Antiretroviral Therapy ART for Patients with TB David

Antiretroviral Therapy (ART) for Patients with TB David L. Cohn, MD Denver Public Health University of Colorado Health Sciences Center Denver, Colorado, USA 5 June 2003


Antiretroviral Therapy for Patients with TB Introduction • ART will be used increasingly in developing countries • In many HIV-infected patients, TB will be the presenting illness and/or will occur commonly • In addition to anti-TB therapy, many patients may also require ART; the optimal timing of ART is uncertain • Treatment with both anti-TB and ARV drugs is complex, especially in newly-diagnosed patients • Recommendations on when to treat and how to manage ART in patients with TB are not consistent or absent in different regions of the world

Antiretroviral Therapy for Patients with TB Approach • Review evidence from published and presented data, ongoing studies, and other field experience where ART is being implemented • Assess recommendations in light of what is known and what is not known • Provide practical information, especially when laboratory support is lacking • What is most relevant to the provider when seeing a patient?

Antiretroviral Therapy for Patients with TB Proposed Chapter • When to start ART • Drug-drug interactions and overlapping toxicities • Adherence • Clinical management • Research agenda To be consistent with and complementary to other WHO documents

Antiretroviral Therapy for Patients with TB When to Start: Resource-Limited Settings • Pulmonary TB and CD 4 count <50/mm 3 or extrapulmonary TB: Start as soon as anti-TB therapy is tolerated • Pulmonary TB and CD 4 count 50 -200/mm 3 or total lymphocyte count <1200/mm 3: Start after 2 months of anti-TB therapy • Pulmonary TB and CD 4 count >200/mm 3 or total lymphocyte count >1200/mm 3: Don’t start. Monitor CD 4 counts if available and start ART if WHO Stage 4 or CD 4 count falls to <200/mm 3 WHO, 2002

Antiretroviral Therapy for Patients with TB When to Start: Brazil • CD 4 count <200/mm 3: Treat TB and initiate ART • CD 4 count 200 -350/mm 3: Treat TB and if appropriate initiate ART • CD 4 count >350/mm 3: Treat TB, do not initiate ART; re-evaluate after 30 -60 days • CD 4 count and viral load unknown: Treat TB, await clinical stability, and then check CD 4 count and viral load Need to assess risk of toxicity and adherence; in cases of less severe immunodeficiency postponement of ART might be considered STD/AIDS Program, Mo. H, 2001

Antiretroviral Therapy in Patients with TB When to Start: United States • Do not delay treatment of TB; optimal time for initiating ART is unknown • Avoid beginning simultaneous administration of anti-TB therapy and ART • Delaying initiation of ART for 4 -8 weeks after anti-TB therapy has potential advantages of assessing cause of side effects, decreasing paradoxical reactions, and enhancing adherence • Decision should be individualized based on response to anti-TB therapy, side effects, and readiness for ART • Need for clinical trials USPHS/IDSA, 2003

Recommended Antiretroviral Regimens to be Used with Rifampin-Containing Anti-TB Treatment • ZDV / 3 TC / ABC • ZDV / 3 TC / EFZ • ZDV / 3 TC / SQV / RTV • ZDV / 3 TC / NVP Or where available: • 2 NRTIs (e. g. , ZDV/3 TC, d 4 T/dd. I) + EFZ • 2 NRTIs + SQV/RTV • 2 NRTIs + NVP

