Antipsychotic Drugs and Risk of Idiopathic Venous Thromboembolism

Antipsychotic Drugs and Risk of Idiopathic Venous Thromboembolism: A Nested Case-Control Study Using the CPRD Valentin Angles Chelsea Thompson 04 -21 -2016

Background • Antipsychotics drugs are used primarily to treat schizophrenia • Antipsychotics drugs can also be used for other mental health conditions such as agitation, anxiety, mania, and aggression • They can be classified into 2 groups: • Typical antipsychotics (1 st generation) lower DA levels • Atypical antipsychotics (2 nd generation) lower DA and increase 5 HT 2 A levels Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175
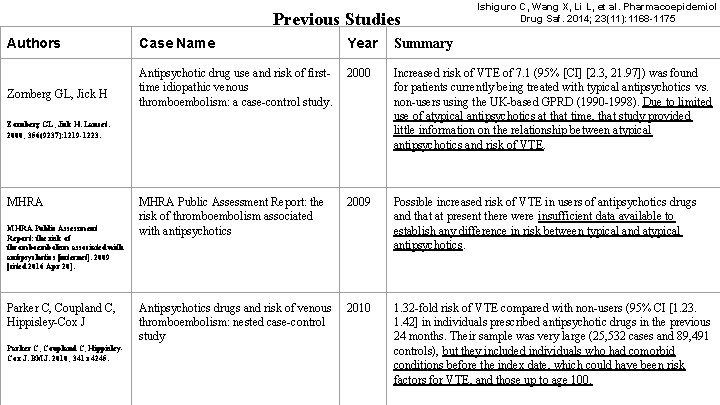
Previous Studies Authors Zornberg GL, Jick H Case Name Year Summary Antipsychotic drug use and risk of firsttime idiopathic venous thromboembolism: a case-control study. 2000 Increased risk of VTE of 7. 1 (95% [CI] [2. 3, 21. 97]) was found for patients currently being treated with typical antipsychotics vs. non-users using the UK-based GPRD (1990 -1998). Due to limited use of atypical antipsychotics at that time, that study provided little information on the relationship between atypical antipsychotics and risk of VTE. MHRA Public Assessment Report: the risk of thromboembolism associated with antipsychotics 2009 Possible increased risk of VTE in users of antipsychotics drugs and that at present there were insufficient data available to establish any difference in risk between typical and atypical antipsychotics. Antipsychotics drugs and risk of venous thromboembolism: nested case-control study 2010 1. 32 -fold risk of VTE compared with non-users (95% CI [1. 23. 1. 42] in individuals prescribed antipsychotic drugs in the previous 24 months. Their sample was very large (25, 532 cases and 89, 491 controls), but they included individuals who had comorbid conditions before the index date, which could have been risk factors for VTE, and those up to age 100. Zornberg GL, Jick H. Lancet. 2000; 356(9237): 1219 -1223. MHRA Public Assessment Report: the risk of thromboembolism associated with antipsychotics [internet]. 2009 [cited 2016 Apr 20]. Parker C, Coupland C, Hippisley-Cox J Parker C, Coupland C, Hippisley. Cox J. BMJ. 2010; 341: c 4245. Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Background on Current Study • Due to previous inconclusive studies, a nested case-control study was performed using the Clinical Practice Research Datalink (CPRD) to assess the risk of VTE in relation to: • Exposure to antipsychotics in a young adult population (ages 20 -59 years), where the presence of strong risk factors/competing causes is low • Including only idiopathic cases to eliminate bias and confounding • Exposure according to class of antipsychotic drug, individual antipsychotic drug, dose, and duration, in addition to looking at recent and current use. Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Inclusion Criteria • All people with incident VTE including DVT and PE that occurred at any time between 1998 and 2012 which required anticoagulation treatment or was fatal • Age 20 -59 years at diagnosis • All subjects were required to have at least 1 year of medical history in the CPRD before the index date (The date of 1 st diagnosis of VTE) Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Exclusion Criteria *Other proximate causes for VTE in the 3 months prior to the index date: • Lower-limb injury • Invasive surgery • Severe trauma • Pregnancy *Patients with important clinical risk factors any time before the index date including: • History of cancer (except non-melanoma cancer) • Renal failure • Epilepsy • Insulin-dependent DM • Multiple sclerosis • MI, CVA, other CV disease • Alcohol abuse or drug abuse Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Exclusion Criteria Continued • Patients with Down’s syndrome • Ulcerative and other colitis • Lupus erythematosus • RA • Ankylosing spondylitis and other spondylopathies • Psoriatic arthritis • Coagulation defects (including a history of anticoagulation therapy) • Cystic fibrosis, or antiphospholipid syndrome • Cases that had other causes for VTE in the last 3 months (surgery, trauma, and pregnancy) Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Primary Endpoint Development of incident VTE upon use of typical and atypical antipsychotics Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175
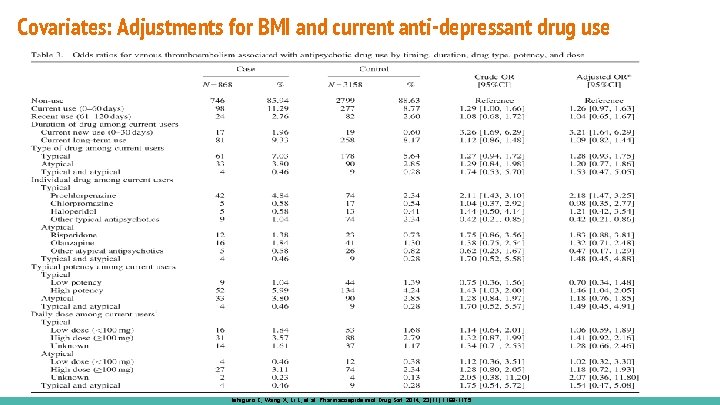
Covariates: Adjustments for BMI and current anti-depressant drug use Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175
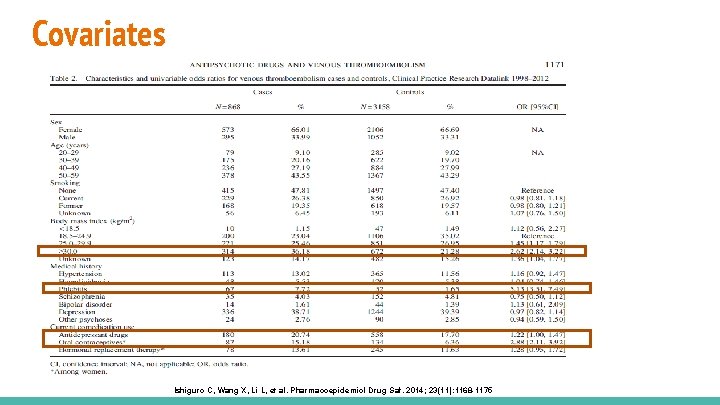
Covariates Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Study Design • Nested case-control study was conducted among the population of people in the CPRD who had filled at least 1 prescription for an antipsychotic drug (Table 1) between Jan 1, 1998 and the end date of data collection in 2012 • Identified those aged 20 -59 at diagnosis with DVT or PE with minimal baseline risk of VTE to assess the risk of antipsychotics • VTE was defined as first time DVT or PE that was fatal or required anticoagulation Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Study Design • The rationale for this study design was to: • Limit the effects of psychiatric diagnoses (such as schizophrenia) on the development of VTE • Optimize the exposure prevalence in the base population, thus ensuring an efficient ratio of exposed to unexposed individuals Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Study Design CONTROLS: • From the base population, up to 4 controls to match each case were selected based on sex, general practice, time, and age (by using the same index date and calander time as their matched case). Meaning cases could be used as controls before they had VTE. • The same exclusion criteria were applied to the controls • For each case and control they defined current, recent, and past exposure to antipsychotic drugs • Current = prescription within 60 days • Recent= 61 -120 • Past exposure (121 -365) and non exposure were combined due to the long timeline Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Statistical Analyses • The data was analyzed using conditional logistic regression to estimate ORs and 95% CIs for the risk of idiopathic VTE in relation to antipsychotic drugs, taking into account the matching factors of age, sex, general practice, calendar time, and years of recorded history • All statistical analyses were performed using SAS Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175
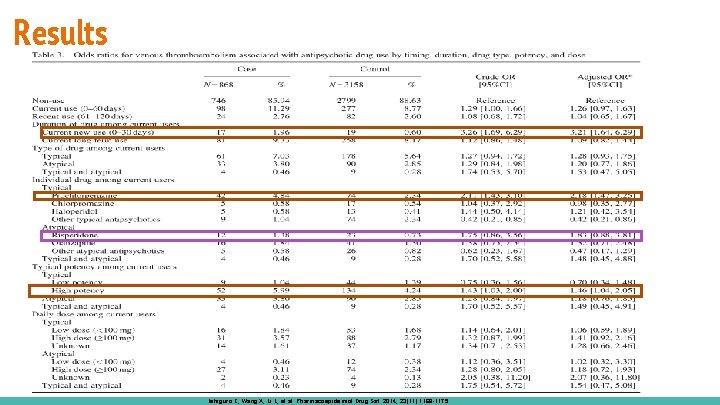
Results Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Author’s Conclusions • The findings were similar to previous studies yielding minimally increased risk of VTE, however a stronger affect was found in current new users (started within the last 30 days of index) • The risk was elevated or users of prochlorperazine and risperidone Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Author’s Discussion • Prochlorperazine accounted for 81% of all cases currently exposed to highpotency typical antipsychotic drugs at diagnoses, prochloperazine is also associated with nausea, vomiting, and vertigo • Thus, other conditions associated with nausea and vertigo might have increased risk of VTE, and the risk of high-potency typical antipsychotics could not be established Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Discussion

