ANTITHROMBOTIC THERAPY R Whorton Mechanisms of Thrombus Formation

ANTITHROMBOTIC THERAPY R. Whorton
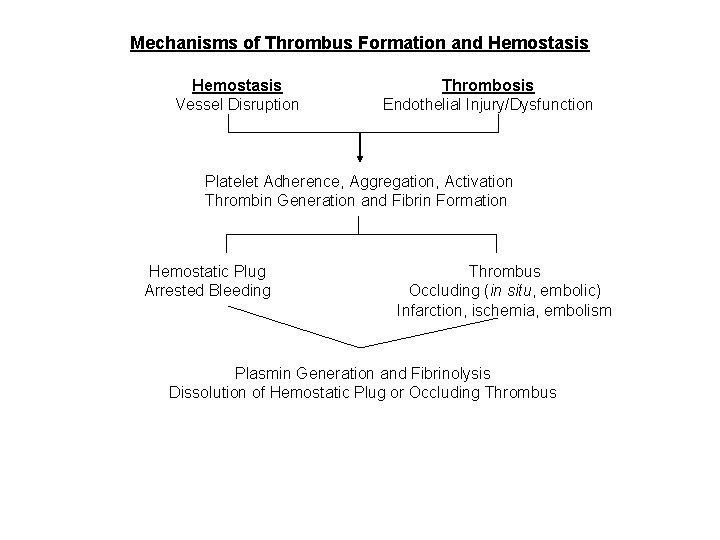
Mechanisms of Thrombus Formation and Hemostasis Vessel Disruption Thrombosis Endothelial Injury/Dysfunction Platelet Adherence, Aggregation, Activation Thrombin Generation and Fibrin Formation Hemostatic Plug Arrested Bleeding Thrombus Occluding (in situ, embolic) Infarction, ischemia, embolism Plasmin Generation and Fibrinolysis Dissolution of Hemostatic Plug or Occluding Thrombus
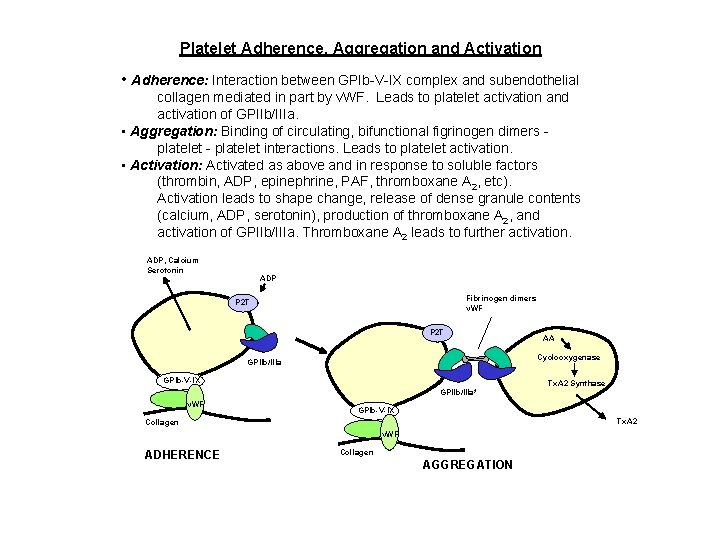
Platelet Adherence, Aggregation and Activation • Adherence: Interaction between GPIb-V-IX complex and subendothelial collagen mediated in part by v. WF. Leads to platelet activation and activation of GPIIb/IIIa. • Aggregation: Binding of circulating, bifunctional figrinogen dimers platelet - platelet interactions. Leads to platelet activation. • Activation: Activated as above and in response to soluble factors (thrombin, ADP, epinephrine, PAF, thromboxane A 2, etc). Activation leads to shape change, release of dense granule contents (calcium, ADP, serotonin), production of thromboxane A 2, and activation of GPIIb/IIIa. Thromboxane A 2 leads to further activation. ADP, Calcium Serotonin ADP Fibrinogen dimers v. WF P 2 T AA Cyclooxygenase GPIIb/IIIa GPIb-V-IX Tx. A 2 Synthase GPIIb/IIIa* v. WF GPIb-V-IX Tx. A 2 Collagen v. WF ADHERENCE Collagen AGGREGATION
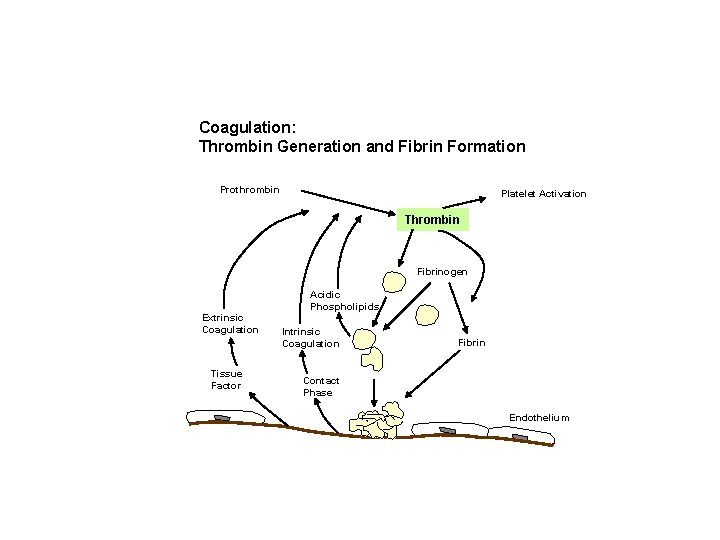
Coagulation: Thrombin Generation and Fibrin Formation Prothrombin Platelet Activation Thrombin Fibrinogen Extrinsic Coagulation Tissue Factor Acidic Phospholipids Intrinsic Coagulation Fibrin Contact Phase Endothelium
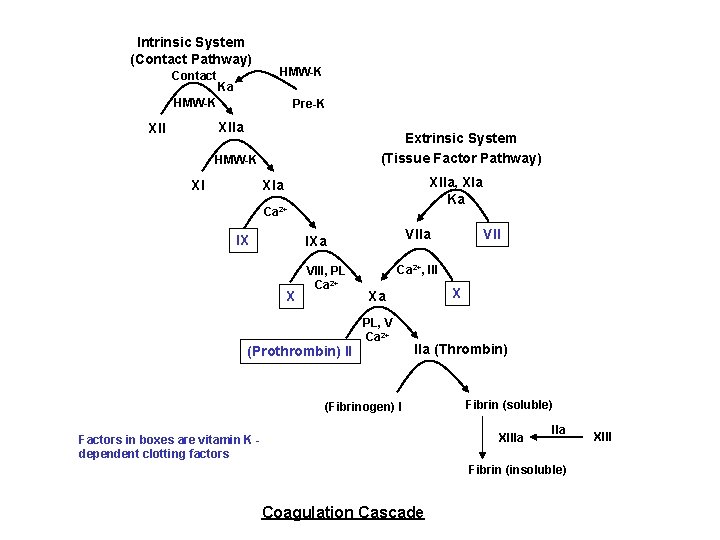