Initiation of Antiretroviral Therapy for Patients with TB To Start or Delay? Reasons to Start ART • Decrease morbidity and mortality related to HIV/AIDS Reasons to delay ART • Overlapping side effects from ART and anti-TB therapy • Complex drug-drug interactions • Immune reconstitution inflammatory syndrome (paradoxical reactions) • Difficulties with adherence to multiple medications
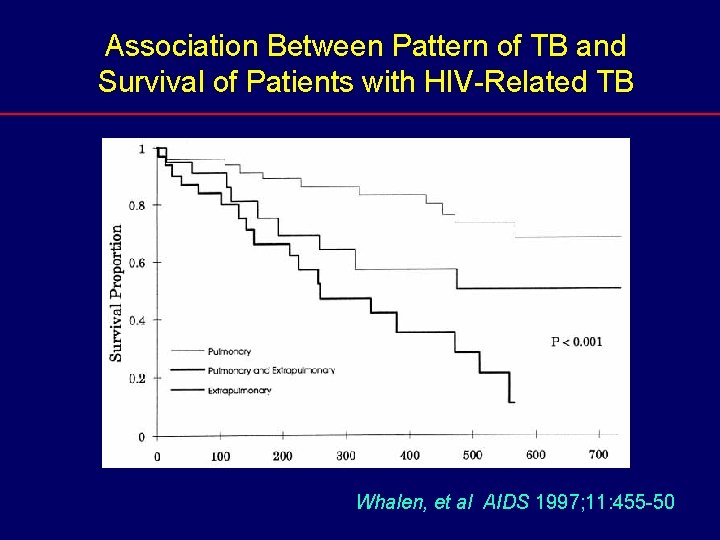
Association Between Pattern of TB and Survival of Patients with HIV-Related TB Whalen, et al AIDS 1997; 11: 455 -50
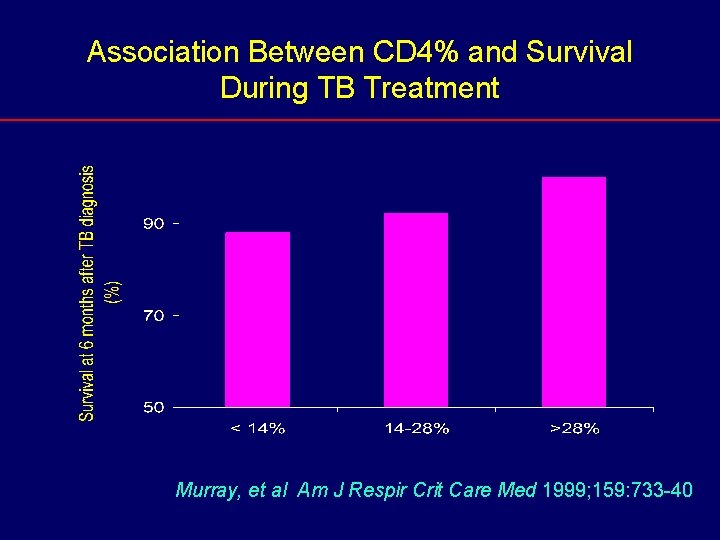
Association Between CD 4% and Survival During TB Treatment Murray, et al Am J Respir Crit Care Med 1999; 159: 733 -40

