AP BIOLOGY Chapter 6 Metabolism Metabolism The total

AP BIOLOGY Chapter 6 Metabolism
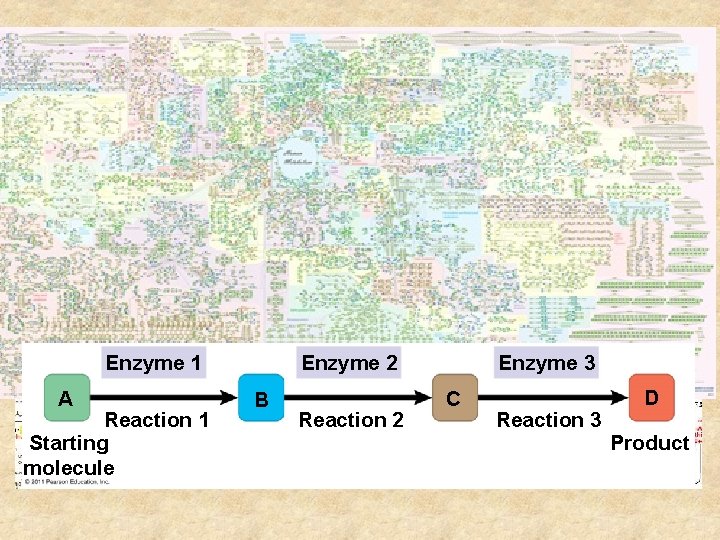
Metabolism • The total of all chemical rxn in an organism – Aranged in pathways – molecule is altered step by step to a product • Manage material and energy resources of the cell Enzyme 2 Enzyme 1 A Reaction 1 Starting molecule B Reaction 2 Enzyme 3 C Reaction 3 D Product
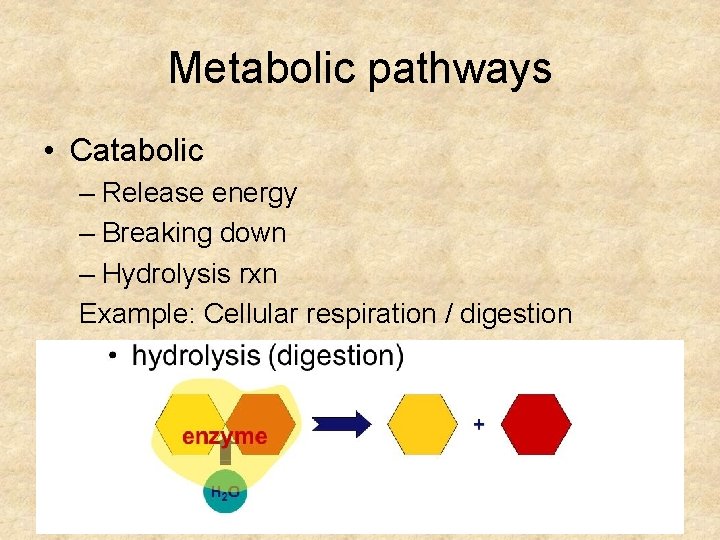
Metabolic pathways • Catabolic – Release energy – Breaking down – Hydrolysis rxn Example: Cellular respiration / digestion
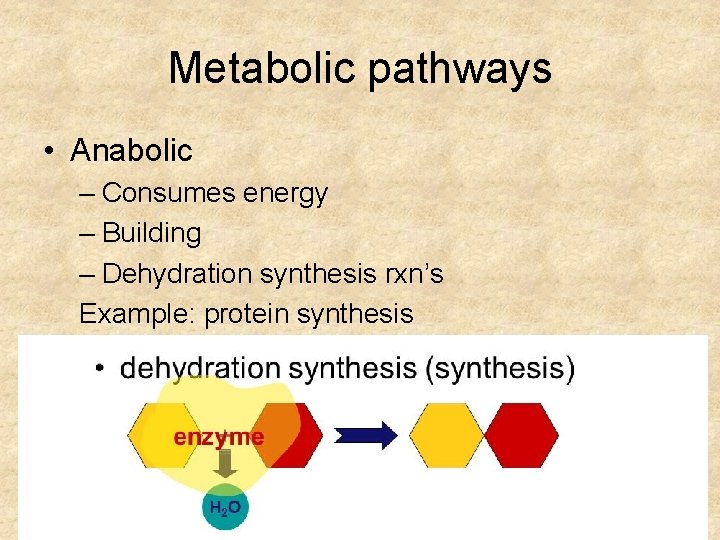
Metabolic pathways • Anabolic – Consumes energy – Building – Dehydration synthesis rxn’s Example: protein synthesis