Intrinsic System (Contact Pathway) Contact HMW-K Ka HMW-K Pre-K XIIa XII Extrinsic System (Tissue Factor Pathway) HMW-K XI XIIa, XIa Ka XIa Ca 2+ IX VIIa IXa X VIII, PL Ca 2+ (Prothrombin) II VII Ca 2+, III X Xa PL, V Ca 2+ IIa (Thrombin) (Fibrinogen) I Fibrin (soluble) XIIIa Factors in boxes are vitamin K dependent clotting factors IIa Fibrin (insoluble) Coagulation Cascade XIII

Regulation of Thrombin Activity: 1. Inactivation by binding to ATIII 2. Binding to receptors on endothelial cells 3. Endothelial facilitated ATIII-thrombin interaction (surface) 4. Activation of protein C by thrombin in combination with thrombomodulin. Protein C inactivates factors V and VIII and thus limits thrombin formation

Other Factors Regulating Thrombus Formation and Size Shear forces caused by blood flow activate platelets, activate aggregation and affect adherence High flow rates reduce coagulation factor concentration - Low flow rates (venous side) may allow coagulation factor concentrations to reach levels which activate coagulation leading to thrombus formation Arterial thrombi are comprised mainly of platelets - result of activated platelet aggregation Venous thrombi contain platelets, red cells and fibrin - result, in part, from activation of coagulation cascade
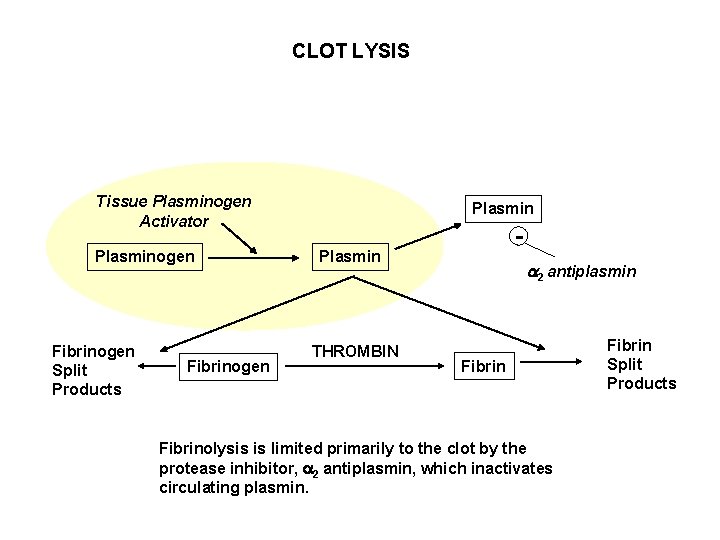
CLOT LYSIS Tissue Plasminogen Activator Plasminogen Fibrinogen Split Products Fibrinogen Plasmin - Plasmin THROMBIN 2 antiplasmin Fibrinolysis is limited primarily to the clot by the protease inhibitor, 2 antiplasmin, which inactivates circulating plasmin. Fibrin Split Products
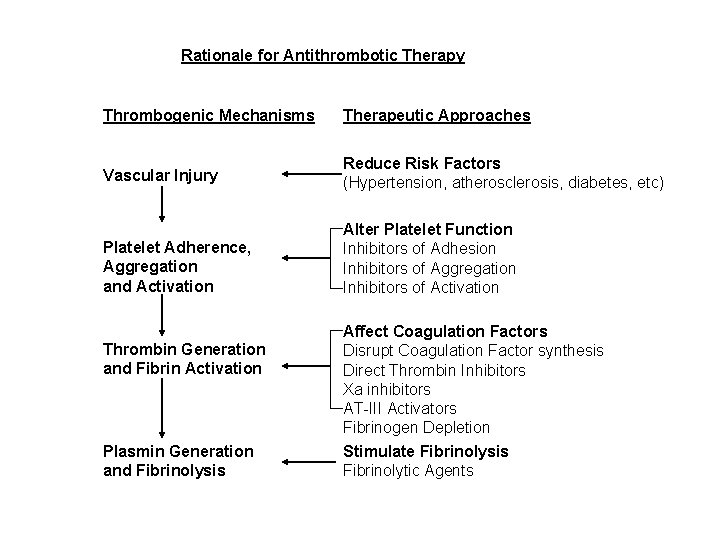
