AP Biology Ch 2 The Chemical Context of

AP Biology Ch. 2 The Chemical Context of Life • Essential elements of life • Structure of atoms • Chemical bonding • Chemical reactions Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Bombardier Beetle’s Defense: Ejecting a spray of hot irritating chemicals, an example of the interconnectedness of biology and chemistry.
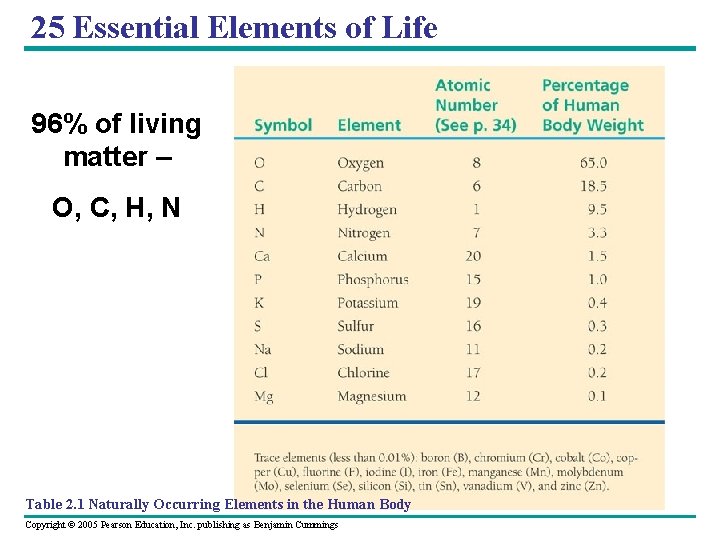
25 Essential Elements of Life 96% of living matter – O, C, H, N Table 2. 1 Naturally Occurring Elements in the Human Body Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Effects of essential-element deficiencies Trace elements – required only in minute quantities deficiency results in great consequences 0. 15 mg iodine required (a) Nitrogen deficiency Figure 2. 3 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings (b) Iodine deficiency – trace element Malaysian woman – can be reversed with iodine supplements

An element’s properties depend on the structure of its atoms. • Atom – smallest unit of matter • Mass number Model of Helium Cloud of negative charge (2 electrons) Electrons Nucleus – sum of protons + neutrons • Atomic number – Number of protons in the nucleus; 2 He • Atomic weight – Mass number; 4 He • Isotopes – 2 atoms of same element; differ in # of neutrons Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Radioactive isotopes • Unstable nuclei decay, emitting particles & energy • Applications: date fossils trace atoms through metabolic processes diagnose disorders Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Figure 2. 5 Research Method Radioactive Tracers APPLICATION Scientists use radioactive isotopes to label certain chemical substances, creating tracers that can be used to follow a metabolic process or locate the substance within an organism. In this example, radioactive tracers are being used to determine the effect of temperature on the rate at which cells make copies of their DNA. TECHNIQUE Ingredients including radioactive tracer (bright blue) 1 2 3 10°C 15°C 20°C 4 5 6 25°C 30°C 35°C 8 9 Human cells 1 2 Ingredients for making DNA are added to human cells. One ingredient is labeled with 3 H, a radioactive isotope of hydrogen. Nine dishes of cells are incubated at different temperatures. The cells make new DNA, incorporating the radioactive tracer with 3 H. The cells are placed in test tubes, their DNA is isolated, and unused ingredients are removed. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Incubators 7 40°C 45°C 50°C DNA (old and new) 1 2 3 4 5 6 7 8 9 Radioactive isotopes – elements with unstable nuclei; give off particles and energy as they decay

