Antiretroviral TherapyInteractive Cases From the Clinicians A CaseBased

Antiretroviral Therapy—Interactive Cases From the Clinic(ians): A Case-Based Panel Discussion Michael S. Saag, MD Professor of Medicine Associate Dean for Global Health University of Alabama at Birmingham, Alabama From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA. San Antonio, Texas: August 21 -23, 2017

Learning Objectives After attending this presentation, participants will be able to: �Select initial ARV Rx in patients who are: Starting initial therapy – Suppressed virologically – Harboring an existing M 184 V mutation – �Discern which individuals are eligible for Pr. EP �Describe the difference between Type I and Type II MI Slide. MS 2 Saag, of XXMD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA. From

HHS Antiretroviral Therapy Guidelines: July, 2017 Initiating Therapy in Treatment-Naïve Patients 2016 HHS Recommendation for Initiating ART Therapy in Treatment-Naïve Patients Antiretroviral Therapy is Recommended for: Rating All HIV-infected individuals, regardless of CD 4 T lymphocyte cell count, to reduce the morbidity and mortality associated with HIV infection. AI For HIV-infected individuals to prevent HIV transmission. AI Source: 2016 HHS Antiretroviral Therapy Guidelines. AIDS Info. Slide 3 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question Seems like we are now starting ARV therapy for about everyone, what about starting therapy for an Elite Controller? Slide 4 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 1 • 30 yo Male was diagnosed with HIV infection 4 years ago • Asymptomatic • Initial: HIV RNA < 50 c/ml (HIV DNA positive) CD 4 count 870 cells/ul • Other labs are normal; HLA-B 57 neg • Genotype determined from DNA is wild-type • No prior medical history. • Ok to start therapy if you think he should Slide 5 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.
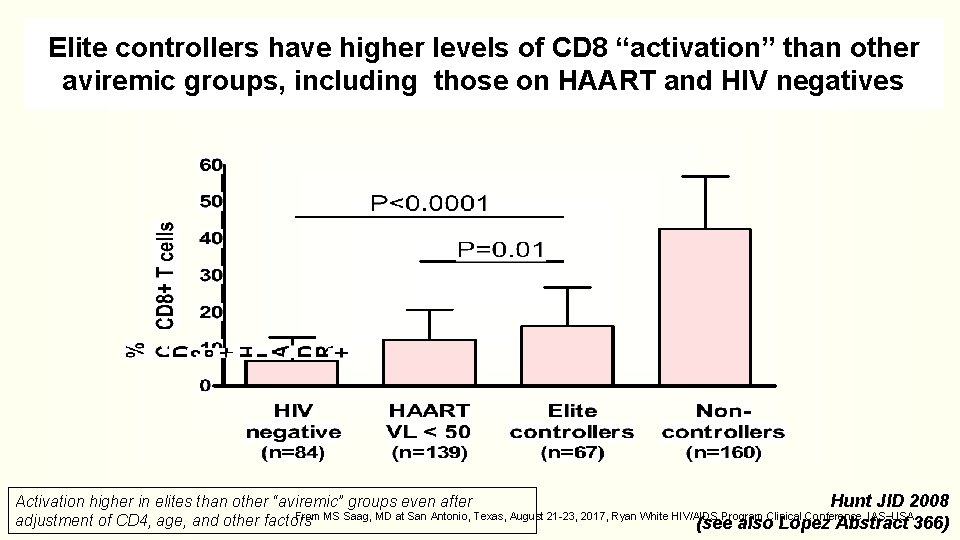
Elite controllers have higher levels of CD 8 “activation” than other aviremic groups, including those on HAART and HIV negatives Activation higher in elites than other “aviremic” groups even after Hunt JID 2008 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA. adjustment of CD 4, age, and other factors (see also Lopez Abstract 366)

Question What regimen should I use as initial therapy? Slide 7 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 2 • 48 yo Male presents with newly diagnosed HIV infection • Asymptomatic • Initial: HIV RNA 28, 000 c/ml • • CD 4 count 650 cells/ul Other labs are normal; HLA-B 57 positive Genotype is Wild-type virus No prior medical history. Normal renal function Ok to start therapy if you think he should Slide 8 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 3 • 48 yo Male presents with newly diagnosed HIV infection • Asymptomatic except for weight loss / fatigue • Initial: HIV RNA 760, 000 c/ml • • CD 4 count 21 cells/ul Other labs are normal; HLA-B 57 negative Genotype is Wild-type virus No prior past medical history. Normal renal function Ok to start therapy if you think he should Slide 9 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

The 6 initial regimens we’ll probably be using most INSTI-based § BIC/FTC/TAF § DTG + FTC/TAF § DTG/3 TC/ABC § RAL + FTC/ TAF (once daily) PI-based § DRV/c + FTC/TAF NNRTI-based § RPV/FTC/TAF Slide 10 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.