Rationale for Antithrombotic Therapy Thrombogenic Mechanisms Therapeutic Approaches Vascular Injury Reduce Risk Factors (Hypertension, atherosclerosis, diabetes, etc) Platelet Adherence, Aggregation and Activation Alter Platelet Function Inhibitors of Adhesion Inhibitors of Aggregation Inhibitors of Activation Thrombin Generation and Fibrin Activation Plasmin Generation and Fibrinolysis Affect Coagulation Factors Disrupt Coagulation Factor synthesis Direct Thrombin Inhibitors Xa inhibitors AT-III Activators Fibrinogen Depletion Stimulate Fibrinolysis Fibrinolytic Agents
![ANTIPLATELET DRUGS Tx. A 2 Thrombin + [Ca 2+]i PL - AA PLA 2 ANTIPLATELET DRUGS Tx. A 2 Thrombin + [Ca 2+]i PL - AA PLA 2](http://slidetodoc.com/presentation_image_h/cf350f0d23db53be75ad592a974906c6/image-10.jpg)
ANTIPLATELET DRUGS Tx. A 2 Thrombin + [Ca 2+]i PL - AA PLA 2 Aspirin Cyclooxygenase Inhibitors of Aggregation Clopidogrel Abciximab (Reopro) Epifibatide Tirobifan Tx. A 2 Synthase PGI 2 + Adenylate Cyclase AMP - Phosphodiesterase Dipyridamole GMP Contraction Clopidogrel Release c. GMP + NO Tx. A 2 c. AMP Guanylate Cyclase - Inhibitors of Activation: Aspirin Dipyridamole P 2 T - + GPIIb/IIIa - Abciximab, Epifibatide, Tirofiban General Use: Prevention of arterial thrombogenesis

Antiplatelet Drugs Inhibitors of Activation: These drugs interfere with platelet function and are used primarily for arterial thrombotic diseases to prevent the formation of platelet thrombi and platelet-derived vasoactive substances (Thromboxane A 2 and serotonin). Aspirin - Irreversible inhibition of cyclooxygenase which lasts the lifetime of the platelet. Dipyridamole - Inhibits adenosine uptake and cyclic nucleotide phosphodiesterase and elevates c. AMP levels. Usually used in combination with other drugs, especially warfarin, for prevention of thrombus/embolus formation in patients with prosthetic heart valves.
