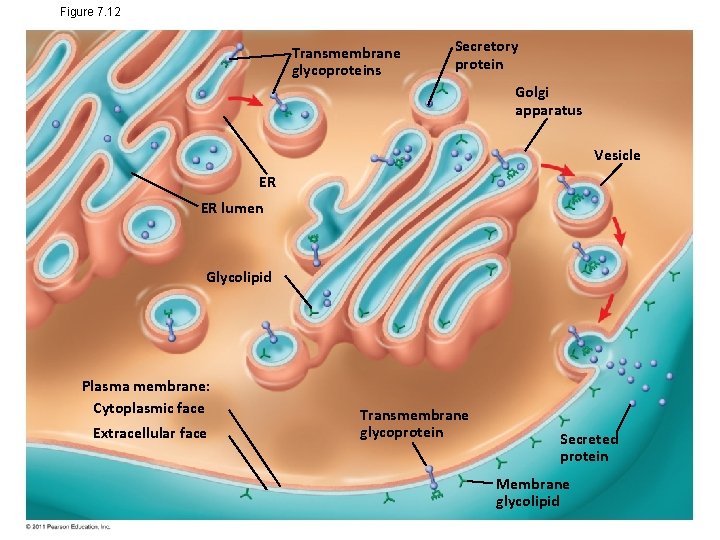
Figure 7. 12 Transmembrane glycoproteins Secretory protein Golgi apparatus Vesicle ER ER lumen Glycolipid Plasma membrane: Cytoplasmic face Extracellular face Transmembrane glycoprotein Secreted protein Membrane glycolipid
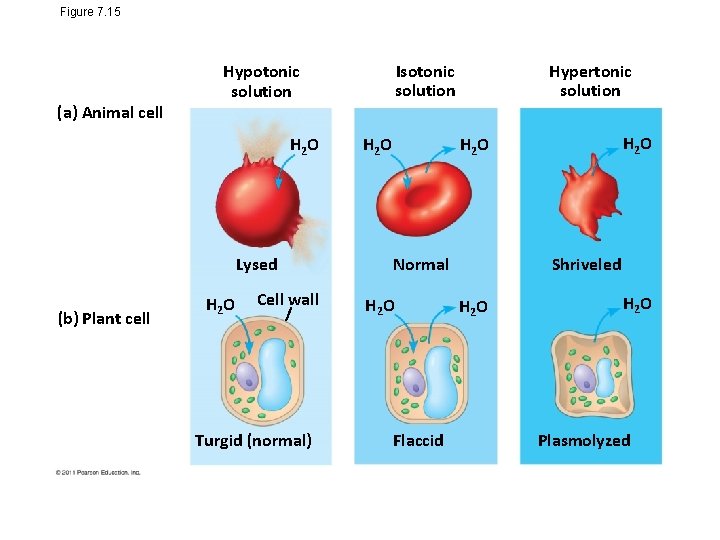
Figure 7. 15 (a) Animal cell H 2 O Lysed (b) Plant cell Isotonic solution Hypotonic solution H 2 O Cell wall Turgid (normal) H 2 O Hypertonic solution H 2 O Normal H 2 O Flaccid Osmosis Shriveled H 2 O Plasmolyzed
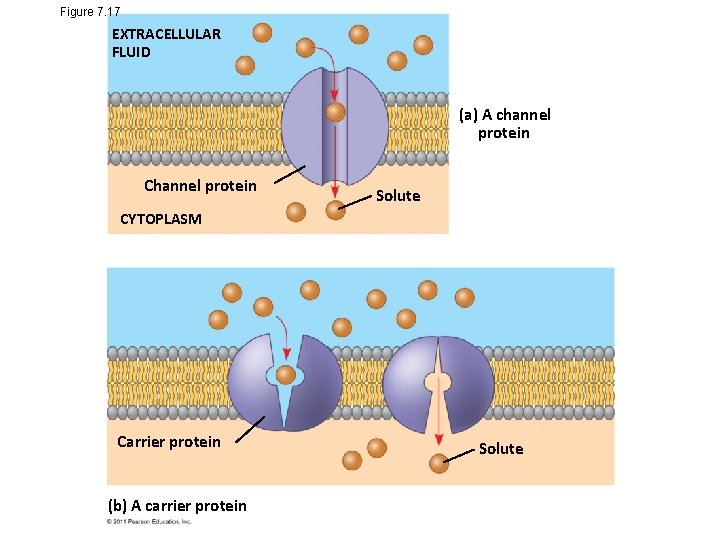
Figure 7. 17 EXTRACELLULAR FLUID (a) A channel protein Channel protein Solute CYTOPLASM Carrier protein (b) A carrier protein Solute
![Figure 7. 18 -6 EXTRACELLULAR FLUID [Na ] high [K ] low Na Na Figure 7. 18 -6 EXTRACELLULAR FLUID [Na ] high [K ] low Na Na](http://slidetodoc.com/presentation_image_h/cda6da02bbb95ed3fafffcf3f1332dc9/image-117.jpg)
Figure 7. 18 -6 EXTRACELLULAR FLUID [Na ] high [K ] low Na Na CYTOPLASM 1 Na [Na ] low [K ] high P ADP 2 ATP P 3 K K K 6 K K K 5 4 P Pi