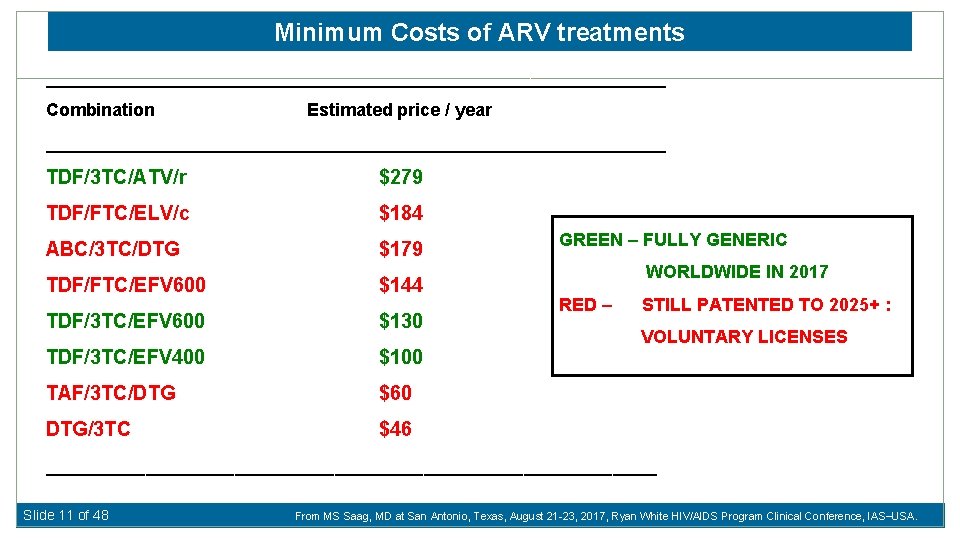
Minimum Costs of ARV treatments _______________________________ Combination Estimated price / year _______________________________ TDF/3 TC/ATV/r $279 TDF/FTC/ELV/c $184 ABC/3 TC/DTG $179 TDF/FTC/EFV 600 $144 TDF/3 TC/EFV 600 $130 TDF/3 TC/EFV 400 $100 TAF/3 TC/DTG $60 DTG/3 TC $46 GREEN – FULLY GENERIC WORLDWIDE IN 2017 RED – STILL PATENTED TO 2025+ : VOLUNTARY LICENSES ____________________________ Slide 11 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question What regimen should be used as initial therapy when an M 184 V mutation is present? Slide 12 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 4 • 30 yo Female presents with newly diagnosed HIV infection • Asymptomatic • Initial: HIV RNA 28, 000 c/ml • • CD 4 count 650 cells/ul Other labs are normal; HLA-B 57 neg Genotype shows M 184 V mutation No prior medical history. No children. Does not plan to become pregnant. Ok to start therapy if you think she should Slide 13 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question Should I switch from EFV / FTC / TDF (fdc) in a patient who has been on it for the last 10 years? Slide 14 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 5 • 45 yo Female referred to you for evaluation • Diagnosed 10 years ago with HIV infection • Initial: HIV RNA 36, 000 c/ml CD 4 count 150 cells/ul • Current: HIV RNA <20 c/ml CD 4 count 525 cells/ul • Started on EFV/ FTC/ TDF (fdc) in Jan 2007. Only regimen. Reports no symptoms currently. • Generally feels well Slide 15 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question Should I give Pr. EP to a sero-negative partner of a successfully treated HIV patient? Slide 16 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 6 • 45 yo Male makes an appointment to request Pr. EP • His partner is HIV positive and has been on successful ARV Rx for • • 17 years (consistently <50 c/ml) Generally feels well No significant PMHx No medications Denies any partners outside of his relationship with his partner Slide 17 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question When, if ever, should I use primary MAC prophylaxis? Slide 18 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 7 • 38 yo female newly diagnosed 6 months ago • HIV RNA 1, 280, 000 c/ml (HLA-B 5701 negative) • CD 4 count 15 cells/ul • Started on ELV / cobi / FTC / TAF 6 weeks ago • Initial HIV resistance assay: Wild-type virus • Admitted for PJP Rx 2 wks after starting ARV Rx (IRIS) • Viral load now 1, 200 c/ml; CD 4 27 cells / ul Slide 19 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question Should I stop abacavir in a patient with CAD? Slide 20 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.