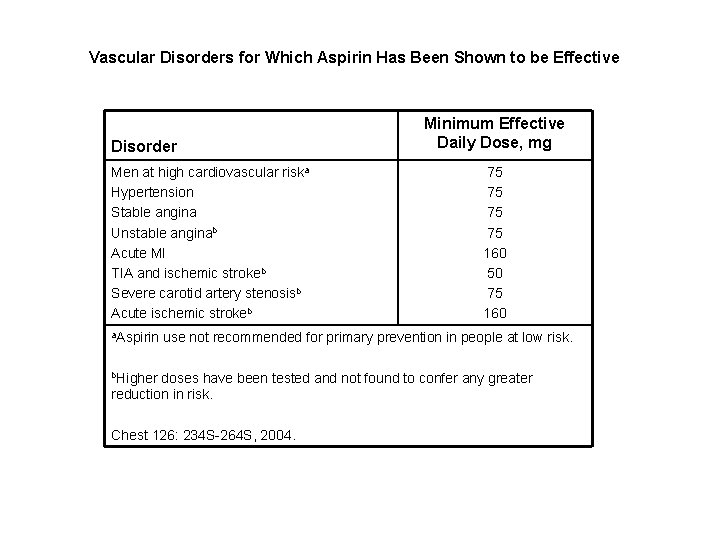
Vascular Disorders for Which Aspirin Has Been Shown to be Effective Disorder Men at high cardiovascular riska Hypertension Stable angina Unstable anginab Acute MI TIA and ischemic strokeb Severe carotid artery stenosisb Acute ischemic strokeb a. Aspirin Minimum Effective Daily Dose, mg 75 75 160 50 75 160 use not recommended for primary prevention in people at low risk. b. Higher doses have been tested and not found to confer any greater reduction in risk. Chest 126: 234 S-264 S, 2004.

Antiplatelet Drugs cont’d Inhibitors of Aggregation: These drugs prevent thrombus/embolus formation by inhibition of aggregation. Used in situations where it is important to prevent arterial thrombogenesis. Clopidogrel (Plavix) and Prasugrel (Effient): P 2 T purinergic receptor antagonists. Uses: Arterial thrombotic complications associated with unstable angina, transient cerebral ischemia, non-cardioembolic stroke, peripheral vascular disease, valvular disease and coronary bypass surgery. Primary prevention in acute coronary syndromes. In some situations may be more effective than aspirin. Especially useful in patients who do not tolerate aspirin. Also used in combination with aspirin in angioplasty or to prevent reocclusion after fibrinolytic treatment of myocardial infarction. Both are prodrugs which require activation by the liver (2 steps for Clopidogrel, 1 for prasugrel making this drug more predictable with fewer potential interactions). Side effects: Diarrhea, skin rash, occasional severe but reversible neutropenia, thrombotic thrombocytopenic pupurea.