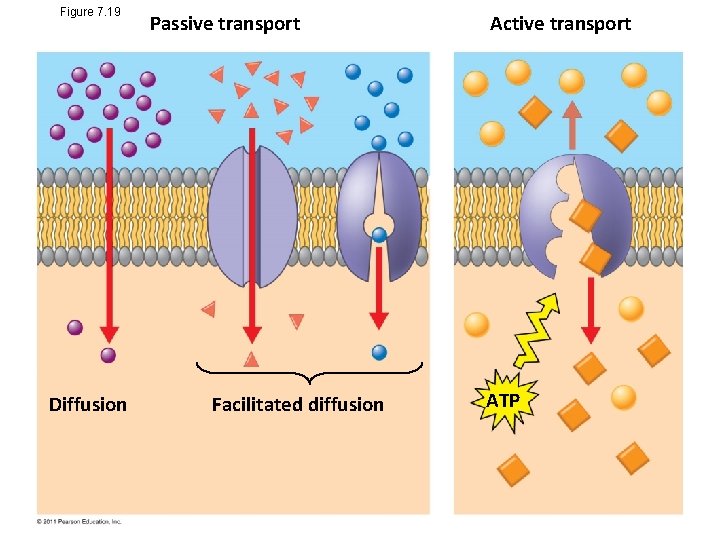
Figure 7. 19 Diffusion Passive transport Facilitated diffusion Active transport ATP

How Ion Pumps Maintain Membrane Potential • Membrane potential is the voltage difference across a membrane • Voltage is created by differences in the distribution of positive and negative ions across a membrane © 2011 Pearson Education, Inc.

• Two combined forces, collectively called the electrochemical gradient, drive the diffusion of ions across a membrane – A chemical force (the ion’s concentration gradient) – An electrical force (the effect of the membrane potential on the ion’s movement) © 2011 Pearson Education, Inc.

• An electrogenic pump is a transport protein that generates voltage across a membrane • The sodium-potassium pump is the major electrogenic pump of animal cells • The main electrogenic pump of plants, fungi, and bacteria is a proton pump • Electrogenic pumps help store energy that can be used for cellular work © 2011 Pearson Education, Inc.