Use of ART During TB Treatment CDC TB Trials Consortium (TBTC) Study 23 TBTC Study 23: Prospective, single-arm study of the efficacy and safety of a largely twice-weekly rifabutinbased regimen for HIV-related TB • 38 patients (22%) on ART when diagnosed with TB • 97 patients (57%) started ART during TB treatment Summary - 135 of 169 patients (80%) received ART during TB treatment
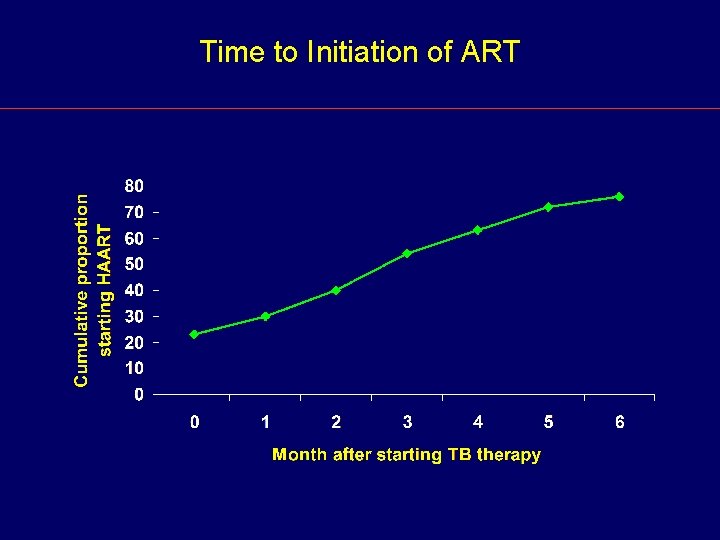
Time to Initiation of ART
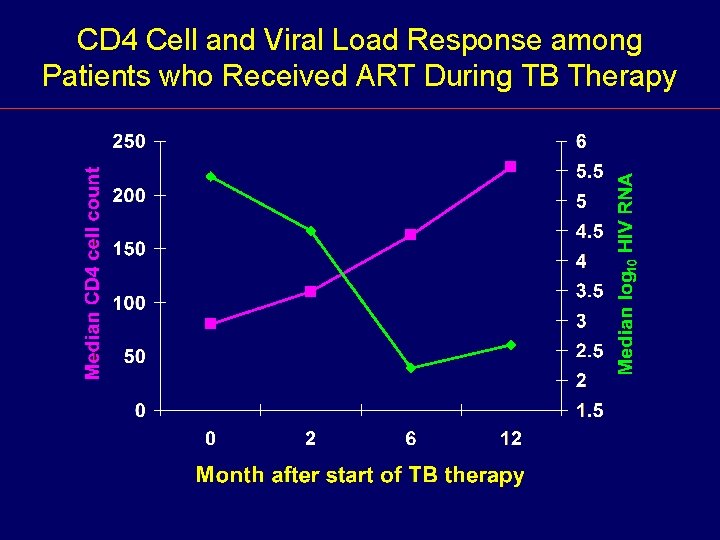
CD 4 Cell and Viral Load Response among Patients who Received ART During TB Therapy
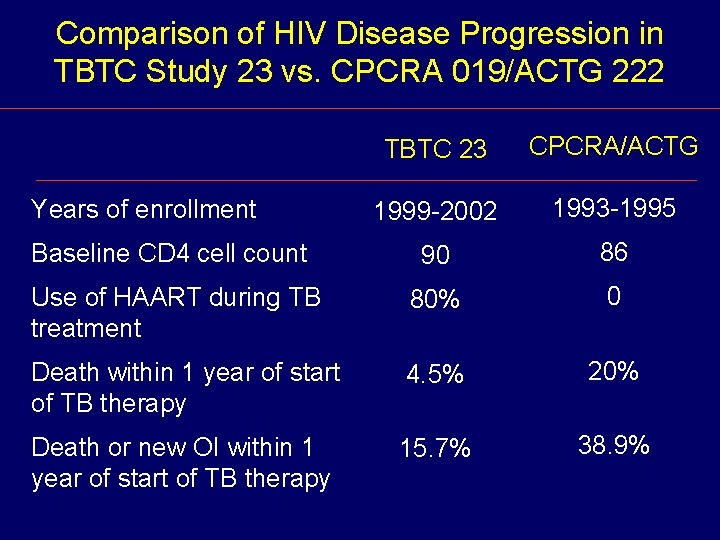
Comparison of HIV Disease Progression in TBTC Study 23 vs. CPCRA 019/ACTG 222 TBTC 23 CPCRA/ACTG 1999 -2002 1993 -1995 Baseline CD 4 cell count 90 86 Use of HAART during TB treatment 80% 0 Death within 1 year of start of TB therapy 4. 5% 20% Death or new OI within 1 year of start of TB therapy 15. 7% 38. 9% Years of enrollment

Overlapping Side Effect Profiles of First-line Antituberculosis Drugs and Antiretroviral Drugs Possible causes Side effect Antituberculosis drugs Antiretroviral drugs Skin rash PZA, RIF, INH NVP, EFZ, ABC Nausea, vomiting PZA, RIF, RBT, INH ZDV, RTV, AMP, IDV Hepatitis PZA, RIF, RBT, INH NVP, PIs, Immune reconstitution Leukopenia, anemia RBT, RIF ZDV

Drug-Drug Interactions • Effects of rifamycins on levels of ART drugs • Effects of ART on levels of rifamycins • Effects of rifamycins on other drugs commonly used in HIV care

Effect of Rifampin on Serum Concentrations of Protease Inhibitors and Non-Nucleoside Reverse Transcriptase Inhibitors PI NNRTI Saquinavir 80% Nevirapine 37 -58% Ritonavir 35% Efavirenz 13 -26% Indinavir 90% Nelfinavir 82% Amprenavir 81% Lopinavir/ritonavir 75% Atazanavir not done

Types of Immune Reconstitution Inflammatory Syndrome among Patients with HIV-Related TB • Hectic fever • New or worsening adenitis - peripheral or central nodes • New or worsening pulmonary infiltrates, including respiratory failure • New or worsening pleuritis, pericarditis, or ascites • Intracranial tuberculomas, worsening meningitis • Disseminated skin lesions • Epididymitis, hepatosplenomegaly, soft tissue abscesses