Antiplatelet Drugs cont’d Abciximab (Reo. Pro): Chimeric antibody with binding regions of a mouse monoclonal antibody directed against GPIIb/IIIa and the constant region of human Ig. G (to reduce antigenicity). Must be given iv. Eptifibatide (Intergilin) and Tirofiban (aggarstat): Epifibatide is a cyclic heptapeptide with a KGD (lys-gly-asp) sequence similar to that in barbourin. Tirofiban is a non-peptide drug which binds to RGD recognition domains in integrin receptors. Both bind rapidly to and block the GPIIb/IIIa receptor. Both drugs must be given iv. As these drugs are relatively specific as inhibitors of aggregation, adherence is not affected and bleeding times are minimally elongated. Eptifibatide is rapidly cleared from circulation. Uses: These drugs are primarily used in conjunction with aspirin and/or heparin in preventing reocclusion following angioplasty. Also used to prevent reocclusion after fibrinolytic therapy in patients with acute MI.
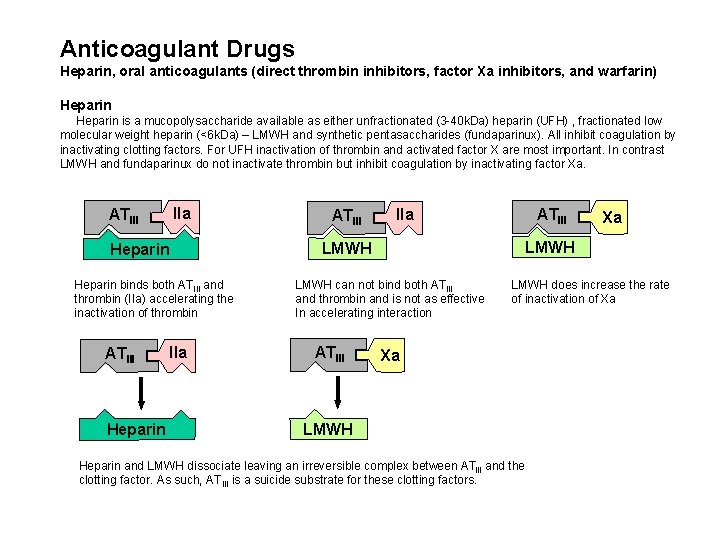
Anticoagulant Drugs Heparin, oral anticoagulants (direct thrombin inhibitors, factor Xa inhibitors, and warfarin) Heparin is a mucopolysaccharide available as either unfractionated (3 -40 k. Da) heparin (UFH) , fractionated low molecular weight heparin (<6 k. Da) – LMWH and synthetic pentasaccharides (fundaparinux). All inhibit coagulation by inactivating clotting factors. For UFH inactivation of thrombin and activated factor X are most important. In contrast LMWH and fundaparinux do not inactivate thrombin but inhibit coagulation by inactivating factor Xa. IIa ATIII Heparin binds both ATIII and thrombin (IIa) accelerating the inactivation of thrombin ATIII Heparin IIa ATIII LMWH can not bind both ATIII and thrombin and is not as effective In accelerating interaction ATIII Xa LMWH does increase the rate of inactivation of Xa Xa LMWH Heparin and LMWH dissociate leaving an irreversible complex between ATIII and the clotting factor. As such, ATIII is a suicide substrate for these clotting factors.

