AOSC 620 Ozone in the Stratosphere R Dickerson

























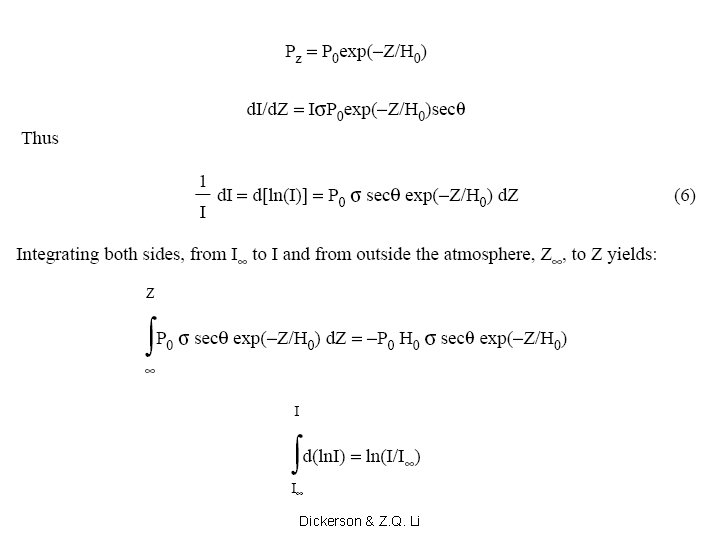







- Slides: 40

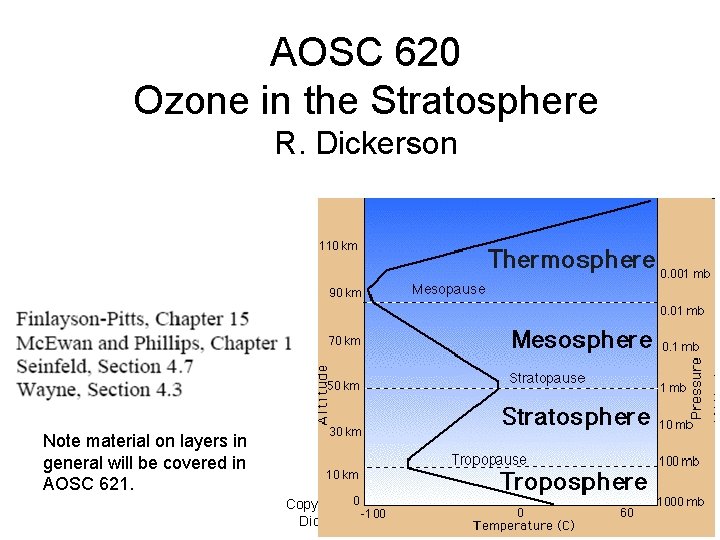
AOSC 620 Ozone in the Stratosphere R. Dickerson Note material on layers in general will be covered in AOSC 621. Copyright © 2010 R. R. Dickerson & Z. Q. Li 1

Montzka et al. , Nature, 2018. Unexpected increase in CFC-11 used for polyurethane foam.

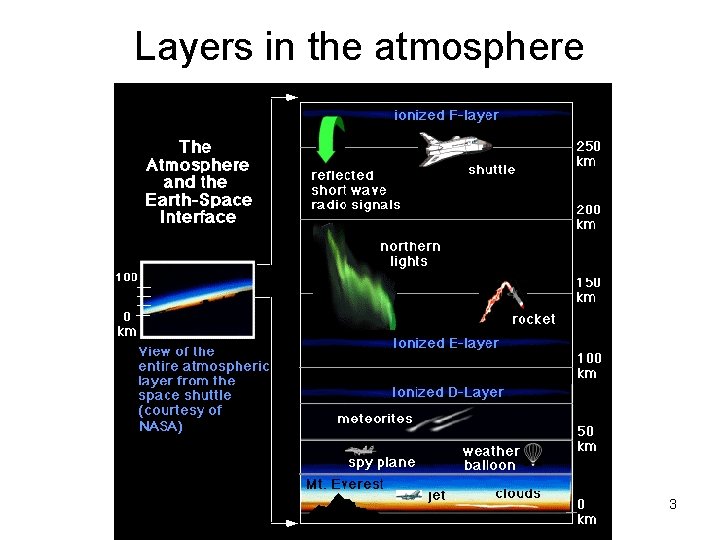
Layers in the atmosphere Copyright © 2013 R. R. Dickerson & Z. Q. Li 3

Actual depth of atmospheric layers. Copyright © 2013 R. R. Dickerson & Z. Q. Li 4