Now…. Lets Talk Energy • Ability to do work – Kinetic energy is NRG associated with motion • Heat (thermal energy) = kinetic NRG associated with random movement of atoms or molecules – Potential energy = NRG of location or structure • Chemical energy is P. E. (potential energy) available in a chemical reaction • Energy can be converted from one form to another


A diver has more potential energy on the platform than in the water. Climbing up converts the kinetic energy of muscle movement to potential energy. Diving converts potential energy to kinetic energy. A diver has less potential energy in the water than on the platform.

Thermodynamics • Study of energy transformations within matter • Lingo to know – System = matter under study – Surrounding = everything outside the system – Isolated system = cannot transfer matter or nrg (ex: thermos) – Open system = nrg and matter can transfer between system and surrounding (ex: organsims)

1 st Law of Thermo’d • Energy can be transferred and transformed, but it cannot be created or destroyed. • AKA the law of “Conservation of Energy”

2 nd Law of Thermo’d • During NRG transfer/transform some NRG is made unusable and lost as heat Therefore • Each energy transfer or transformation increases the entropy of the universe.

Entropy • The measure of disorder or randomness (symbolized by S) • What has higher entropy?

Entropy • Cells unavoidably covert organized forms of nrg to heat – Spontaneous processes occur without nrg input • Energetically favorable (release energy) • Must increase entropy of universe

Heat Chemical energy (b) Second law of thermodynamics (a) First law of thermodynamics

Summary • The quantity of energy in the universe is constant, but its quality is not.

Question? • Does evolution, the creation of ever more complex organisms, violate the 2 nd law of thermodynamics? • NOPE – Entropy (disorder) may decrease in an organism, but the universe’s total entropy increases

Spontaneous or not spontaneous that is the question

Free-Energy Change, G • The portion of a system's energy that can perform work. ∆G = ∆H – T∆S ∆G = free energy of a system ∆H = total energy of a system (enthalpy) T = temperature in o. K ∆S = entropy of a system

Free-Energy Change, G • Only rxn with negative G are spontaneous – Can be harnessed to do work • Measure of a systems instability, tendency to become more stable – Decrease in free nrg = increase in stability – Chemical equilibrium = max stability

• More free nrg (higher G) • Less stable • Greater work capacity • More free energy (higher G) • Less stable • Greater work capacity In a spontaneous change • free nrg of the system In a decreases spontaneous change ( G 0) • The free energy of the system decreases ( G 0) • • The system becomes more stable • The released free energy can harnessed to do work • be. The released free nrg can be harnessed to do work • Less free energy (lower G) • More stable • Less work capacity (a) Gravitational • Less free energy (lowermotion G) • More stable • Less work capacity (b) Diffusion (c) Chemical reaction

Free-Energy and Metabolism • Chemical rxn = source of nrg for living systems – Based on Free-Energy Change, G

Rxn Types • Exergonic – rxn with a net release of free energy – Spontaneous • Endergonic – rxn that absorb free nrg from environment – Non-spontaneous


(a) Exergonic reaction: energy released, spontaneous Free energy Reactants Amount of energy released ( G 0) Energy Products Progress of the reaction

(b) Endergonic reaction: energy required, nonspontaneous Free energy Products Energy Reactants Progress of the reaction Amount of energy required ( G 0)

Bio examples • Exergonic = digestion • Endergonic = protein synthesis

Equilibrium and Metabolism • Metabolism is NEVER at equilibrium • Catabolic pathway releases free NRG in a series of RXN – Reversable RXN are pulled in one direction • kept out of equilibrium • Product of one RXN is reactant for the next
- Slides: 26