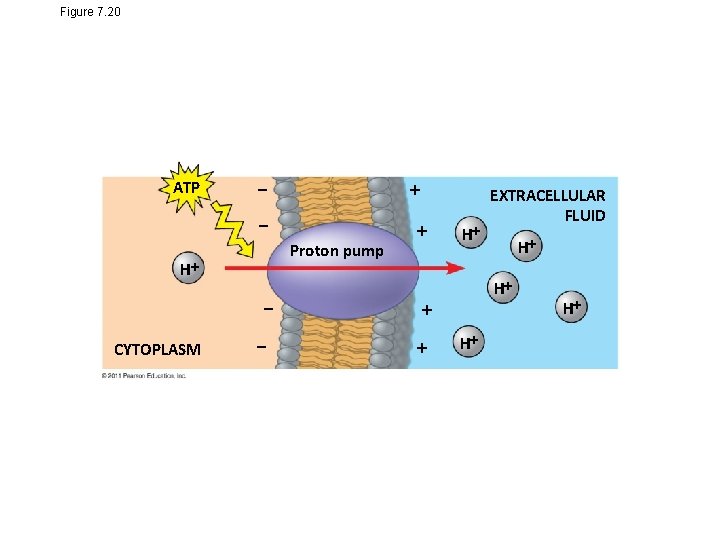
Figure 7. 20 ATP Proton pump H CYTOPLASM H H H EXTRACELLULAR FLUID H H

Cotransport: Coupled Transport by a Membrane Protein • Cotransport occurs when active transport of a solute indirectly drives transport of other solutes • Plants commonly use the gradient of hydrogen ions generated by proton pumps to drive active transport of nutrients into the cell © 2011 Pearson Education, Inc.
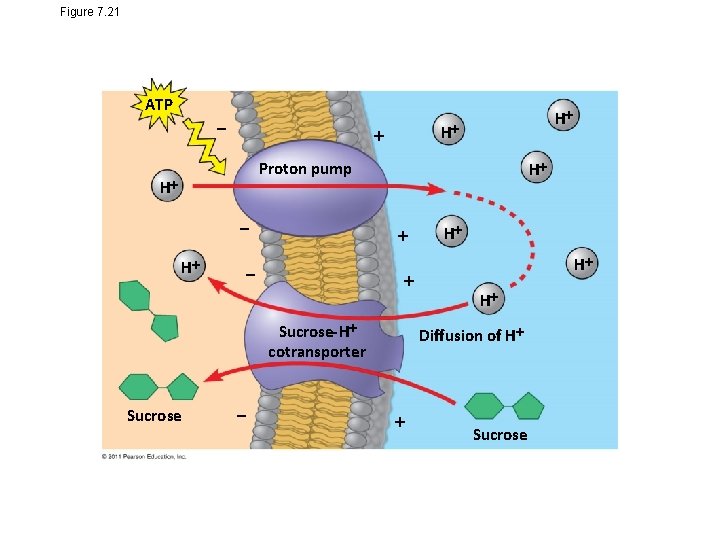
Figure 7. 21 ATP H H Proton pump H H H H H Sucrose-H cotransporter Sucrose Diffusion of H Sucrose
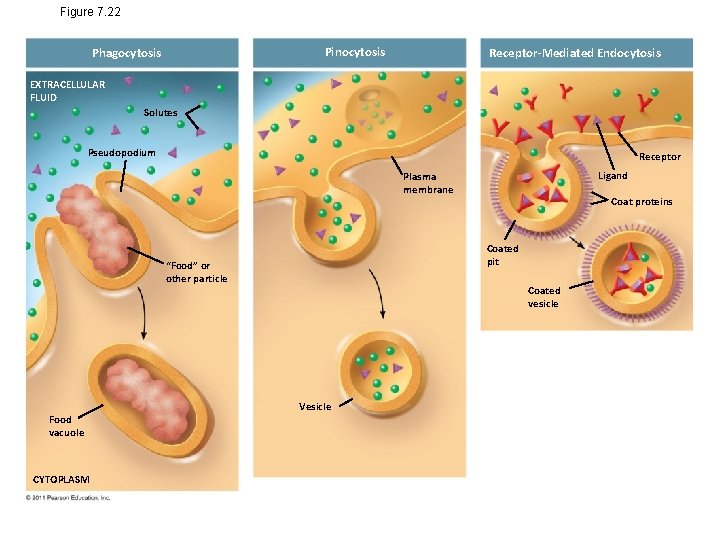
Figure 7. 22 Pinocytosis Phagocytosis Receptor-Mediated Endocytosis EXTRACELLULAR FLUID Solutes Pseudopodium Receptor Ligand Plasma membrane Coated pit “Food” or other particle Food vacuole CYTOPLASM Coat proteins Coated vesicle Vesicle
- Slides: 125