Comparison Unfractionated Heparin – LMWH and Fondaparinux Route of administration Unfractionated Heparin – IV or subcutaneously. Bioavailability by subcutaneous route is only 30%. LMWH (enoxaparin) and Fondaparinux) – Subcutaneously. Bioavailability is at least 90% - longer half life than heparin. Pharmacokinetics Unfractionated Heparin – rapid saturable binding to proteins and vascular surface reduces effectiveness and may lead to sudden unexpected increase in antithrombotic effect – must monitor a. PTT. LMWH and Fondaparinux – predictable clearance by renal and hepatic mechanisms (minor) Anticoagulant effect Unfractionated Heparin – must be carefully titrated and followed by laboratory evaluation of coagulation – complex pharmacokinetics make it difficult to predict plasma levels LMWH and Fondaparinux – Effects and plasma levels are predictable (dose related) and usually do not require monitoring. Uses Used for venous thromboembolism (DVT, PE), unstable angina, cardiac vascular surgery, angioplasty. Because LMWH and Fondaparinux can be administered subcutaneously with predictable anticoagulation, often preferred and can be used in outpatient setting. These drugs are also used in combination with aspirin or other antiplatelet drugs with appropriate monitoring of clotting activity. Risks Bleeding (bigger problem with UFH). Up to 5% of patients receiving UFH or LMWH develop an immunologic reaction leading to thrombocytpenia (heparin induced thrombocytopenia – HIT) – not seen with fondaparinux making it useful in patients with HIT. Antidote – Protoamine sulfate – not as effective with LMWH

Anticoagulant Drugs Cont’d – Direct Thrombin Inhibitors: Hirudin, Bivalirudin: Peptides mimics of anticoagulant isolated from medicinal leach. Produced by recombinant techniques. Hirudin binds to the fibrinogen recognition site of thrombin blocking both protease activity and access to substrate. Bivalirudin is a synthetic derivative of hirudin. Two forms of recombinant hirudin – lepirudin and desirudin. Uses: Hirudin and bivalirudin are used in patients with or at risk for heparin induced thrombocytopenia. Bivalirudin is also used as a adjunct to thrombolytic therapy in acute MI, for treatment of unstable angina and as an antithrombotic agent in conjunction with GP IIb/IIIa inhibitor in high risk angioplasty. Pharmocokinetics: Given either iv (half-life of 1 -2 hrs) or sc where peak plasma concentrations occur in 2 -3 hrs.

Anticoagulant Drugs – Direct Thrombin Inhibitors: Argatroban: Argatroban is a competitive inhibitor of thrombin. It is administered iv and has a half-life of 50 minutes. Used in patients with heparin induced thrombocytopenia and in high risk angioplasty (especially in patients with HIT). Dabigatran: Dabigatran (Pradaxa) is a newly approved, orally available, potent, irreversible inhibitor of thrombin. Approved for use in stroke prevention in patients with non valvular atrial fibrillation and for prevention of venous thromboembolism after hip or knee replacement. More effective and more predictable than warafrin. Anticoagulant Drugs - Factor Xa inhibitors Rivaroxaban and Apixaban: Rivaroxaban (Xarelto) and Apixaban (Eliquis) are inhibitors of factor Xa. They are given orally and like dabigatran are essentially irreversible inhibors. They are approved for prevention of stroke and systemic embolism in patients with nonvalvular artrial fibrillation and for prevention of venous thromboembolism after knee or hip relacement.
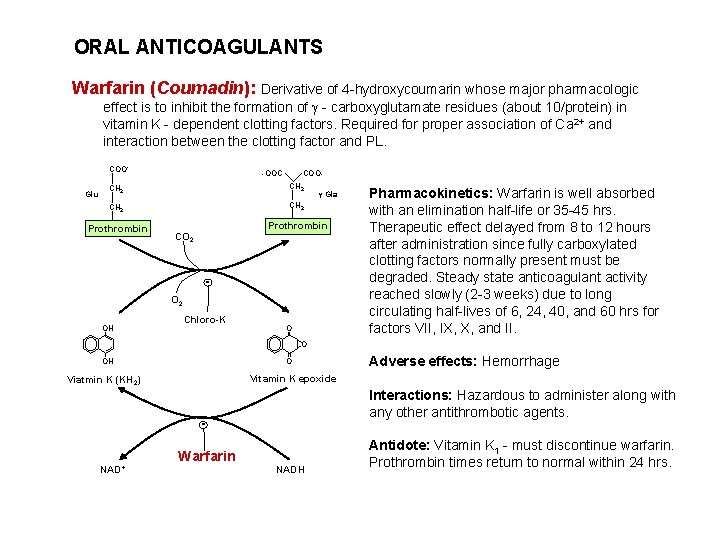
ORAL ANTICOAGULANTS Warfarin (Coumadin): Derivative of 4 -hydroxycoumarin whose major pharmacologic effect is to inhibit the formation of - carboxyglutamate residues (about 10/protein) in vitamin K - dependent clotting factors. Required for proper association of Ca 2+ and interaction between the clotting factor and PL. COOGlu -OOC COO- CH 2 Prothrombin Gla Prothrombin CO 2 OH Chloro-K O Pharmacokinetics: Warfarin is well absorbed with an elimination half-life or 35 -45 hrs. Therapeutic effect delayed from 8 to 12 hours after administration since fully carboxylated clotting factors normally present must be degraded. Steady state anticoagulant activity reached slowly (2 -3 weeks) due to long circulating half-lives of 6, 24, 40, and 60 hrs for factors VII, IX, X, and II. O OH O Vitamin K epoxide Viatmin K (KH 2) Interactions: Hazardous to administer along with any other antithrombotic agents. NAD+ Adverse effects: Hemorrhage Warfarin NADH Antidote: Vitamin K 1 - must discontinue warfarin. Prothrombin times return to normal within 24 hrs.