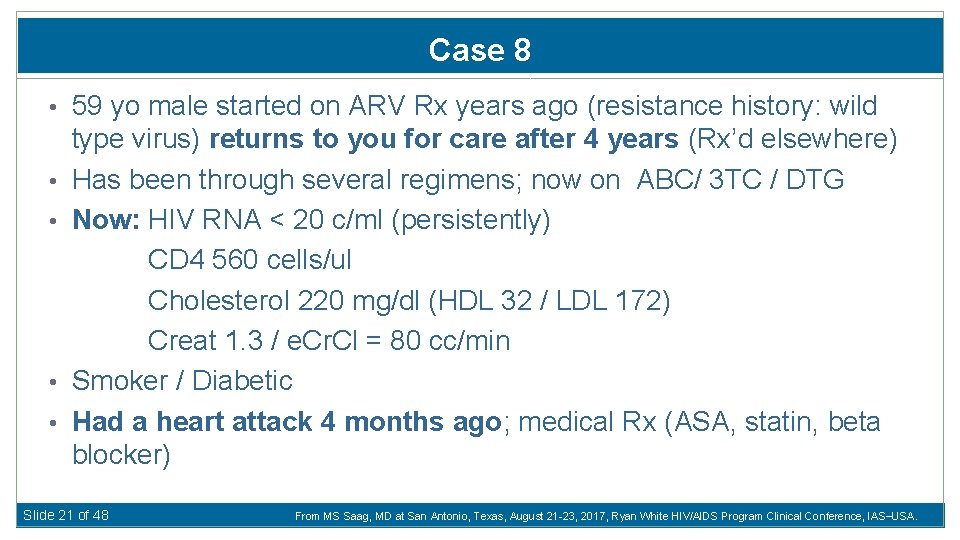
Case 8 • 59 yo male started on ARV Rx years ago (resistance history: wild • • type virus) returns to you for care after 4 years (Rx’d elsewhere) Has been through several regimens; now on ABC/ 3 TC / DTG Now: HIV RNA < 20 c/ml (persistently) CD 4 560 cells/ul Cholesterol 220 mg/dl (HDL 32 / LDL 172) Creat 1. 3 / e. Cr. Cl = 80 cc/min Smoker / Diabetic Had a heart attack 4 months ago; medical Rx (ASA, statin, beta blocker) Slide 21 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.