Strengths of Study • Data was retrieved from CPRD • Exclusion of strong risk factors for VTE • Restriction of the study to people 20 -59 y. o. • There was at least one prescription for an antipsychotic • The researchers established sampling with replacement by adjusting for covariates • Independently funded Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Limitations of Study • Small number of cases which places limitations on ability to examine the effects of drug and dose categories • Cases were eligible to be selected as controls before developing VTE • Many of the sample groups were not analyzed, including some that may have yielded interesting data such as birth control use • The true length of time the individual was on antipsychotic is sometimes inaccurate in the database Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Statistics • Use of SAS to analyze data from CPRD was appropriate • OR was the appropriate method of statistical analysis • Incidence-density sampling with replacement was appropriate Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Author’s Conclusions • There was an increased risk of VTE in current users of antipsychotic drugs compared with non-users, which was strongest within the 1 st month of use • The effects were strongest in prochlorperazine and risperidone users • There was a “dose effect” in typical antipsychotics drug users only Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Our Conclusion • The increased risk of VTE is minimal with concurrent use of antipsychotics, this is not novel evidence, and is consistent with previous studies • While the intended method of statistical analysis was appropriate, the fact that a control may have at some point had a VTE bring the validity of the data into question • It would have been of clinical interest to see the data on concomitant use of oral contraceptives Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Impact and/or Application to Current Clinical Practice • Initial use of an antipsychotic is associated with increased risk of VTE, thus in patients with other risk factors for VTE this may be an important counseling point Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Further Studies Needed • The mechanism of action for increased VTE risk for antipsychotic drugs • Larger scale/population • Further assess the risk of risperidone and early antipsychotic use on subsequent VTE Ishiguro C, Wang X, Li L, et al. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175

Citations Ishiguro C, Wang X, Li L, et al. Antipsychotic drugs and risk of idiopathic venous thromboembolism: a nested casecontrol study using the CPRD. Pharmacoepidemiol Drug Saf. 2014; 23(11): 1168 -1175. Zornberg GL, Jick H. Antipsychotic drug use and risk of first-time idiopathic venous thromboembolism: a case-control study. Lancet. 2000; 356(9237): 1219 -1223. MHRA Public Assessment Report: the risk of thromboembolism associated with antipsychotics [internet]. 2009 [cited 2016 Apr 20]. Available from: http: //webarchive. nationalarchives. gov. uk/20141205150130/http: //www. mhra. gov. uk/home/groups/plp/documents/websiteresources/con 049075. pdf Parker C, Coupland C, Hippisley-Cox J. Antipsychotics drugs and risk of venous thromboembolism: nested casecontrol study. BMJ. 2010; 341: c 4245.
- Slides: 27