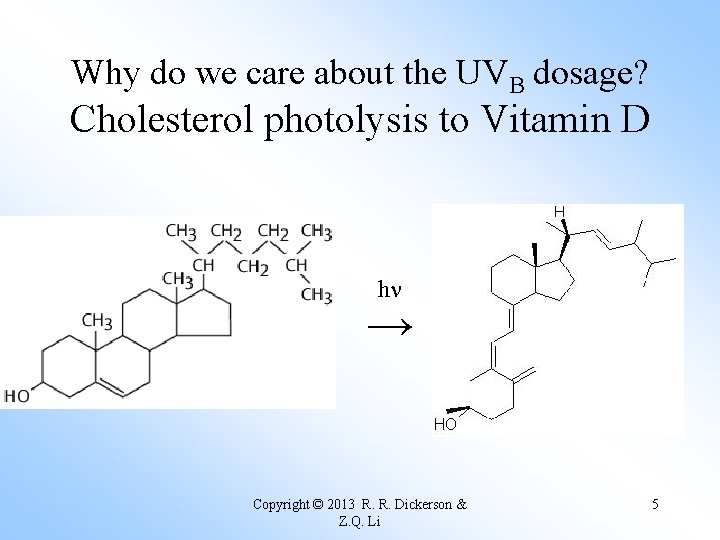
Why do we care about the UVB dosage? Cholesterol photolysis to Vitamin D hn → Copyright © 2013 R. R. Dickerson & Z. Q. Li 5

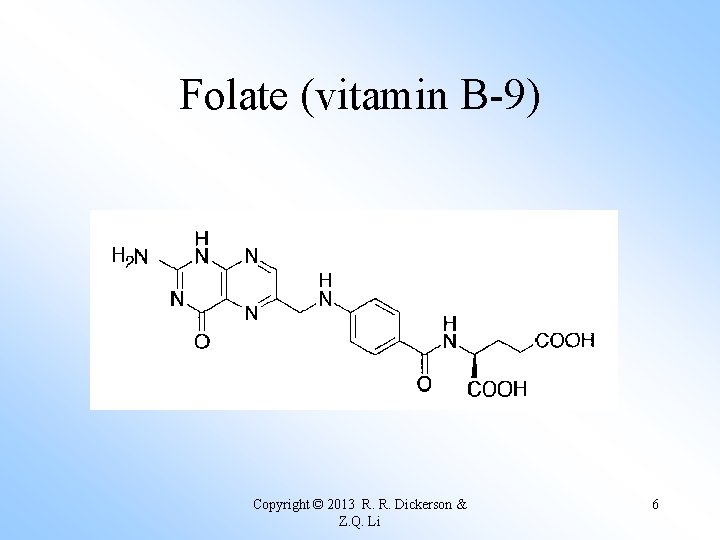
Folate (vitamin B-9) Copyright © 2013 R. R. Dickerson & Z. Q. Li 6

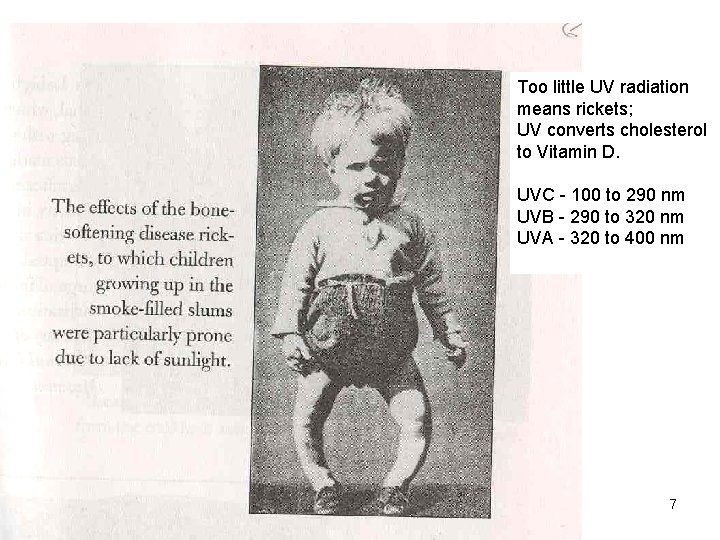
Too little UV radiation means rickets; UV converts cholesterol to Vitamin D. UVC - 100 to 290 nm UVB - 290 to 320 nm UVA - 320 to 400 nm Copyright © 2013 R. R. Dickerson & Z. Q. Li 7

The National Museum of Health and Medicine Silver Spring, MD Normal Femur Rickets