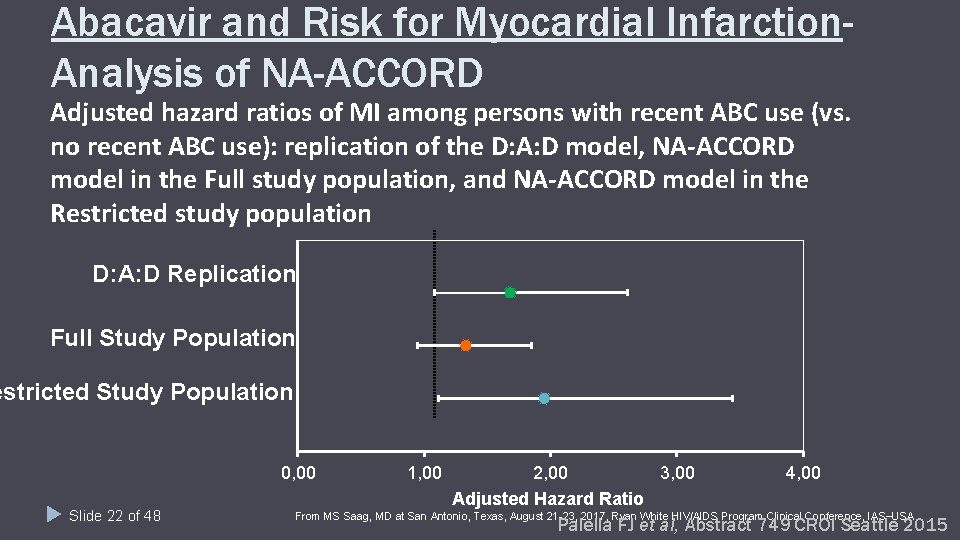
Abacavir and Risk for Myocardial Infarction. Analysis of NA-ACCORD Adjusted hazard ratios of MI among persons with recent ABC use (vs. no recent ABC use): replication of the D: A: D model, NA-ACCORD model in the Full study population, and NA-ACCORD model in the Restricted study population D: A: D Replication Full Study Population estricted Study Population 0, 00 Slide 22 of 48 1, 00 2, 00 3, 00 Adjusted Hazard Ratio 4, 00 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA. Palella FJ et al, Abstract 749 CROI Seattle 2015
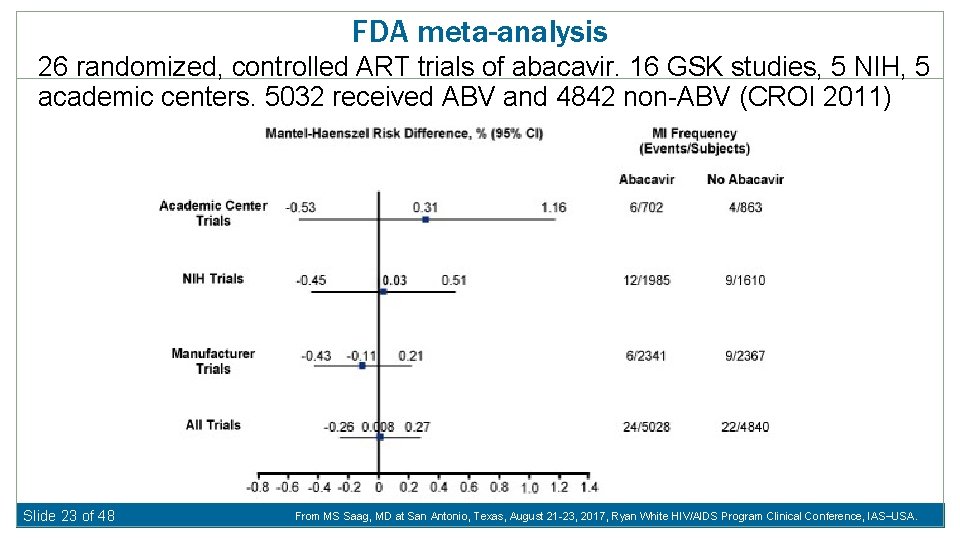
FDA meta-analysis 26 randomized, controlled ART trials of abacavir. 16 GSK studies, 5 NIH, 5 academic centers. 5032 received ABV and 4842 non-ABV (CROI 2011) Slide 23 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.