Cough, Fevers, and Chest Pain During Treatment for TB - 1 • 43 yr old man from Ethiopia – admitted with fevers and cough • CXR – Left lower lobe infiltrate • AFB smear-positive, drug-susceptible M. tuberculosis • HIV-positive, CD 4 cell count – 12/mm 3, VL – 548, 000 copies/ml • Marked improvement with INH, RBT, PZA, EMB



Cough, Fevers, and Chest Pain During Treatment for TB - 2 • Started on d 4 T, 3 TC, nelfinavir after 2 months of anti-TB therapy • 7 days later – fevers, cough, Lt pleuritic chest pain • Sputum – AFB smear-positive (eventually culture-negative)

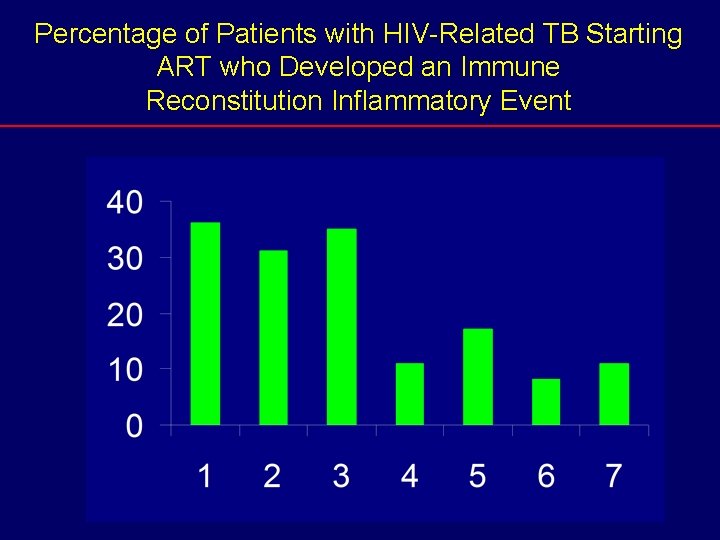
Percentage of Patients with HIV-Related TB Starting ART who Developed an Immune Reconstitution Inflammatory Event

Challenges Related to Adherence When Starting Multiple Medications Would you like to get started on the following medications on the same day? Isoniazid, rifampin, ethambutol, pyrazinamide And: Zidovudine, lamivudine, efavirenz And possibly: Cotrimoxazole, Fluconazole

Clinical Management Issues - 1 • Management with or without laboratory support, e. g. , Complete blood counts, CD 4 cell counts, liver function tests, chest x-rays • Adverse drug reactions/toxicity – Minor vs Major – Treat through or Discontinue – Rechallenge or Change drugs • Paradoxical reactions – How to recognize – Symptomatic treatment, e. g. , use of non-steroidal anti-inflammatory drugs or corticosteroids

Clinical Management Issues - 2 • Use of other HIV-related medications, e. g. , cotrimoxazole, fluconazole • When to refer patients • Use of the Integrated Management of Adolescent/Adult Illness (IMAI) modules

Antiretroviral Therapy for Patients with TB Research Agenda • When to start ART? – Clinical trials - e. g. , START Study, South Africa: Randomize to start ART at same time or at completion of anti-TB therapy (6 months) • Operational research – Descriptive studies of field experiences – Focus on co-morbidities, mortality, adverse events, paradoxical reactions – Use of CD 4 cell counts in decision making • Outcomes with and without laboratory support • Determinants of adherence in patients on multiple drugs; adherence interventions

Antiretroviral Therapy for Patients with TB Proposed Chapter - Process and Timeline June-August 2003: Select experts to write sections of the chapter September-October 2003: Circulate a draft to Scientific Panel of TB/HIV Working Group for critique and to generate questions December 2003: Consultation meeting to further discuss the chapter and specific recommendations February-March 2004: Finalize chapter

Questions for Discussion • Is the proposed chapter a good idea, i. e. , will it be of use in addition to current guidelines or manuals? • If no, what would be more helpful? • If yes, what components are most useful? Least useful? • Should we add anything? Delete anything?
- Slides: 33