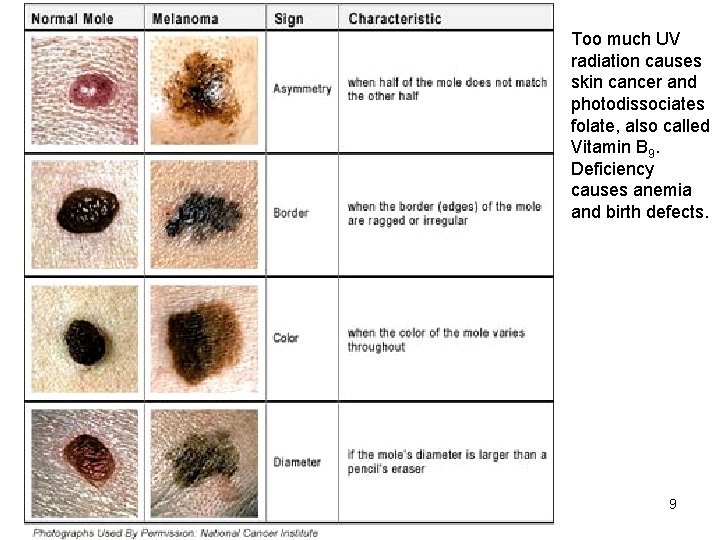
Too much UV radiation causes skin cancer and photodissociates folate, also called Vitamin B 9. Deficiency causes anemia and birth defects. Copyright © 2013 R. R. Dickerson & Z. Q. Li 9

Copyright © 2013 R. R. Dickerson & Z. Q. Li 10

Copyright © 2013 R. R. Dickerson & Z. Q. Li 11

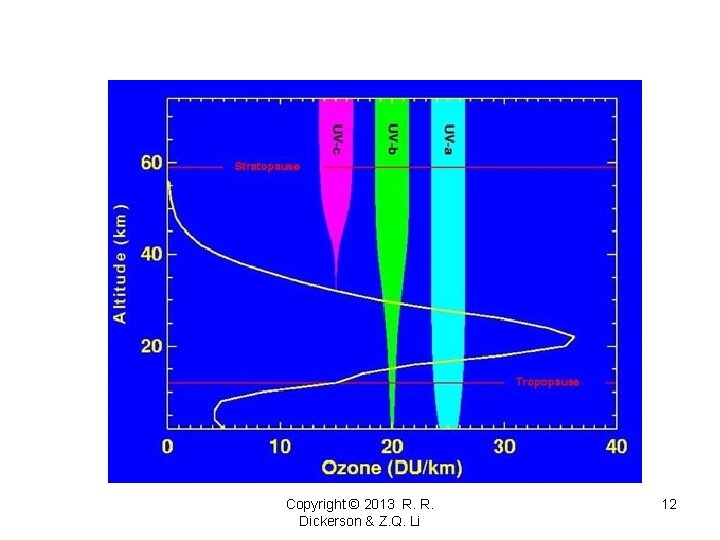
Copyright © 2013 R. R. Dickerson & Z. Q. Li 12

The Stratospheric Ozone Layer Without a protective ozone layer there can be no protein-based life on the surface of the Earth. Here we examine the catalytic cycles that control the level of ozone in the stratosphere. We will calculate the O 3 abundances expected for various levels of understanding. 1) Chapman Reactions (1931) O 2 + hv 2 O O + O 2 + M O 3 + hv O + O 3 (1) O 3 + M* O 2 + O (2) (3) 2 O 2 (4) Copyright © 2013 R. R. Dickerson & Z. Q. Li 13

Copyright © 2013 R. R. Dickerson & Z. Q. Li 14

Copyright © 2013 R. R. Dickerson & Z. Q. Li 15

Copyright © 2013 R. R. Dickerson & Z. Q. Li 16

Copyright © 2013 R. R. Dickerson & Z. Q. Li 17

From The Stratosphere 1981 R. D. Hudson Editor 18

Copyright © 2013 R. R. Dickerson & Z. Q. Li 19

O/O 3 = j(O 3)/(k 2 M O 2) Copyright © 2013 R. R. Dickerson & Z. Q. Li 20

Copyright © 2013 R. R. Dickerson & Z. Q. Li 21

Copyright © 2013 R. R. Dickerson & Z. Q. Li 22

Copyright © 2013 R. R. Dickerson & Z. Q. Li 23

Copyright © 2013 R. R. Dickerson & Z. Q. Li 24

The second catalytic cycle speeds up Reaction 4, that is it effectively increases k₄. Note that any loss of odd oxygen is the same as loss of ozone. These catalytic losses are still insufficient to explain the observed ozone concentration. 3) Crutzen (1970); Johnston (1971) “NOx” Odd nitrogen or “NOx” is the sum of NO and NO₂. Often “NOx” is used as “odd nitrogen” which includes NO₃, HNO₃, 2 N₂O₅, HONO, PAN and other species. This total of “odd nitrogen” is better called “NOy” or “total reactive nitrogen. ” N₂ and N₂O are unreactive. NO + O₃ → NO₂ + O₂ O + NO₂ → NO + O₂ O + O₃ → 2 O₂ NET This is the major means of destruction of stratospheric ozone. The NOx cycle accounts for about 70% of the ozone loss at 30 km. We will calculate the implied steady ozone concentration later. Copyright © 2013 R. R. Dickerson & Z. Q. Li 25