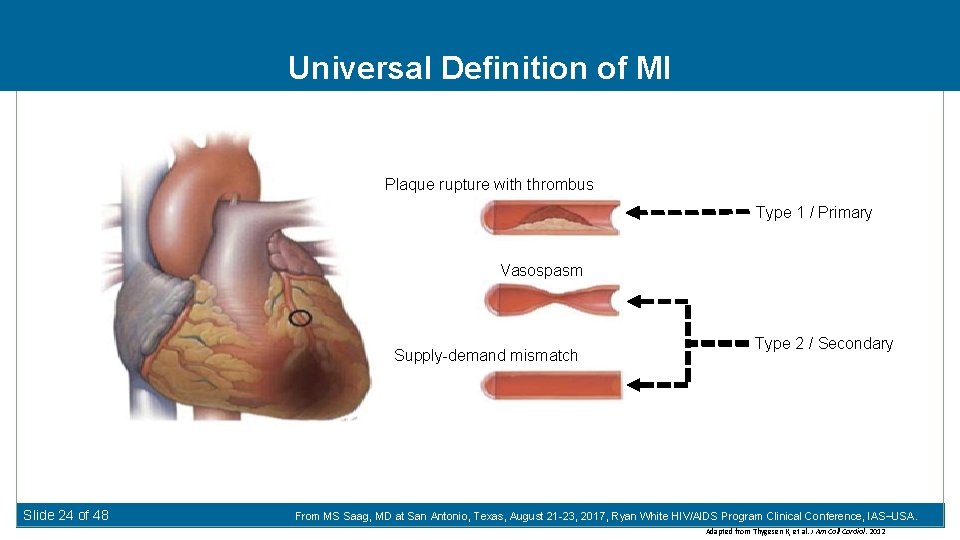
Universal Definition of MI Plaque rupture with thrombus Type 1 / Primary Vasospasm Supply-demand mismatch Slide 24 of 48 Type 2 / Secondary From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA. Adapted from Thygesen K, et al. J Am Coll Cardiol. 2012

MI Classification Protocol Universal Definition of MI: Causes of Secondary MI in HIV-infected Individuals* N (%) Primary MI (Type 1 ‘traditional’ MI atherosclerosis) Plaque rupture with thrombus Secondary MI (Type 2 supply-demand mismatch) Vasospasm Secondary MIs common in HIV-infected individuals before age 50 Slide 25 of 48 Sepsis/bacteremia 100 (35%) Cocaine induced/illicit drug 39 (14%) Hypertensive urgency/emergency 28 (10%) Respiratory failure 26 (9%) Non-coronary cardiac 23 (8%) Hypotension 15 (5%) Procedure related 12 (4%) GI bleed 11 (4%) Neurologic 6 (2%) Overdose 5 (2%) Other/unknown 23 (8%) From MS Saag, MD 15 at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA. *Crane et al. Am J Epidemiol Apr 2014

Question Should I change a regimen when low level detectable virus is present? Slide 26 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 9 • 55 yo male referred to you for evaluation • Diagnosed 18 years ago with HIV infection • Initial: HIV RNA 936, 000 c/ml CD 4 count 70 cells/ul • Current: HIV RNA 85 c/ml (prior value 62 c/ml) CD 4 count 525 cells/ul • Started on NEL/D 4 T/3 TC; subsequently treated with • • • LOP-r / TDF/FTC, EFV/ FTC/ TDF (fdc). Now DTG / DRV/c / 3 TC • No historical resistance tests are available Slide 27 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question What regimen should I use as initial therapy in a pregnant patient? Slide 28 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 10 • 30 yo Female presents with newly diagnosed HIV infection • Asymptomatic, 2. 5 months pregnant • Initial: HIV RNA 28, 000 c/ml • • CD 4 count 650 cells/ul Other labs are normal; HLA-B 57 neg Genotype is Wild-type virus No prior medical history. First pregnancy Ok to start therapy if you think she should Slide 29 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