Placed in a Scintillation Chamber In scintillators the part of the energy lost by a particle is converted into the energy of a light flash Counts per minute (x 1, 000) RESULTS The frequency of flashes, - counts per minute, proportional to the amount of the radioactive tracer present, indicating the amount of new DNA. In this experiment, when the counts per minute are plotted against temperature, it is clear that temperature affects the rate of DNA synthesis—the most DNA was made at 35°C. 30 20 Optimum temperature for DNA synthesis 10 0 10 20 30 40 Temperature (°C) 50 Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Chemical Behavior - determined by it’s electron configuration. Valence electrons – only unpaired electrons are involved Hydrogen 1 H First shell Atomic mass 2 He 4. 00 Atoms with the same # of valence e- have similar chemical behaviors. Lithium 3 Li Beryllium 4 Be Sodium 11 Na Magnesium 12 Mg Boron 3 B Carbon 6 C Nitrogen 7 N Silicon 14 Si Phosphorus 15 P Atomic number Helium 2 He Element symbol Electron-shell diagram Oxygen 8 O Fluorine 9 F Neon 10 Ne Sulfur 16 S Chlorine 17 Cl Argon 18 Ar Second shell Aluminum 13 Al Third shell Figure 2. 8 Electron-shell diagrams of the first 18 elements in the periodic table. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings nonreactive

Formation & function of molecules depend on chemical bonding between atoms. • Chemical bonds (4) – Covalent bond – – Ionic bond – – Hydrogen bonds – – van der Waals interactions – Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Covalent bonding – atoms sharing epolar (e- shared unequally) vs. nonpolar (e- shared equally) Name (molecular formula) (a) Hydrogen (H 2). Two hydrogen atoms can form a single bond. H (b) Oxygen (O 2). Two oxygen atoms share two pairs of electrons to form a double bond. (c) Water (H 2 O). Two hydrogen atoms and one oxygen atom are joined by covalent bonds to produce a molecule of water. (d) Methane (CH 4). Four hydrogen atoms can satisfy the valence of one carbon atom, forming methane. Structural formula Electronshell diagram H O O O H H Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings Pair of equally shared e- Pair of unequally shared e- H H H C H Spacefilling model H

Polar / Nonpolar Covalent Rules • Nonpolar – e- shared equally -b/t 2 atoms of same element – H-H -b/t atoms w/similar electronegativity – CH 4 • Polar – e- shared unequally ex. H 2 O; oxygen – higher electronegativity • Electronegativity – attraction of an atom for the shared e- of a covalent bond (strong electronegative atoms attempt to pull the shared e- toward themselves) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Figure 2. 12 Polar covalent bonds in a water molecule Because oxygen (O) is more electronegative than hydrogen (H), shared electrons are pulled more toward oxygen. – This results in a partial negative charge on the oxygen and a partial positive charge on the hydrogens. O H H + H 2 O Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings +

Ionic bond – – one atom strips e- from another cations (atom w/+ charge) (atom w/- charge) anions The attraction between oppositely charged atoms, or ions, is an ionic bond. 1 The lone valence electron of a sodium 2 Each resulting ion has a completed atom is transferred to join the 7 valence electrons of a chlorine atom. Na Na Sodium atom (an uncharged atom) Cl Cl Chlorine atom (an uncharged atom) Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings valence shell. An ionic bond can form between the oppositely charged ions. + – Na Cl Na+ Sodium ion (a cation) Cl– Chloride ion (an anion) Sodium chloride (Na. Cl)

Hydrogen bonds – – H atom of one compound is attracted to another compound – Holds water together – + H Water (H 2 O) O H + – Ammonia (NH 3) N H + H H + Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings + A hydrogen bond results from the attraction between the partial positive charge on the hydrogen atom of water and the partial negative charge on the nitrogen atom of ammonia.

van der Waals interactions – Molecules in close proximity exert attraction in concert Hair tip molecules and the molecules of the wall’s surface are so numerous, collectively they form strong bonding patterns temporarily. Gecko lizard – walks up walls Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Molecule’s biological function -related to its shape • Biological molecules: – Recognize, interact with one another with a specificity based on molecular shape. Ex. morphine affects pain perception and emotional state by mimicking the brain’s natural endorphins. Natural endorphin Morphine Endorphin receptors Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings

Chemical reactions make / break chemical bonds. • Change reactants into products conserving matter • Most are reversible. • Chemical equilibrium is reached when the forward and reverse reaction rates are equal Elodea, a freshwater plant, produces sugar by rearranging the atoms of carbon dioxide and water in the chemical process known as photosynthesis. Much of the sugar is then converted to other food molecules. Oxygen gas is a by-product of photosynthesis; hence the bubbles of oxygen escaping from the leaves. Copyright © 2005 Pearson Education, Inc. publishing as Benjamin Cummings
- Slides: 17