Warfarin Uses: DVT (venous thromboembolic disease), pulmonary embolism, valvular disease, atrial stasis, atrial fibrillation, prophylaxis in patients with prostheric heart valves and in patients with a history of stroke. In DVT, warfarin therapy is initiated within the first 24 hours in patients receiving heparin. Warfarin is continued for 3 -6 months or longer depending on presence of risk factors. Factors Which Increase Risk for Bleeding in Patients Taking Warfarin 1. Vitamin K deficiency – Diseases which affect vitamin K uptake, certain antibiotics which inhibit vitamin K synthesis by the intestinal flora. 2. Liver Disease – Altered synthesis of clotting factors 3. Drug Interactions (serious bleeding complications are possible) Antiplatelet drugs including aspirin, NSAIDS, others: clopidogrel, ticlopidine 4. Drugs which inhibit warfarin metabolism – cimetidine, salicylates, imipramine, antifungal azoles. 5. Drugs that inhibit reduction of vitamin K – cephalosporines Individual responses to warfarin vary greatly - thus it is important to monitor coagulation.
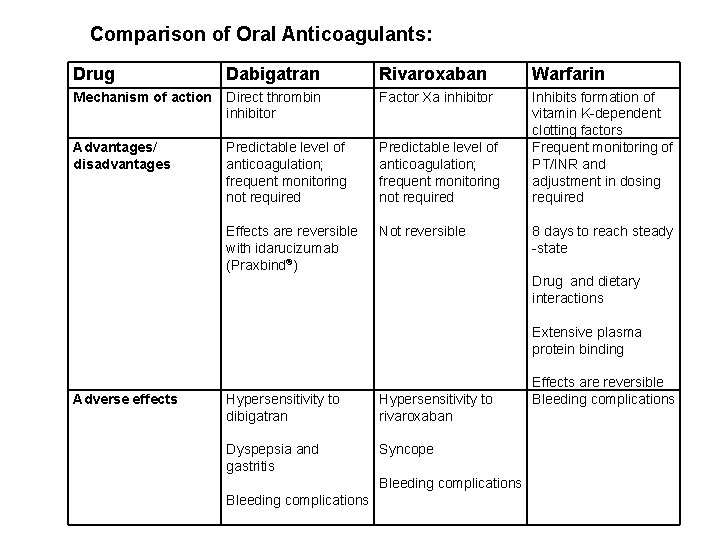
Comparison of Oral Anticoagulants: Drug Dabigatran Rivaroxaban Warfarin Mechanism of action Direct thrombin inhibitor Factor Xa inhibitor Advantages/ disadvantages Predictable level of anticoagulation; frequent monitoring not required Inhibits formation of vitamin K-dependent clotting factors Frequent monitoring of PT/INR and adjustment in dosing required Effects are reversible with idarucizumab (Praxbind®) Not reversible 8 days to reach steady -state Drug and dietary interactions Extensive plasma protein binding Adverse effects Hypersensitivity to dibigatran Hypersensitivity to rivaroxaban Dyspepsia and gastritis Syncope Bleeding complications Effects are reversible Bleeding complications