TAF PK - Fetus �Intracellular concentration of Tenofovir-DP is 4 -5 times higher for TAF compared to TDF �Does this expose the fetus to a higher risk of birth abnormalities? �Does this lower the risk of vertical transmission? Andrew Hill, 2016 WHO meeting Slide 30 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Dolutegravir in pregnancy: Background �No fetal toxicity or teratogenicity in animal studies described in manufacturer’s submission for regulatory approval 1 �High placental transfer of DTG relative to other ARVs in an ex vivo study 2 �“Unexpected placental transfer of DTG with fetal accumulation and then slow neonatal clearance” 3 1. European Medicines Agency. INN-dolutegravir. Annex I: Summary of Product Characteristics. http: //www. ema. europa. eu/docs/en_GB/document_library/EPAR__Product_Information/human/002753/WC 500160680. pdf 2. Schalkwijk S, Greupink R, Colbers AP, Wouterse AC, Verweij VGM, van Drongelen J, et al. Placental transfer of the HIV integrase inhibitor dolutegravir in an ex vivo human cotyledon perfusion model. J Antimicrob Chemother. 2015 Nov 3 pii: dkv 358. [Epub ahead of print] Available from: http: //www. jac. oxfordjournals. org/lookup/doi/10. 1093/jac/dkv 358 3. Pain JB, Lê MP, Caseris M, Amiel C, Lassel L, Charpentier C, et al. Pharmacokinetics of Dolutegravir in a Premature Neonate after HIV Treatment Intensification during Pregnancy: FIG 1. Antimicrob Agents Chemother [Internet]. 2015 Jun; 59(6): 3660– 2. Available from: http: //aac. asm. org/lookup/doi/10. 1128/AAC. 00173 -15 Slide 31 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Conclusion • Debate about whether to treat Elite Controllers • Presence of M 184 V does not effect initial Rx much (except for use of ABC at higher viral load) • Primary and Secondary MIs are distributed equally in HIV patients • Hold off on using TAF or DTG in pregnant women, pending more data • Do not need to change ARV therapy if persistent low level viremia Slide 32 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Question How can I simplify a complex regimen in a highly treatmentexperienced patient? Slide 33 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.

Case 412 � 57 year old man diagnosed with HIV in 1991; multiple opportunistic infections and complains of ‘Pill Fatigue’ �Has taken most existing antiretroviral drugs available; no exposure to maraviroc, DTG, or ELV �Currently on TDF / FTC / ETV / DRV-r / RAL (1 st In. STI) – CD 4+ count 330 /u. L (nadir CD 4 = 6) – HIV RNA <40 c/m. L (max VL 667, 000) Slide 34 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.
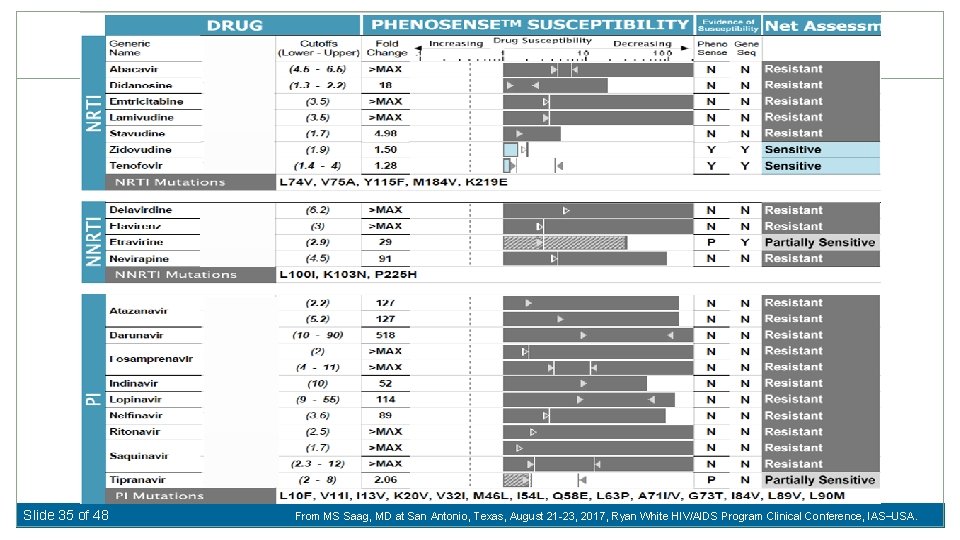
Slide 35 of 48 From MS Saag, MD at San Antonio, Texas, August 21 -23, 2017, Ryan White HIV/AIDS Program Clinical Conference, IAS USA.
- Slides: 35