4) Stolarski & Cicerone (1974); Wofsy & Mc. Elroy (1974) “Cl. Ox” Cl + O₃ → Cl. O + O₂ Cl. O + O → Cl + O₂ O + O₃ → 2 O₂ NET This reaction scheme is very fast, but there is not much Cl. Ox in the stratosphere … yet. Molina and Rowland (1974) pointed out that CFC’s release Cl atoms. Today Cl. Ox accounts for about 8% of the ozone loss at 30 km. If all these catalytic destruction cycles are added together, they are still insufficient to explain the present stratosphere O₃ level. The general for of a catalytic ozone destruction cycle is: X + O₃ → XO + O₂ XO + O → X + O₂ O + O₃ → 2 O₂ NET Copyright © 2013 R. R. Dickerson & Z. Q. Li 26

The Stratospheric Ozone Layer Copyright © 2013 R. R. Dickerson & Z. Q. Li 27

How did Farby and Buisson do? At 3000 Å (300 nm) 1/100 = I/Io = ln(-ecl) e 300 = 9. 5 atm-1 cm-1 -ln(0. 01)/9. 5 = 0. 485 atm cm (compare to 0. 50) 0. 485 atm cm = 485 DU Correct for zenith angle of 27 o for Paris? (cos 27 o = 0. 9 so W = 432 DU. ) [O 3] = 0. 6 cm 3 m-3 = 6 x 10 -7 Is that right? [O 3] for uniform mixing ratio = column content/scale height 5 mm/8 x 106 mm = 6. 25 x 10– 7 = 625 ppb As Fabry said this is too much for surface air, and the ozone layer lies above. Copyright © 2013 R. R. Dickerson & Z. Q. Li 28

Slides 27 on will be presented in AOSC 621. Copyright © 2010 R. R. Dickerson & Z. Q. Li 29

Copyright © 2010 R. R. Dickerson & Z. Q. Li 30

Copyright © 2010 R. R. Dickerson & Z. Q. Li 31

Copyright © 2010 R. R. Dickerson & Z. Q. Li 32
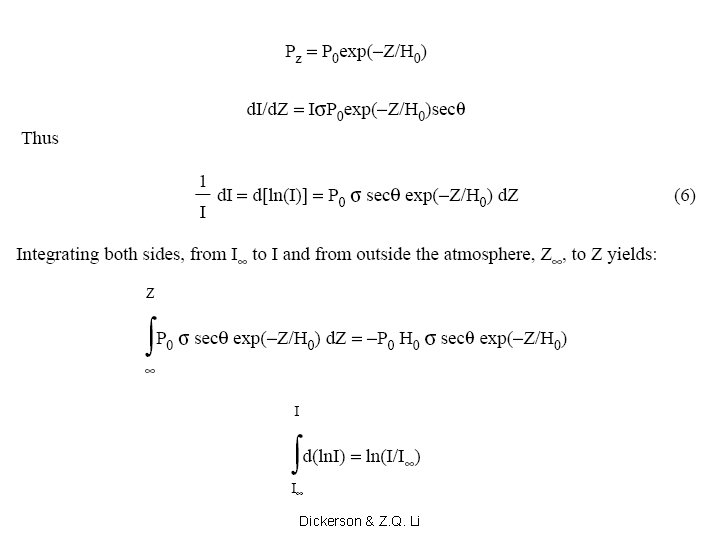
Copyright © 2010 R. R. Dickerson & Z. Q. Li 33

Copyright © 2010 R. R. Dickerson & Z. Q. Li 34

Copyright © 2010 R. R. Dickerson & Z. Q. Li 35

Copyright © 2013 R. R. Dickerson & Z. Q. Li 36

Copyright © 2013 R. R. Dickerson & Z. Q. Li 37

Copyright © 2013 R. R. Dickerson & Z. Q. Li 38

Copyright © 2013 R. R. Dickerson & Z. Q. Li 39

Copyright © 2010 R. R. Dickerson & Z. Q. Li 40