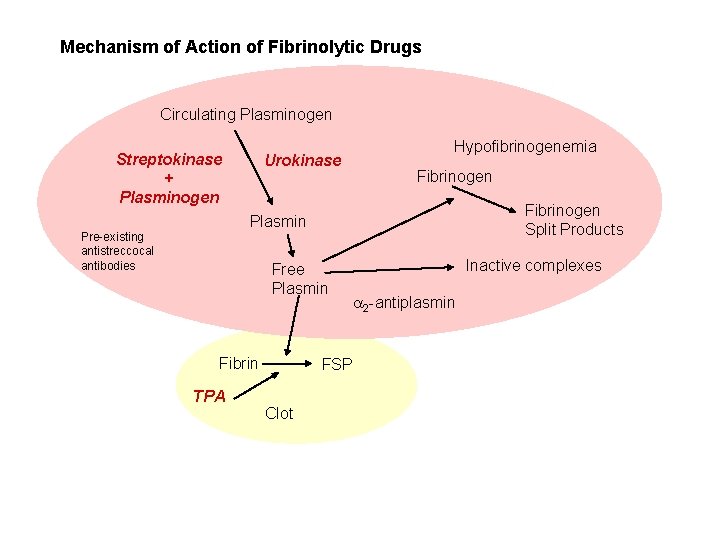
Mechanism of Action of Fibrinolytic Drugs Circulating Plasminogen Streptokinase + Plasminogen Urokinase Hypofibrinogenemia Fibrinogen Split Products Plasmin Pre-existing antistreccocal antibodies Free Plasmin Fibrin TPA FSP Clot Inactive complexes 2 -antiplasmin

Fibrinolytic Agents Primary use of fibrinolytic agents Acute myocardial infarction SK, t. PA Acute ischemic stroke t. PA Acute pulmonary embolism SK, UK, t. PA Acute deep vein thrombosis SK Given intra-arterially. During continuous infusion of streptokinase (DVT, PE) plasminogen levels fall, reducing the effectiveness (partially due to requirement of complex formation).
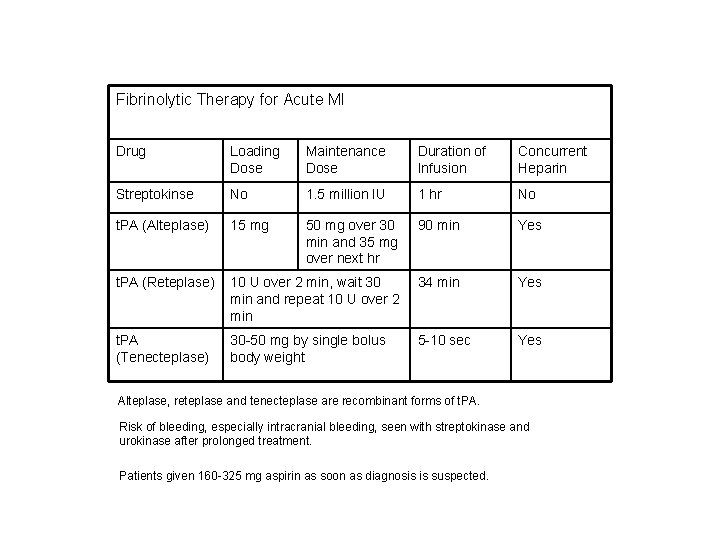
Fibrinolytic Therapy for Acute MI Drug Loading Dose Maintenance Dose Duration of Infusion Concurrent Heparin Streptokinse No 1. 5 million IU 1 hr No t. PA (Alteplase) 15 mg 50 mg over 30 min and 35 mg over next hr 90 min Yes t. PA (Reteplase) 10 U over 2 min, wait 30 min and repeat 10 U over 2 min 34 min Yes t. PA (Tenecteplase) 30 -50 mg by single bolus body weight 5 -10 sec Yes Alteplase, reteplase and tenecteplase are recombinant forms of t. PA. Risk of bleeding, especially intracranial bleeding, seen with streptokinase and urokinase after prolonged treatment. Patients given 160 -325 mg aspirin as soon as diagnosis is suspected.
- Slides: 24